Field Imaging Spectroscopy of Beech Seedlings under Dryness Stress
Abstract
:1. Introduction
2. Material and Methods
2.1. Planting
2.2. Dryness Stress
2.3. Imaging Spectroscopy
2.4. Reference Spectroscopic Leaf Measurements
2.5. Estimation of Equivalent Water Thickness from Imaging Spectroscopy
2.6. Estimation of Chlorophyll Content from Imaging Spectroscopy
3. Results and Discussion
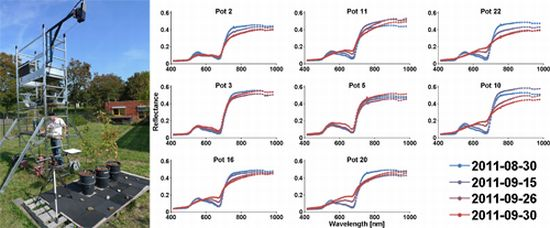
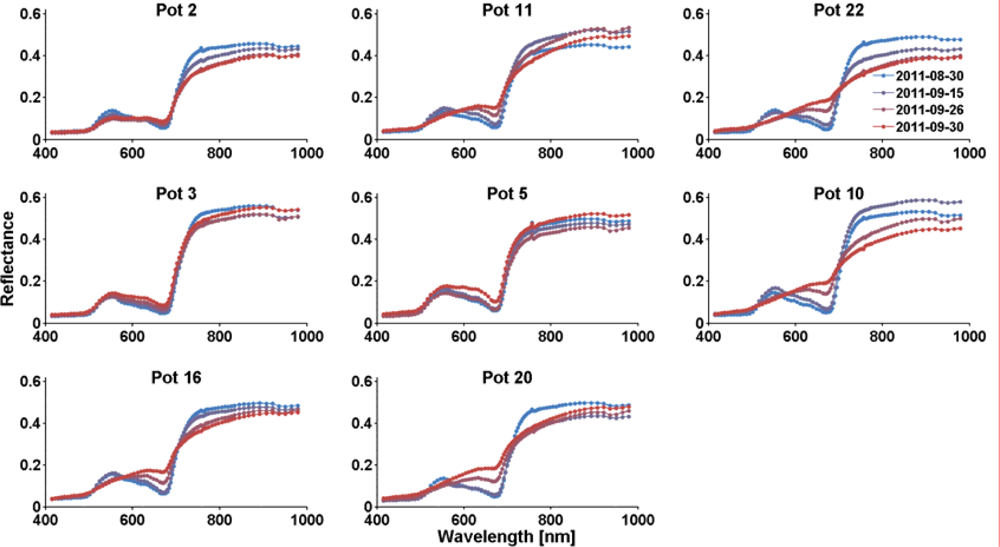
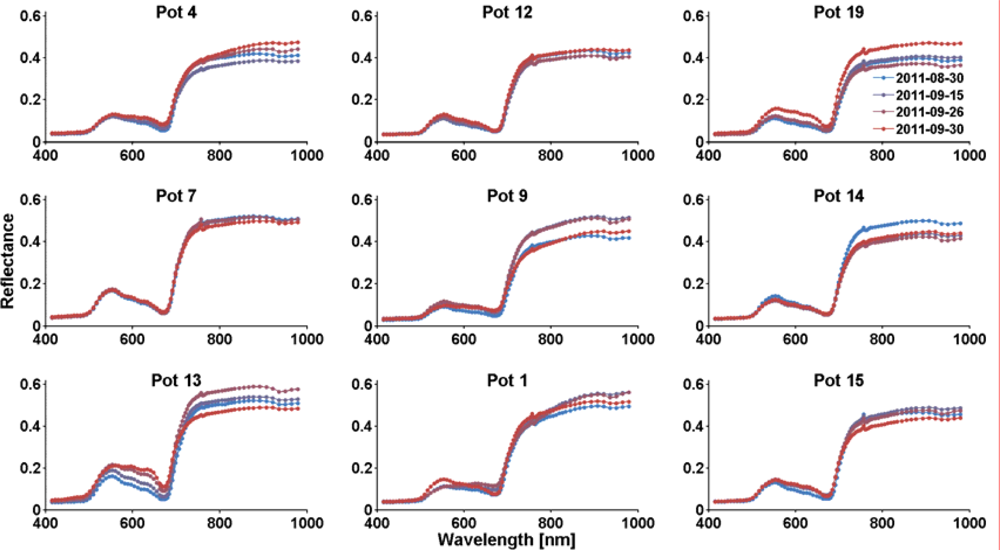
3.1. Analysis of the Hyperspectral Images and Spectra
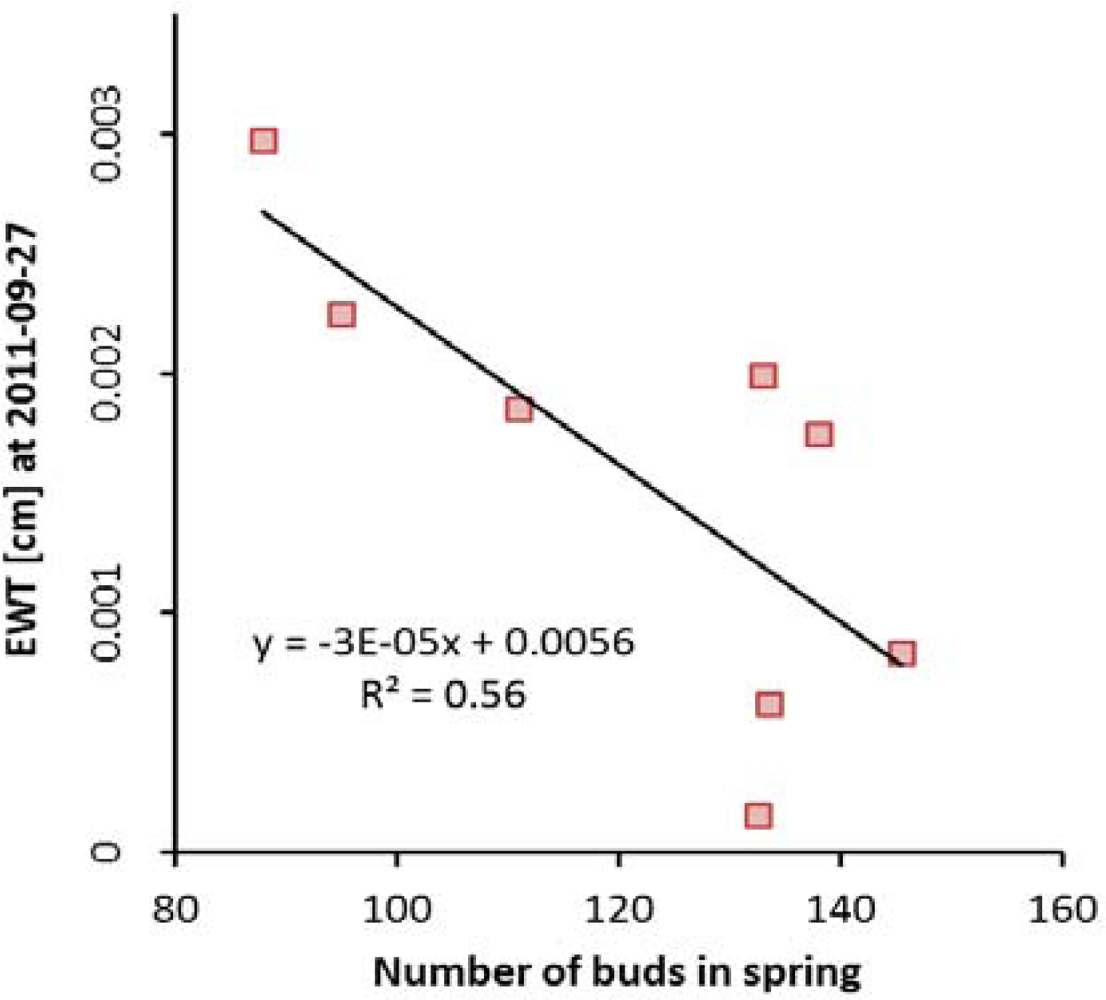
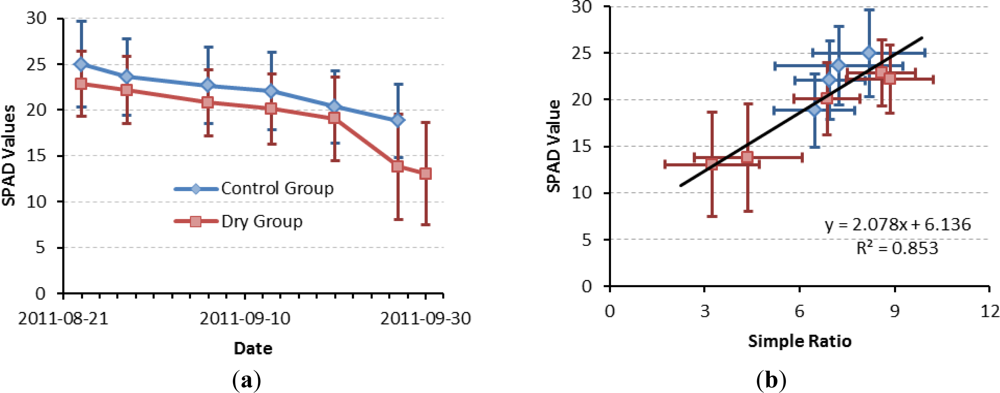
3.2. Dryness Stress
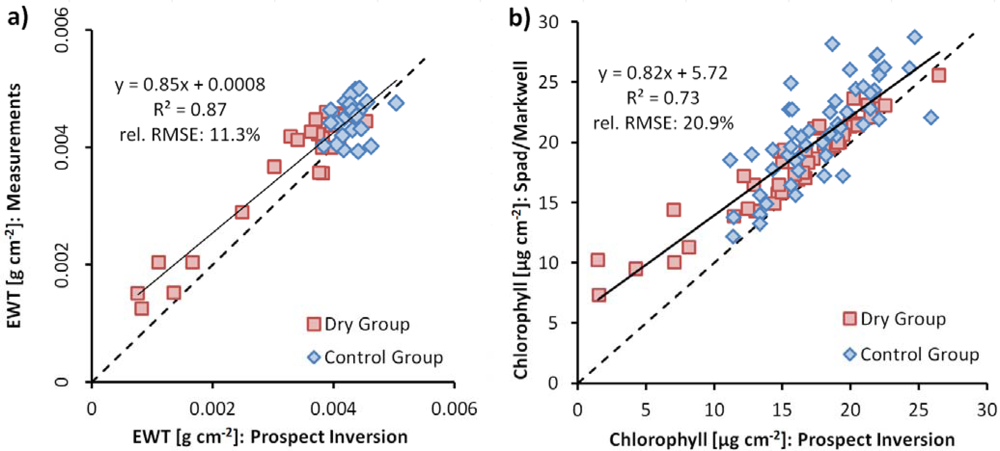
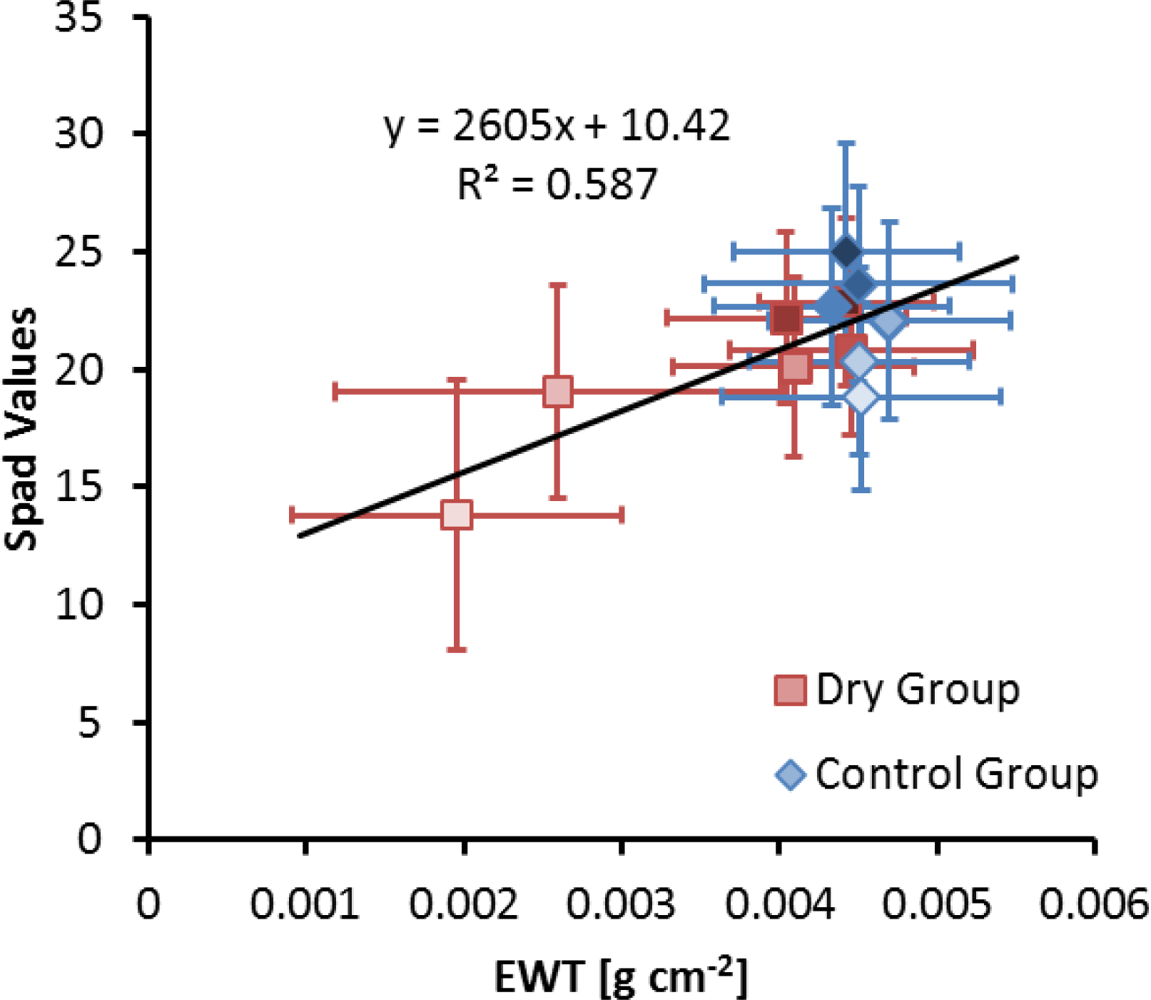
3.3. Estimation of Equivalent Water Thickness from Imaging Spectroscopy
3.4. Chlorophyll Content
4. Conclusions
Acknowledgments
References
- Kaufmann, H.; Segl, K.; Chabrillat, S.; Müller, A.; Richter, R.; Schreier, G.; Hofer, S.; Stuffler, T.; Haydn, R.; Bach, H.; et al. EnMAP—An Advanced Hyperspectral Mission. Proceedings of 4th EARSeL Workshop on Imaging Spectroscopy, Warsaw, Poland, 27–29 April 2005; pp. 55–60.
- Segl, K.; Guanter, L.; Kaufmann, H.; Schubert, J.; Kaiser, S.; Sang, B.; Hofer, S. Simulation of spatial sensor characteristics in the context of the EnMAP hyperspectral mission. IEEE Trans. Geosci. Remote Sens 2010, 48, 3046–3054. [Google Scholar]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manage 2010, 259, 698–709. [Google Scholar]
- Bréda, N.; Huc, R.; Granier, A.; Dreyer, E. Temperate forest trees and stands under severe drought: A review of ecophysiological responses, adaptation processes and long-term consequences. Ann. For. Sci 2006, 63, 625–644. [Google Scholar]
- Gobron, N.; Pinty, B.; Mélin, F.; Taberner, M.; Verstraete, M.M.; Belward, A.; Lavergne, T.; Widlowski, J.L. The state of vegetation in Europe following the 2003 drought. Int. J. Remote Sens 2005, 26, 2013–2020. [Google Scholar]
- Milton, E.J.; Schaepman, M.E.; Anderson, K.; Kneubühler, M.; Fox, N. Progress in field spectroscopy. Remote Sens. Environ 2009, 113, S92–S109. [Google Scholar]
- Blackburn, G.A. Quantifying chlorophylls and caroteniods at leaf and canopy scales: An evaluation of some hyperspectral approaches. Remote Sens. Environ 1998, 66, 273–285. [Google Scholar]
- Yoder, B.J.; Pettigrew-Crosby, R.E. Predicting nitrogen and chlorophyll content and concentrations from reflectance spectra (400–2500 nm) at leaf and canopy scales. Remote Sens. Environ 1995, 53, 199–211. [Google Scholar]
- Verhoef, W.; Bach, H. Coupled soil-leaf-canopy and atmosphere radiative transfer modeling to simulate hyperspectral multi-angular surface reflectance and TOA radiance data. Remote Sens. Environ 2007, 109, 166–182. [Google Scholar]
- Asner, G.P.; Martin, R.E.; Knapp, D.E.; Tupayachi, R.; Anderson, C.; Carranza, L.; Martinez, P.; Houcheime, M.; Sinca, F.; Weiss, P. Spectroscopy of canopy chemicals in humid tropical forests. Remote Sens. Environ 2011, 115, 3587–3598. [Google Scholar]
- Curran, P.J. Remote sensing of foliar chemistry. Remote Sens. Environ 1989, 30, 271–278. [Google Scholar]
- Buddenbaum, H.; Pueschel, P.; Stellmes, M.; Werner, W.; Hill, J. Measuring water and Chlorophyll content on the leaf and canopy scale. EARSeL eProc 2011, 10, 66–72. [Google Scholar]
- Schlerf, M.; Atzberger, C.; Hill, J.; Buddenbaum, H.; Werner, W.; Schüler, G. Retrieval of chlorophyll and nitrogen in Norway spruce (Picea abies L. Karst.) using imaging spectroscopy. Int. J. Appl. Earth Obs. Geoinf 2010, 12, 17–26. [Google Scholar]
- d’Oleire-Oltmanns, S.; Marzolff, I.; Peter, K.; Ries, J. Unmanned Aerial Vehicle (UAV) for monitoring soil erosion in Morocco. Remote Sens 2012, 4, 3390–3416. [Google Scholar]
- Hill, J.; Stellmes, M.; Stoffels, J.; Werner, W.; Shtern, O.; Frantz, D. Assessing the Sensitivity of European Beech (Fagus sylvatica L.) Stands to Severe Drought Based on Measurements from Earth Observation Satellites. Proceedings of 1st Forestry Workshop: Operational Remote Sensing in Forest Management, Prague, Czech Republic, 2–3 June 2011.
- Sims, D.A.; Gamon, J.A. Estimation of vegetation water content and photosynthetic tissue area from spectral reflectance: A comparison of indices based on liquid water and chlorophyll absorption features. Remote Sens. Environ 2003, 84, 526–537. [Google Scholar]
- Peñuelas, J.; Piñol, J.; Ogaya, R.; Filella, I. Estimation of plant water concentration by the reflectance Water Index WI (R900/R970). Int. J. Remote Sens 1997, 18, 2869–2875. [Google Scholar]
- Datt, B. Remote Sensing of water content in Eucalyptus leaves. Aust. J. Bot 1999, 47, 909–923. [Google Scholar]
- Markwell, J.; Osterman, J.C.; Mitchell, J.L. Calibration of the Minolta SPAD-502 leaf chlorophyll meter. Photosynth. Res 1995, 46, 467–472. [Google Scholar]
- Uddling, J.; Gelang-Alfredsson, J.; Piikki, K.; Pleijel, H. Evaluating the relationship between leaf chlorophyll concentration and SPAD-502 chlorophyll meter readings. Photosynth. Res 2007, 91, 37–46. [Google Scholar]
- Buddenbaum, H.; Steffens, M. Laboratory imaging spectroscopy of soil profiles. J. Spect. Imag 2011, 2, 1–5. [Google Scholar]
- Castro-Esau, K.L.; Sánchez-Azofeifa, G.A.; Rivard, B. Comparison of spectral indices obtained using multiple spectroradiometers. Remote Sens. Environ 2006, 103, 276–288. [Google Scholar]
- Delalieux, S.; Auwerkerken, A.; Verstraeten, W.; Somers, B.; Valcke, R.; Lhermitte, S.; Keulemans, J.; Coppin, P. Hyperspectral reflectance and fluorescence imaging to detect scab induced stress in apple leaves. Remote Sens 2009, 1, 858–874. [Google Scholar]
- Feret, J.-B.; François, C.; Asner, G.P.; Gitelson, A.A.; Martin, R.E.; Bidel, L.P.R.; Ustin, S.L.; le Maire, G.; Jacquemoud, S. PROSPECT-4 and 5: Advances in the leaf optical properties model separating photosynthetic pigments. Remote Sens. Environ. 2008, 112, 3030–3043. [Google Scholar]
- Jacquemoud, S.; Baret, F. PROSPECT: a model of leaf optical properties spectra. Remote Sens. Environ 1990, 34, 75–91. [Google Scholar]
- Jacquemoud, S. Prospect + Sail = Prosail. Available online: http://teledetection.ipgp.jussieu.fr/prosail/ (accessed on 23 November 2012).
- Peñuelas, J.; Filella, I.; Biel, C.; Serrano, L.; Savé, R. The reflectance at the 950–970 nm region as an indicator of plant water status. Int. J. Remote Sens 1993, 14, 1887–1905. [Google Scholar]
- Gao, B.-C. NDWI—A normalized difference water index for remote sensing of vegetation liquid water from space. Remote Sens. Environ 1996, 58, 257–266. [Google Scholar]
- Zarco-Tejada, P.J.; Rueda, C.A.; Ustin, S.L. Water content estimation in vegetation with MODIS reflectance data and model inversion methods. Remote Sens. Environ 2003, 85, 109–124. [Google Scholar]
- Thenkabail, P.S.; Lyon, J.G.; Huete, A. Optical Remote Sensing of Vegetation Water Content. In Hyperspectral Remote Sensing of Vegetation; CRC Press: Boca Raton, FL, USA/London, UK/New York, NY, USA, 2012; pp. 227–244. [Google Scholar]
- Schlerf, M.; Atzberger, C.; Hill, J. Remote sensing of forest biophysical variables using HyMap imaging spectrometer data. Remote Sens. Environ 2005, 95, 177–194. [Google Scholar]
- Gamon, J.A.; Peñuelas, J.; Field, C.B. A narrow-waveband spectral index that tracks diurnal changes in photosynthetic efficiency. Remote Sens. Environ 1992, 41, 35–44. [Google Scholar]
- Gamon, J.A.; Serrano, L.; Surfus, J.S. The photochemical reflectance index: An optical indicator of photosynthetic radiation use efficiency across species, functional types, and nutrient levels. Oecologia 1997, 112, 492–501. [Google Scholar]
- Wold, S.; Sjöström, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemometr. Intell. Lab. Syst 2001, 58, 109–130. [Google Scholar]
- Wold, S.; Trygg, J.; Berglund, A.; Antti, H. Some recent developments in PLS modeling. Chemometr. Intell. Lab. Syst 2001, 58, 131–150. [Google Scholar]
- Vohland, M.; Emmerling, C. Determination of total soil organic C and hot water-extractable C from VIS-NIR soil reflectance with partial least squares regression and spectral feature selection techniques. Eur. J. Soil Sci. 2011, 598–606. [Google Scholar]
- Atzberger, C.; Guérif, M.; Baret, F.; Werner, W. Comparative analysis of three chemometric techniques for the spectroradiometric assessment of canopy chlorophyll content in winter wheat. Comput. Electron. Agr 2010, 73, 165–173. [Google Scholar]
- Eisele, A.; Lau, I.; Hewson, R.; Carter, D.; Wheaton, B.; Ong, C.; Cudahy, T.J.; Chabrillat, S.; Kaufmann, H. Applicability of the thermal infrared spectral region for the Prediction of soil properties across semi-arid agricultural landscapes. Remote Sens 2012, 4, 3265–3286. [Google Scholar]
- Féret, J.-B.; François, C.; Gitelson, A.; Asner, G.P.; Barry, K.M.; Panigada, C.; Richardson, A.D.; Jacquemoud, S. Optimizing spectral indices and chemometric analysis of leaf chemical properties using radiative transfer modeling. Remote Sens. Environ 2011, 115, 2742–2750. [Google Scholar]
- Nansen, C. Use of variogram parameters in analysis of hyperspectral imaging data acquired from dual-stressed crop leaves. Remote Sens 2012, 4, 180–193. [Google Scholar]
- Eitel, J.U.H.; Vierling, L.A.; Long, D.S. Simultaneous measurements of plant structure and chlorophyll content in broadleaf saplings with a terrestrial laser scanner. Remote Sens. Environ 2010, 114, 2229–2237. [Google Scholar]




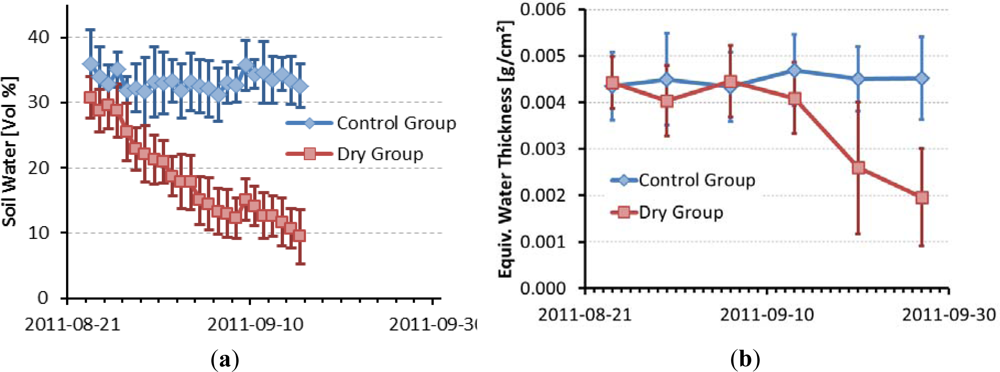




| Date | 2011-08-24 | 2011-08-30 | 2011-09-06 | 2011-09-13 | 2011-09-20 | 2011-09-27 |
|---|---|---|---|---|---|---|
| Mean Dry Group | 0.00442 | 0.00404 | 0.00445 | 0.00409 | 0.00259 | 0.00195 |
| StDev Dry Group | 0.00055 | 0.00076 | 0.00077 | 0.00076 | 0.00141 | 0.00104 |
| Mean Control Group | 0.00442 | 0.00450 | 0.00433 | 0.00469 | 0.00450 | 0.00452 |
| StDev Control Group | 0.00072 | 0.00098 | 0.00074 | 0.00076 | 0.00069 | 0.00089 |
| Significance of t-test | 0.4996 | 0.0492* | 0.3090 | 0.0092** | 4.87E-06*** | 2.89E-09*** |
| Date | 2011-08-23 | 2011-08-28 | 2011-09-06 | 2011-09-13 | 2011-09-20 | 2011-09-27 |
|---|---|---|---|---|---|---|
| Mean Dry Group | 22.86 | 22.18 | 20.81 | 20.12 | 19.04 | 13.80 |
| StDev Dry Group | 3.56 | 3.66 | 3.61 | 3.83 | 4.53 | 5.73 |
| Mean Control Group | 25.00 | 23.62 | 22.68 | 22.07 | 20.35 | 18.84 |
| StDev Control Group | 4.63 | 4.18 | 4.18 | 4.22 | 3.95 | 3.97 |
| Significance of t-test | 0.050* | 0.125 | 0.063 | 0.059 | 0.156 | 0.0007*** |
| Measuring Dates | 08-23/24 | 08-30/31 | 09-06/07 | 09-13/14 | 09-20/21 | 09-27/28 |
|---|---|---|---|---|---|---|
| Correlation all pots | 0.45** | 0.46** | 0.37* | 0.56*** | 0.63*** | 0.77*** |
| Correlation dry group | 0.30 | 0.34 | 0.38° | 0.55** | 0.56** | 0.77*** |
| Correlation control group | 0.63** | 0.63** | 0.37° | 0.56** | 0.56** | 0.53* |
Share and Cite
Buddenbaum, H.; Stern, O.; Stellmes, M.; Stoffels, J.; Pueschel, P.; Hill, J.; Werner, W. Field Imaging Spectroscopy of Beech Seedlings under Dryness Stress. Remote Sens. 2012, 4, 3721-3740. https://doi.org/10.3390/rs4123721
Buddenbaum H, Stern O, Stellmes M, Stoffels J, Pueschel P, Hill J, Werner W. Field Imaging Spectroscopy of Beech Seedlings under Dryness Stress. Remote Sensing. 2012; 4(12):3721-3740. https://doi.org/10.3390/rs4123721
Chicago/Turabian StyleBuddenbaum, Henning, Oksana Stern, Marion Stellmes, Johannes Stoffels, Pyare Pueschel, Joachim Hill, and Willy Werner. 2012. "Field Imaging Spectroscopy of Beech Seedlings under Dryness Stress" Remote Sensing 4, no. 12: 3721-3740. https://doi.org/10.3390/rs4123721