Characterization of the Anti-Influenza Activity of the Chinese Herbal Plant Paeonia lactiflora
Abstract
:1. Introduction
2. Results and Discussion
2.2. Inhibitory Mechanism Determined by a Time-of-Addition Assay
| Concentration (mg/mL) | |||
|---|---|---|---|
| CC50 a | IC50 b | SI c | |
| Cytotoxic effect | |||
| MDCK | 0.210 ± 0.022 | ||
| RD | 0.029 ± 0.001 | ||
| Influenza viruses | |||
| A/WSN/33 (H1N1) | 0.016 ± 0.005 b | 13.5 | |
| 0.015 ± 0.001 d | 14 | ||
| A/TW/6663/09 (H1N1) * | 0.005 ± 0.001 | 42 | |
| A/TW/7717/09 (H1N1) * | 0.005 ± 0.001 | 42 | |
| A/TW/206/09 (H1N1) | 0.007 ± 0.001 | 23 | |
| A/TW/167/09 (H1N1) | 0.005 ± 0.001 | 42 | |
| A/udorn/72 (H3N2) | 0.050 ± 0.001 | 4.2 | |
| A/3446/02 (H3N2) | 0.003 ± 0.0004 | 70 | |
| A/TW/2289/12 (H3N2) | 0.017 ± 0.008 d | 12.4 | |
| A/TW/3003/12 (H3N2) | 0.026 ± 0.006 d | 8.1 | |
| B/TW/70325/05 | 0.036 ± 0.004 | 5.8 | |
| B/TW/99/07 | 0.042 ± 0.015 | 5 | |
| Enteroviruses | |||
| EV71/Tainan/4643/98 | >1 | ||
| Echovirus 9 | >1 | ||
| Coxsackievirus A16 | >1 | ||
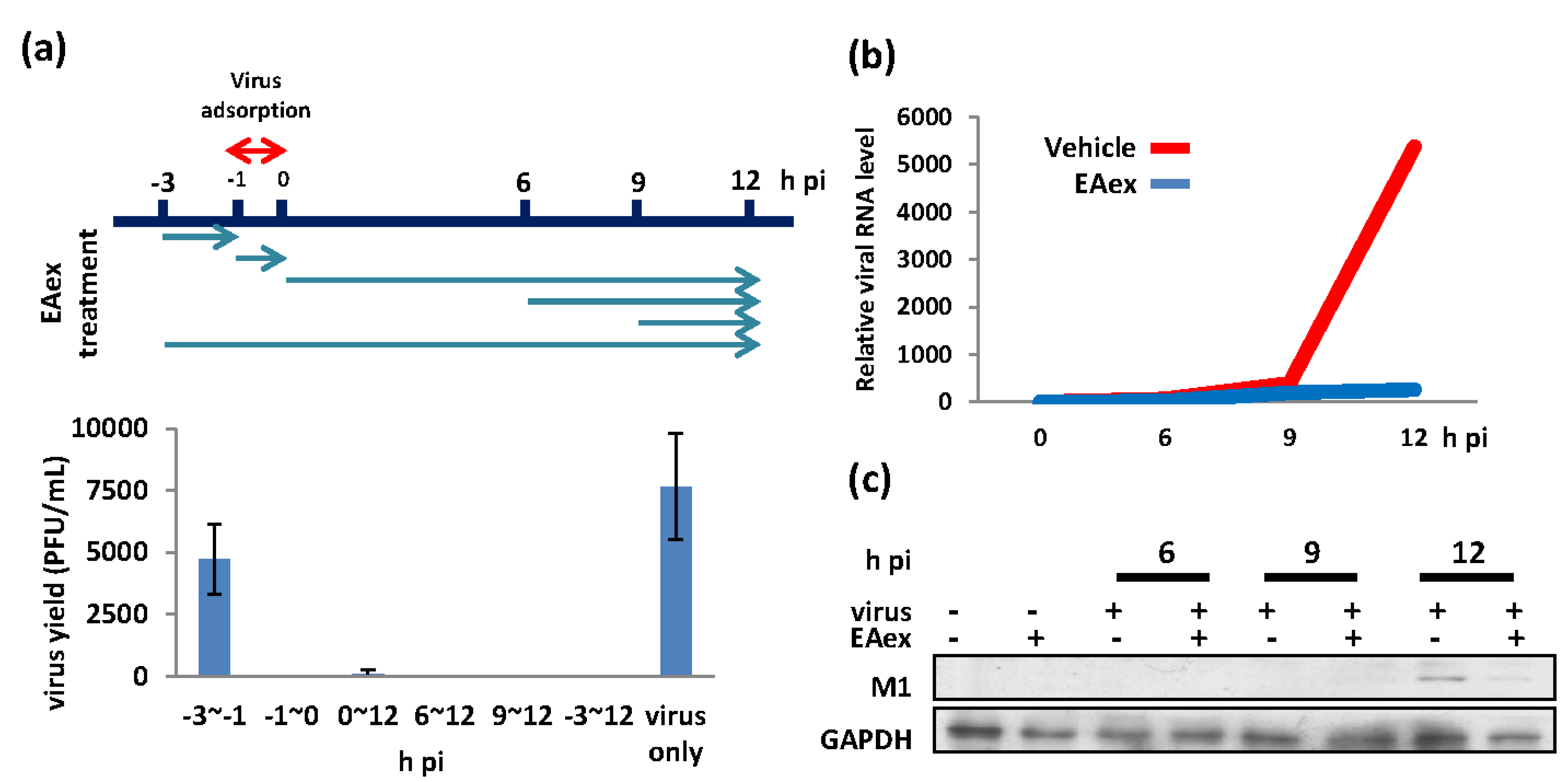
2.3. EAex Inhibits the PI3K-AKT Pathway

2.4. EAex Inhibits Viral Entry

2.5. Effect of EAex on HA and NA Activity
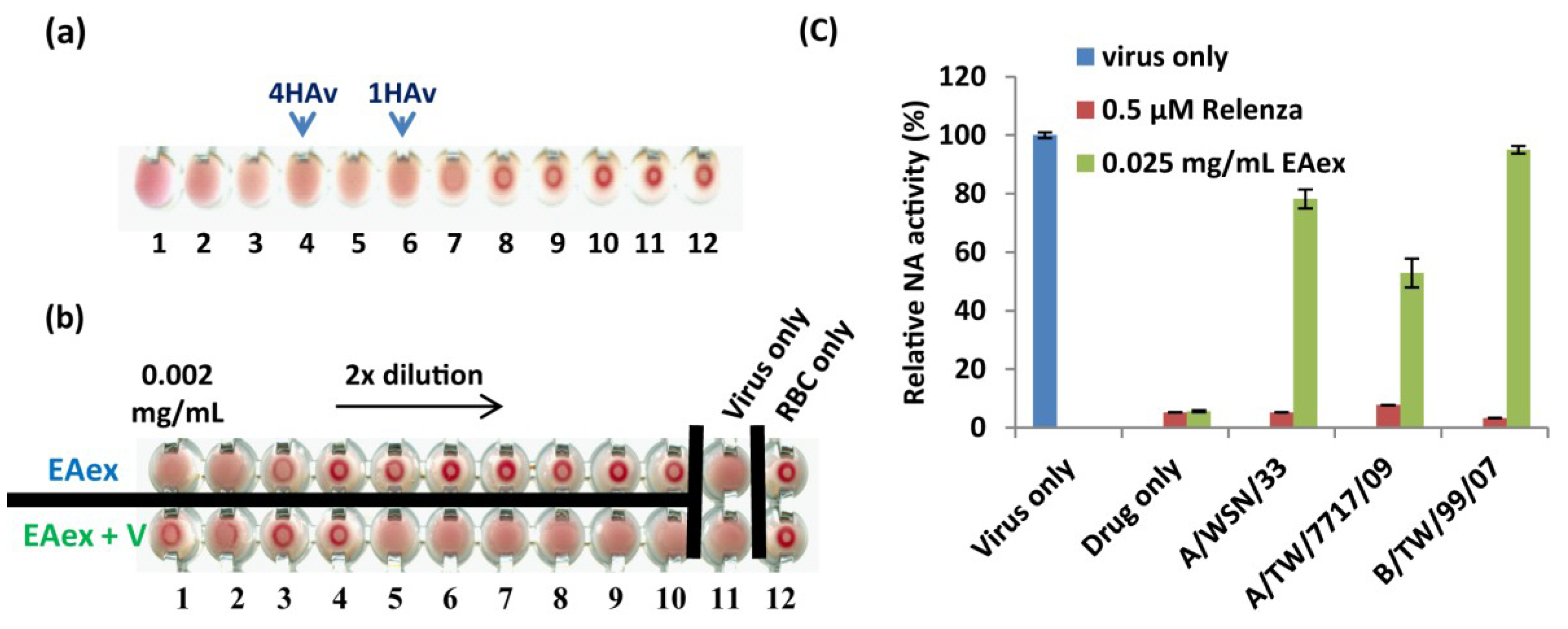
2.6. In Vivo Efficacy Test Using a Mouse Model of Influenza
2.7. Discussion

3. Experimental
3.1. Cell Culture and Virus Infection
3.2. Preparation of EA Extract from BS
3.3. IC50 Determination by Anti-CPE (neutralization) Assay
3.4. IC50 Determination by Plaque Reduction Assay
3.5. Cytotoxicity Assay and MTT Assay
3.6. Time-of-Addition Assay
3.7. RNA Extraction and Quantitative Reverse Transcription-PCR (RT-qPCR)
3.8. Detection of Viral Protein Synthesis and AKT Phosphorylation by Western Blotting
3.9. Attachment Assay
3.10. Penetration Assay
3.11. Hemagglutination Assay (HAv) and Hemagglutination Inhibition (HAI) Assay
3.12. NA Assay
3.13. Mouse Experiment
3.14. Indirect Immunofluorescence Assay
3.15. RNA Polymerase (RdRp) Activity Assay (Minigenome Reporter Assay)
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Nellore, A.; Fishman, J. Pandemic Swine flu 2009. Xenotransplantation 2009, 16, 463–465. [Google Scholar] [CrossRef]
- Sambhara, S.; Poland, G.A. H5N1 Avian influenza: Preventive and therapeutic strategies against a pandemic. Annu. Rev. Med. 2010, 61, 187–198. [Google Scholar] [CrossRef]
- Gao, R.; Cao, B.; Hu, Y.; Feng, Z.; Wang, D.; Hu, W.; Chen, J.; Jie, Z.; Qiu, H.; Xu, K.; et al. Human infection with a novel avian-origin influenza A (H7N9) virus. N. Engl. J. Med. 2013, 368, 1888–1897. [Google Scholar] [CrossRef]
- Guan, Y.; Vijaykrishna, D.; Bahl, J.; Zhu, H.; Wang, J.; Smith, G.J. The emergence of pandemic influenza viruses. Protein Cell 2010, 1, 9–13. [Google Scholar] [CrossRef]
- Muramoto, Y.; Noda, T.; Kawakami, E.; Akkina, R.; Kawaoka, Y. Identification of novel influenza A virus proteins translated from PA mRNA. J. Virol. 2013, 87, 2455–2462. [Google Scholar] [CrossRef]
- Zhu, X.; Yang, H.; Guo, Z.; Yu, W.; Carney, P.J.; Li, Y.; Chen, L.M.; Paulson, J.C.; Donis, R.O.; Tong, S.; et al. Crystal structures of two subtype N10 neuraminidase-like proteins from bat influenza A viruses reveal a diverged putative active site. Proc. Natl. Acad. Sci. USA 2012, 109, 18903–18908. [Google Scholar] [CrossRef]
- Nayak, D.P.; Balogun, R.A.; Yamada, H.; Zhou, Z.H.; Barman, S. Influenza virus morphogenesis and budding. Virus Res. 2009, 143, 147–161. [Google Scholar] [CrossRef]
- Nicholls, J.M.; Chan, R.W.; Russell, R.J.; Air, G.M.; Peiris, J.S. Evolving complexities of influenza virus and its receptors. Trends Microbiol. 2008, 16, 149–157. [Google Scholar] [CrossRef]
- Air, G.M.; Laver, W.G. The neuraminidase of influenza virus. Proteins 1989, 6, 341–356. [Google Scholar] [CrossRef]
- Su, B.; Wurtzer, S.; Rameix-Welti, M.A.; Dwyer, D.; van der Werf, S.; Naffakh, N.; Clavel, F.; Labrosse, B. Enhancement of the influenza A hemagglutinin (HA)-mediated cell-cell fusion and virus entry by the viral neuraminidase (NA). PLoS One 2009, 4, e8495. [Google Scholar] [CrossRef]
- Matrosovich, M.N.; Matrosovich, T.Y.; Gray, T.; Roberts, N.A.; Klenk, H.D. Neuraminidase is important for the initiation of influenza virus infection in human airway epithelium. J. Virol. 2004, 78, 12665–12667. [Google Scholar] [CrossRef]
- Noda, T.; Sagara, H.; Yen, A.; Takada, A.; Kida, H.; Cheng, R.H.; Kawaoka, Y. Architecture of ribonucleoprotein complexes in influenza A virus particles. Nature 2006, 439, 490–492. [Google Scholar] [CrossRef]
- Wang, C.; Takeuchi, K.; Pinto, L.H.; Lamb, R.A. Ion channel activity of influenza A virus M2 protein: Characterization of the amantadine block. J. Virol. 1993, 67, 5585–5594. [Google Scholar]
- Englund, J.A. Antiviral therapy of influenza. Sem. Pediatr. Infect. Dis. 2002, 13, 120–128. [Google Scholar] [CrossRef]
- Whitley, R.J.; Boucher, C.A.; Lina, B.; Nguyen-Van-Tam, J.S.; Osterhaus, A.; Schutten, M.; Monto, A.S. Global Assessment of Resistance to Neuraminidase Inhibitors: 2008–2011. The Influenza Resistance Information Study (IRIS). Clin. Infect. Dis. 2013, 56, 1197–1205. [Google Scholar] [CrossRef]
- Ilyushina, N.A.; Hay, A.; Yilmaz, N.; Boon, A.C.; Webster, R.G.; Govorkova, E.A. Oseltamivir-ribavirin combination therapy for highly pathogenic H5N1 influenza virus infection in mice. Antimicrob. Agents Chemother. 2008, 52, 3889–3897. [Google Scholar]
- Nguyen, J.T.; Hoopes, J.D.; Le, M.H.; Smee, D.F.; Patick, A.K.; Faix, D.J.; Blair, P.J.; de Jong, M.D.; Prichard, M.N.; Went, G.T. Triple combination of amantadine, ribavirin, and oseltamivir is highly active and synergistic against drug resistant influenza virus strains in vitro. PLoS One 2010, 5, e9332. [Google Scholar] [CrossRef]
- Wu, M.S.; Yen, H.R.; Chang, C.W.; Peng, T.Y.; Hsieh, C.F.; Chen, C.J.; Lin, T.Y.; Horng, J.T. Mechanism of action of the suppression of influenza virus replication by Ko-Ken Tang through inhibition of the phosphatidylinositol 3-kinase/Akt signaling pathway and viral RNP nuclear export. J. Ethnopharmacol. 2011, 134, 614–623. [Google Scholar] [CrossRef]
- Hsieh, C.F.; Lo, C.W.; Liu, C.H.; Lin, S.; Yen, H.R.; Lin, T.Y.; Horng, J.T. Mechanism by which ma-xing-shi-gan-tang inhibits the entry of influenza virus. J. Ethnopharmacol. 2012, 143, 57–67. [Google Scholar] [CrossRef]
- Ngan, L.T.; Moon, J.K.; Shibamoto, T.; Ahn, Y.J. Growth-inhibiting, bactericidal, and urease inhibitory effects of Paeonia lactiflora root constituents and related compounds on antibiotic-susceptible and -resistant strains of Helicobacter pylori. J. Agric. Food Chem. 2012, 60, 9062–9073. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, H.K.; Jung, M.K.; Mar, W. In vitro antiviral activity of 1,2,3,4,6-penta-O-galloyl-beta-d-glucose against hepatitis B virus. Biol. Pharm. Bull. 2006, 29, 2131–2134. [Google Scholar] [CrossRef]
- Lin, T.J.; Wang, K.C.; Lin, C.C.; Chiang, L.C.; Chang, J.S. Anti-viral activity of water extract of Paeonia lactiflora pallas against human respiratory syncytial virus in human respiratory tract cell lines. Am. J. Chin. Med. 2013, 41, 585–599. [Google Scholar] [CrossRef]
- Yang, J.R.; Huang, Y.P.; Lin, Y.C.; Su, C.H.; Kuo, C.Y.; Hsu, L.C.; Wu, H.S.; Liu, M.T. Early findings of oseltamivir-resistant pandemic (H1N1) 2009 influenza A viruses in Taiwan. Antiviral Res. 2010, 88, 256–262. [Google Scholar] [CrossRef]
- Ehrhardt, C.; Marjuki, H.; Wolff, T.; Nurnberg, B.; Planz, O.; Pleschka, S.; Ludwig, S. Bivalent role of the phosphatidylinositol-3-kinase (PI3K) during influenza virus infection and host cell defence. Cell. Microbiol. 2006, 8, 1336–1348. [Google Scholar] [CrossRef]
- Ehrhardt, C.; Wolff, T.; Ludwig, S. Activation of phosphatidylinositol 3-kinase signaling by the nonstructural NS1 protein is not conserved among type A and B influenza viruses. J. Virol. 2007, 81, 12097–12100. [Google Scholar] [CrossRef]
- Shin, Y.K.; Liu, Q.; Tikoo, S.K.; Babiuk, L.A.; Zhou, Y. Effect of the phosphatidylinositol 3-kinase/Akt pathway on influenza A virus propagation. J. Gen. Virol. 2007, 88, 942–950. [Google Scholar] [CrossRef]
- Kratz, J.M.; Andrighetti-Frohner, C.R.; Kolling, D.J.; Leal, P.C.; Cirne-Santos, C.C.; Yunes, R.A.; Nunes, R.J.; Trybala, E.; Bergstrom, T.; Frugulhetti, I.C.; et al. Anti-HSV-1 and anti-HIV-1 activity of gallic acid and pentyl gallate. Mem. Inst. Oswaldo Cruz 2008, 103, 437–442. [Google Scholar] [CrossRef]
- Choi, H.J.; Song, J.H.; Bhatt, L.R.; Baek, S.H. Anti-human rhinovirus activity of gallic acid possessing antioxidant capacity. Phytother. Res. 2010, 24, 1292–1296. [Google Scholar] [CrossRef]
- He, D.Y.; Dai, S.M. Anti-inflammatory and immunomodulatory effects of Paeonia lactiflora pall., a traditional chinese herbal medicine. Front. Pharmacol. 2011, 2, e10. [Google Scholar]
- Hsu, A.C.; See, H.V.; Wark, P.A. Innate immunity to influenza in chronic airways diseases. Respirology 2012, 17, 1166–1175. [Google Scholar] [CrossRef]
- Teijaro, J.R.; Walsh, K.B.; Cahalan, S.; Fremgen, D.M.; Roberts, E.; Scott, F.; Martinborough, E.; Peach, R.; Oldstone, M.B.; Rosen, H. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell 2011, 146, 980–991. [Google Scholar] [CrossRef]
- Oldstone, M.B.; Teijaro, J.R.; Walsh, K.B.; Rosen, H. Dissecting influenza virus pathogenesis uncovers a novel chemical approach to combat the infection. Virology 2013, 435, 92–101. [Google Scholar] [CrossRef]
- Hsu, J.T.; Yeh, J.Y.; Lin, T.J.; Li, M.L.; Wu, M.S.; Hsieh, C.F.; Chou, Y.C.; Tang, W.F.; Lau, K.S.; Hung, H.C.; et al. Identification of BPR3P0128 as an inhibitor of cap-snatching activities of influenza virus. Antimicrob. Agents Chemother. 2012, 56, 647–657. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ho, J.-Y.; Chang, H.-W.; Lin, C.-F.; Liu, C.-J.; Hsieh, C.-F.; Horng, J.-T. Characterization of the Anti-Influenza Activity of the Chinese Herbal Plant Paeonia lactiflora. Viruses 2014, 6, 1861-1875. https://doi.org/10.3390/v6041861
Ho J-Y, Chang H-W, Lin C-F, Liu C-J, Hsieh C-F, Horng J-T. Characterization of the Anti-Influenza Activity of the Chinese Herbal Plant Paeonia lactiflora. Viruses. 2014; 6(4):1861-1875. https://doi.org/10.3390/v6041861
Chicago/Turabian StyleHo, Jin-Yuan, Hui-Wen Chang, Chwan-Fwu Lin, Chien-Jou Liu, Chung-Fan Hsieh, and Jim-Tong Horng. 2014. "Characterization of the Anti-Influenza Activity of the Chinese Herbal Plant Paeonia lactiflora" Viruses 6, no. 4: 1861-1875. https://doi.org/10.3390/v6041861




