Pretreatment of Mice with Oligonucleotide prop5 Protects Them from Influenza Virus Infections
Abstract
:1. Introduction
2. Results and Discussion
2.1. Prop5 Protected Animals from Influenza Virus Infections
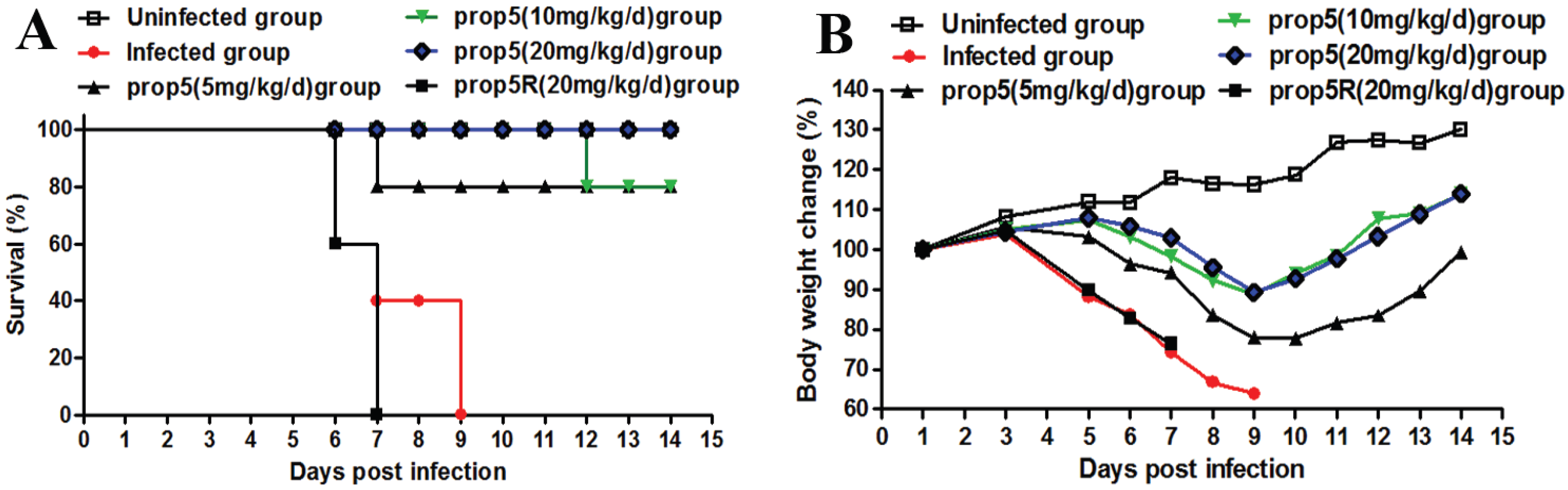
| Pretreatment | No. of survives/total no. | MSD, (days) | Lung index (%) | Virus titre (Log10 TCID50/g lung ± SD) | ||||
|---|---|---|---|---|---|---|---|---|
| Day 2 | Day 4 | Day 6 | Day 2 | Day 4 | Day 6 | |||
| Prop5 (5 mg/kg/d) | 8/10 | 12.6 ± 3.13 * | 0.79 ± 0.06 | 1.22 ± 0.64 | 1.15 ± 0.21 | 3.31 ± 0.09 | 3.03 ± 0.17 ** | 2.91 ± 0.52 ** |
| Prop5 (10 mg/kg/d) | 8/10 | 13.6± 0.89 ** | 0.83 ± 0.03 | 0.87 ± 0.17 | 0.98 ± 0.03 | 2.26 ± 0.02 * | 2.81 ± 0.31 ** | 2.72 ± 0.44 ** |
| Prop5 (20 mg/kg/d) | 10/10 | 14.0 ** | 0.92 ± 0.07 | 0.95 ± 0.14 | 0.81 ± 0.07 * | 1.41 ± 0.35 ** | 2.66 ± 0.27 ** | 2.40 ± 0.13 ** |
| Prop5R (20 mg/kg/d) | 0/10 | 6.6 ± 0.54 | 0.75 ± 0.05 | 1.17 ± 0.68 | 1.31 ± 0.43 | 3.48 ± 0.10 | 4.27 ± 1.19 | 4.92 ± 0.99 |
| Infected control | 0/10 | 7.4 ± 1.51 | 0.75 ± 0.08 | 1.38 ± 0.98 | 1.27 ± 0.24 | 4.12 ± 0.07 | 5.45 ± 0.50 | 5.74 ± 1.14 |
| Uninfected control | 10/10 | 14.0 | 0.67 ± 0.05 | 0.82 ± 0.06 | 0.80 ± 0.09 | NA | NA | NA |
2.2. Prop5 Decreased the Lung Infection Parameters
2.3. Reduction of PDCD5 Protein Expression Levels by prop5 in BALB/c Mice
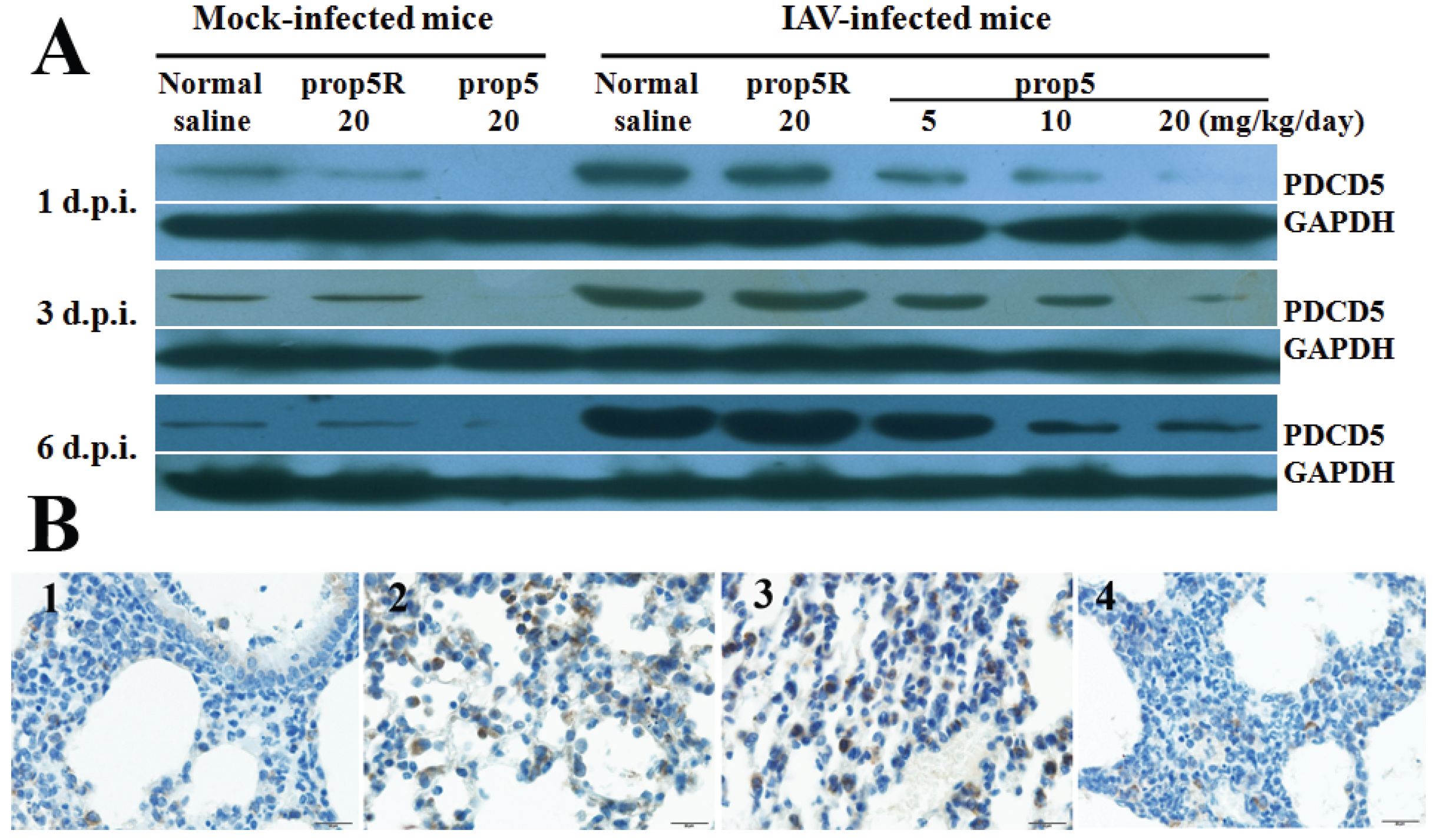
3. Experimental Section
3.1. Virus, Cell, Mice, and ASODNs
3.2. Detecting Expression of PDCD5 Protein by Western Blotting and Immunohistochemical Staining
3.4. Lung Infection Parameters
3.5. Statistics Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Smith, G.J.D.; Vijaykrishna, D.; Bahl, J.; Lycett, S.J.; Worobey, M.; Pybus, O.G.; Ma, S.K.; Cheung, C.L.; Raghwani, J.; Bhatt, S.; et al. Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature 2009, 459, 1122–1125. [Google Scholar] [CrossRef]
- Yang, F.; Wang, J.; Jiang, L.; Jin, J.; Shao, L.; Zhang, Y.; Zhang, J.; Weng, X.; Chen, S.; Zhang, W. A fatal case caused by novel H7N9 avian influenza A virus in China. Emerg. Microbes. Infect. 2013, 2, e19. [Google Scholar] [CrossRef]
- Haaheim, L.; Madhun, A.; Cox, R. Pandemic influenza vaccines–The challenges. Viruses 2009, 1, 1089–1109. [Google Scholar] [CrossRef]
- Rowe, T.; Banner, D.; Farooqui, A.; Ng, D.C.K.; Kelvin, A.A.; Rubino, S.; Huang, S.S.H.; Fang, Y.; Kelvin, D.J. In vivo ribavirin activity against severe pandemic H1N1 influenza A/Mexico/4108/2009. J. Gen. Virol. 2010, 91, 2898–2906. [Google Scholar] [CrossRef]
- Ujike, M.; Shimabukuro, K.; Mochizuki, K.; Obuchi, M.; Kageyama, T.; Shirakura, M. Oseltamivir-resistant influenza A (H1N1) viruses during 2007–2009 influenza seasons, Japan. Emerg. Infect. Dis. 2010, 16. [Google Scholar] [CrossRef]
- Dharan, N.J.; Gubareva, L.V.; Meyer, J.J.; Okomo-Adhiambo, M.; McClinton, R.C.; Marshall, S.A.; St George, K.; Epperson, S.; Brammer, L.; Klimov, A.; et al. Infections with oseltamivir-resistant influenza A (H1N1) virus in the United States. JAMA 2009, 301, 1034–1041. [Google Scholar] [CrossRef]
- Hurt, A.C.; Holien, J.K.; Parker, M.; Kelso, A.; Barr, I.G. Zanamivir-resistant influenza viruses with a novel neuraminidase mutation. J. Virol. 2009, 83, 10366–10373. [Google Scholar]
- Konig, R.; Stertz, S.; Zhou, Y.; Inoue, A.; Hoffmann, H.H.; Bhattacharyya, S.; Alamares, J.G.; Tscherne, D.M.; Ortigoza, M.B.; Liang, Y.; et al. Human host factors required for influenza virus replication. Nature 2010, 463, 813–817. [Google Scholar] [CrossRef]
- Karlas, A.; Machuy, N.; Shin, Y.; Pleissner, K.P.; Artarini, A.; Heuer, D.; Becker, D.; Khalil, H.; Ogilvie, L.A.; Hess, S.; et al. Genome-wide RNAi screen identifies human host factors crucial for influenza virus replication. Nature 2010, 463, 818–822. [Google Scholar] [CrossRef]
- Liu, H.; Wang, Y.; Zhang, Y.; Song, Q.; Di, C.; Chen, G.; Tang, J.; Ma, D. TFAR19, a novel apoptosis-related gene cloned from human leukemia cell line TF-1, could enhance apoptosis of some tumor cells induced by growth factor withdrawal. Biochem. Biophys. Res. Commun. 1999, 254, 203–210. [Google Scholar] [CrossRef]
- Zhao, H.; Yang, J.; Li, K.; Ding, X.; Lin, R.; Ma, Y.; Lu, J.; Zhong, Z.; Qian, X.; Bo, X.; et al. Proteomic analysis at the subcellular level for host targets against influenza A virus (H1N1). Antivir. Res. 2013. [Google Scholar] [CrossRef]
- Everett, H.; McFadden, G. Apoptosis: An innate immune response to virus infection. Trends Microbiol. 1999, 7, 160–165. [Google Scholar] [CrossRef]
- Takizawa, T.; Matsukawa, S.; Higuchi, Y.; Nakamura, S.; Nakanishi, Y.; Fukuda, R. Induction of programmed cell death (apoptosis) by influenza virus infection in tissue culture cells. J. Gen. Virol. 1993, 74, 2347–2355. [Google Scholar] [CrossRef]
- Mori, I.; Komatsu, T.; Takeuchi, K.; Nakakuki, K.; Sudo, M.; Kimura, Y. In vivo induction of apoptosis by influenza virus. J. Gen. Virol. 1995, 76, 2869–2873. [Google Scholar] [CrossRef]
- Price, G.E.; Smith, H.; Sweet, C. Differential induction of cytotoxicity and apoptosis by influenza virus strains of differing virulence. J. Gen. Virol. 1997, 78, 2821–2829. [Google Scholar]
- Ludwig, S.; Pleschka, S.; Planz, O.; Wolff, T. Ringing the alarm bells: Signalling and apoptosis in influenza virus infected cells. Cell Microbiol. 2006, 8, 375–386. [Google Scholar] [CrossRef]
- Wurzer, W.J.; Planz, O.; Ehrhardt, C.; Giner, M.; Silberzahn, T.; Pleschka, S.; Ludwig, S. Caspase 3 activation is essential for efficient influenza virus propagation. EMBO. J. 2003, 22, 2717–2728. [Google Scholar] [CrossRef]
- Tripathi, S.; Batra, J.; Cao, W.; Sharma, K.; Patel, J.R.; Ranjan, P.; Kumar, A.; Katz, J.M.; Cox, N.J.; Lal, R.B.; et al. Influenza A virus nucleoprotein induces apoptosis in human airway epithelial cells: Implications of a novel interaction between nucleoprotein and host protein Clusterin. Cell Death. Dis. 2013, 4, e562. [Google Scholar] [CrossRef]
- Brydon, E.W.; Morris, S.J.; Sweet, C. Role of apoptosis and cytokines in influenza virus morbidity. FEMS Microbiol. Rev. 2005, 29, 837–850. [Google Scholar] [CrossRef]
- Herold, S.; Ludwig, S.; Pleschka, S.; Wolff, T. Apoptosis signaling in influenza virus propagation, innate host defense, and lung injury. J. Leukoc. Biol. 2012, 92, 75–82. [Google Scholar] [CrossRef]
- Wang, X.; Tan, J.; Zoueva, O.; Zhao, J.; Ye, Z.; Hewlett, I. Novel Pandemic Influenza A (H1N1) Virus infection modulates apoptotic pathways that impact its replication in A549 cells. Microbes Infect. 2013, 18, 00224-4. [Google Scholar] [CrossRef]
- Chen, L.N.; Wang, Y.; Ma, D.L.; Chen, Y.Y. Short interfering RNA against the PDCD5 attenuates cell apoptosis and caspase-3 activity induced by Bax overexpression. Apoptosis 2006, 11, 101–111. [Google Scholar] [CrossRef]
- Morris, S.J.; Price, G.E.; Barnett, J.M.; Hiscox, S.A.; Smith, H.; Sweet, C. Role of neuraminidase in influenza virus-induced apoptosis. J. Gen. Virol. 1999, 80, 137–146. [Google Scholar]
- Daidoji, T.; Koma, T.; Du, A.; Yang, C.-S.; Ueda, M.; Ikuta, K.; Nakaya, T. H5N1 avian influenza virus induces apoptotic cell death in mammalian airway epithelial cells. J. Virol. 2008, 82, 11294–11307. [Google Scholar] [CrossRef]
- Schultz-Cherry, S.; Dybdahl-Sissoko, N.; Neumann, G.; Kawaoka, Y.; Hinshaw, V.S. Influenza virus NS1 protein induces apoptosis in cultured cells. J. Virol. 2001, 75, 7875–7881. [Google Scholar] [CrossRef]
- Chanturiya, A.N.; Basañez, G.; Schubert, U.; Henklein, P.; Yewdell, J.W.; Zimmerberg, J. PB1-F2, an Influenza A virus-encoded proapoptotic mitochondrial protein, creates variably sized pores in planar lipid membranes. J. Virol. 2004, 78, 6304–6312. [Google Scholar] [CrossRef]
- Pietrantoni, A.; Dofrelli, E.; Tinari, A.; Ammendolia, M.G.; Puzelli, S.; Fabiani, C.; Donatelli, I.; Superti, F. Bovine lactoferrin inhibits influenza A virus induced programmed cell death in vitro. Biometals 2010, 23, 465–475. [Google Scholar] [CrossRef]
- Srivastava, V.; Rawall, S.; Vijayan, V.K.; Khanna, M. Influenza a virus induced apoptosis: Inhibition of DNA laddering & caspase-3 activity by zinc supplementation in cultured HeLa cells. Indian. J. Med. Res. 2009, 129, 579–586. [Google Scholar]
- Tran, A.T.; Cortens, J.P.; Du, Q.; Wilkins, J.A.; Coombs, K.M. Influenza virus induces apoptosis via BAD-mediated mitochondrial dysregulation. J. Virol. 2013, 87, 1049–1060. [Google Scholar] [CrossRef]
- Ding, X.; Yang, J.; Wang, S. Antisense oligonucleotides targeting abhydrolase domain containing 2 block human hepatitis B virus propagation. Oligonucleotides 2011, 21, 77–84. [Google Scholar] [CrossRef]
- Duan, M.; Zhou, Z.; Lin, R.; Yang, J.; Xia, X.; Wang, S.Q. In vitro and in vivo protection against the highly pathogenic H5N1 influenza virus by an antisense phosphorothioate oligonucleotide. Antivir. Ther. 2008, 13, 109–114. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, K.; Zhou, Z.; Wang, Y.O.; Liu, J.; Zhao, H.B.; Yang, J.; Wang, S.Q. Pretreatment of Mice with Oligonucleotide prop5 Protects Them from Influenza Virus Infections. Viruses 2014, 6, 573-581. https://doi.org/10.3390/v6020573
Li K, Zhou Z, Wang YO, Liu J, Zhao HB, Yang J, Wang SQ. Pretreatment of Mice with Oligonucleotide prop5 Protects Them from Influenza Virus Infections. Viruses. 2014; 6(2):573-581. https://doi.org/10.3390/v6020573
Chicago/Turabian StyleLi, Kang, Zhe Zhou, Yu Ou Wang, Juan Liu, Hai Bao Zhao, Jing Yang, and Sheng Qi Wang. 2014. "Pretreatment of Mice with Oligonucleotide prop5 Protects Them from Influenza Virus Infections" Viruses 6, no. 2: 573-581. https://doi.org/10.3390/v6020573




