Nucleocytoplasmic Shuttling of Viral Proteins in Borna Disease Virus Infection
Abstract
:1. Introduction
2. Nucleocytoplasmic Shuttling of BDV Proteins
2.1. N Protein
2.2. P Protein

2.3. X Protein
2.4. L Protein
3. Implication of Nucleocytoplasmic Shuttling for the BDV Replication Cycle
3.1. Nuclear Import of Incoming Viral RNPs
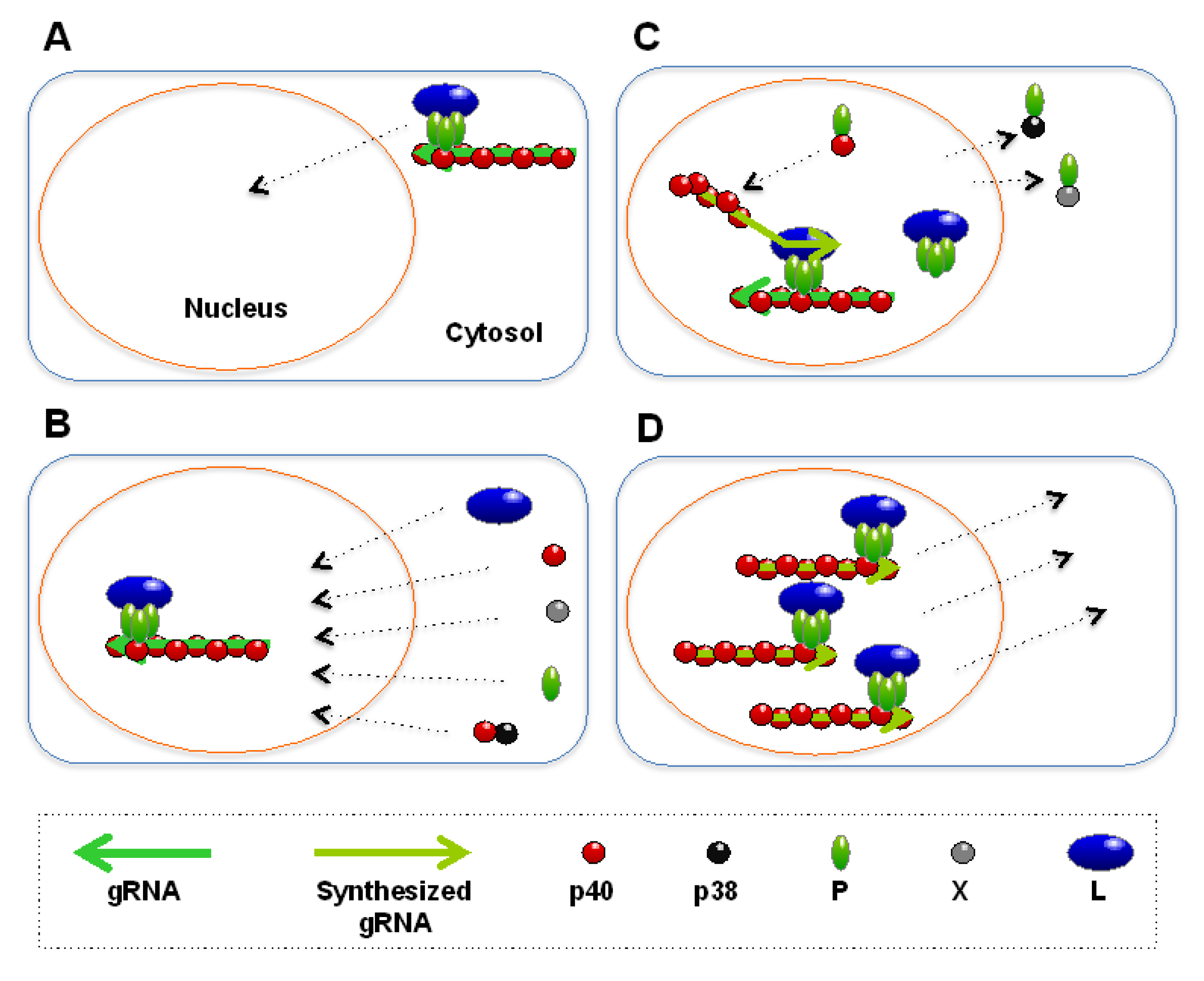
3.2. Replication and Transcription of RNPs in the Nucleus
3.3. Egress of Nascent RNPs from the Nucleus
3.3.1. Regulation by p38
3.3.2. Regulation by P
3.3.3. Regulation by M
4. Perspective
Acknowledgments
Conflict of Interest
References and Notes
- Pletnikov, M.V.; Rubin, S.A.; Vasudevan, K.; Moran, T.H.; Carbone, K.M. Developmental brain injury associated with abnormal play behavior in neonatally Borna disease virus-infected Lewis rats: A model of autism. Behav. Brain Res. 1999, 100, 43–50. [Google Scholar] [CrossRef]
- Hornig, M.; Solbrig, M.; Horscroft, N.; Weissenböck, H.; Lipkin, W.I. Borna disease virus infection of adult and neonatal rats: Models for neuropsychiatric disease. Curr. Top. Microbiol. Immunol. 2001, 253, 157–177. [Google Scholar] [CrossRef]
- Schwemmle, M. Interactions of the borna disease virus P, N, and X proteins and their functional implications. J. Biol. Chem. 1998, 273, 9007–9012. [Google Scholar] [CrossRef]
- Rudolph, M.G.; Kraus, I.; Dickmanns, A.; Eickmann, M.; Garten, W.; Ficner, R. Crystal structure of the borna disease virus nucleoprotein. Structure 2003, 11, 1219–1226. [Google Scholar] [CrossRef]
- Schneider, U.; Naegele, M.; Staeheli, P.; Schwemmle, M. Active borna disease virus polymerase complex requires a distinct nucleoprotein-to-phosphoprotein ratio but no viral X protein. J. Virol. 2003, 77, 11781–11789. [Google Scholar] [CrossRef]
- Perez, M.; Sanchez, A.; Cubitt, B.; Rosario, D.; de la Torre, J.C. A reverse genetics system for Borna disease virus. J. Gen. Virol. 2003, 84, 3099–3104. [Google Scholar] [CrossRef]
- Kraus, I.; Eickmann, M.; Kiermayer, S.; Scheffczik, H.; Fluess, M.; Richt, J.A.; Garten, W. Open reading frame III of borna disease virus encodes a nonglycosylated matrix protein. J. Virol. 2001, 75, 12098–12104. [Google Scholar] [CrossRef]
- Bajramovic, J.J.; Münter, S.; Syan, S.; Nehrbass, U.; Brahic, M.; Gonzalez-Dunia, D. Borna disease virus glycoprotein is required for viral dissemination in neurons. J. Virol. 2003, 77, 12222–12231. [Google Scholar] [CrossRef]
- Honda, T.; Horie, M.; Daito, T.; Ikuta, K.; Tomonaga, K. Molecular chaperone BiP interacts with Borna disease virus glycoprotein at the cell surface. J. Virol. 2009, 83, 12622–12625. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Hayashi, Y.; Omori, H.; Honda, T.; Daito, T.; Horie, M.; Ikuta, K.; Fujino, K.; Nakamura, S.; Schneider, U.; Chase, G.; Yoshimori, T.; Schwemmle, M.; Tomonaga, K. Bornavirus closely associates and segregates with host chromosomes to ensure persistent intranuclear infection. Cell Host Microbe 2012, 11, 492–503. [Google Scholar] [CrossRef] [Green Version]
- Fulcher, A.J.; Jans, D.A. Regulation of nucleocytoplasmic trafficking of viral proteins: an integral role in pathogenesis? Biochim. Biophys. Acta 2011, 1813, 2176–2190. [Google Scholar]
- Nakielny, S.; Dreyfuss, G. Transport of proteins and RNAs in and out of the nucleus. Cell 1999, 99, 677–690. [Google Scholar] [CrossRef]
- Whittaker, G.R.; Helenius, A. Nuclear import and export of viruses and virus genomes. Virology 1998, 246, 1–23. [Google Scholar] [CrossRef]
- Hutchinson, E.C.; Fodor, E. Nuclear import of the influenza A virus transcriptional machinery. Vaccine 2012, 30, 7353–7358. [Google Scholar] [CrossRef]
- Tomonaga, K.; Kobayashi, T.; Ikuta, K. Molecular and cellular biology of Borna disease virus infection. Microb. Infect. 2002, 4, 491–500. [Google Scholar] [CrossRef]
- Yanai, H.; Kobayashi, T.; Hayashi, Y.; Ohtaki, N.; Zhang, G.; De, J.C.; Ikuta, K.; Tomonaga, K.; Watanabe, Y.; Carlos, J.; Torre, D. A methionine-rich domain mediates CRM1-dependent nuclear export activity of borna disease virus phosphoprotein a methionine-rich domain mediates CRM1-dependent nuclear export activity of borna disease virus phosphoprotein. J. Virol. 2006, 80, 1121–1129. [Google Scholar] [CrossRef]
- Kobayashi, T.; Zhang, G.; Lee, B.-J.; Baba, S.; Yamashita, M.; Kamitani, W.; Yanai, H.; Tomonaga, K.; Ikuta, K. Modulation of borna disease virus phosphoprotein nuclear localization by the viral protein X encoded in the overlapping open reading frame. J. Virol. 2003, 77, 8099–8107. [Google Scholar] [CrossRef]
- Pyper, J.M.; Gartner, A.E. Molecular basis for the differential subcellular localization of the 38- and 39-kilodalton structural proteins of Borna disease virus. J. Virol. 1997, 71, 5133–5139. [Google Scholar]
- Kobayashi, T.; Shoya, Y.; Koda, T.; Takashima, I.; Lai, P.K.; Ikuta, K.; Kakinuma, M.; Kishi, M. Nuclear targeting activity associated with the amino terminal region of the Borna disease virus nucleoprotein. Virology 1998, 243, 188–197. [Google Scholar] [CrossRef]
- Kobayashi, T.; Kamitani, W.; Zhang, G.; Watanabe, M.; Tomonaga, K.; Ikuta, K. Borna disease virus nucleoprotein requires both nuclear localization and export activities for viral nucleocytoplasmic shuttling. J. Virol. 2001, 75, 3404–3412. [Google Scholar] [CrossRef]
- Berg, M.; Ehrenborg, C.; Blomberg, J.; Pipkorn, R.; Berg, A.L. Two domains of the Borna disease virus p40 protein are required for interaction with the p23 protein. J. Gen. Virol. 1998, 79, 2957–2963. [Google Scholar]
- Schwemmle, M. Borna disease virus P-protein is phosphorylated by protein kinase cepsilon and casein kinase II. J. Biol. Chem. 1997, 272, 21818–21823. [Google Scholar] [CrossRef]
- Schmid, S.; Mayer, D.; Schneider, U.; Schwemmle, M. Functional characterization of the major and minor phosphorylation sites of the P protein of Borna disease virus. J. Virol. 2007, 81, 5497–5507. [Google Scholar] [CrossRef]
- Kamitani, W.; Ono, E.; Yoshino, S.; Kobayashi, T.; Taharaguchi, S.; Lee, B.-J.; Yamashita, M.; Kobayashi, T.; Okamoto, M.; Taniyama, H.; Tomonaga, K.; Ikuta, K. Glial expression of Borna disease virus phosphoprotein induces behavioral and neurological abnormalities in transgenic mice. Proc. Natl. Acad. Sci. USA 2003, 100, 8969–8974. [Google Scholar] [CrossRef]
- Honda, T.; Fujino, K.; Okuzaki, D.; Ohtaki, N.; Matsumoto, Y.; Horie, M.; Daito, T.; Itoh, M.; Tomonaga, K. Upregulation of insulin-like growth factor binding protein 3 in astrocytes of transgenic mice that express Borna disease virus phosphoprotein. J. Virol. 2011, 85, 4567–4571. [Google Scholar] [CrossRef]
- Shoya, Y.; Kobayashi, T.; Koda, T.; Ikuta, K.; Kakinuma, M.; Kishi, M. Two proline-rich nuclear localization signals in the amino- and Carboxyl-terminal regions of the borna disease virus phosphoprotein. J. Virol. 1998, 72, 9755–9762. [Google Scholar]
- Schwemmle, M.; Jehle, C.; Shoemaker, T.; Lipkin, W.I. Characterization of the major nuclear localization signal of the Borna disease virus phosphoprotein. J. Gen. Virol. 1999, 80, 97–100. [Google Scholar]
- Schneider, U.; Blechschmidt, K.; Schwemmle, M.; Staeheli, P. Overlap of interaction domains indicates a central role of the P protein in assembly and regulation of the Borna disease virus polymerase complex. J. Biol. Chem. 2004, 279, 55290–55296. [Google Scholar]
- Poenisch, M.; Unterstab, G.; Wolff, T.; Staeheli, P.; Schneider, U. The X protein of Borna disease virus regulates viral polymerase activity through interaction with the P protein. J. Gen. Virol. 2004, 85, 1895–1898. [Google Scholar] [CrossRef]
- Poenisch, M.; Wille, S.; Ackermann, A.; Staeheli, P.; Schneider, U. The X protein of borna disease virus serves essential functions in the viral multiplication cycle. J. Virol. 2007, 81, 7297–7299. [Google Scholar] [CrossRef]
- Hayashi, Y.; Horie, M.; Daito, T.; Honda, T.; Ikuta, K.; Tomonaga, K. Heat shock cognate protein 70 controls Borna disease virus replication via interaction with the viral non-structural protein X. Microb. Infect. 2009, 11, 394–402. [Google Scholar] [CrossRef]
- Wolff, T.; Unterstab, G.; Heins, G.; Richt, J.A.; Kann, M. Characterization of an unusual importin alpha binding motif in the borna disease virus p10 protein that directs nuclear import. J. Biol. Chem. 2002, 277, 12151–12157. [Google Scholar]
- Wolff, T.; Pfleger, R.; Wehner, T.; Reinhardt, J.; Richt, J.A. A short leucine-rich sequence in the Borna disease virus p10 protein mediates association with the viral phospho- and nucleoproteins. J. Gen. Virol. 2000, 81, 939–947. [Google Scholar]
- Malik, T.H.; Kishi, M.; Lai, P.K. Characterization of the P protein-binding domain on the 10-kilodalton protein of Borna disease virus. J. Virol. 2000, 74, 3413–3417. [Google Scholar] [CrossRef]
- Watanabe, Y.; Ohtaki, N.; Hayashi, Y.; Ikuta, K.; Tomonaga, K. Autogenous translational regulation of the Borna disease virus negative control factor X from polycistronic mRNA using host RNA helicases. PLoS Pathog. 2009, 5, e1000654. [Google Scholar] [CrossRef]
- Briese, T.; Schneemann, A.; Lewis, A.J.; Park, Y.S.; Kim, S.; Ludwig, H.; Lipkin, W.I. Genomic organization of Borna disease virus. Proc. Natl. Acad. Sci. USA 1994, 91, 4362–4366. [Google Scholar] [CrossRef]
- Walker, M.P.; Jordan, I.; Briese, T.; Fischer, N.; Lipkin, W.I. Expression and characterization of the Borna disease virus polymerase. J. Virol. 2000, 74, 4425–4428. [Google Scholar] [CrossRef]
- Walker, M.P.; Lipkin, W.I. Characterization of the nuclear localization signal of the borna disease virus polymerase. J. Virol. 2002, 76, 8460–8467. [Google Scholar] [CrossRef]
- Gonzalez-Dunia, D.; Cubitt, B.; de la Torre, J.C. Mechanism of Borna disease virus entry into cells. J. Virol. 1998, 72, 783–788. [Google Scholar]
- Vreede, F.T.; Jung, T.E.; Brownlee, G.G. Model suggesting that replication of influenza virus is regulated by stabilization of replicative intermediates. J. Virol. 2004, 78, 9568–9572. [Google Scholar] [CrossRef]
- Resa-Infante, P.; Jorba, N.; Zamarreño, N.; Fernández, Y.; Juárez, S.; Ortín, J. The host-dependent interaction of alpha-importins with influenza PB2 polymerase subunit is required for virus RNA replication. PLoS One 2008, 3, e3904. [Google Scholar] [CrossRef]
- Hutchinson, E.C.; Orr, O.E.; Man Liu, S.; Engelhardt, O.G.; Fodor, E. Characterization of the interaction between the influenza A virus polymerase subunit PB1 and the host nuclear import factor Ran-binding protein 5. J. Gen. Virol. 2011, 92, 1859–1869. [Google Scholar] [CrossRef]
- Schneider, U. Novel insights into the regulation of the viral polymerase complex of neurotropic Borna disease virus. Virus Res. 2005, 111, 148–160. [Google Scholar] [CrossRef]
- Curran, J. A role for the Sendai virus P protein trimer in RNA synthesis. J. Virol. 1998, 72, 4274–4280. [Google Scholar]
- Watanabe, M.; Zhong, Q.; Kobayashi, T.; Kamitani, W.; Tomonaga, K.; Ikuta, K. Molecular ratio between borna disease viral-p40 and -p24 proteins in infected cells determined by quantitative antigen capture ELISA. Microbiol. Immunol. 2000, 44, 765–772. [Google Scholar]
- Schneider, U.; Naegele, M.; Staeheli, P. Regulation of the Borna disease virus polymerase complex by the viral nucleoprotein p38 isoform. Brief Report. Arch. Virol. 2004, 149, 1409–1414. [Google Scholar]
- Bui, M.; Wills, E.G.; Helenius, A.; Whittaker, G.R. Role of the influenza virus M1 protein in nuclear export of viral ribonucleoproteins. J. Virol. 2000, 74, 1781–1786. [Google Scholar] [CrossRef]
- Mayer, D.; Baginsky, S.; Schwemmle, M. Isolation of viral ribonucleoprotein complexes from infected cells by tandem affinity purification. Proteomics 2005, 5, 4483–4487. [Google Scholar] [CrossRef]
- Chase, G.; Mayer, D.; Hildebrand, A.; Frank, R.; Hayashi, Y.; Tomonaga, K.; Schwemmle, M. Borna disease virus matrix protein is an integral component of the viral ribonucleoprotein complex that does not interfere with polymerase activity. J. Virol. 2007, 81, 743–749. [Google Scholar] [CrossRef]
- Neumann, P.; Lieber, D.; Meyer, S.; Dautel, P.; Kerth, A.; Kraus, I.; Garten, W.; Stubbs, M.T. Crystal structure of the Borna disease virus matrix protein (BDV-M) reveals ssRNA binding properties. Proc. Natl. Acad. Sci. USA 2009, 106, 3710–3715. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Honda, T.; Tomonaga, K. Nucleocytoplasmic Shuttling of Viral Proteins in Borna Disease Virus Infection. Viruses 2013, 5, 1978-1990. https://doi.org/10.3390/v5081978
Honda T, Tomonaga K. Nucleocytoplasmic Shuttling of Viral Proteins in Borna Disease Virus Infection. Viruses. 2013; 5(8):1978-1990. https://doi.org/10.3390/v5081978
Chicago/Turabian StyleHonda, Tomoyuki, and Keizo Tomonaga. 2013. "Nucleocytoplasmic Shuttling of Viral Proteins in Borna Disease Virus Infection" Viruses 5, no. 8: 1978-1990. https://doi.org/10.3390/v5081978



