Latest Insights on Adenovirus Structure and Assembly
Abstract
:1. AdV Structure: A Historical Perspective
| Protein | Species 1 | Ligands/Modifications | Reference |
|---|---|---|---|
| Hexon | HAdV-2 | [61] | |
| HAdV-5 | [61] | ||
| FAdV-1 (CELO) | [62] | ||
| SAdV-25 | [63] | ||
| Penton base | HAdV-2 | [42] | |
| HAdV-2 | N-terminal fiber peptide | [42] | |
| HAdV-2 | Chimera with RGD loop from HAdV-12 | [64] | |
| Fiber shaft | HAdV-2 | [27] | |
| HAdV-2 | Fused to bacteriophage T4 fibritin trimerization motif | [65] | |
| Fiber knob | HAdV-2 | [66] | |
| HAdV-3 | [67] | ||
| HAdV-5 | [41] | ||
| HAdV-7 | [68] | ||
| HAdV-11 | [68] | ||
| HAdV-11 | Consensus repeats SCR1-SCR2 of CD46 | [69] | |
| HAdV-11 | Consensus repeats SCR1 to 4 of CD46 | [70] | |
| HAdV-12 | [71] | ||
| HAdV-12 | Domain 1 of CAR | [71] | |
| HAdV-12 | Knob mutants in complex with domain 1 of CAR | [72] | |
| HAdV-14 | [68] | ||
| HAdV-16 | [73] | ||
| HAdV-19 | Sialyl-lactose | [74] | |
| HAdV-21 | [75] | ||
| HAdV-21 | Consensus repeats SCR1-SCR2 of CD46 | [75] | |
| HAdV-35 | [76,77] | ||
| HAdV-37 | [74] | ||
| HAdV-37 | Sialyl-lactose | [74] | |
| HAdV-37 | Domain 1 of CAR | [78] | |
| HAdV-37 | Sialic acid derivatives | [79] | |
| HAdV-37 | GD1a glycan | [80] | |
| HAdV-37 | Trivalent sialic acid inhibitor | [81] | |
| CAdV-2 | [78] | ||
| CAdV-2 | Domain 1 of CAR | [78] | |
| CAdV-2 | Sialic acid | [82] | |
| CAdV-2 | Domain 1 of CAR and sialic acid | [82] | |
| Fiber knob (short fiber) | HAdV-41 | [83] | |
| Fiber knob (head domain) | PAdV-4 | [84] | |
| Fiber knob (galectin domain) | PAdV-4 | [84] | |
| Fiber knob (galectin domain) | PAdV-4 | Carbohydrates | [84] |
| Fiber knob (short fiber) | FAdV-1 (CELO) | [85] | |
| Fiber knob (long fiber) | FAdV-1 (CELO) | [86] |
2. Reaching Atomic Detail in the Complete AdV Virion
2.1. X-Ray Diffraction
2.2. Cryo-EM
3. Atomic Structure of the Mature Adenovirus Virion
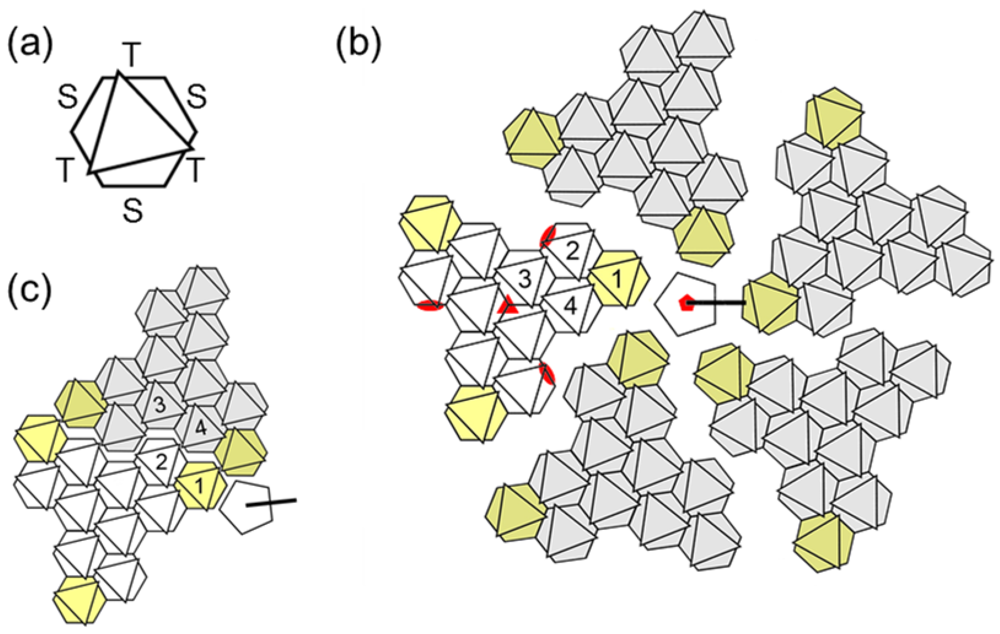
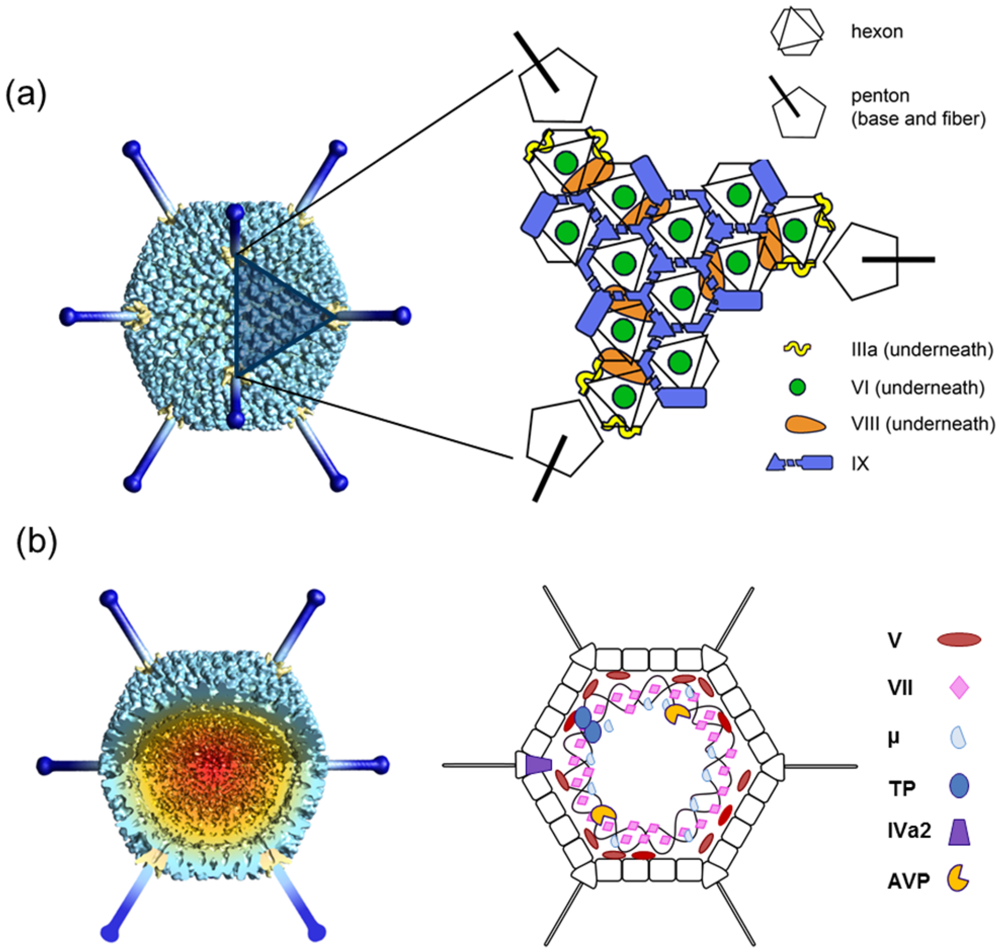
3.1. Hexon
3.2. Penton Base and Fiber
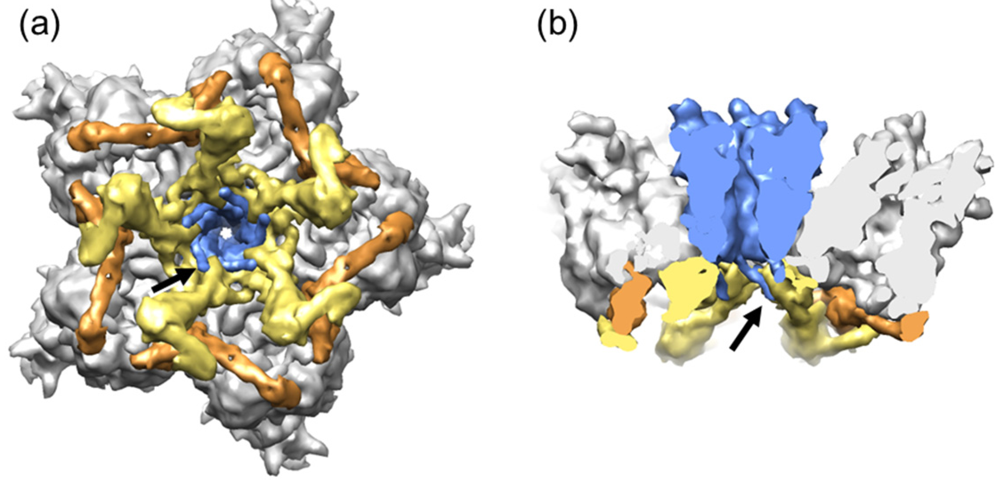
3.3. Polypeptide IIIa
3.4. Polypeptide VI

3.5. Polypeptide VIII

3.6. Polypeptide IX
4. Structural Changes Involved in Maturation
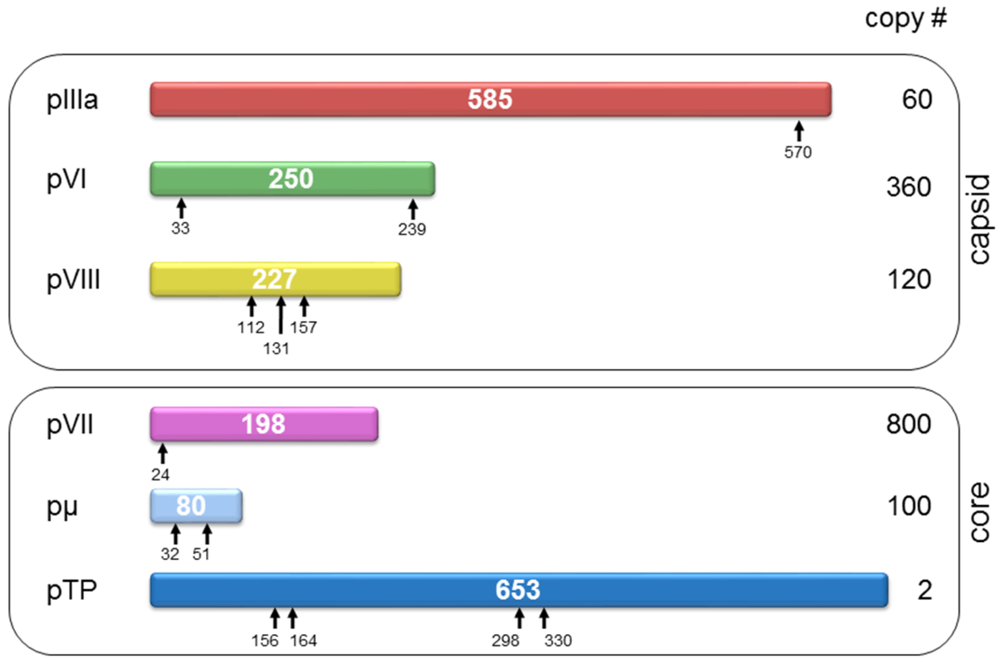

5. Remaining Questions
Acknowledgments
Conflict of Interest
References
- Silvestry, M.; Lindert, S.; Smith, J.G.; Maier, O.; Wiethoff, C.M.; Nemerow, G.R.; Stewart, P.L. Cryo-electron microscopy structure of adenovirus type 2 temperature-sensitive mutant 1 reveals insight into the cell entry defect. J. Virol. 2009, 83, 7375–7383. [Google Scholar]
- Pérez-Berná, A.J.; Marabini, R.; Scheres, S.H.W.; Menéndez-Conejero, R.; Dmitriev, I.P.; Curiel, D.T.; Mangel, W.F.; Flint, S.J.; San Martín, C. Structure and uncoating of immature adenovirus. J. Mol. Biol. 2009, 392, 547–557. [Google Scholar] [CrossRef]
- Reddy, V.S.; Natchiar, S.K.; Stewart, P.L.; Nemerow, G.R. Crystal structure of human adenovirus at 3.5 A resolution. Science 2010, 329, 1071–1075. [Google Scholar] [CrossRef]
- Liu, H.; Jin, L.; Koh, S.B.; Atanasov, I.; Schein, S.; Wu, L.; Zhou, Z.H. Atomic structure of human adenovirus by cryo-EM reveals interactions among protein networks. Science 2010, 329, 1038–1043. [Google Scholar] [CrossRef]
- Berk, A.J. Adenoviridae: The Viruses and Their Replication. In Fields Virology, 5th; Knipe, D.M., Howley, P.M., Griffin, D.E., Lamb, R.A., Martin, M.A., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 2355–2394. [Google Scholar]
- Harrach, B.; Benkő, M.; Both, G.; Brown, M.; Davison, A.; Echavarría, M.; Hess, M.; Jones, M.; Kajon, A.; Lehmkuhl, H.; et al. Family Adenoviridae. In Virus Taxonomy: Classification and Nomenclature of Viruses. Ninth Report of the International Committee on Taxonomy of Viruses; King, A., Adams, M., Carstens, E., Lefkowitz, E., Eds.; Elsevier: San Diego, CA, USA, 2011; pp. 95–111. [Google Scholar]
- Leen, A.M.; Rooney, C.M. Adenovirus as an emerging pathogen in immunocompromised patients. Br. J. Haematol. 2005, 128, 135–144. [Google Scholar] [CrossRef]
- Kinchington, P.R.; Romanowski, E.G.; Jerold Gordon, Y. Prospects for adenovirus antivirals. J. Antimicrob. Chemother. 2005, 55, 424–429. [Google Scholar] [CrossRef]
- Shiver, J.W.; Fu, T.-M.; Chen, L.; Casimiro, D.R.; Davies, M.-E.; Evans, R.K.; Zhang, Z.-Q.; Simon, A.J.; Trigona, W.L.; Dubey, S.A.; et al. Replication-incompetent adenoviral vaccine vector elicits effective anti-immunodeficiency-virus immunity. Nature 2002, 415, 331–335. [Google Scholar]
- Dobbelstein, M. Replicating adenoviruses in cancer therapy. Curr. Top. Microbiol. Immunol. 2004, 273, 291–334. [Google Scholar]
- Gonçalves, M.A.F.V.; de Vries, A.A.F. Adenovirus: From foe to friend. Rev. Med. Virol. 2006, 16, 167–186. [Google Scholar] [CrossRef]
- Stadtfeld, M.; Nagaya, M.; Utikal, J.; Weir, G.; Hochedlinger, K. Induced pluripotent stem cells generated without viral integration. Science 2008, 322, 945–949. [Google Scholar]
- Lasaro, M.O.; Ertl, H.C. New insights on adenovirus as vaccine vectors. Mol. Ther. 2009, 17, 1333–1339. [Google Scholar] [CrossRef]
- Draper, S.J.; Heeney, J.L. Viruses as vaccine vectors for infectious diseases and cancer. Nat. Rev. Microbiol. 2010, 8, 62–73. [Google Scholar] [CrossRef]
- Yamamoto, M.; Curiel, D.T. Current issues and future directions of oncolytic adenoviruses. Mol. Ther. 2010, 18, 243–250. [Google Scholar] [CrossRef]
- Alba, R.; Bosch, A.; Chillon, M. Gutless adenovirus: Last-generation adenovirus for gene therapy. Gene Ther. 2005, 12 Suppl 1, S18–S27. [Google Scholar]
- Singh, R.; Kostarelos, K. Designer adenoviruses for nanomedicine and nanodiagnostics. Trends Biotechnol. 2009, 27, 220–229. [Google Scholar] [CrossRef]
- Rowe, W.P.; Huebner, R.J.; Gillmore, L.K.; Parrott, R.H.; Ward, T.G. Isolation of a cytopathogenic agent from human adenoids undergoing spontaneous degeneration in tissue culture. Proc. Soc. Exp. Biol. Med. 1953, 84, 570–573. [Google Scholar]
- Russell, W. Adenoviruses: Update on structure and function. J. Gen. Virol. 2009, 90, 1–20. [Google Scholar] [CrossRef]
- Horne, R.W.; Brenner, S.; Waterson, A.P.; Wildy, P. The icosahedral form of an adenovirus. J. Mol. Biol. 1959, 1, 84–86. [Google Scholar] [CrossRef]
- Crick, F.H.; Watson, J.D. Structure of small viruses. Nature 1956, 177, 473–475. [Google Scholar]
- Caspar, D.L.D.; Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb. Symp. Quant. Biol. 1962, 27, 1–24. [Google Scholar] [CrossRef]
- Valentine, R.C.; Pereira, H.G. Antigens and structure of the adenovirus. J. Mol. Biol. 1965, 13, 13–20. [Google Scholar] [CrossRef]
- Valentine, R.C.; Pereira, H.G. Antigens and structure of the adenovirus. Reprinted from J. Mol. Biol. 1965, 13, 13-20. Rev Med Virol 2003, 13, 71–82, discussion 80-73.. [Google Scholar] [CrossRef]
- Ginsberg, H.S.; Pereira, H.G.; Valentine, R.C.; Wilcox, W.C. A proposed terminology for the adenovirus antigens and virion morphological subunits. Virology 1966, 28, 782–783. [Google Scholar] [CrossRef]
- Maizel, J.V., Jr.; White, D.O.; Scharff, M.D. The polypeptides of adenovirus. I. Evidence for multiple protein components in the virion and a comparison of types 2, 7A, and 12. Virology 1968, 36, 115–125. [Google Scholar] [CrossRef]
- van Raaij, M.J.; Mitraki, A.; Lavigne, G.; Cusack, S. A triple β-spiral in the adenovirus fibre shaft reveals a new structural motif for a fibrous protein. Nature 1999, 401, 935–938. [Google Scholar]
- Christensen, J.B.; Byrd, S.A.; Walker, A.K.; Strahler, J.R.; Andrews, P.C.; Imperiale, M.J. Presence of the adenovirus IVa2 protein at a single vertex of the mature virion. J. Virol. 2008, 82, 9086–9093. [Google Scholar]
- Prage, L.; Pettersson, U.; Höglund, S.; Lonberg-Holm, K.; Philipson, L. Structural proteins of adenoviruses. IV. Sequential degradation of the adenovirus type 2 virion. Virology 1970, 42, 341–358. [Google Scholar] [CrossRef]
- Crowther, R.A.; Franklin, R.M. The structure of the groups of nine hexons from adenovirus. J. Mol. Biol. 1972, 68, 181–184. [Google Scholar] [CrossRef]
- Grütter, M.; Franklin, R.M. Studies on the molecular weight of the adenovirus type 2 hexon and its subunit. J. Mol. Biol. 1974, 89, 163–178. [Google Scholar] [CrossRef]
- Burnett, R.M.; Grutter, M.G.; White, J.L. The structure of the adenovirus capsid. 1. An envelope model of hexon at 6 Å resolution. J. Mol. Biol. 1985, 185, 105–123. [Google Scholar] [CrossRef]
- Burnett, R.M. The structure of the adenovirus capsid. II. The packing symmetry of hexon and its implications for viral architecture. J. Mol. Biol. 1985, 185, 125–143. [Google Scholar] [CrossRef]
- Van Oostrum, J.; Smith, P.R.; Mohraz, M.; Burnett, R.M. The structure of the adenovirus capsid. III. Hexon packing determined from electron micrographs of capsid fragments. J. Mol. Biol. 1987, 198, 73–89. [Google Scholar]
- Roberts, M.M.; White, J.L.; Grutter, M.G.; Burnett, R.M. 3-dimensional structure of the adenovirus major coat protein hexon. Science 1986, 232, 1148–1151. [Google Scholar]
- Everitt, E.; Sundquist, B.; Pettersson, U.; Philipson, L. Structural proteins of adenoviruses. X. Isolation and topography of low molecular weight antigens from the virion of adenovirus type 2. Virology 1973, 52, 130–147. [Google Scholar] [CrossRef]
- Van Oostrum, J.; Burnett, R.M. Molecular composition of the adenovirus type-2 virion. J. Virol. 1985, 56, 439–448. [Google Scholar]
- Furcinitti, P.S.; van Oostrum, J.; Burnett, R.M. Adenovirus polypeptide-IX revealed as capsid cement by difference images from electron-microscopy and crystallography. EMBO J. 1989, 8, 3563–3570. [Google Scholar]
- Stewart, P.L.; Burnett, R.M.; Cyrklaff, M.; Fuller, S.D. Image-reconstruction reveals the complex molecular-organization of adenovirus. Cell 1991, 67, 145–154. [Google Scholar]
- Stewart, P.L.; Fuller, S.D.; Burnett, R.M. Difference imaging of adenovirus—Bridging the resolution gap between x-ray crystallography and electron-microscopy. EMBO J. 1993, 12, 2589–2599. [Google Scholar]
- Xia, D.; Henry, L.J.; Gerard, R.D.; Deisenhofer, J. Crystal structure of the receptor-binding domain of adenovirus type 5 fiber protein at 1.7 Å resolution. Structure 1994, 2, 1259–1270. [Google Scholar] [CrossRef]
- Zubieta, C.; Schoehn, G.; Chroboczek, J.; Cusack, S. The structure of the human adenovirus 2 penton. Mol. Cell 2005, 17, 121–135. [Google Scholar] [CrossRef]
- Saban, S.D.; Nepomuceno, R.R.; Gritton, L.D.; Nemerow, G.R.; Stewart, P.L. CryoEM structure at 9Å resolution of an adenovirus vector targeted to hematopoietic cells. J. Mol. Biol. 2005, 349, 526–537. [Google Scholar] [CrossRef]
- Saban, S.D.; Silvestry, M.; Nemerow, G.R.; Stewart, P.L. Visualization of α-helices in a 6 Å resolution cryoEM structure of adenovirus allows refinement of capsid protein assignments. J. Virol. 2006, 80, 12049–12059. [Google Scholar]
- Fabry, C.M.; Rosa-Calatrava, M.; Conway, J.F.; Zubieta, C.; Cusack, S.; Ruigrok, R.W.; Schoehn, G. A quasi-atomic model of human adenovirus type 5 capsid. EMBO J. 2005, 24, 1645–1654. [Google Scholar]
- Scheres, S.H.W.; Marabini, R.; Lanzavecchia, S.; Cantele, F.; Rutten, T.; Fuller, S.D.; Carazo, J.M.; Burnett, R.M.; San Martín, C. Classification of single-projection reconstructions for cryo-electron microscopy data of icosahedral viruses. J. Struct. Biol. 2005, 151, 79–91. [Google Scholar] [CrossRef]
- Marsh, M.P.; Campos, S.K.; Baker, M.L.; Chen, C.Y.; Chiu, W.; Barry, M.A. CryoEM of protein IX-modified adenoviruses suggests a new position for the C-terminus of protein IX. J. Virol. 2006, 80, 11881–11886. [Google Scholar]
- Fabry, C.M.; Rosa-Calatrava, M.; Moriscot, C.; Ruigrok, R.W.; Boulanger, P.; Schoehn, G. The C-terminal domains of adenovirus serotype 5 protein IX assemble into an antiparallel structure on the facets of the capsid. J. Virol. 2009, 83, 1135–1139. [Google Scholar]
- San Martín, C.; Glasgow, J.N.; Borovjagin, A.V.; Beatty, M.S.; Kashentseva, E.A.; Curiel, D.T.; Marabini, R.; Dmitriev, I.P. Localization of the N-terminus of minor coat protein IIIa in the adenovirus capsid. J. Mol. Biol. 2008, 383, 923–934. [Google Scholar] [CrossRef]
- Pantelic, R.S.; Lockett, L.J.; Rothnagel, R.; Hankamer, B.; Both, G.W. Cryoelectron microscopy map of Atadenovirus reveals cross-genus structural differences from human adenovirus. J. Virol. 2008, 82, 7346–7356. [Google Scholar] [CrossRef]
- Schoehn, G.; El Bakkouri, M.; Fabry, C.M.; Billet, O.; Estrozi, L.F.; Le, L.; Curiel, D.T.; Kajava, A.V.; Ruigrok, R.W.; Kremer, E.J. Three-dimensional structure of canine adenovirus serotype 2 capsid. J. Virol. 2008, 82, 3192–3203. [Google Scholar]
- Lindert, S.; Silvestry, M.; Mullen, T.M.; Nemerow, G.R.; Stewart, P.L. Cryo-electron microscopy structure of an adenovirus-integrin complex indicates conformational changes in both penton base and integrin. J. Virol. 2009, 83, 11491–11501. [Google Scholar] [CrossRef]
- Wu, E.; Pache, L.; von Seggern, D.J.; Mullen, T.M.; Mikyas, Y.; Stewart, P.L.; Nemerow, G.R. Flexibility of the adenovirus fiber is required for efficient receptor interaction. J. Virol. 2003, 77, 7225–7235. [Google Scholar]
- Chiu, C.Y.; Wu, E.; Brown, S.L.; von Seggern, D.J.; Nemerow, G.R.; Stewart, P.L. Structural analysis of a fiber-pseudotyped adenovirus with ocular tropism suggests differential modes of cell receptor interactions. J. Virol. 2001, 75, 5375–5380. [Google Scholar]
- Von Seggern, D.J.; Chiu, C.Y.; Fleck, S.K.; Stewart, P.L.; Nemerow, G.R. A helper-independent adenovirus vector with E1, E3, and fiber deleted: Structure and infectivity of fiberless particles. J. Virol. 1999, 73, 1601–1608. [Google Scholar]
- Chiu, C.Y.; Mathias, P.; Nemerow, G.R.; Stewart, P.L. Structure of adenovirus complexed with its internalization receptor, αvβ5 integrin. J. Virol. 1999, 73, 6759–6768. [Google Scholar]
- Stewart, P.L.; Chiu, C.Y.; Huang, S.; Muir, T.; Zhao, Y.; Chait, B.; Mathias, P.; Nemerow, G.R. Cryo-EM visualization of an exposed RGD epitope on adenovirus that escapes antibody neutralization. EMBO J. 1997, 16, 1189–1198. [Google Scholar]
- Waddington, S.N.; McVey, J.H.; Bhella, D.; Parker, A.L.; Barker, K.; Atoda, H.; Pink, R.; Buckley, S.M.; Greig, J.A.; Denby, L.; et al. Adenovirus serotype 5 hexon mediates liver gene transfer. Cell 2008, 132, 397–409. [Google Scholar] [CrossRef]
- Kalyuzhniy, O.; di Paolo, N.C.; Silvestry, M.; Hofherr, S.E.; Barry, M.A.; Stewart, P.L.; Shayakhmetov, D.M. Adenovirus serotype 5 hexon is critical for virus infection of hepatocytes in vivo. Proc. Natl. Acad. Sci. USA 2008, 105, 5483–5488. [Google Scholar]
- Smith, J.G.; Silvestry, M.; Lindert, S.; Lu, W.; Nemerow, G.R.; Stewart, P.L. Insight into the mechanisms of adenovirus capsid disassembly from studies of defensin neutralization. PLoS Pathog. 2010, 6. [Google Scholar]
- Rux, J.J.; Kuser, P.R.; Burnett, R.M. Structural and phylogenetic analysis of adenovirus hexons by use of high-resolution x-ray crystallographic, molecular modeling, and sequence-based methods. J. Virol. 2003, 77, 9553–9566. [Google Scholar] [CrossRef]
- Xu, L.; Benson, S.D.; Burnett, R.M. Nanoporous crystals of chicken embryo lethal orphan (CELO) adenovirus major coat protein, hexon. J. Struct. Biol. 2007, 157, 424–431. [Google Scholar] [CrossRef]
- Pichla-Gollon, S.L.; Drinker, M.; Zhou, X.; Xue, F.; Rux, J.J.; Gao, G.P.; Wilson, J.M.; Ertl, H.C.; Burnett, R.M.; Bergelson, J.M. Structure-based identification of a major neutralizing site in an adenovirus hexon. J. Virol. 2007, 81, 1680–1689. [Google Scholar]
- Zubieta, C.; Blanchoin, L.; Cusack, S. Structural and biochemical characterization of a human adenovirus 2/12 penton base chimera. FEBS J. 2006, 273, 4336–4345. [Google Scholar] [CrossRef]
- Papanikolopoulou, K.; Teixeira, S.; Belrhali, H.; Forsyth, V.T.; Mitraki, A.; van Raaij, M.J. Adenovirus fibre shaft sequences fold into the native triple beta-spiral fold when N-terminally fused to the bacteriophage T4 fibritin foldon trimerisation motif. J. Mol. Biol. 2004, 342, 219–227. [Google Scholar] [CrossRef]
- Van Raaij, M.J.; Louis, N.; Chroboczek, J.; Cusack, S. Structure of the human adenovirus serotype 2 fiber head domain at 1.5 Å resolution. Virology 1999, 262, 333–343. [Google Scholar] [CrossRef]
- Durmort, C.; Stehlin, C.; Schoehn, G.; Mitraki, A.; Drouet, E.; Cusack, S.; Burmeister, W.P. Structure of the fiber head of Ad3, a non-CAR-binding serotype of adenovirus. Virology 2001, 285, 302–312. [Google Scholar] [CrossRef]
- Persson, B.D.; Muller, S.; Reiter, D.M.; Schmitt, B.B.; Marttila, M.; Sumowski, C.V.; Schweizer, S.; Scheu, U.; Ochsenfeld, C.; Arnberg, N.; et al. An arginine switch in the species B adenovirus knob determines high-affinity engagement of cellular receptor CD46. J. Virol. 2009, 83, 673–686. [Google Scholar]
- Persson, B.D.; Reiter, D.M.; Marttila, M.; Mei, Y.F.; Casasnovas, J.M.; Arnberg, N.; Stehle, T. Adenovirus type 11 binding alters the conformation of its receptor CD46. Nat. Struct. Mol. Biol. 2007, 14, 164–166. [Google Scholar] [CrossRef]
- Persson, B.D.; Schmitz, N.B.; Santiago, C.; Zocher, G.; Larvie, M.; Scheu, U.; Casasnovas, J.M.; Stehle, T. Structure of the extracellular portion of CD46 provides insights into its interactions with complement proteins and pathogens. PLoS Pathog. 2010, 6. [Google Scholar]
- Bewley, M.C.; Springer, K.; Zhang, Y.-B.; Freimuth, P.; Flanagan, J.M. Structural analysis of the mechanism of adenovirus binding to its human cellular receptor, CAR. Science 1999, 286, 1579–1583. [Google Scholar]
- Howitt, J.; Bewley, M.C.; Graziano, V.; Flanagan, J.M.; Freimuth, P. Structural basis for variation in adenovirus affinity for the cellular coxsackievirus and adenovirus receptor. J. Biol. Chem. 2003, 278, 26208–26215. [Google Scholar]
- Pache, L.; Venkataraman, S.; Reddy, V.S.; Nemerow, G.R. Structural variations in species B adenovirus fibers impact CD46 association. J. Virol. 2008, 82, 7923–7931. [Google Scholar]
- Burmeister, W.P.; Guilligay, D.; Cusack, S.; Wadell, G.; Arnberg, N. Crystal structure of species D adenovirus fiber knobs and their sialic acid binding sites. J. Virol. 2004, 78, 7727–7736. [Google Scholar]
- Cupelli, K.; Muller, S.; Persson, B.D.; Jost, M.; Arnberg, N.; Stehle, T. Structure of adenovirus type 21 knob in complex with CD46 reveals key differences in receptor contacts among species B adenoviruses. J. Virol. 2010, 84, 3189–3200. [Google Scholar]
- Wang, H.; Liaw, Y.C.; Stone, D.; Kalyuzhniy, O.; Amiraslanov, I.; Tuve, S.; Verlinde, C.L.; Shayakhmetov, D.; Stehle, T.; Roffler, S.; Lieber, A. Identification of CD46 binding sites within the adenovirus serotype 35 fiber knob. J. Virol. 2007, 81, 12785–12792. [Google Scholar]
- Pache, L.; Venkataraman, S.; Nemerow, G.R.; Reddy, V.S. Conservation of fiber structure and CD46 usage by subgroup B2 adenoviruses. Virology 2008, 375, 573–579. [Google Scholar] [CrossRef]
- Seiradake, E.; Lortat-Jacob, H.; Billet, O.; Kremer, E.J.; Cusack, S. Structural and mutational analysis of human Ad37 and canine adenovirus 2 fiber heads in complex with the D1 domain of coxsackie and adenovirus receptor. J. Biol. Chem. 2006, 281, 33704–33716. [Google Scholar]
- Johansson, S.; Nilsson, E.; Qian, W.; Guilligay, D.; Crepin, T.; Cusack, S.; Arnberg, N.; Elofsson, M. Design, synthesis, and evaluation of N-acyl modified sialic acids as inhibitors of adenoviruses causing epidemic keratoconjunctivitis. J. Med. Chem. 2009, 52, 3666–3678. [Google Scholar]
- Nilsson, E.C.; Storm, R.J.; Bauer, J.; Johansson, S.M.; Lookene, A.; Angstrom, J.; Hedenstrom, M.; Eriksson, T.L.; Frangsmyr, L.; Rinaldi, S.; et al. The GD1a glycan is a cellular receptor for adenoviruses causing epidemic keratoconjunctivitis. Nat. Med. 2011, 17, 105–109. [Google Scholar]
- Spjut, S.; Qian, W.; Bauer, J.; Storm, R.; Frangsmyr, L.; Stehle, T.; Arnberg, N.; Elofsson, M. A potent trivalent sialic Acid inhibitor of adenovirus type 37 infection of human corneal cells. Angew. Chem. Int. Ed. Engl. 2011, 50, 6519–6521. [Google Scholar]
- Seiradake, E.; Henaff, D.; Wodrich, H.; Billet, O.; Perreau, M.; Hippert, C.; Mennechet, F.; Schoehn, G.; Lortat-Jacob, H.; Dreja, H.; et al. The cell adhesion molecule “CAR” and sialic acid on human erythrocytes influence adenovirus in vivo biodistribution. PLoS Pathog. 2009, 5. [Google Scholar]
- Seiradake, E.; Cusack, S. Crystal structure of enteric adenovirus serotype 41 short fiber head. J. Virol. 2005, 79, 14088–14094. [Google Scholar] [CrossRef]
- Guardado-Calvo, P.; Munoz, E.M.; Llamas-Saiz, A.L.; Fox, G.C.; Kahn, R.; Curiel, D.T.; Glasgow, J.N.; van Raaij, M.J. Crystallographic structure of porcine adenovirus type 4 fiber head and galectin domains. J. Virol. 2010, 84, 10558–10568. [Google Scholar]
- El Bakkouri, M.; Seiradake, E.; Cusack, S.; Ruigrok, R.W.; Schoehn, G. Structure of the C-terminal head domain of the fowl adenovirus type 1 short fibre. Virology 2008, 378, 169–176. [Google Scholar] [CrossRef]
- Guardado-Calvo, P.; Llamas-Saiz, A.L.; Fox, G.C.; Langlois, P.; van Raaij, M.J. Structure of the C-terminal head domain of the fowl adenovirus type 1 long fiber. J. Gen. Virol. 2007, 88, 2407–2416. [Google Scholar] [CrossRef]
- Benson, S.D.; Bamford, J.K.H.; Bamford, D.H.; Burnett, R.M. Viral evolution revealed by bacteriophage PRD1 and human adenovirus coat protein structures. Cell 1999, 98, 825–833. [Google Scholar] [CrossRef]
- Krupovic, M.; Bamford, D.H. Virus evolution: How far does the double beta-barrel viral lineage extend? Nat. Rev. Microbiol. 2008, 6, 941–948. [Google Scholar] [CrossRef]
- Xiao, C.; Kuznetsov, Y.G.; Sun, S.; Hafenstein, S.L.; Kostyuchenko, V.A.; Chipman, P.R.; Suzan-Monti, M.; Raoult, D.; McPherson, A.; Rossmann, M.G. Structural studies of the giant mimivirus. PLoS Biol. 2009, 7. [Google Scholar]
- Bahar, M.W.; Graham, S.C.; Stuart, D.I.; Grimes, J.M. Insights into the evolution of a complex virus from the crystal structure of vaccinia virus D13. Structure 2011, 19, 1011–1020. [Google Scholar]
- Pereira, H.G.; Valentine, R.C.; Russell, W.C. Crystallization of an adenovirus protein (the hexon). Nature 1968, 219, 946–947. [Google Scholar] [CrossRef]
- Van Oostrum, J.; Smith, P.R.; Mohraz, M.; Burnett, R.M. Interpretation of electron-micrographs of adenovirus hexon arrays using a crystallographic molecular-model. J. Ultrastruct. Mol. Struct. Res. 1986, 96, 77–90. [Google Scholar] [CrossRef]
- Grigorieff, N.; Harrison, S.C. Near-atomic resolution reconstructions of icosahedral viruses from electron cryo-microscopy. Curr. Opin. Struct. Biol. 2011, 21, 265–273. [Google Scholar] [CrossRef]
- Reddy, V.S.; Natchiar, S.K.; Gritton, L.; Mullen, T.M.; Stewart, P.L.; Nemerow, G.R. Crystallization and preliminary X-ray diffraction analysis of human adenovirus. Virology 2010, 402, 209–214. [Google Scholar] [CrossRef]
- Cockburn, J.J.; Bamford, J.K.; Grimes, J.M.; Bamford, D.H.; Stuart, D.I. Crystallization of the membrane-containing bacteriophage PRD1 in quartz capillaries by vapour diffusion. Acta Crystallogr. D Biol. Crystallogr. 2003, 59, 538–540. [Google Scholar]
- Abrescia, N.G.; Cockburn, J.J.; Grimes, J.M.; Sutton, G.C.; Diprose, J.M.; Butcher, S.J.; Fuller, S.D.; San Martín, C.; Burnett, R.M.; Stuart, D.I.; et al. Insights into assembly from structural analysis of bacteriophage PRD1. Nature 2004, 432, 68–74. [Google Scholar]
- Bamford, J.K.; Cockburn, J.J.; Diprose, J.; Grimes, J.M.; Sutton, G.; Stuart, D.I.; Bamford, D.H. Diffraction quality crystals of PRD1, a 66-MDa dsDNA virus with an internal membrane. J. Struct. Biol. 2002, 139, 103–112. [Google Scholar] [CrossRef]
- Crowther, R.A. From envelopes to atoms: The remarkable progress of biological electron microscopy. Adv. Protein Chem. Struct. Biol. 2010, 81, 1–32. [Google Scholar] [CrossRef]
- Zhou, Z.H. Atomic resolution cryo electron microscopy of macromolecular complexes. Adv. Protein Chem. Struct. Biol. 2011, 82, 1–35. [Google Scholar] [CrossRef]
- San Martín, C.; Burnett, R.M.; de Haas, F.; Heinkel, R.; Rutten, T.; Fuller, S.D.; Butcher, S.J.; Bamford, D.H. Combined EM/X-ray imaging yields a quasi-atomic model of the adenovirus-related bacteriophage PRD1, and shows key capsid and membrane interactions. Structure 2001, 9, 917–930. [Google Scholar] [CrossRef]
- Rux, J.J.; Burnett, R.M. Type-specific epitope locations revealed by X-ray crystallographic study of adenovirus type 5 hexon. Mol. Ther. 2000, 1, 18–30. [Google Scholar] [CrossRef]
- Athappilly, F.K.; Murali, R.; Rux, J.J.; Cai, Z.P.; Burnett, R.M. The refined crystal-structure of hexon, the major coat protein of adenovirus type-2, at 2.9 Å resolution. J. Mol. Biol. 1994, 242, 430–455. [Google Scholar] [CrossRef]
- Shortridge, K.F.; Biddle, F. The proteins of adenovirus type 5. Arch. Gesamte Virusforsch. 1970, 29, 1–24. [Google Scholar] [CrossRef]
- Jornvall, H.; Philipson, L. Limited proteolysis and a reactive cysteine residue define accesible regions in the native conformation of the adenovirus hexon protein. Eur. J. Biochem. 1980, 104, 237–247. [Google Scholar] [CrossRef]
- San Martín, C.; Huiskonen, J.T.; Bamford, J.K.; Butcher, S.J.; Fuller, S.D.; Bamford, D.H.; Burnett, R.M. Minor proteins, mobile arms and membrane-capsid interactions in the bacteriophage PRD1 capsid. Nat. Struct. Biol. 2002, 9, 756–763. [Google Scholar]
- Olson, A.J.; Bricogne, G.; Harrison, S.C. Structure of tomato busy stunt virus IV. The virus particle at 2.9 Å resolution. J. Mol. Biol. 1983, 171, 61–93. [Google Scholar] [CrossRef]
- Stehle, T.; Gamblin, S.J.; Yan, Y.; Harrison, S.C. The structure of simian virus 40 refined at 3.1 Å resolution. Structure 1996, 4, 165–182. [Google Scholar] [CrossRef]
- Luque, D.; Saugar, I.; Rodríguez, J.F.; Verdaguer, N.; Garriga, D.; San Martín, C.; Velázquez-Muriel, J.A.; Trus, B.L.; Carrascosa, J.L.; Castón, J.R. Infectious bursal disease virus capsid assembly and maturation by structural rearrangements of a transient molecular switch. J. Virol. 2007, 81, 6869–6878. [Google Scholar]
- Bergelson, J.M.; Cunningham, J.A.; Droguett, G.; Kurt-Jones, E.A.; Krithivas, A.; Hong, J.S.; Horwitz, M.S.; Crowell, R.L.; Finberg, R.W. Isolation of a common receptor for Coxsackie B viruses and adenoviruses 2 and 5. Science 1997, 275, 1320–1323. [Google Scholar]
- Wickham, T.J.; Mathias, P.; Cheresh, D.A.; Nemerow, G.R. Integrins αvβ3 and αvβ5 promote adenovirus internalization but not virus attachment. Cell 1993, 73, 309–319. [Google Scholar] [CrossRef]
- Burckhardt, C.J.; Suomalainen, M.; Schoenenberger, P.; Boucke, K.; Hemmi, S.; Greber, U.F. Drifting motions of the adenovirus receptor CAR and immobile integrins initiate virus uncoating and membrane lytic protein exposure. Cell Host Microbe 2011, 10, 105–117. [Google Scholar] [CrossRef] [Green Version]
- San Martín, C.; Glasgow, J.N.; Borovjagin, A.; Beatty, M.S.; Kashentseva, E.A.; Curiel, D.T.; Marabini, R.; Dmitriev, I.P. Localization of the N-terminus of minor coat protein IIIa in the adenovirus capsid. J. Mol. Biol. 2008, 383, 923–934. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Liu, H.; Wu, L.; Zhou, Z.H. Model of the trimeric fiber and its interactions with the pentameric penton base of human adenovirus by cryo-electron microscopy. J. Mol. Biol. 2011, 406, 764–774. [Google Scholar] [CrossRef]
- Schoehn, G.; Fender, P.; Chroboczek, J.; Hewat, E.A. Adenovirus 3 penton dodecahedron exhibits structural changes of the base on fibre binding. EMBO J. 1996, 15, 6841–6846. [Google Scholar]
- D’Halluin, J.C.; Milleville, M.; Boulanger, P.A.; Martin, G.R. Temperature-sensitive mutant of adenovirus type 2 blocked in virion assembly: Accumulation of light intermediate particles. J. Virol. 1978, 26, 344–356. [Google Scholar]
- Ma, H.C.; Hearing, P. Adenovirus structural protein IIIa is involved in the serotype specificity of viral DNA packaging. J. Virol. 2011, 85, 7849–7855. [Google Scholar]
- Moyer, C.L.; Wiethoff, C.M.; Maier, O.; Smith, J.G.; Nemerow, G.R. Functional genetic and biophysical analyses of membrane disruption by human adenovirus. J. Virol. 2011, 85, 2631–2641. [Google Scholar] [CrossRef]
- Maier, O.; Galan, D.L.; Wodrich, H.; Wiethoff, C.M. An N-terminal domain of adenovirus protein VI fragments membranes by inducing positive membrane curvature. Virology 2010, 402, 11–19. [Google Scholar] [CrossRef]
- Wiethoff, C.M.; Wodrich, H.; Gerace, L.; Nemerow, G.R. Adenovirus protein VI mediates membrane disruption following capsid disassembly. J. Virol. 2005, 79, 1992–2000. [Google Scholar]
- Wodrich, H.; Henaff, D.; Jammart, B.; Segura-Morales, C.; Seelmeir, S.; Coux, O.; Ruzsics, Z.; Wiethoff, C.M.; Kremer, E.J. A capsid-encoded PPxY-motif facilitates adenovirus entry. PLoS Pathog. 2010, 6. [Google Scholar]
- Schreiner, S.; Martinez, R.; Groitl, P.; Rayne, F.; Vaillant, R.; Wimmer, P.; Bossis, G.; Sternsdorf, T.; Marcinowski, L.; Ruzsics, Z.; et al. Transcriptional activation of the adenoviral genome is mediated by capsid protein VI. PLoS Pathog. 2012, 8. [Google Scholar]
- Wodrich, H.; Guan, T.; Cingolani, G.; von Seggern, D.; Nemerow, G.; Gerace, L. Switch from capsid protein import to adenovirus assembly by cleavage of nuclear transport signals. EMBO J. 2003, 22, 6245–6255. [Google Scholar]
- Mangel, W.F.; Toledo, D.L.; Brown, M.T.; Martin, J.H.; McGrath, W.J. Characterization of three components of human adenovirus proteinase activity in vitro. J. Biol. Chem. 1996, 271, 536–543. [Google Scholar]
- Mangel, W.F.; McGrath, W.J.; Toledo, D.L.; Anderson, C.W. Viral DNA and a viral peptide can act as cofactors of adenovirus virion proteinase activity. Nature 1993, 361, 274–275. [Google Scholar]
- Russell, W.C.; Precious, B. Nucleic acid-binding properties of adenovirus structural polypeptides. J. Gen. Virol. 1982, 63, 69–79. [Google Scholar] [CrossRef]
- Liu, G.Q.; Babiss, L.E.; Volkert, F.C.; Young, C.S.; Ginsberg, H.S. A thermolabile mutant of adenovirus 5 resulting from a substitution mutation in the protein VIII gene. J. Virol. 1985, 53, 920–925. [Google Scholar]
- Singh, M.; Shmulevitz, M.; Tikoo, S.K. A newly identified interaction between IVa2 and pVIII proteins during porcine adenovirus type 3 infection. Virology 2005, 336, 60–69. [Google Scholar]
- Remaut, H.; Waksman, G. Protein-protein interaction through beta-strand addition. Trends Biochem. Sci. 2006, 31, 436–444. [Google Scholar] [CrossRef]
- Takahashi, E.; Cohen, S.L.; Tsai, P.K.; Sweeney, J.A. Quantitation of adenovirus type 5 empty capsids. Anal. Biochem. 2006, 349, 208–217. [Google Scholar]
- Vellekamp, G.; Porter, F.W.; Sutjipto, S.; Cutler, C.; Bondoc, L.; Liu, Y.H.; Wylie, D.; Cannon-Carlson, S.; Tang, J.T.; Frei, A.; et al. Empty capsids in column-purified recombinant adenovirus preparations. Hum. Gene Ther. 2001, 12, 1923–1936. [Google Scholar]
- Diouri, M.; Keyvani-Amineh, H.; Geoghegan, K.F.; Weber, J.M. Cleavage efficiency by adenovirus protease is site-dependent. J. Biol. Chem. 1996, 271, 32511–32514. [Google Scholar]
- Lehmberg, E.; Traina, J.A.; Chakel, J.A.; Chang, R.J.; Parkman, M.; McCaman, M.T.; Murakami, P.K.; Lahidji, V.; Nelson, J.W.; Hancock, W.S.; et al. Reversed-phase high-performance liquid chromatographic assay for the adenovirus type 5 proteome. J. Chromatogr. B Biomed. Sci. Appl. 1999, 732, 411–423. [Google Scholar] [CrossRef]
- Blanche, F.; Monegier, B.; Faucher, D.; Duchesne, M.; Audhuy, F.; Barbot, A.; Bouvier, S.; Daude, G.; Dubois, H.; Guillemin, T.; et al. Polypeptide composition of an adenovirus type 5 used in cancer gene therapy. J. Chromatogr. A 2001, 921, 39–48. [Google Scholar] [CrossRef]
- Chelius, D.; Huhmer, A.F.; Shieh, C.H.; Lehmberg, E.; Traina, J.A.; Slattery, T.K.; Pungor, E., Jr. Analysis of the adenovirus type 5 proteome by liquid chromatography and tandem mass spectrometry methods. J. Proteome Res. 2002, 1, 501–513. [Google Scholar] [CrossRef]
- Liu, Y.H.; Vellekamp, G.; Chen, G.D.; Mirza, U.A.; Wylie, D.; Twarowska, B.; Tang, J.T.; Porter, F.W.; Wang, S.H.; Nagabhushan, T.L.; et al. Proteomic study of recombinant adenovirus 5 encoding human p53 by matrix-assisted laser desorption/ionization mass spectrometry in combination with database search. Int. J. Mass Spectrom. 2003, 226, 55–69. [Google Scholar] [CrossRef]
- Colby, W.W.; Shenk, T. Adenovirus type 5 virions can be assembled in vivo in the absence of detectable polypeptide IX. J. Virol. 1981, 39, 977–980. [Google Scholar]
- Vellinga, J.; van den Wollenberg, D.J.; van der Heijdt, S.; Rabelink, M.J.; Hoeben, R.C. The coiled-coil domain of the adenovirus type 5 protein IX is dispensable for capsid incorporation and thermostability. J. Virol. 2005, 79, 3206–3210. [Google Scholar]
- Qin, L.; Fokine, A.; O’Donnell, E.; Rao, V.B.; Rossmann, M.G. Structure of the small outer capsid protein, Soc: A clamp for stabilizing capsids of T4-like phages. J. Mol. Biol. 2010, 395, 728–741. [Google Scholar] [CrossRef]
- Ishii, T.; Yanagida, M. The two dispensable structural proteins (soc and hoc) of the T4 phage capsid; their purification and properties, isolation and characterization of the defective mutants, and their binding with the defective heads in vitro. J. Mol. Biol. 1977, 109, 487–514. [Google Scholar] [CrossRef]
- De Vrij, J.; van den Hengel, S.K.; Uil, T.G.; Koppers-Lalic, D.; Dautzenberg, I.J.; Stassen, O.M.; Barcena, M.; Yamamoto, M.; de Ridder, C.M.; Kraaij, R.; et al. Enhanced transduction of CAR-negative cells by protein IX-gene deleted adenovirus 5 vectors. Virology 2011, 410, 192–200. [Google Scholar] [CrossRef]
- Parks, R.J. Adenovirus protein IX: A new look at an old protein. Mol. Ther. 2005, 11, 19–25. [Google Scholar] [CrossRef]
- The UniProt Consortium. Reorganizing the protein space at the Universal Protein Resource (UniProt). Nucleic Acids Res. 2012, 40, D71–D75. [CrossRef]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Soding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7. [Google Scholar]
- Waterhouse, A.M.; Procter, J.B.; Martin, D.M.; Clamp, M.; Barton, G.J. Jalview version 2—A multiple sequence alignment editor and analysis workbench. Bioinformatics 2009, 25, 1189–1191. [Google Scholar]
- Strunze, S.; Engelke, M.F.; Wang, I.H.; Puntener, D.; Boucke, K.; Schleich, S.; Way, M.; Schoenenberger, P.; Burckhardt, C.J.; Greber, U.F. Kinesin-1-mediated capsid disassembly and disruption of the nuclear pore complex promote virus infection. Cell Host Microbe 2011, 10, 210–223. [Google Scholar] [CrossRef] [Green Version]
- Suomalainen, M.; Nakano, M.Y.; Keller, S.; Boucke, K.; Stidwill, R.P.; Greber, U.F. Microtubule-dependent plus- and minus end-directed motilities are competing processes for nuclear targeting of adenovirus. J. Cell Biol. 1999, 144, 657–672. [Google Scholar] [CrossRef] [Green Version]
- D’Halluin, J.C. Virus assembly. Curr. Top. Microbiol. Immunol. 1995, 199, 47–66. [Google Scholar]
- Weber, J.M. Role of Endoprotease in Adenovirus Infection. In Adenoviruses: Basic Biology to Gene Therapy; Seth, P., Ed.; R.G. Landes: Austin, TX, USA, 1999; pp. 79–83. [Google Scholar]
- Cotten, M.; Weber, J.M. The adenovirus protease is required for virus entry into host-cells. Virology 1995, 213, 494–502. [Google Scholar] [CrossRef]
- Gastaldelli, M.; Imelli, N.; Boucke, K.; Amstutz, B.; Meier, O.; Greber, U.F. Infectious adenovirus type 2 transport through early but not late endosomes. Traffic 2008, 9, 2265–2278. [Google Scholar] [CrossRef]
- Baniecki, M.L.; McGrath, W.J.; McWhirter, S.M.; Li, C.; Toledo, D.L.; Pellicena, P.; Barnard, D.L.; Thorn, K.S.; Mangel, W.F. Interaction of the human adenovirus proteinase with its 11-amino acid cofactor pVIc. Biochemistry 2001, 40, 12349–12356. [Google Scholar]
- McGrath, W.J.; Baniecki, M.L.; Li, C.; McWhirter, S.M.; Brown, M.T.; Toledo, D.L.; Mangel, W.F. Human adenovirus proteinase: DNA binding and stimulation of proteinase activity by DNA. Biochemistry 2001, 40, 13237–13245. [Google Scholar]
- Weber, J. Genetic analysis of adenovirus type 2 III. Temperature sensitivity of processing viral proteins. J. Virol. 1976, 17, 462–471. [Google Scholar]
- Rancourt, C.; Keyvani-Amineh, H.; Sircar, S.; Labrecque, P.; Weber, J.M. Proline 137 is critical for adenovirus protease encapsidation and activation but not enzyme activity. Virology 1995, 209, 167–173. [Google Scholar] [CrossRef]
- Imelli, N.; Ruzsics, Z.; Puntener, D.; Gastaldelli, M.; Greber, U.F. Genetic reconstitution of the human adenovirus type 2 temperature-sensitive 1 mutant defective in endosomal escape. Virol. J. 2009, 6. [Google Scholar]
- Nguyen, E.K.; Nemerow, G.R.; Smith, J.G. Direct evidence from single-cell analysis that human {alpha}-defensins block adenovirus uncoating to neutralize infection. J. Virol. 2010, 84, 4041–4049. [Google Scholar] [CrossRef]
- Perez-Berna, A.J.; Marabini, R.; Scheres, S.H.W.; Menendez-Conejero, R.; Dmitriev, I.P.; Curiel, D.T.; Mangel, W.F.; Flint, S.J.; San Martin, C. Structure and uncoating of immature adenovirus. J. Mol. Biol. 2009, 392, 547–557. [Google Scholar]
- Ostapchuk, P.; Hearing, P. Control of adenovirus packaging. J. Cell. Biochem. 2005, 96, 25–35. [Google Scholar] [CrossRef]
- Ostapchuk, P.; Almond, M.; Hearing, P. Characterization of Empty adenovirus particles assembled in the absence of a functional adenovirus IVa2 protein. J. Virol. 2011, 85, 5524–5531. [Google Scholar] [CrossRef]
- 1 HAdV: human adenovirus. FAdV: fowl adenovirus. CAdV: canine adenovirus. SAdV: simian adenovirus. PAdV: porcine adenovirus.
- 2 Strictly speaking, “atomic resolution” would be ~1 Å. However it is frequent to use this term, like here, meaning “a resolution such that the map quality allows tracing of the polypeptide chain, generally thanks to the visibility of bulky side chains”.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
San Martín, C. Latest Insights on Adenovirus Structure and Assembly. Viruses 2012, 4, 847-877. https://doi.org/10.3390/v4050847
San Martín C. Latest Insights on Adenovirus Structure and Assembly. Viruses. 2012; 4(5):847-877. https://doi.org/10.3390/v4050847
Chicago/Turabian StyleSan Martín, Carmen. 2012. "Latest Insights on Adenovirus Structure and Assembly" Viruses 4, no. 5: 847-877. https://doi.org/10.3390/v4050847






