Accountable Accounting: Carbon-Based Management on Marginal Lands
Abstract
:1. Introduction
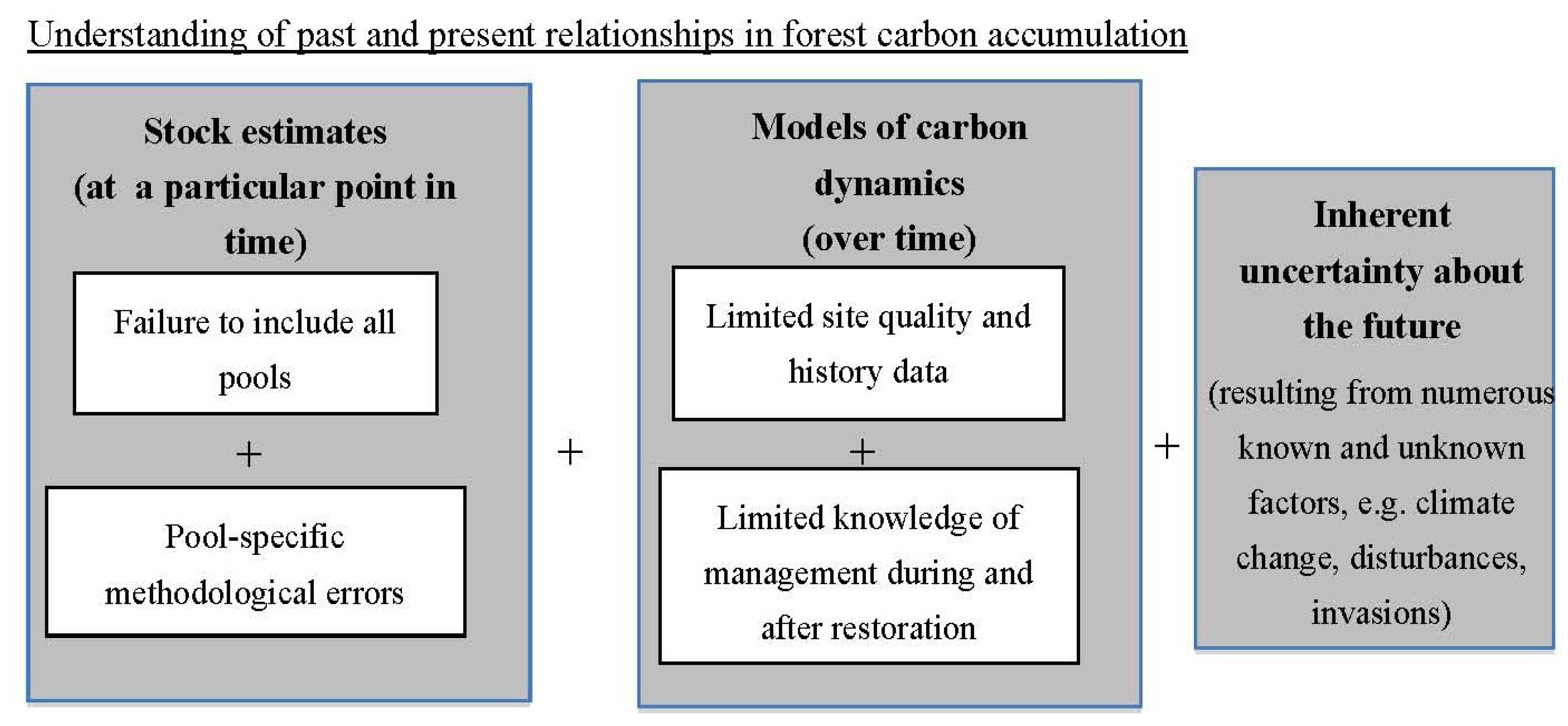
2. Quantification of Forest Carbon Stocks
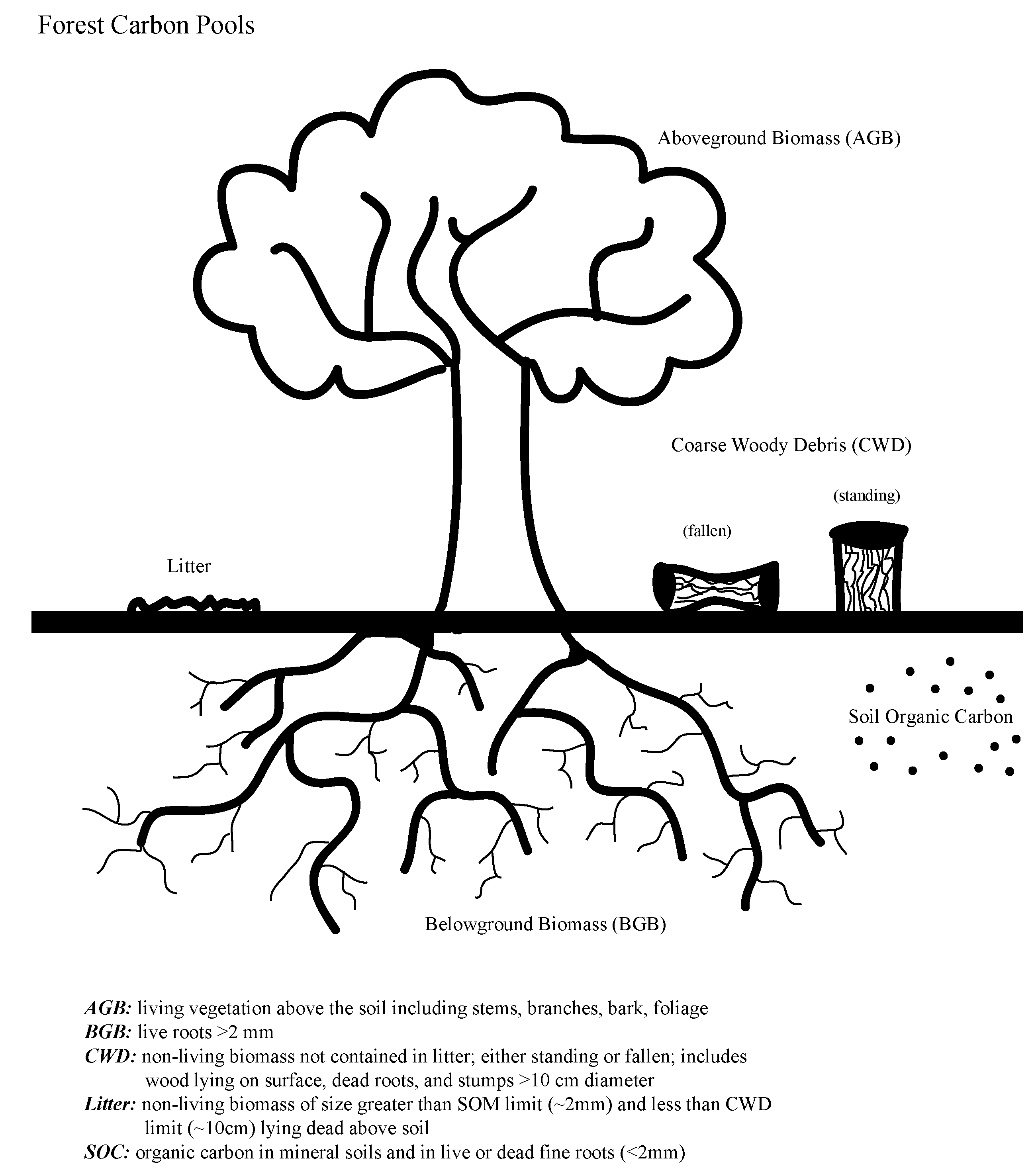
2.1. Failure to Include All C Pools
2.2. Pool-Specific Methodological Errors
2.3. The Importance of Standardized and Scientifically Sound Methodology
3. Barriers to Effective Prediction of C Recovery Dynamics
3.1. Broad Categories of Land Use History
| Prior Land Use Characteristics | Effect on Forest Recovery |
|---|---|
| Fertilized > Not Fertilized | Fertilized cropland will likely experience higher rates of biomass accumulation than unfertilized land, due to increased soil nutrient availability. |
| Cropland > Pasture | Pasture lands tend to be more degraded than cropland, as low fertility and soil compaction cause reduced aeration and soil biological activity. However, this trend could be due to the preferential selection of less productive sites for pasture in the first place. |
| Young Pasture > Old Pasture | Younger pastures are less degraded than older pastures since they have not experienced as much soil compaction and nutrient loss from pastoral activity. Therefore, there is faster biomass accumulation on younger pastures than older pastures. |
| Longer fallow > Shorter fallow | Although an ideal fallow period to maintain site fertility has yet to be established in the literature, longer fallow periods allow the replenishment of site nutrients. Increased site nutrients are associated with increased biomass growth and subsequent C sequestration. |
| Fewer cycles > More cycles | Fewer cycles of cultivation means a less degraded landscape. A less degraded landscape has better site quality with the nutrients necessary for higher carbon accumulation rates. |
3.2. Effect of Current and Post-Restoration Management
3.3. Problems with Meta-Analysis
4. Research, Policy, and Decision-Making Implications
4.1. Research Implications
| Data Category | Relevant Variables |
|---|---|
| Site Quality | A range of physical (slope, aspect, altitude) and soil characteristics including pH, bulk density, N, Ca, P, K, micronutrient levels, soil texture and structure, stone content (pre-restoration data) |
| Land Use History | Type, duration, number of cycles, number of types, fertilization, remnant vegetation, other detailed practices (pre-restoration data) |
| Management | Fertilizer applications during planting/natural succession, ploughing, tilling, mycorrhizal inoculations, thinning, planting of N-fixing species, any other strategies for assisted regeneration (data during restoration) |
4.2. Policy and Decision-Making Implications
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Campbell, J.E.; Lobell, D.B.; Genova, R.C.; Field, C.B. The global potential of bioenergy on abandoned agriculture lands. Environ. Sci. Technol. 2008, 42, 5791–5794. [Google Scholar] [CrossRef]
- Cai, X.; Zhang, X.; Wang, D. Land availability for biofuel production. Environ. Sci. Technol. 2011, 45, 334–339. [Google Scholar] [CrossRef]
- Righelato, R.; Spracklen, D.V. Environment. Carbon mitigation by biofuels or by saving and restoring forests? Science 2007, 317, 902. [Google Scholar] [CrossRef]
- Kauffman, J.B.; Hughes, R.F.; Heider, C. Carbon pool and biomass dynamics associated with deforestation, land use, and agricultural abandonment in the neotropics. Ecol. Appl. 2009, 19, 1211–1222. [Google Scholar] [CrossRef]
- Omeja, P.A.; Obua, J.; Rwetsiba, A.; Chapman, C.A. Biomass accumulation in tropical lands with different disturbance histories: Contrasts within one landscape and across regions. For. Ecol. Manag. 2012, 269, 293–300. [Google Scholar] [CrossRef]
- Ngo, K.M.; Turner, B.L.; Muller-Landau, H.C.; Davies, S.J.; Larjavaara, M.; Nik Hassan, N.F.B.; Lum, S. Carbon stocks in primary and secondary tropical forests in Singapore. For. Ecol. Manag. 2013, 296, 81–89. [Google Scholar] [CrossRef]
- Scown, C.D.; Nazaroff, W.W.; Mishra, U.; Strogen, B.; Lobscheid, A.B.; Masanet, E.; Santero, N.J.; Horvath, A.; Mckone, T.E. Lifecycle greenhouse gas implications of US national scenarios for cellulosic ethanol production. Environ. Res. Lett. 2012, 7, 1–9. [Google Scholar]
- Edwards, R.; Larive, J.F.; Rickeard, D.; Weindorf, W. Well-to-Tank Version 4 JEC Well-to-Wheels Analysis; European Commission Joint Research Center: Brussels, Belgium, 2013; p. 134. [Google Scholar]
- Fischer, G.; Hiznyik, E.; Priler, S.; Shana, M.; van Velthuizen, H. Global Agro-Ecological Assessment for Agriculture in the 21st Century: Methodology and Results; IIASA and FAO: Laxenburg, Austria; Rome, Italy, 2002. [Google Scholar]
- Ramankutty, N.; Gibbs, H.K.; Achard, F.; Defries, R.; Foley, J.A.; Houghton, R.A. Challenges to estimating carbon emissions from tropical deforestation. Glob. Chang. Biol. 2007, 13, 51–66. [Google Scholar] [CrossRef]
- Woodall, C.W.; Domke, G.M.; Riley, K.L.; Oswalt, C.M.; Crocker, S.J.; Yohe, G.W. A framework for assessing global change risks to forest carbon stocks in the United States. PLoS One 2013, 8, e73222. [Google Scholar]
- Silver, W.L.; Ostertag, R.; Lugo, A.E. The Potential for carbon sequestration through reforestation of abandoned tropical agricultural and pasture lands. Restor. Ecol. 2000, 8, 394–407. [Google Scholar] [CrossRef]
- Bonner, M.T.L.; Schmidt, S.; Shoo, L.P. A meta-analytical global comparison of aboveground biomass accumulation between tropical secondary forests and monoculture plantations. For. Ecol. Manag. 2013, 291, 73–86. [Google Scholar] [CrossRef]
- Cuevas, E.; Brown, S.; Lugo, A.E. Above- and belowground organic matter storage and production in tropical pine plantation and a paired broadleaf secondary forest. Plant Soil 1991, 135, 257–268. [Google Scholar] [CrossRef]
- Davis, M.R.; Condron, L.M. Impact of grassland afforestation on soil carbon in New Zealand: A review of paired-site studies. Aust. J. Soil Res. 2002, 40, 675–690. [Google Scholar] [CrossRef]
- Rhoades, C.C.; Eckert, G.E.; Coleman, D.C. Soil carbon differences among forest, agriculture, and secondary vegetation in Lower Montane Ecuador. Ecol. Appl. 2000, 10, 497–505. [Google Scholar] [CrossRef]
- Paul, K.I.; Polglase, P.J.; Nyakuengama, J.G.; Khanna, P.K. Change in soil carbon following afforestation. For. Ecol. Manag. 2002, 168, 241–257. [Google Scholar] [CrossRef]
- Powers, J.S. Changes in soil carbon and nitrogen after contrasting land-use transitions in northeastern Costa Rica. Ecosystems 2004, 7, 134–146. [Google Scholar] [CrossRef]
- Schedlbauer, J.L.; Kavanagh, K.L. Soil carbon dynamics in a chronosequence of secondary forests in northeastern Costa Rica. For. Ecol. Manag. 2008, 255, 1326–1335. [Google Scholar] [CrossRef]
- Lima, A.M.N.; Silva, I.R.; Neves, J.C.L.; Novais, R.F.; Barros, N.F.; Mendonça, E.S.; Smyth, T.J.; Moreira, M.S.; Leite, F.P. Soil organic carbon dynamics following afforestation of degraded pastures with eucalyptus in southeastern Brazil. For. Ecol. Manag. 2006, 235, 219–231. [Google Scholar] [CrossRef]
- Sauer, T.J.; James, D.E.; Cambardella, C.A.; Hernandez-Ramirez, G. Soil properties following reforestation or afforestation of marginal cropland. Plant Soil 2012, 360, 375–390. [Google Scholar] [CrossRef]
- Li, D.; Niu, S.; Luo, Y. Global patterns of the dynamics of soil carbon and nitrogen stocks following afforestation: A meta-analysis. New Phytol. 2012, 195, 172–181. [Google Scholar] [CrossRef]
- Don, A.; Schumacher, J.; Freibauer, A. Impact of tropical land-use change on soil organic carbon stocks-a meta-analysis. Glob. Chang. Biol. 2011, 17, 1658–1670. [Google Scholar] [CrossRef]
- Neumann-Cosel, L.; Zimmermann, B.; Hall, J.S.; van Breugel, M.; Elsenbeer, H. Soil carbon dynamics under young tropical secondary forests on former pastures—A case study from Panama. For. Ecol. Manag. 2011, 261, 1625–1633. [Google Scholar] [CrossRef]
- Vesterdal, L.; Rosenqvist, L. Carbon sequestration in soil and biomass following afforestation: Experiences from oak and Norway spruce chronosequences in Denmark, Sweden and the Netherlands. Eff. Afforestation 2007, 1, 19–51. [Google Scholar]
- Wang, J.; Epstein, H.E. Estimating carbon source-sink transition during secondary succession in a Virginia valley. Plant Soil 2012, 362, 135–147. [Google Scholar] [CrossRef]
- Sang, P.M.; Lamb, D.; Bonner, M.; Schmidt, S. Carbon sequestration and soil fertility of tropical tree plantations and secondary forest established on degraded land. Plant Soil 2012, 362, 187–200. [Google Scholar]
- Sierra, C.A.; del Valle, J.I.; Orrego, S.A.; Moreno, F.H.; Harmon, M.E.; Zapata, M.; Coorado, G.J.; Herrera, M.A.; Lara, W.; Restrepo, D.E.; et al. Total carbon stocks in a tropical forest landscape of the Porce region, Colombia. For. Ecol. Manag. 2007, 243, 299–309. [Google Scholar] [CrossRef]
- Dierkes, C. Accounting for carbon in Great Lakes Forests. In OSU Climate Change Webinar Series; OSU Climate Change: Columbus, OH, USA, 2011; pp. 1–3. [Google Scholar]
- Hooker, T.D.; Compton, J.E. Forest ecosystem carbon and nitrogen accumulation during the first century after agricultural abandonment. Ecol. Appl. 2003, 13, 299–313. [Google Scholar] [CrossRef]
- Jaramillo, V.J.; Ahedo-Hernndez, R.; Kauffman, J.B. Root biomass and carbon in a tropical evergreen forest of Mexico: Changes with secondary succession and forest conversion to pasture. J. Trop. Ecol. 2003, 19, 457–464. [Google Scholar] [CrossRef]
- Nunes, L.; Patricio, M.; Tomé, J.; Tomé, M. Carbon and nutrients stocks in even-aged maritime pine stands from Portugal. For. Syst. 2010, 19, 434–448. [Google Scholar]
- Robinson, D. Implications of a large global root biomass for carbon sink estimates and for soil carbon dynamics. Proc. R. Soc. B: Biol. Sci. 2007, 274, 2753–2759. [Google Scholar] [CrossRef]
- Preece, N.D.; Crowley, G.M.; Lawes, M.J.; van Oosterzee, P. Comparing above-ground biomass among forest types in the Wet Tropics: Small stems and plantation types matter in carbon accounting. For. Ecol. Manag. 2012, 264, 228–237. [Google Scholar] [CrossRef]
- Brown, S. Measuring carbon in forests: Current status and future challenges. Environ. Pollut. 2002, 116, 363–372. [Google Scholar] [CrossRef]
- Chave, J.; Andalo, C.; Brown, S.; Cairns, M.A.; Chambers, J.Q.; Eamus, D.; Folster, H.; Fromard, F.; Higuchi, N.; Kira, T.; et al. Tree allometry and improved estimation and balance in tropical forests of carbon stocks. Oecologia 2005, 145, 87–99. [Google Scholar] [CrossRef]
- Martin, A.R.; Thomas, S.C. A reassessment of carbon content in tropical trees. PLoS One 2011, 6, e23533. [Google Scholar] [CrossRef]
- Elias, M.; Potvin, C. Assessing inter- and intra-specific variation in trunk carbon concentration for 32 neotropical tree species. Can. J. For. Res. 2003, 33, 1039–1045. [Google Scholar] [CrossRef]
- Gifford, R.M. Carbon Contents of Above-Ground Tissues of Forest and Woodland Tress; National Carbon Accounting System, Technical Report No 22; Australian Greenhouse Office: Canberra, Australia, September 2000. [Google Scholar]
- Milakovsky, B.; Frey, B.; James, T. Carbon dynamics in the boreal forest. In Managing Forest Carbon in a Changing Climate; Ashton, M.S., Tyrrell, M.L., Spalding, D., Gentry, B., Eds.; Springer: New York, NY, USA, 2012; p. 115. [Google Scholar]
- Robinson, D. Scaling the depths: Below-ground allocation in plants, forests and biomes. Funct. Ecol. 2004, 18, 290–295. [Google Scholar] [CrossRef]
- Taylor, B.N.; Beidler, K.V.; Cooper, E.R.; Strand, A.E.; Pritchard, S.G. Sampling volume in root studies: The pitfalls of under-sampling exposed using accumulation curves. Ecol. Lett. 2013, 16, 862–869. [Google Scholar] [CrossRef]
- Mokany, K.; Raison, R.J.; Prokushkin, A.S. Critical analysis of root: Shoot ratios in terrestrial biomes. Glob. Chang. Biol. 2006, 12, 84–96. [Google Scholar] [CrossRef]
- Ping, X.; Zhou, G.; Zhuang, Q.; Wang, Y.; Zuo, W.; Shi, G.; Lin, X.; Wang, Y. Effects of sample size and position from monolith and core methods on the estimation of total root biomass in a temperate grassland ecosystem in Inner Mongolia. Geoderma 2010, 155, 262–268. [Google Scholar] [CrossRef]
- Aalde, H.; Gonzalez, P.; Gytarsky, M.; Krug, T.; Kurz, W.A.; Ogle, S.; Somogyi, Z. Forest land. In 2006 IPCC Guidelines for National Greenhouse Gas Inventories (Vol. 4: Agriculture, Forestry, and Other Land Use); Eggleston, H.S., Buendia, L., Miwa, K., Ngara, T., Tanabe, K., Eds.; Institute for Global Environmental Strategies: Hayama, Japan, 2006. [Google Scholar]
- MacDicken, K. A Guide to Monitoring Carbon Storage in Forestry and Agroforestry Projects; Winrock International Institute for Agricultural Development: Little Rock, AR, USA, 1997. [Google Scholar]
- Center for Tropical Forest Science. Soil Carbon Sampling Protocol; Center for Tropical Forest Science, Smithsonian Tropical Research Institute: Washington, DC, USA, 2010. [Google Scholar]
- Fahey, T.J.; Woodbury, P.B.; Battles, J.J.; Goodale, C.L.; Hamburg, S.; Ollinger, S.; Woodall, C.W. Forest carbon storage: Ecology, management, and policy. Front. Ecol. Environ. 2010, 8, 245–252. [Google Scholar] [CrossRef]
- Lemenih, M.; Olsson, M.; Karltun, E. Comparison of soil attributes under Cupressus lusitanica and Eucalyptus saligna established on abandoned farmlands with continuously cropped farmlands and natural forest in Ethiopia. For. Ecol. Manag. 2004, 195, 57–67. [Google Scholar] [CrossRef]
- Anderson, K.J.; Allen, A.P.; Gillooly, J.F.; Brown, J.H. Temperature-dependence of biomass accumulation rates during secondary succession. Ecol. Lett. 2006, 9, 673–682. [Google Scholar] [CrossRef]
- Holl, K.D.; Aide, T.M. When and where to actively restore ecosystems? For. Ecol. Manag. 2011, 261, 1558–1563. [Google Scholar] [CrossRef]
- Niu, X.; Duiker, S.W. Carbon sequestration potential by afforestation of marginal agricultural land in the Midwestern U.S. For. Ecol. Manag. 2006, 223, 415–427. [Google Scholar] [CrossRef]
- Letcher, S.G.; Chazdon, R.L. Rapid recovery of biomass, species richness, and species composition in a forest chronosequence in northeastern Costa Rica. Biotropica 2009, 41, 608–617. [Google Scholar] [CrossRef]
- Van Rooyen, M.W.; van Rooyen, N.; Stoffberg, G.H. Carbon sequestration potential of post-mining reforestation activities on the KwaZulu-Natal coast, South Africa. Forestry 2012, 86, 211–223. [Google Scholar]
- Kasel, S.; Bennett, L.T. Land-use history, forest conversion, and soil organic carbon in pine plantations and native forests of south eastern Australia. Geoderma 2007, 137, 401–413. [Google Scholar] [CrossRef]
- Guo, L.B.; Gifford, R.M. Soil carbon stocks and land use change: A meta analysis. Glob. Chang. Biol. 2002, 8, 345–360. [Google Scholar] [CrossRef]
- Kotto-same, J.; Woomer, P.L.; Appolinaire, M.; Louis, Z. Carbon dynamics in slash-and-bum agriculture and land use alternatives of the humid forest zone in Cameroon. Agric. Ecosyst. Environ. 1997, 809, 245–256. [Google Scholar]
- Feldpausch, T.R.; Rondon, M.A.; Fernandes, E.C.M.; Riha, S.J.; Wandelli, E. Carbon and nutrient accumulation in secondary forests regenerating on pastures in central Amazonia. Ecol. Appl. 2004, 14, S164–S176. [Google Scholar]
- Hughes, R.F.; Kauffmann, J.B.; Jaramillo, V.J. Biomass, carbon, and nutrient dynamics of secondary forests in a humid tropical region of Mexico. Ecology 1999, 80, 1892–1907. [Google Scholar]
- De Koning, G.H.J.; Veldkamp, E.; López-Ulloa, M. Quantification of carbon sequestration in soils following pasture to forest conversion in northwestern Ecuador. Glob. Biogeochem. Cycles 2003, 17. [Google Scholar] [CrossRef]
- Powers, J.S.; Veldkamp, E. Regional variation in soil carbon and d13C in forests and pastures of northeastern Costa Rica. Biogeochemistry 2005, 72, 315–336. [Google Scholar] [CrossRef]
- Klanderud, K.; Mbolatiana, H.Z.H.; Vololomboahangy, M.N.; Radimbison, M.A.; Roger, E.; Totland, Ø.; Rajeriarison, C. Recovery of plant species richness and composition after slash-and-burn agriculture in a tropical rainforest in Madagascar. Biodivers. Conserv. 2009, 19, 187–204. [Google Scholar]
- Fearnside, P.M.; Guimaraes, W.M. Carbon uptake by secondary forests in Brazilian Amazonia. For. Ecol. Manag. 1996, 80, 35–46. [Google Scholar] [CrossRef]
- Holl, K.D. Old field vegetation succession in the Neotropics. In Old Fields; Cramer, V.A., Hobbs, R.J., Eds.; Island Press: Washington, DC, USA, 2007; pp. 93–117. [Google Scholar]
- Peichl, M.; Arain, M.A.; Brodeur, J.J. Age effects on carbon fluxes in temperate pine forests. Agric. For. Meteorol. 2010, 150, 1090–1101. [Google Scholar] [CrossRef]
- Fonseca, W.; Rey Benayas, J.M.; Alice, F.E. Carbon accumulation in the biomass and soil of different aged secondary forests in the humid tropics of Costa Rica. For. Ecol. Manag. 2011, 262, 1400–1408. [Google Scholar] [CrossRef]
- Black, K.; Byrne, K.A.; Mencuccini, M.; Tobin, B.; Nieuwenhuis, M.; Reidy, B.; Bolger, T.; Saiz, G.; Green, C.; Farrell, E.T.; et al. Carbon stock and stock changes across a Sitka spruce chronosequence on surface-water gley soils. Forestry 2009, 82, 255–272. [Google Scholar] [CrossRef]
- Lasco, R.D.; Pulhin, F.B. Carbon budgets of forest ecosystems in the Philippines. J. Eviron. Sci. Manag. 2009, 12, 1–13. [Google Scholar]
- Macedo, M.O.; Resende, A.S.; Garcia, P.C.; Boddey, R.M.; Jantalia, C.P.; Urquiaga, S.; Campello, E.F.C.; Franco, A.A. Changes in soil C and N stocks and nutrient dynamics 13 years after recovery of degraded land using leguminous nitrogen-fixing trees. For. Ecol. Manag. 2008, 255, 1516–1524. [Google Scholar] [CrossRef]
- Kanowski, J.; Catterall, C.P. Carbon stocks in above-ground biomass of monoculture plantations, mixed species plantations and environmental restoration plantings in north-east Australia. Ecol. Manag. Restor. 2010, 11, 119–126. [Google Scholar] [CrossRef]
- Mekuria, W.; Veldkamp, E.; Corre, M.D. Restoration of ecosystem carbon stocks following exclosure establishment in communal. For. Range Wildland Soils 2011, 75, 246–256. [Google Scholar]
- Gamboa, A.M.; Hidalgo, C.; de León, F.; Etchevers, J.D.; Gallardo, J.F.; Campo, J. Nutrient addition differentially affects soil carbon sequestration in secondary tropical dry forests: Early- versus Late-Succession Stages. Restor. Ecol. 2010, 18, 252–260. [Google Scholar] [CrossRef]
- Ziegler, A.D.; Phelps, J.; Yuen, J.Q.; Webb, E.L.; Lawrence, D.; Fox, J.M.; Bruun, T.B.; Leisz, S.J.; Ryan, C.; Dressler, W.; et al. Carbon outcomes of major land-cover transitions in SE Asia: Great uncertainties and REDD+ policy implications. Glob. Chang. Biol. 2012, 18, 3087–3099. [Google Scholar] [CrossRef]
- Hall, J.M.; Holt, T.; Daniels, A.E.; Balthazar, V.; Lambin, E.F. Trade-offs between tree cover, carbon storage and floristic biodiversity in reforesting landscapes. Landsc. Ecol. 2012, 27, 1135–1147. [Google Scholar] [CrossRef]
- Thompson, I.; Mackey, B.; McNulty, S.; Mosseler, A. Forest resilience, biodiversity, and climate change. A synthesis of the biodiversity/resilience/stability relationship in forest ecosystems. In CBD Technical Series; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2009. [Google Scholar]
- Hobbs, R.J.; Higgs, E.; Harris, J.A. Novel ecosystems: Implications for conservation and restoration. Trends Ecol. Evol. 2009, 24, 599–605. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
DiRocco, T.L.; Ramage, B.S.; Evans, S.G.; Potts, M.D. Accountable Accounting: Carbon-Based Management on Marginal Lands. Forests 2014, 5, 847-861. https://doi.org/10.3390/f5040847
DiRocco TL, Ramage BS, Evans SG, Potts MD. Accountable Accounting: Carbon-Based Management on Marginal Lands. Forests. 2014; 5(4):847-861. https://doi.org/10.3390/f5040847
Chicago/Turabian StyleDiRocco, Tara L., Benjamin S. Ramage, Samuel G. Evans, and Matthew D. Potts. 2014. "Accountable Accounting: Carbon-Based Management on Marginal Lands" Forests 5, no. 4: 847-861. https://doi.org/10.3390/f5040847




