Seed Size, the Only Factor Positively Affecting Direct Seeding Success in an Abandoned Field in Quebec, Canada
Abstract
:1. Introduction
2. Methods
2.1. Study Area
2.2. Experimental Design
2.3. Seeding
| Species | Species development in natural conditions | Average dry seed mass (g) | Shade tolerance | Germination in laboratory |
|---|---|---|---|---|
| Betula papyrifera Marsh. (Paper birch) | Fast-growing tree, often established after fire or logging; develops best on well-drained sandy loams | 0.00045 | Low | 82% |
| Betula alleghaniensis Britton (Yellow birch) | Slow-growing tree, often found on moist well-drained soils | 0.0012 | Intermediate | 88% |
| Larix laricina (Du Roi) K. Koch (Tamarack) | Fast-growing tree, often found on wet to moist organic soils | 0.0027 | Low | 81% |
| Pinus resinosa Ait. (Red pine) | Moderate to fast growing tree, usually established following fire; often found on sandy soils | 0.01 | Low | 93% |
| Acer saccharum Marsh. (Sugar maple) | Slow-growing tree; grows on a variety of sites and soils | 0.06 | High | 88% |
| Quercus rubra L. (Northern red oak) | Moderate to fast growing tree; found on a variety of soils | 3.14 | Intermediate | 88% |
2.4. Measurements
2.4.1. Seedling Emergence, Survival and Growth
2.4.2. Red Oak Seedling Measurements
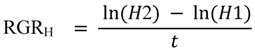
2.4.3. Environmental Measurements: Soil Moisture and Light
2.5. Data Analysis
2.6. Second Seeding Experiment
3. Results
3.1. Establishment Success
| Species | Date of maximum emergence | Maximum emergence (%) | Seedling survival in 2010 (%) | Seedling survival in 2011 (%) | Proportion of live seedlings in June 2011 | Height in June 2011 (cm) |
|---|---|---|---|---|---|---|
| Red pine | 21 June, 2010 | 34.0 ± 17.6 | 32.1 | 16.3 | 5.5 ± 5.9 | 3.8 ± 0.6 |
| Sugar maple | 6 July, 2010 | 5.8 ± 4.6 | 71.4 | 57.1 | 3.3 ± 3.1 | 11.5 ± 0.7 |
| Red oak | 25 August, 2010 | 57.2 ± 13.8 | 100 | 91.7 | 52.5 ± 14.8 | 10.6 ± 0.2 |
3.2. Effects of Treatments

| Growth variables | No vegetation | Vegetation | Fdf; p-values |
|---|---|---|---|
| Height (cm) | 10.65 ± 0.58 | 12.53 ± 0.57 | F1,1.98 = 7.82; p = 0.1086 |
| RGRH (cm cm−1 yr−1) | 0.50 ± 0.03 | 0.45 ± 0.03 | F1,1 = 0.0005; p = 0.9860 |
| Diameter (mm) | 3.51 ± 0.19 | 2.64 ± 0.19 | F1,1.11 = 118.47; p = 0.0486 |
| Shoot biomass (g) | 1.36 ± 0.08 | 0.78 ± 0.07 | F1,1.85 = 29.42; p = 0.0381 |
| SLA (mm2·mg−1) | 14.75 ± 0.53 | 17.13 ± 0.53 | F1,1.96 = 21.24; p = 0.0456 |
| Date | Precipitation during the last 3 days (mm) | No vegetation | Vegetation | Fdf; p-values |
|---|---|---|---|---|
| 15 June, 2011 | 41 | 22.5 ± 0.4 | 21.1 ± 0.4 | F1,2.08 = 21.42; p = 0.0405 |
| 12 July, 2011 | 2 | 22.4 ± 0.4 | 18.6 ± 0.4 | F1,1.96 = 31.14; p = 0.0321 |
| 18 July, 2011 | 1 | 23.8 ± 0.5 | 17.3 ± 0.5 | F1,2 = 83.39; p = 0.0117 |
4. Discussion
4.1. Establishment Success and Seed Size
4.2. Effects of Vegetation
4.3. Effects of Predation
5. Conclusions and Restoration Implications
Acknowledgments
Conflict of Interest
References
- MacDonald, D.; Crabtree, J.R.; Wiesinger, G.; Dax, T.; Stamou, N.; Fleury, P.; Gutierrez Lazpita, J.; Gibon, A. Agricultural abandonment in mountain areas of Europe: Environmental consequences and policy response. J. Environ. Manag. 2000, 59, 47–69. [Google Scholar] [CrossRef]
- Ramankutty, N.; Foley, J.A. Estimating historical changes in land cover: North American croplands from 1850 to 1992. Glob. Ecol. Biogeogr. 1999, 8, 381–396. [Google Scholar] [CrossRef]
- Ramankutty, N.; Foley, J.A. Estimating historical changes in global land cover: Croplands from 1700 to 1992. Glob. Biogeochem. Cycles 1999, 13, 997–1027. [Google Scholar] [CrossRef]
- Domon, G.; Bouchard, A.; Gariepy, M. The dynamics of the forest landscape of Haut-Saint-Laurent (Quebec, Canada): Interactions between biophysical factors, perceptions and policy. Landsc. Urban Plan. 1993, 25, 53–74. [Google Scholar] [CrossRef]
- Niering, W.A.; Goodwin, R.H. Creation of relatively stable shrublands with herbicides: Arresting “Succession” on rights-of-way and pastureland. Ecology 1974, 55, 784–795. [Google Scholar] [CrossRef]
- Benjamin, K.; Domon, G.; Bouchard, A. Vegetation composition and succession of abandoned farmland: Effects of ecological, historical and spatial factors. Landsc. Ecol. 2005, 20, 627–647. [Google Scholar] [CrossRef]
- D’Orangeville, L.; Bouchard, A.; Cogliastro, A. Post-Agricultural forests: Landscape patterns add to stand-scale factors in causing insufficient hardwood regeneration. For. Ecol. Manag. 2008, 255, 1637–1646. [Google Scholar]
- Hill, J.D.; Canham, C.D.; Wood, D.M. Patterns and causes of resistance to tree invasion in rights-of-way. Ecol. Appl. 1995, 5, 459–470. [Google Scholar] [CrossRef]
- Engel, V.L.; Parrotta, J.A. An evaluation of direct seeding for reforestation of degraded lands in central São Paulo State, Brazil. For. Ecol. Manag. 2001, 152, 169–181. [Google Scholar] [CrossRef]
- Hooper, E.; Condit, R.; Legendre, P. Responses of 20 native tree species to reforestation strategies for abandoned farmland in Panama. Ecol. Appl. 2002, 12, 1626–1641. [Google Scholar] [CrossRef]
- Sampaio, A.B.; Holl, K.D.; Scariot, A. Does restoration enhance regeneration of seasonal deciduous forests in pastures in central Brazil? Restor. Ecol. 2007, 15, 462–471. [Google Scholar] [CrossRef]
- King, S.L.; Keeland, B.D. Evaluation of reforestation in the Lower Mississippi River Alluvial Valley. Restor. Ecol. 1999, 7, 348–359. [Google Scholar] [CrossRef]
- Bullard, S.; Hodges, J.D.; Johnson, R.L.; Straka, T.J. Economics of direct seeding and planting for establishing oak stands on old-field sites in the south. South. J. Appl. For. 1992, 16, 34–40. [Google Scholar]
- De Steven, D. Experiments on mechanisms of tree establishment in old-field succession: Seedling emergence. Ecology 1991, 72, 1066–1075. [Google Scholar] [CrossRef]
- Gill, D.S.; Marks, P.L. Tree and shrub seedling colonization of old fields in central New York. Ecol. Monogr. 1991, 61, 183–205. [Google Scholar] [CrossRef]
- Burton, P.J.; Bazzaz, F.A. Tree seedling emergence on interactive temperature and moisture gradients and in patches of old-field vegetation. Am. J. Bot. 1991, 78, 131–149. [Google Scholar] [CrossRef]
- Doust, S.J.; Erskine, P.D.; Lamb, D. Direct seeding to restore rainforest species: Microsite effects on the early establishment and growth of rainforest tree seedlings on degraded land in the wet tropics of Australia. For. Ecol. Manag. 2006, 234, 333–343. [Google Scholar] [CrossRef]
- Myster, R.W. Contrasting litter effects on old field tree germination and emergence. Plant Ecol. 1994, 114, 169–174. [Google Scholar]
- Gómez-Aparicio, L. The role of plant interactions in the restoration of degraded ecosystems: A meta-analysis across life-forms and ecosystems. J. Ecol. 2009, 97, 1202–1214. [Google Scholar] [CrossRef]
- De Steven, D. Experiments on mechanisms of tree establishment in old-field succession: Seedling survival and growth. Ecology 1991, 72, 1076–1088. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Canham, C.D. Effects of meadow vole population density on tree seedling survival in old fields. Ecology 1993, 74, 1792–1801. [Google Scholar] [CrossRef]
- Ostfeld, R.S.; Manson, R.H.; Canham, C.D. Effects of rodents on survival of tree seeds and seedlings invading old fields. Ecology 1997, 78, 1531–1542. [Google Scholar] [CrossRef]
- Manson, R.H.; Ostfeld, R.S.; Canham, C.D. Long-Term effects of rodent herbivores on tree invasion dynamics along forest-field edges. Ecology 2001, 82, 3320–3329. [Google Scholar]
- Herrera, C.M. Plant-vertebrate seed dispersal systems in the Mediterranean: Ecological, evolutionary, and historical determinants. Annu. Rev. Ecol. Syst. 1995, 26, 705–727. [Google Scholar]
- Kollmann, J.; Schill, H.P. Spatial patterns of dispersal, seed predation and germination during colonization of abandoned grassland by Quercus petraea and Corylus avellana. Vegetatio 1996, 125, 193–205. [Google Scholar] [CrossRef]
- Hulme, P.E.; Borelli, T. Variability in post-dispersal seed predation in deciduous woodland: Relative importance of location, seed species, burial and density. Plant Ecol. 1999, 145, 149–156. [Google Scholar] [CrossRef]
- Woods, K.; Elliott, S. Direct seeding for forest restoration on abandoned agricultural land in northern Thailand. J. Trop. For. Sci. 2004, 16, 248–259. [Google Scholar]
- Moles, A.T.; Warton, D.I.; Westoby, M. Do small-seeded species have higher survival through seed predation than large-seeded species? Ecology 2003, 84, 3148–3161. [Google Scholar]
- Environment Canada. Monthly Data Report for 2010. Montréal/Pierre Elliott Trudeau Intl A, Québec. Available online: http://www.climate.weatheroffice.gc.ca/climateData/monthlydata_ e.html?timeframe=3&Prov=XX&StationID=5415&Year=2010&Month=1&Day=1 (accessed on 22 January 2013).
- Environment Canada. Monthly Data Report for 2011. Montréal/Pierre Elliott Trudeau Intl A, Québec. Available online: http://www.climate.weatheroffice.gc.ca/climateData/monthlydata_ e.html?timeframe=3&Prov=XX&StationID=5415&Year=2011&Month=1&Day=1 (accessed on 22 January 2013).
- Institut de recherche et de développement en agroenvironnement (IRDA). Études pédologiques. Available online: http://www.irda.qc.ca/fr/Etudes-pedologiques (accessed on 12 December 2012).
- Burns, R.M.; Honkala, B.H. Silvics of North America: 1. Conifers; 2. Hardwoods; U.S. Department of Agriculture, Forest Service: Washington, DC, USA, 1990; Volume 2, p. 877.
- Centre des semences forestières de Berthier (CSFB). Quebec Ministry of Natural Resources. Personal communication, Sainte-Geneviève-de-Berthier (Québec): Canada, 2010.
- Royal Botanic Gardens Kew. Seed Information Database (SID). Version 7.1. Available online: http://data.kew.org/sid/ (accessed on 17 December 2012).
- U.S. Department of Agriculture. The PLANTS Database. Available online: http://plants.usda.gov (accessed on 11 February 2013).
- Cornelissen, J.H.C.; Lavorel, S.; Garnier, E.; Díaz, S.; Buchmann, N.; Gurvich, D.E.; Reich, P.B.; Ter Steege, H.; Morgan, H.D.; van der Heijden, M.G.A.; et al. A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust. J. Bot. 2003, 51, 335–380. [Google Scholar] [CrossRef]
- Messier, C.; Puttonen, P. Spatial and temporal variation in the light environment of developing scots pine stands: The basis for a quick and efficient method of characterizing light. Can. J. For. Res. 1995, 25, 343–354. [Google Scholar] [CrossRef]
- JMP Software. In JMP Statistical Discovery Software; Version 10.0; SAS Institute Inc.: Cary, NC, USA, 2007.
- Ray, G.J.; Brown, B.J. Restoring Caribbean dry forests: Evaluation of tree propagation techniques. Restor. Ecol. 1995, 3, 86–94. [Google Scholar] [CrossRef]
- Doust, S.J.; Erskine, P.D.; Lamb, D. Restoring rainforest species by direct seeding: Tree seedling establishment and growth performance on degraded land in the wet tropics of Australia. For. Ecol. Manag. 2008, 256, 1178–1188. [Google Scholar] [CrossRef]
- Environment Canada. Canadian Climate Normals 1971–2000. Montréal/Pierre Elliott Trudeau Intl A, Québec. Available online: http://www.climate.weatheroffice.gc.ca/climate_normals/ results_e.html?stnID=5415&lang=f&dCode=1&StationName=MONTREAL&SearchType=Contains&province=ALL&provBut=&month1=0&month2=12 (accessed on 22 January 2013).
- Jinks, R.L.; Willoughby, I.; Baker, C. Direct seeding of ash (Fraxinus excelsior L.) and sycamore (Acer pseudoplatanus L.): The effects of sowing date, pre-emergent herbicides, cultivation, and protection on seedling emergence and survival. For. Ecol. Manag. 2006, 237, 373–386. [Google Scholar]
- Willoughby, I.; Jinks, R.L.; Kerr, G.; Gosling, P.G. Factors affecting the success of direct seeding for lowland afforestation in the UK. Forestry 2004, 77, 467–482. [Google Scholar] [CrossRef]
- Mendoza, I.; Zamora, R.; Castro, J. A seeding experiment for testing tree-community recruitment under variable environments: Implications for forest regeneration and conservation in mediterranean habitats. Biol. Conserv. 2009, 142, 1491–1499. [Google Scholar] [CrossRef]
- Beckage, B.; Clark, J.S. Seedling survival and growth of three forest tree species: The role of spatial heterogeneity. Ecology 2003, 84, 1849–1861. [Google Scholar] [CrossRef]
- Tripathi, R.; Khan, M. Effects of seed weight and microsite characteristics on germination and seedling fitness in two species of Quercus in a subtropical wet hill forest. Oikos 1990, 57, 289–296. [Google Scholar] [CrossRef]
- Berkowitz, A.R.; Canham, C.D.; Kelly, V.R. Competition vs. Facilitation of tree seedling growth and survival in early successional communities. Ecology 1995, 76, 1156–1168. [Google Scholar] [CrossRef]
- Poorter, L. Growth responses of 15 rain-forest tree species to a light gradient: The relative importance of morphological and physiological traits. Funct. Ecol. 1999, 13, 396–410. [Google Scholar] [CrossRef]
- Van Hees, A.F.M. Growth and morphology of pedunculate oak (Quercus robur L.) and beech (Fagus sylvatica L.) seedlings in relation to shading and drought. Ann. For. Sci. 1997, 54, 9–18. [Google Scholar]
- Löf, M.; Thomsen, A.; Madsen, P. Sowing and transplanting of broadleaves (Fagus sylvatica L., Quercus robur L., Prunus avium L. and Crataegus monogyna Jacq.) for afforestation of farmland. For. Ecol. Manag. 2004, 188, 113–123. [Google Scholar]
- Laliberté, E.; Cogliastro, A.; Bouchard, A. Spatiotemporal patterns in seedling emergence and early growth of two oak species direct-seeded on abandoned pastureland. Ann. For. Sci. 2008, 65. [Google Scholar] [CrossRef]
- Davis, M.A.; Wrage, K.J.; Reich, P.B. Competition between tree seedlings and herbaceous vegetation: Support for a theory of resource supply and demand. J. Ecol. 1998, 86, 652–661. [Google Scholar] [CrossRef]
- Davis, M.A.; Wrage, K.J.; Reich, P.B.; Tjoelker, M.G.; Schaeffer, T.; Muermann, C. Survival, growth, and photosynthesis of tree seedlings competing with herbaceous vegetation along a water-light-nitrogen gradient. Plant Ecol. 1999, 145, 341–350. [Google Scholar] [CrossRef]
- St-Denis, A.; Messier, C.; Kneeshaw, D. Predation, competition and facilitation in a forest restoration project of abandoned fields. 2013; in preparation. [Google Scholar]
- Garcia-Orth, X.; Martinez-Ramos, M. Seed dynamics of early and late successional tree species in tropical abandoned pastures: Seed burial as a way of evading predation. Restor. Ecol. 2008, 16, 435–443. [Google Scholar] [CrossRef]
- Andersen, A.N. Effects of seed predation by ants on seedling densities at a woodland site in SE Australia. Oikos 1987, 48, 171–174. [Google Scholar] [CrossRef]
- Vander Wall, S.B.; Kuhn, K.M.; Beck, M.J. Seed removal, seed predation, and secondary dispersal. Ecology 2005, 86, 801–806. [Google Scholar] [CrossRef]
- Doust, S.J. Seed removal and predation as factors affecting seed availability of tree species in degraded habitats and restoration plantings in rainforest areas of Queensland, Australia. Restor. Ecol. 2011, 19, 617–626. [Google Scholar] [CrossRef]
- Willoughby, I.H.; Jinks, R.L.; Morgan, G.W.; Pepper, H.; Budd, J.; Mayle, B. The use of repellents to reduce predation of tree seed by wood mice (Apodemus sylvaticus L.) and grey squirrels (Sciurus carolinensis Gmelin). Eur. J. For. Res. 2011, 130, 601–611. [Google Scholar]
- Hulme, P.E. Post-dispersal seed predation: Consequences for plant demography and evolution. Perspect. Plant Ecol. 1998, 1, 32–46. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
St-Denis, A.; Messier, C.; Kneeshaw, D. Seed Size, the Only Factor Positively Affecting Direct Seeding Success in an Abandoned Field in Quebec, Canada. Forests 2013, 4, 500-516. https://doi.org/10.3390/f4020500
St-Denis A, Messier C, Kneeshaw D. Seed Size, the Only Factor Positively Affecting Direct Seeding Success in an Abandoned Field in Quebec, Canada. Forests. 2013; 4(2):500-516. https://doi.org/10.3390/f4020500
Chicago/Turabian StyleSt-Denis, Annick, Christian Messier, and Daniel Kneeshaw. 2013. "Seed Size, the Only Factor Positively Affecting Direct Seeding Success in an Abandoned Field in Quebec, Canada" Forests 4, no. 2: 500-516. https://doi.org/10.3390/f4020500




