1. Introduction
Since the 1930s, the historical woodland-savanna-glade mosaic in the Ozark Highlands of northern Arkansas and southern Missouri has been gradually replaced by large contiguous blocks of high-density oak-hickory forests. This is the result of extensive fire suppression and widespread application of other management practices that have relatively little impact on the forest overstory. As upland oak-hickory forests in the region have matured, large-scale oak decline events have increased in extent and severity. Among oaks, the red oak group species (
Quercus Section
Lobatae) mainly including scarlet oak (
Q. coccinea Muenchh.), black oak (
Q. velutina Lam.), northern red oak (
Q. rubra L.), blackjack oak (
Q. marilandica Munchh.) and southern red aok (
Q. falcate Michx.) have suffered moderate to severe “oak decline syndrome” in the upland oak forests [
1,
2,
3]. According to USDA Forest Service’s Forest Inventory and Analysis (FIA) database, approximately 98% of all scarlet oak volume, 83% of all black oak volume, and 54% all northern red oak volume found in Arkansas and Missouri occur in the Ozark Highlands. Declining oak trees first show foliage wilt and browning followed by progressive branch dieback in the middle and/or upper crown. Many trees eventually die if crown dieback continues. Oak decline and mortality in red oak species occurred throughout the 1970s [
4] and earlier [
5] and is prevalent throughout the Ozarks Highlands as forests reach physiological maturity [
6,
7]. Analysis of recent FIA data showed that decline-caused annual oak mortality had exceeded two percent, on average, by 2004, which is three to five times larger than regular (competition-induced) mortality. The observed higher mortality of red oak group species, to varying degrees, occurred in all diameter classes compared with the steeply decreasing reverse-J mortality pattern (
i.e., mortality occurred predominately within small-diameter trees) of white oak group species (e.g.,
Quercus alba L.) [
3]. A 14-year study conducted in southern Missouri found that improvement harvests did not mitigate oak decline [
8]. High red oak group mortality and the unprecedented abundance of oak borer populations associated with oak decline pose significant threats to the health and management of forests in the Ozark Highlands.
Oak decline occurs over an extended temporal scale and is associated with forest succession [
9]. No single cause is responsible for the oak decline syndrome and escalating mortality. Rather, oak decline is an on-going process, resulting from the interaction of multiple factors over a set of temporal and spatial scales [
4,
10]. It has been hypothesized that current oak decline and mortality in the Ozark highlands is a result of poor site quality, high stand density and/or advanced tree age (collectively called predisposing factors); triggered by environmental stresses including periodic droughts, frost, and temperature extremes (e.g., inciting factors); and exacerbated by secondary disease and insect pests (e.g., root fungi, insect borers,
etc., known as contributing factors) [
2,
4,
6,
10,
11,
12].
Whether a normal tree becomes a declining tree or how soon it will die of or recover from decline depends not only on its physiological condition or vigor but also on the duration and intensity of inciting factors like drought and secondary insects/diseases and varies by individual species. For instance, white oaks exhibiting moderate to severe crown dieback usually are less likely to recover compared to red oaks in a 14-year monitoring study in the Missouri Ozarks [
8]. Some argue that the attack of insects like red oak borers may degrade the quality of oak trees due to the tunnels they excavated but not cause tree mortality directly [
13], and improvement harvests conducted in declining oak stands may have no effect on the decline process. A mechanistic study of oak decline and mortality is necessary to explore the relationship between risk factors and symptoms and to guide forest management to restore forest health or vigor.
Oak decline and mortality are inherently multi-scale issues, because the predisposing factors, inciting factors and contributing factors may interact across a variety of spatial scales. From a forest management perspective, oak decline and mortality studies mainly focus on three interrelated spatial scales: individual tree, stand, and forest landscape/region. At the individual tree level, foresters need to identify primary risk (predisposing) factors associated with tree decline and quantify their influence on the probability that a tree will decline and/or die [
2,
10]. This information will allow resource managers to preemptively remove at-risk trees during intermediate harvest treatments. At the stand level, knowledge of which stands are most susceptible to oak decline will allow at-risk stands to be ranked and prioritized for treatment [
14]. At the landscape level, identification of the potential extent and condition of oak decline and mortality—particularly locating future oak decline hot spots—is a top priority for resource assessment and forest planning [
15,
16].
The major objective of this study is to assess the spatial and temporal trends of oak decline and mortality in the Ozark Highlands by using USDA Forest Service FIA data. Specifically, we intended to (1) map the extent and condition of oak decline and mortality, in terms of relative standing basal area and/or stem density of dead trees by species group and inventory time; and (2) correlate dead basal area and stem density with the Palmer drought severity index (PDSI) to evaluate the effect of regional drought events on oak decline and mortality. This information will be useful to forest planning and management aimed at mitigating oak decline and mortality.
2. Methods
Starting from 1999, the USDA Forest Service’s FIA program adopted an annual inventory plan with about one-fifth of the forest inventory plots in each survey unit remeasured annually on a rotating basis to evaluate the temporal trend of forest change in addition to spatial condition and extent of forest resources [
17]. The data for this study included 6,997 FIA plots in the Ozark Highlands of Arkansas and Missouri that contained oak species and were measured and remeasured from 1999 through 2010 (
Table 1). On each 0.4-ha FIA plots, trees > 11 cm dbh (diameter at breast height) were measured. Tree dbh, species, crown ratio, damage class, plot slope, aspect and elevation were measured or recorded.
Table 1.
Number of plots within each species group used in analysis.
Table 1.
Number of plots within each species group used in analysis.
| Year | Species group |
|---|
| Red oak | White oak | Other |
|---|
| 1999 | 231 | 265 | 292 |
| 2000 | 324 | 403 | 442 |
| 2001 | 419 | 496 | 541 |
| 2002 | 589 | 674 | 729 |
| 2003 | 595 | 716 | 782 |
| 2004 | 463 | 568 | 639 |
| 2005 | 445 | 550 | 611 |
| 2006 | 483 | 590 | 654 |
| 2007 | 446 | 553 | 631 |
| 2008 | 366 | 487 | 549 |
| 2009 | 418 | 533 | 595 |
| 2010 | 361 | 461 | 532 |
Each FIA plot was spatially referenced by the latitude and longitude of the plot center. Within each plot, trees were classified into one of three species groups: white oak group, red oak group and non-oak group. Both the white oak group and non-oak species had no or very low levels of decline and served as the reference for the decline-prone red oak group. We calculated percentage of dead trees by number and basal area for each of the three species groups as an indirect measure of oak decline and mortality. Semivariogram analyses of mortality showed a weak trend of spatial continuity. This is either because of the mixed effect of risk factors across spatial scales or the huge sampling variation due to the small plot size (0.4 ha) and large distances between plots. Thus, kernel smoothing was used to map the regional patterns of oak decline and mortality in terms of percent of basal area or tree number of standing dead trees in the Ozarks. As described in Wand and Jones [
18], kernel smoothing (or kernel-based estimators) is a nonparametric “weighted moving averages via the kernel” method to estimate the true density (probability) of a random variable. The standardized isotropic two-dimension Gaussian kernel density estimator at a location of
x is
where
K is the kernel function,
e is the base of natural logarithm, and
h is the bandwidth (smoothing parameter, here, it refers to the standard deviation of Gaussian distribution) with
n = 2 for the two-dimension density smoothing and based on a random sample
xi (relative mortality) of a specific size. Exploratory data analysis indicated that the kernel density smoothing method was more appropriate than kriging to map the spatial trend of the relative mortality. Details of kernel smoothing methods were recorded in Fan
et al. [
19].
It has been observed that oak decline and escalating mortality were preceded by periodic droughts in the Ozarks (
Figure 1 and
Figure 2). To detect the temporal trend of oak decline and mortality and its relationship with drought events, the average relative mortality of red oak group species was calculated for each climate division (Missouri climate division 4 and 5, Arkansas climate division 1 and 2) by inventory year from 1999 to 2010. PDSI was used to measure drought status for each climate division. The PDSI measures the departure of the moisture supply based on precipitation and temperature data, as well as the local available water content (AWC) of the soil, and ranges from −4 (extreme drought) to 4 (extremely wet) [
20]. The monthly PDSIs for each of the four climate divisions in the Ozarks were downloaded from the National Climatic Data Center. Exploratory analysis showed that droughts in growing seasons (from May to October) are especially critical to oak decline and mortality. The drought severity in a single growing season from 1990 to 2010 is represented by the cumulative monthly PDSI (cmPDSI) which is the sum of PDSI’s from May to October. Considering temporal variation of drought events in duration (from several months to several years) and magnitude in the Ozarks, the cumulative yearly PDSI (cyPDSI) for the previous
n (
n = 0, 1, 2, …) years was calculated as the sum of cmPDSIs.
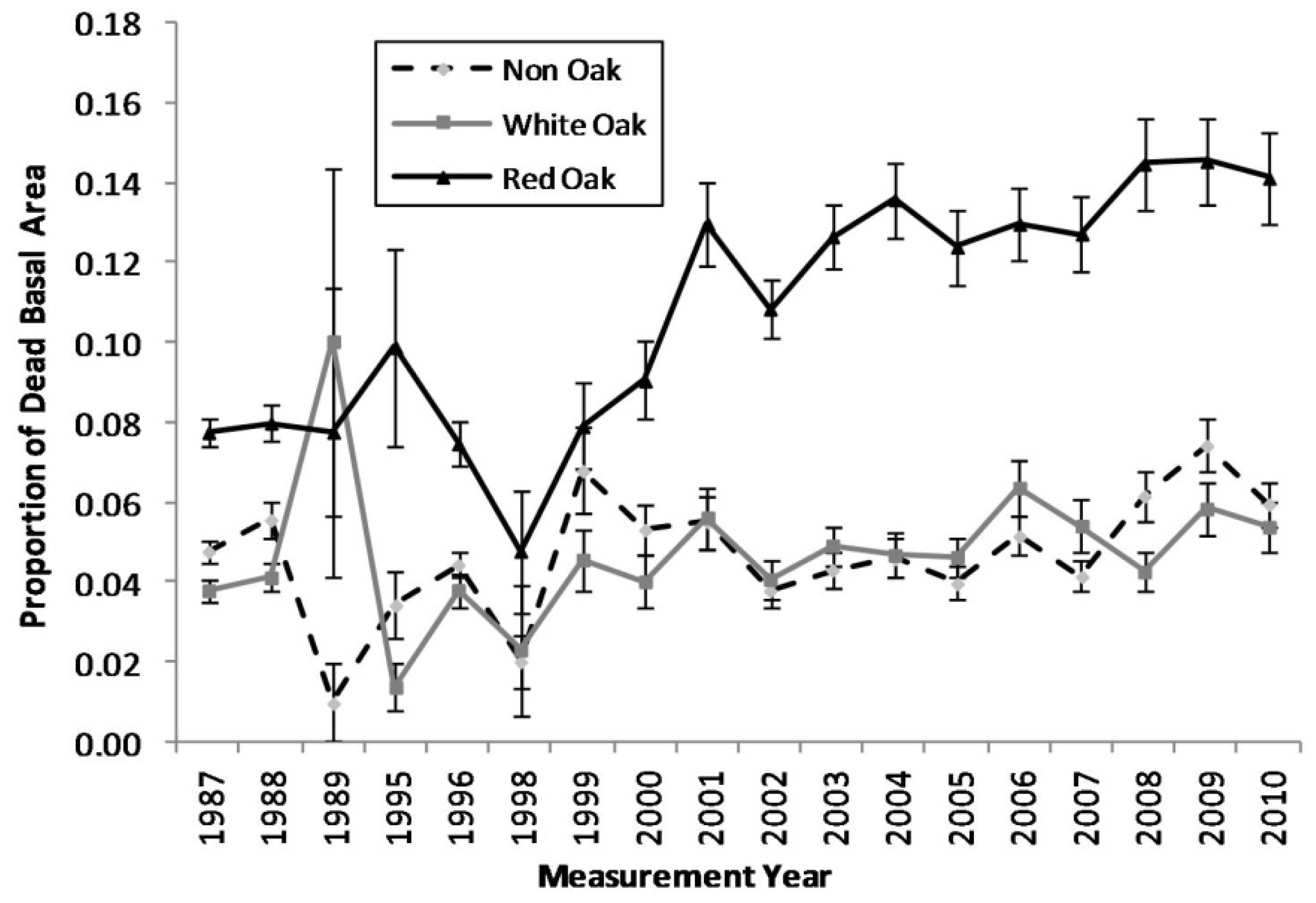
Figure 1.
Proportion of dead basal area for each measurement year for red oak, white oak, and non oak species (The vertical bars represent the standard error of dead basal area for each species group).
Figure 1.
Proportion of dead basal area for each measurement year for red oak, white oak, and non oak species (The vertical bars represent the standard error of dead basal area for each species group).
Figure 2.
Growing season, non-growing season, and annual average Palmer drought severity index (PDSI) values, 1987–2010.
Figure 2.
Growing season, non-growing season, and annual average Palmer drought severity index (PDSI) values, 1987–2010.
The relative mortality of declining red oak species in each climate division from 1999 to 2010 was transformed using arc sin square root function and then was cross correlated with corresponding cmPDSIs and cyPDSIs. The cross correlation coefficient r(d) at delay d (0 ≤ d < N years) is defined as
where series x(i) and y(i) (i = 0, 1, 2, ..., N − 1) represent the relative mortality and PDSI series, respectively.
3. Results
Comparing the mortality trends between the red oak species, white oak species, and non-oak species groups, an escalating trend in mortality in the red oak species group becomes apparent (
Figure 1). The proportions of standing dead trees in the white oak and non-oak species fluctuates between approximately 1%–7%, while the red oak species increases from approximately 8% in the late 1990s to near 15% by 2010. Cross-correlation analyses indicated that red oak mortality was significantly correlated with growing season PDSI with a general lag of two to three years following a single drought (
Table 2,
Figure 2). Moreover, the cumulative impact of drought could potentially last up to 10 years based on the cross correlation coefficients (
Table 2). The region-wide drought events in the Ozark Highlands during the 1998–2001 and 2005–2007 periods triggered an escalation in mortality in red oak species.
Table 2.
Cross correlation coefficients between mortality and single and cumulative growing season PDSI.
Table 2.
Cross correlation coefficients between mortality and single and cumulative growing season PDSI.
| Delay (d) year | Single year | Cumulative years |
|---|
| r | p | r | p |
|---|
| 0 | –0.0256 | 0.8752 | 0.1624 | 0.3912 |
| 1 | –0.1446 | 0.3733 | –0.1666 | 0.3790 |
| 2 | –0.3928 | 0.0121 * | –0.2980 | 0.1097 |
| 3 | –0.2971 | 0.0626 | –0.3247 | 0.0799 |
| 4 | –0.2508 | 0.1186 | –0.3950 | 0.0303 * |
| 5 | –0.3927 | 0.0122 * | –0.4214 | 0.0203 * |
| 6 | –0.1830 | 0.2583 | –0.4394 | 0.0151 * |
| 7 | –0.1026 | 0.5286 | –0.4058 | 0.0260 * |
| 8 | 0.0233 | 0.8865 | –0.4025 | 0.0274 * |
| 9 | 0.0002 | 0.9882 | –0.3146 | 0.0904 |
| 10 | 0.2237 | 0.1652 | –0.1384 | 0.4656 |
| 11 | | | –0.1819 | 0.3361 |
| 12 | | | –0.2017 | 0.2851 |
| 13 | | | –0.2557 | 0.1726 |
| 14 | | | –0.2309 | 0.2196 |
| 15 | | | –0.2286 | 0.2244 |
| 16 | | | –0.2703 | 0.1485 |
The results of our spatial analysis (
Figure 3,
Figure 4,
Figure 5), via kernel smoothing, align well with the results of the temporal analysis (
Figure 1). For the 1999–2004 time period (
Figure 3A), high mortality rates (greater than 9%) were widespread in the red oak species and appeared to lesser degrees for white oak and non-oak species. Plots with high mortality in the red oak group were widespread across the study area, but plots with high mortality levels for white oak and non-oak species were less common and more widely dispersed across the landscape. Approximately 72% of forest areas had greater than 9% mortality for red oak species while this number is 6% and 3% for white oak and non-oak groups, respectively (
Figure 3B). For the 2005–2010 time period (
Figure 4A), higher mortality rates (greater than 9%) were more common for all species groups. The areas with greater than 9% mortality increased for all three species groups and reached up to 80%, 15% and 17%, respectively for the red oak group, white oak group and non-oak group species (
Figure 4B).
A closer inspection of mortality for the red oak species group shows the changing mortality patterns in response to drought events (
Figure 5A). The pattern during the period of the first drought event shows that just over 20% of the area had mortality rates higher than 12%, occurring primarily in clusters throughout the central portion of study area. This increased to approximately 46%, 55% and 56% of the area having greater than 12% mortality during years 2001–2003 (within 3 years following the first severe drought period), 2004–2007 (the second, milder drought period), and 2008–2010 (within 3 years following the second drought period), respectively (
Figure 5B).
Figure 3.
(A) Spatial trend, by mortality group for the Ozark Highlands and (B) area in each mortality group by species group, 1999–2005.
Figure 3.
(A) Spatial trend, by mortality group for the Ozark Highlands and (B) area in each mortality group by species group, 1999–2005.
Figure 4.
(A) Spatial trend, by mortality group for the Ozark Highlands and (B) area in each mortality group by species group, 2006–2010.
Figure 4.
(A) Spatial trend, by mortality group for the Ozark Highlands and (B) area in each mortality group by species group, 2006–2010.
Figure 5.
(A) Spatial trend for the red oak species group in the Ozark Highlands and (B) area in each mortality group by time period.
Figure 5.
(A) Spatial trend for the red oak species group in the Ozark Highlands and (B) area in each mortality group by time period.
4. Discussion
In the Ozark Highlands, episodes of oak decline and mortality have occurred sporadically since 1960s [
1,
2,
3,
5,
9,
21]. As millions of acres of drought-prone oak forests are maturing physiologically, oak decline and mortality, particularly in red oak species, has reached unprecedented levels [
22]. A mechanistic interpretation of oak decline and mortality in the Ozark Highlands is the three-step chain reaction theory of predisposing, inciting, and contributing factors [
23,
24]. Advanced age, species, dbh, crown class, basal area in larger trees (a measure of competition from above), and site index have been identified as major predisposing factors which predispose oak trees to decline and thereby classify oak trees into different risk (declining) groups [
2,
10,
12,
25]. Widespread red oak borer populations in the Ozark highlands, particularly in Arkansas, were found to be an important contributing factor which is significantly related to oak decline (crown dieback) [
3,
26]. However, the relationship between red oak borer infestation and mortality suggests that other factors such as severe ice storms and
Armillaria root rot may also be involved in the process of oak decline [
27].
Region-wide drought is believed to be one of the most important inciting factors that plague the Ozark Highlands periodically and may trigger and exacerbate oak decline [
2,
6,
28,
29]. To study the relationship between oak mortality and drought, we used relative mortality which was aggregated by climate division and correlated with PDSI to filter out certain non-climatic factors such as species composition, stand age, oak basal area and site condition. As shown by Kabrick
et al. [
13,
21], red oak mortality measured in terms of dead tree basal area or stem density may differ significantly by ecological land type, slope or aspect, but relative mortality calculated as a percentage of the stem density or basal area of dead oak trees to all oak trees does not. On many sites, large accumulations of dead oak trees on south-facing slopes and ridge tops is the consequence of the prevalence of oaks on those sites; sites that naturally have fewer oaks may have a comparably high oak mortality rate but less visual impact [
14]. The cross correlation between relative mortality and historical PDSI confirms the role of regional droughts in oak decline and mortality (
Table 2). The Ozark Highlands have suffered through drought periods from 1998–2001 and from 2005–2007. These drought events can be discerned from the region-averaged PDSI data (
Figure 2). The region-wide drought events in the Ozark Highlands during the 1998–2001 and 2005–2007 periods triggered an escalation in relative mortality in red oak species. This relationship between drought and red oak species is in agreement with previous findings in the region using dendrochronology [
30]. Of particular interest are the peaks in mortality that occur in 2001, 2004, and 2008. These peaks seem to follow, after a lag of 2–3 years, drought events that impact the region [
23].
To further explore the mortality trends for each species group, mortality was analyzed in two consecutive inventory cycles (periods), 1999–2005 and 2006–2010. While each period was impacted by a drought event, the 2006–2010 time period could show the cumulative impact of two drought periods. For the 1999–2005 time period (
Figure 3A), high mortality rates (greater than 9%) were widespread in the red oak species and appeared to lesser degrees for white oak and non-oak species. Approximately 72% of forest areas had greater than 9% mortality for red oak species while this number is 6% and 3% for white oak and non-oak groups, respectively (
Figure 3B). For the 2006–2010 time period, high mortality rates (> 9%) were more widespread for all species groups (
Figure 4A). The areas with greater than 9% mortality increased for all three species groups, with red oak species increasing from 72% in the 1999–2005 period to over 80% in the 2006–2010 period, white oaks from 6% to 15%, and non-oaks from 3% to 17% between these two time periods (
Figure 4B). The differences in mortality rates between the red oaks and white and non-oak species aligns with the finding of Fan
et al. [
14] who found mortality rates in red oak species three to five times higher than other species groups.
Significant increases in mortality in red oak species during the two inventory periods led us to further explore the potential link between drought and mortality in the region. We further divided the two inventory periods into four alternate periods based on the changing pattern of PDSI: 1998–2000 (first drought period), 2001–2003 (a 3-year period following the first drought period), 2004–2007 (second drought period), and 2008–2010 (a 3-year period following the second drought period). During the first drought period (1998–2000), the five mortality classes distributed approximately evenly. Following the first drought period, there was a significant transition among mortality classes with more of the area experiencing high mortality rates (
i.e., <9%) (
Figure 5B). Including the two highest mortality classes, the period from 2001–2003 had the most area with mortality rates greater than 9% compared to the other two subsequent analysis periods, reflecting the lag-effect of the severe drought during 1998–2001 (
Figure 5B). The highest rates (>12%) of mortality exhibited a further increase in area accompanied by a corresponding decrease in the area in the 9%–12% mortality class during the 2004–2007 time period, particularly across northern sections of the study area (
Figure 5A). The drought period from 2005–2007 could have resulted in greater cumulative stress in trees still recovering from the earlier drought event that ended in 2001. Further, there would certainly be a mortality signal by 2007 based on the temporal lag between drought and mortality already established (
Figure 3) The final time period of study was between 2008–2010 and had only a slightly larger area that experienced more than 12% mortality than did the 2004–2007 period. The area with the most extreme levels of mortality increased from 56.7% during the years 2004–2007 to 57.1% from 2008–2010 (
Figure 5B). The continual increase in the most extreme mortality class (
i.e., greater than 12%) could indicate a cumulative effect on areas that were impacted by the second drought event from 2005–2007.
The significant correlation coefficients between relative mortality and PDSI vary between −0.3 and −0.5 (
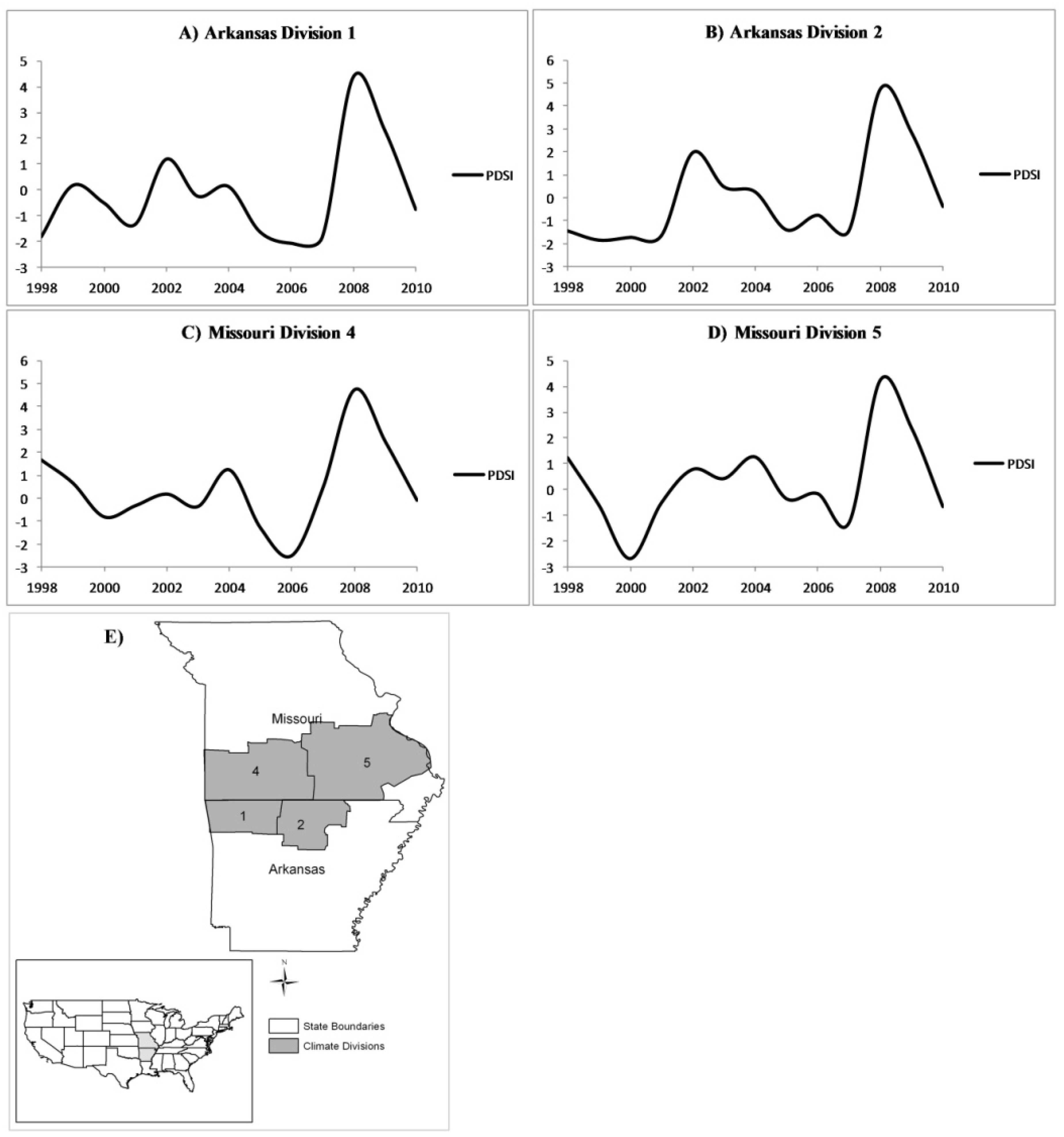
Table 2). One possible cause of low correlation coefficients may be from the difference in forest condition among the four climatic divisions. For example, a larger proportion of forested area belonging to the white oak species group compared to the red oak species group could weaken the correlation coefficient between PDSI and mortality. Also, and perhaps more importantly, the severity and duration of drought varied by climate division (
Figure 6). This could prevent a strong correlation from presenting in the year-to-year analysis but produce a stronger signal in the cumulative coefficients. Forest type differences could also play a role in the variation of mortality relating to drought. The oak-hickory, oak-pine, and loblolly-shortleaf pine forest types dominate the landscape in the Ozark Highlands with varying levels of annual mortality [
27]. Mortality in this study was not considered by forest type; rather, plots were aggregated by climate division. A more detailed, multi-level (hierarchical), assessment may allow for mortality estimates to be further refined and allow managers to assess risk at a variety of scales (e.g., species, forest type, or landscape level). Further, changes at the forest type level could allow for the consideration of additional disturbance events (e.g., red oak borer, extreme weather) or climatic variables (e.g., maximum temperature). Susceptibility to enhanced mortality via additional disturbance events acting in concert with ongoing oak decline, would allow forest managers to adapt management plans to reduce the risk of fuel loading for catastrophic wildfires.
Figure 6.
Annual growing-season PDSI for the years 1998–2010 for (A) Arkansas climate division 1;(B) Arkansas climate division 2;(C) Missouri climate division 4; and (D) Missouri climate division 5. A map depicting the locations of the four climate regions discussed is included for reference (E).
Figure 6.
Annual growing-season PDSI for the years 1998–2010 for (A) Arkansas climate division 1;(B) Arkansas climate division 2;(C) Missouri climate division 4; and (D) Missouri climate division 5. A map depicting the locations of the four climate regions discussed is included for reference (E).
Spatially, the southern portion of the study area had the greatest increase in mortality for white oak and non-oak species, which could indicate that drought was more severe in this region (
Figure 6) or that there are other, localized variables also impacting mortality in these species groups (
Figure 4A). Arkansas experienced a more severe drought, which could explain the higher levels of mortality in those areas. Also, the area was impacted by an ice storm in 2009 which could, with the preceding drought serving as a catalyst, lead to increased mortality in white oak and non-oak species. The Arkansas sites experienced severe red oak borer attack following the first drought period [
26] which could exacerbate ongoing mortality due to drought in the red oak species group leading into the second drought period.
The areal coverage for mortality in red oak species increased across all areas of the Ozark Highlands. The years 2006–2010 show the cumulative effect of two drought periods occurring within a five-year time frame. The species impacted by the 1998–2001 drought event may not have had sufficient time to recover before the onset of further drought in the region, which increased the cumulative effect on red oaks found there. The greatest increase in mortality occurs across southern portions of the Ozark Highlands (
Figure 3,
Figure 4,
Figure 5) for all species where, perhaps, localized conditions exacerbated mortality in all species groups.
Much of this area is dominated by species in the red oak group, particularly black oak (
Quercus velutina Lam.) and scarlet oak (
Quercus coccinea Muenchh.) [
31,
32], which have a known susceptibility to drought induced mortality [
14]. The areas exhibiting the highest levels of mortality also become much more widespread as compared to the years 1998–2000 (
Figure 5B). Such an increase in mortality has been previously linked to extended periods of drought in the region [
6]. The south to north trend in the four time periods (
Figure 5A) could be explained by the more severe drought that occurred there during the first drought period (
Figure 6). Further, red oak borer infestation between the two drought periods could increase the likelihood of mortality [
33].
While the highest mortality rates increased in area in subsequent years, it could be that oak regeneration and regrowth began taking place after the initial drought event ending in 2001. This could account for the increased area experiencing lower mortality rate (0–6%), particularly in northeastern sections of the study area (
Figure 5A) after 2003 (
i.e., the years 2004–2007 and 2008–2010), as younger oak trees are less susceptible to drought compared to older trees [
16].
Multiple drought events, when combined with additional stressors such as red oak borer infestation and extreme weather events, leads to increased tree mortality [
27]. Oak decline and mortality ultimately leads to a build-up of fuels which could lead to catastrophic wildfire. Wildlife habitat could also be impacted as many species depend on mature forests for nesting and/or sustenance [
34]. Carbon sequestration potential is also severely impacted by disturbance [
35], particularly when large, mature oaks are not salvaged leading to gradual emissions of carbon dioxide from dead and dying trees. Future studies could seek to assess the impact of oak decline on the aforementioned areas. Also, a hierarchical study of mortality relating to climatic extremes and disturbance events could provide a clearer picture to forest managers regarding the potential impacts to forests at multiple scales and assist them in refining management strategies and goals.
5. Conclusions
Oak mortality, particularly in red oak species, has reached unprecedented levels in the Ozark Highlands. This has been linked to region wide drought events occurring from 1998–2001 and from 2005–2007. Mortality in red oak species has risen dramatically, from 6–8% in the late 1990s to current levels of approximately 15%. While some factors (e.g., age, high stocking levels, etc.), predispose red oak species to mortality, the region wide drought events have played a significant role in exacerbating the level of mortality. Given that drought is an inciting factor for increased mortality, forest resource managers should plan forest management activities to mitigate the impacts of drought on oak mortality. Forest managers could naturally or artificially regenerate more drought resistant tree species, or in the case of mortality prone red oak species manage for a greater diversity of forest age and density because older and denser stands have been shown to have increased mortality rates.
There is a temporal lag between the onset of a drought and the increased mortality rates seen in the forest. During or immediately after future severe drought events, forest managers could elect to preemptively harvest trees that are more susceptible to mortality to avoid losses or increased forest damage from pests (e.g., red oak borer) or catastrophic wildfires. Mortality rates increased further in the Ozark Highlands after the onset of a second drought event in 2005. It could be that those trees that were impacted by the first drought event did not have sufficient time to recover and were lost as a result of the second drought period. As climatic fluctuations continue across the world, the result could be an increased frequency in drought across the Ozark Highlands. Additional research could attempt to incorporate previous periods of drought into a model to predict the likelihood of mortality associated with sequential drought events.
In the Ozark Highlands, drought has been shown as an inciting factor leading to increased levels of mortality. Future research could attempt to test other climate variables that could occur coincident with drought (e.g., elevated temperatures). It would also be useful to natural resource managers to know if this trend of increasing mortality following drought events holds across larger, contiguous areas of the United States. Periods of prolonged drought could have serious consequences for the biological diversity of forested areas beyond the Ozark Highlands. Continual monitoring of drought indicators (i.e., PDSI) could provide a means of mitigating some of the losses associated with increased levels of mortality.