A Theoretical Study of the Binding of [Re6Se8(OH)2(H2O)4] Rhenium Clusters to DNA Purine Base Guanine
Abstract
:1. Introduction
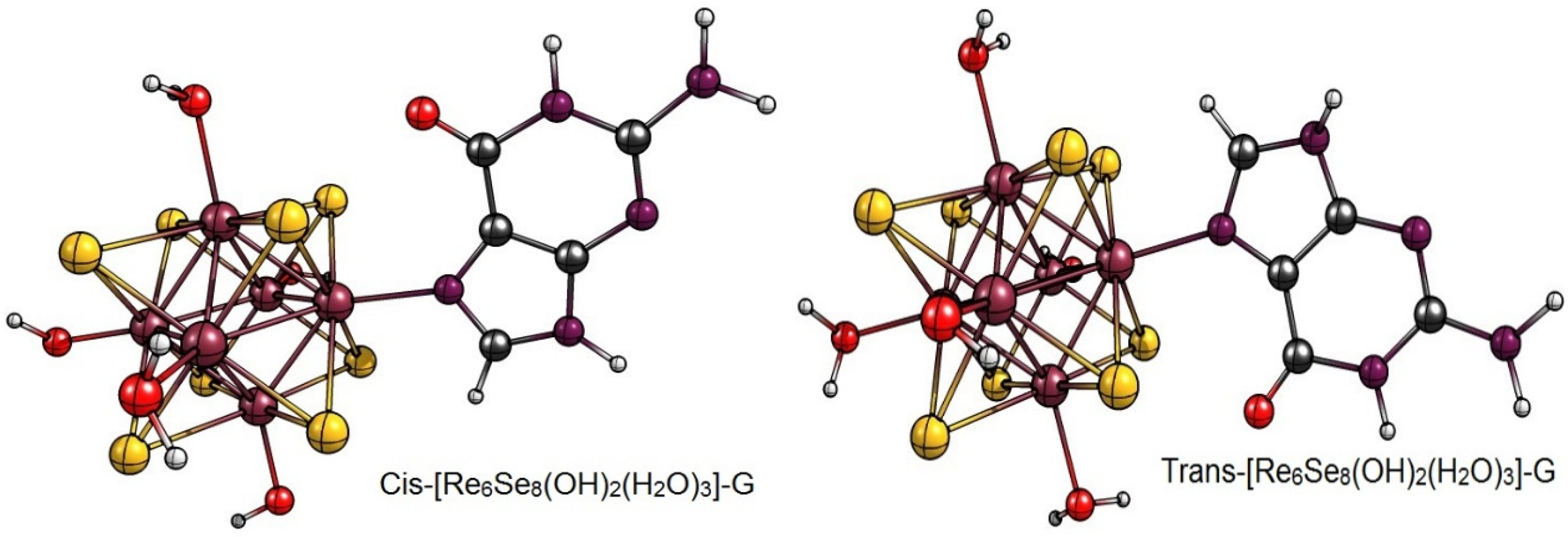
2. Results and Discussion
| Complexes | Binding Energy |
|---|---|
| Cis-[Re6Se8(OH)2(H2O)3]-G | −869.25 kcal/mol |
| Trans-[Re6Se8(OH)2(H2O)3]-G | −332.03 kcal/mol |
| Cis-[Pt(NH3)2(H2O)]-G2+ | −315.17 kcal/mol |
| Cis-[Re6Se8(OH)2(H2O)3] | Trans-[Re6Se8(OH)2(H2O)3] | Cis-[Re6Se8(OH)2(H2O)3]-G | Trans-[Re6Se8(OH)2(H2O)3]-G | Guanine |
|---|---|---|---|---|
 HOMO -1 |  HOMO -1 |  HOMO -1 |  HOMO-1 |  HOMO-1 |
 HOMO |  HOMO |  HOMO |  HOMO |  HOMO |
 LUMO |  LUMO |  LUMO |  LUMO |  LUMO |
 LUMO +1 |  LUMO +1 |  LUMO +1 |  LUMO +1 |  LUMO +1 |
| Cis-[Re6Se8(OH)2(H2O)3] |  |
| Trans-[Re6Se8(OH)2(H2O)3] |  |
| Cis-[Re6Se8(OH)2(H2O)3]-G |  |
| Trans-[Re6Se8(OH)2(H2O)3]-G |  |
| Guanine |  |
3. Experimental Section
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kirakci, K.; Kubát, P.; Dušek, M.; Fejfarová, K.; Šícha, V.; Mosinger, J.; Lang, K. A highly luminescent hexanuclear molybdenum cluster—A promising candidate toward photoactive materials. Eur. J. Inorg. Chem. 2012, 2012, 3107–3111. [Google Scholar] [CrossRef]
- Alvarado-Soto, L. Electronic structure and molecular properties of [Re6−xOsxSe8Cl6](4−x)−(x = 0–3) clusters: A study based on time-dependent density functional theory including spin-orbit and solvent effects. J. Struct. Chem. 2014, 55, 363–367. [Google Scholar] [CrossRef]
- Durham, J.L.; Tirado, J.N.; Knott, S.A.; Oh, M.K.; McDonald, R.; Szczepura, L.F. Preparation of a family of hexanuclear rhenium cluster complexes containing 5-(phenyl)tetrazol-2-yl ligands and alkylation of 5-substituted tetrazolate ligands. Inorg. Chem. 2012, 51, 7825–7836. [Google Scholar] [CrossRef] [PubMed]
- Szczepura, L.F.; Ketcham, K.A.; Ooro, B.A.; Edwards, J.A.; Templeton, J.N.; Cedeño, D.L.; Jircitano, A.J. Synthesis and study of hexanuclear molybdenum clusters containing thiolate ligands. Inorg. Chem. 2008, 47, 7271–7278. [Google Scholar] [CrossRef] [PubMed]
- Brylev, K.A.; Mironov, Y.V.; Fedorov, V.E.; Kim, S.-J.; Pietzsch, H.-J.; Stephan, H.; Ito, A.; Kitamura, N. A new hexanuclear rhenium cluster complex with six terminal acetate ligands: Synthesis, structure, and properties of K4[Re6S8(CH3COO)6]·8H2O. Inorg. Chim. Acta 2010, 363, 2686–2691. [Google Scholar] [CrossRef]
- Arratia-pérez, R.; Hernández-Acevedo, L. The hexanuclear rhenium cluster ions Re6S8X64-(X = Cl, Br, I): Are these clusters luminescent? J. Chem. Phys. 1999, 110, 2529–2532. [Google Scholar] [CrossRef]
- Kirakci, K.; Kubát, P.; Langmaier, J.; Polívka, T.; Fuciman, M.; Fejfarová, K.; Lang, K. A comparative study of the redox and excited state properties of (nBu4N)2[Mo6X14] and (nBu4N)2[Mo6X8(CF3COO)6] (X = Cl, Br, or I). Dalton Trans. 2013, 42, 7224–7232. [Google Scholar] [CrossRef] [PubMed]
- Cordier, S.; Dorson, F.; Grasset, F.; Molard, Y.; Fabre, B.; Haneda, H.; Sasaki, T.; Mortier, M.; Ababou-Girard, S.; Perrin, C. Novel Nanomaterials Based on Inorganic Molybdenum Octahedral Clusters. J. Clust. Sci. 2008, 20, 9–21. [Google Scholar] [CrossRef]
- Gabriel, J.C.; Boubekeur, K.; Uriel, S.; Batail, P. Chemistry of hexanuclear rhenium chalcohalide clusters. Chem. Rev. 2001, 101, 2037–2066. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-J.; Brylev, K.A.; Xu, J.-Z.; Mironov, Y.V.; Fedorov, V.E.; Sohn, Y.S.; Kim, S.-J.; Choy, J.-H. Cellular uptake and cytotoxicity of octahedral rhenium cluster complexes. J. Inorg. Biochem. 2008, 102, 1991–1996. [Google Scholar] [CrossRef] [PubMed]
- Brylev, K.; Shestopalov, M. Biodistribution of Rhenium Cluster Complex K4 in the Body of Laboratory Rats. Bull. Exp. 2013, 155, 741–744. [Google Scholar] [CrossRef]
- Echeverría, C.; Becerra, A.; Nuñez-Villena, F.; Muñoz-Castro, A.; Stehberg, J.; Zheng, Z.; Arratia-Perez, R.; Simon, F.; Ramírez-Tagle, R. The paramagnetic and luminescent [Re6Se8I6]3- cluster. Its potential use as an antitumoral and biomarker agent. New J. Chem. 2012, 36, 927–932. [Google Scholar] [CrossRef]
- Knope, K.E.; Wilson, R.E.; Vasiliu, M.; Dixon, D.A.; Soderholm, L. Thorium (IV) molecular clusters with a hexanuclear Th core. Inorg. Chem. 2011, 50, 9696–9704. [Google Scholar] [CrossRef] [PubMed]
- Shestopalov, M.A.; Zubareva, K.E.; Khripko, O.P.; Khripko, Y.I.; Solovieva, A.O.; Kuratieva, N.V.; Mironov, Y.V.; Kitamura, N.; Fedorov, V.E.; Brylev, K.A. The first water-soluble hexarhenium cluster complexes with a heterocyclic ligand environment: Synthesis, luminescence, and biological properties. Inorg. Chem. 2014, 53, 9006–9013. [Google Scholar] [CrossRef] [PubMed]
- Krasilnikova, A.A.; Shestopalov, M.A.; Brylev, K.A.; Kirilova, I.A.; Khripko, O.P.; Zubareva, K.E.; Khripko, Y.I.; Podorognaya, V.T.; Shestopalova, L.V; Fedorov, V.E.; et al. Prospects of molybdenum and rhenium octahedral cluster complexes as X-ray contrast agents. J. Inorg. Biochem. 2015, 144, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-K.; Sadler, P.J. Metal complexes as DNA intercalators. Acc. Chem. Res. 2011, 44, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Ljungman, M. Targeting the DNA damage response in cancer. Chem. Rev. 2009, 109, 2929–2950. [Google Scholar] [CrossRef] [PubMed]
- Baik, M.; Friesner, R.A.; Lippard, S.J. Theoretical Study of Cisplatin Binding to Purine Bases : Why Does Cisplatin Prefer Guanine over Adenine? J. Am. Chem. Soc. 2003, 125, 14082–14092. [Google Scholar] [CrossRef] [PubMed]
- Chiavarino, B.; Crestoni, M.E.; Fornarini, S.; Scuderi, D.; Salpin, J.-Y. Interaction of cisplatin with adenine and guanine: A combined IRMPD, MS/MS, and theoretical study. J. Am. Chem. Soc. 2013, 135, 1445–1455. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Ruiz, S. New directions in the fight against cancer: From metal complexes to nanostructured materials. In Molecules at Work: Selfassembly, Nanomaterials, Molecular Machinery; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Bruijnincx, P.C.A.; Sadler, P.J. New trends for metal complexes with anticancer activity. Curr. Opin. Chem. Biol. 2008, 12, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Alvarado-Soto, L.; Ramirez-Tagle, R. Tryptophan-[Re6Se8I6]3-Cluster Interaction: A Computational Study. J. Clust. Sci. 2015, 26, 623–630. [Google Scholar] [CrossRef]
- Zarate, X.; Schott, E.; Alvarado-Soto, L.; Ramirez-Tagle, R. A family of octahedral molybdenum cluster complexes [Mo6Cl8(H2O)n(OH)6−n]n−2 with n = 0–6 as a pH-sensors: A theoretical study. Chem. Phys. Lett. 2013, 567, 39–42. [Google Scholar] [CrossRef]
- Brylev, K.A.; Mironov, Y.V.; Yarovoi, S.S.; Naumov, N.G.; Fedorov, V.E.; Kim, S.; Kitamura, N.; Kuwahara, Y.; Yamada, K.; Ishizaka, S.; et al. A family of octahedral rhenium cluster complexes [Re6Q8(H2O)n(OH)6−n]n−4 (Q = S, Se; n = 0−6): Structural and pH—Dependent spectroscopic studies. Inorg. Chem. 2007, 46, 7414–7422. [Google Scholar] [CrossRef] [PubMed]
- Van Lenthe, E. Relativistic total energy using regular approximations. J. Chem. Phys. 1994, 101, 9783–9792. [Google Scholar] [CrossRef]
- Perdew, J.; Chevary, J.; Chevary, J.A.; Perdew, J.P.; Vosko, S.; Jackson, K.; Singh, D.J.; Vosko, S.H.; Pederson, M.; Pederson, M.R.; et al. Atoms, molecules, solids, and surfaces: Applications of the generalized gradient approximation for exchange and correlation. Phys. Rev. B 1992, 46, 6671–6687. [Google Scholar] [CrossRef]
- Pye, C.C.; Ziegler, T.; van Lenthe, E.; Louwen, J.N. An implementation of the conductor-like screening model of solvation within the Amsterdam density functional package—Part II. COSMO for real solvents 1. Can. J. Chem. 2009, 87, 790–797. [Google Scholar] [CrossRef]
- Te velde, G.; Bickelhaupt, F.M.; Baerends, E.J.; Fonseca Guerra, C.; van Gisbergen, S.J.A.; Snijders, J.G.; Ziegler, T. Chemistry with ADF. J. Comput. Chem. 2001, 22, 931–967. [Google Scholar] [CrossRef]
- Ramírez-Tagle, R.; Alvarado-Soto, L.; Hernández-Acevedo, L.; Arratia-Pérez, R. Spin-orbit and solvent effects in the luminescent [Re6Q8(Ncs)6]4−, Q = S, Se, Te clusters: Molecular sensors and molecular devices. J. Chil. Chem. Soc. 2010, 55, 39–43. [Google Scholar] [CrossRef]
- Orto, P.J.; Nichol, G.S.; Okumura, N.; Evans, D.H.; Arratia-Pérez, R.; Ramirez-Tagle, R.; Wang, R.; Zheng, Z. Cluster carbonyls of the [Re6(μ3-Se)8]2+ core: synthesis, structural characterization, and computational analysis. Dalton Trans. 2008, 6, 4247–4253. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvarado-Soto, L.; Ramirez-Tagle, R. A Theoretical Study of the Binding of [Re6Se8(OH)2(H2O)4] Rhenium Clusters to DNA Purine Base Guanine. Materials 2015, 8, 3938-3944. https://doi.org/10.3390/ma8073938
Alvarado-Soto L, Ramirez-Tagle R. A Theoretical Study of the Binding of [Re6Se8(OH)2(H2O)4] Rhenium Clusters to DNA Purine Base Guanine. Materials. 2015; 8(7):3938-3944. https://doi.org/10.3390/ma8073938
Chicago/Turabian StyleAlvarado-Soto, Leonor, and Rodrigo Ramirez-Tagle. 2015. "A Theoretical Study of the Binding of [Re6Se8(OH)2(H2O)4] Rhenium Clusters to DNA Purine Base Guanine" Materials 8, no. 7: 3938-3944. https://doi.org/10.3390/ma8073938





