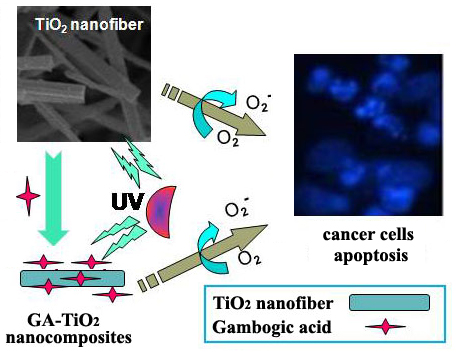
A Novel Exploration of a Combination of Gambogic Acid with TiO2 Nanofibers: The Photodynamic Effect for HepG2 Cell Proliferation
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of TiO2 Nanofibers

2.2. Loading Efficiency and Drug Release Behavior of TiO2 Nanofibers in vitro

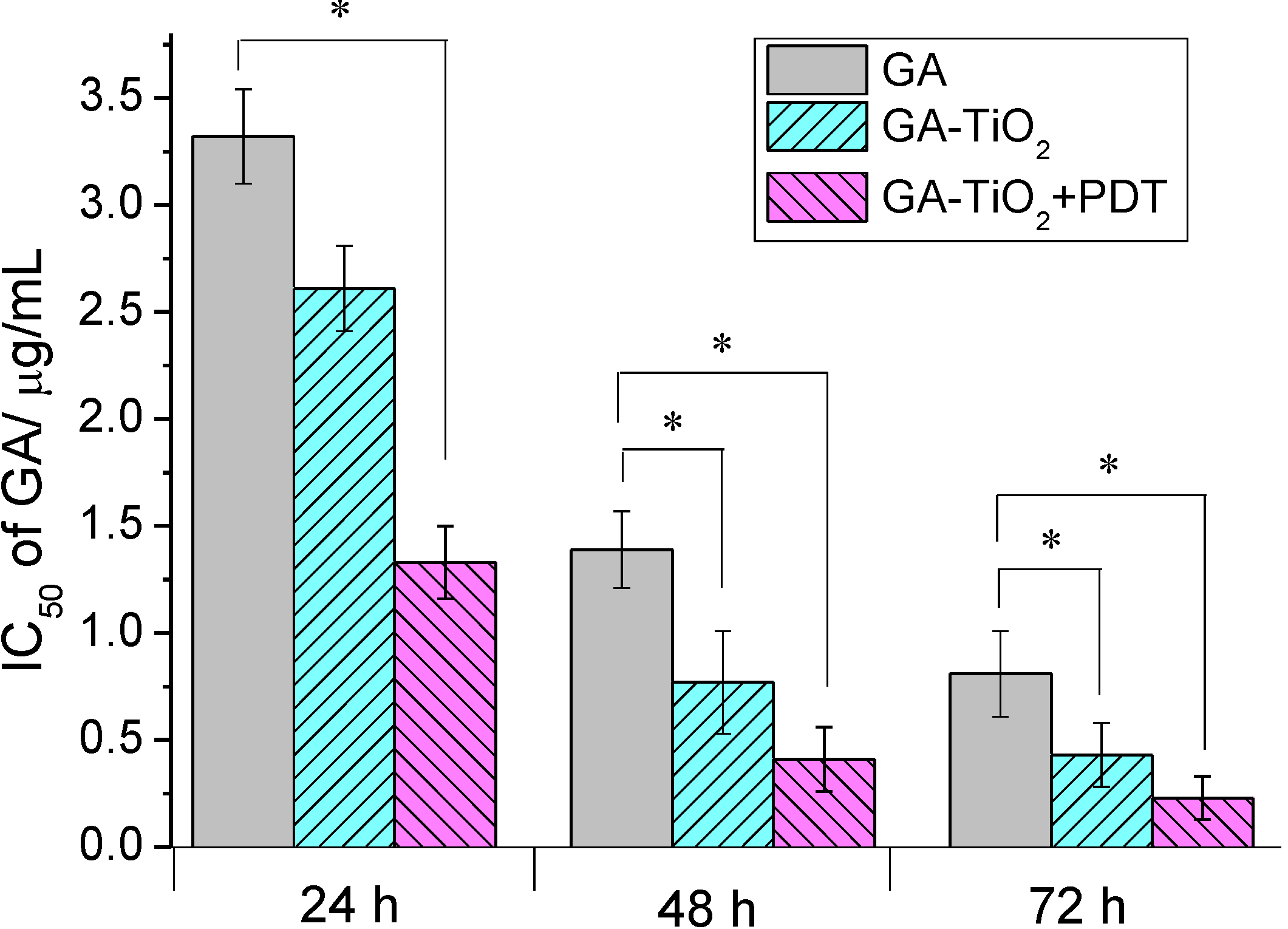
2.3. Cytotoxicity Assay of GA and GA-TiO2 Nanocomposites for HepG2 Cancer Cells
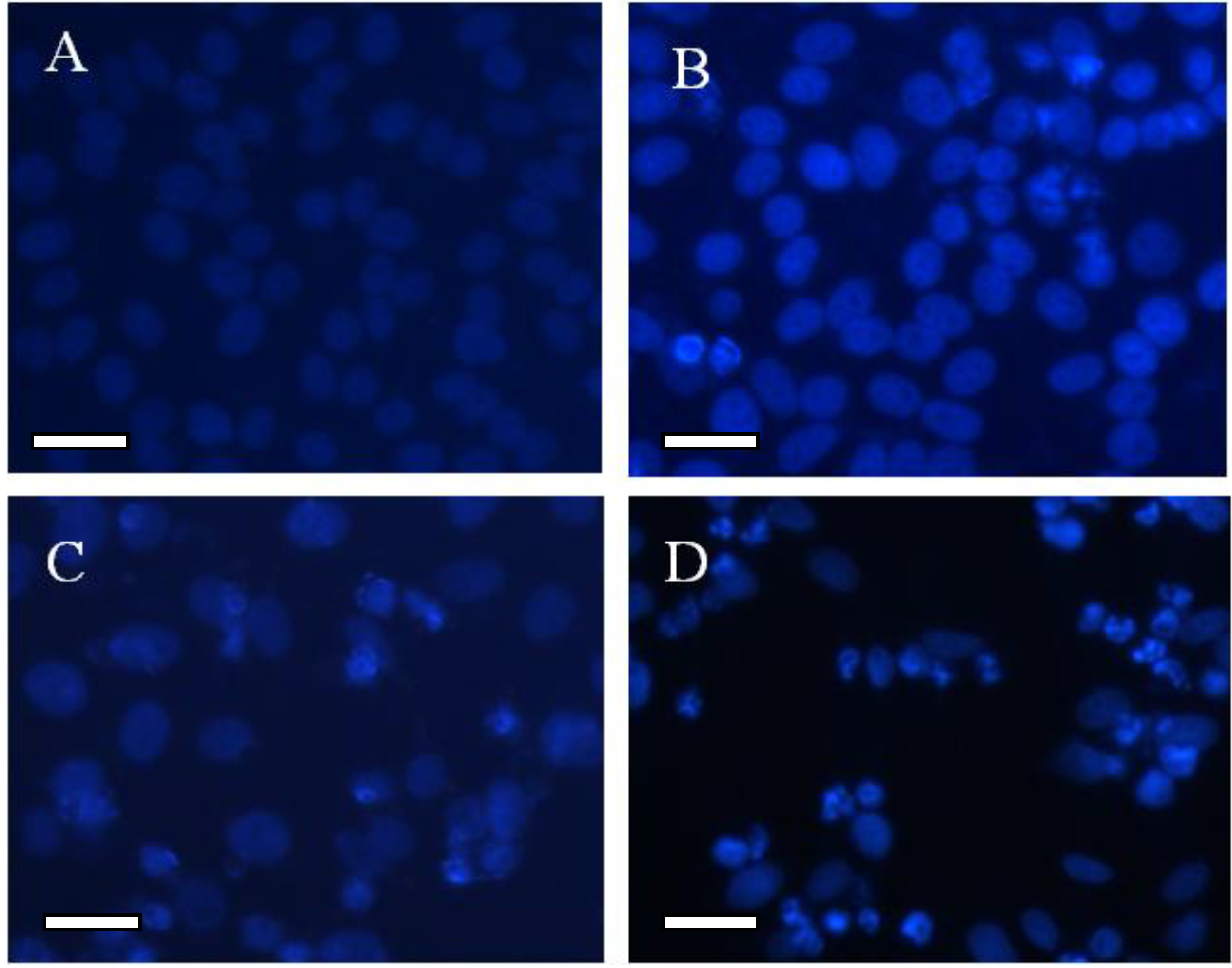
2.4. Morphological Study for the Cytotoxicity of GA and GA-TiO2 Nanocomposites

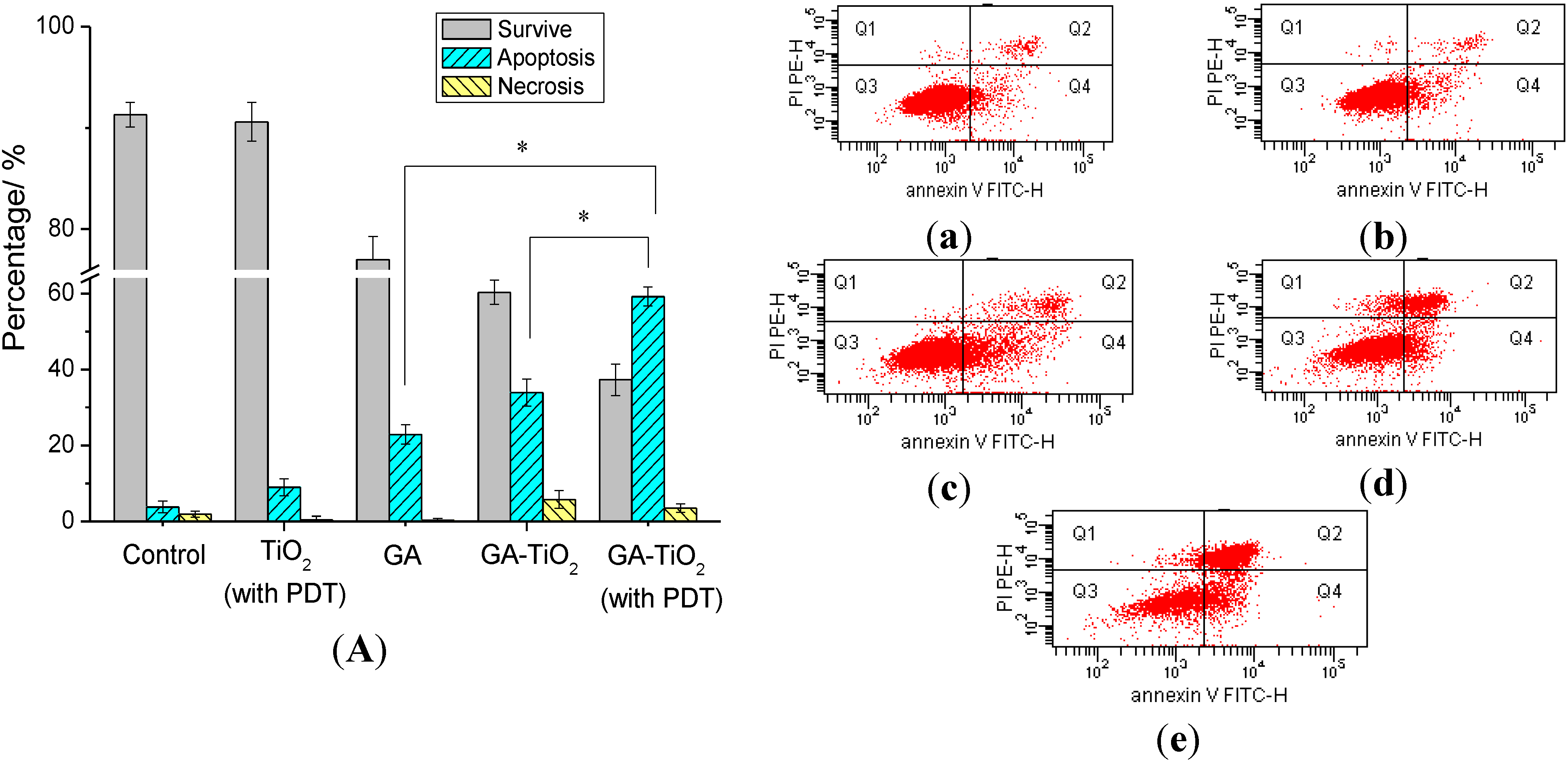
2.5. Flow Cytometry Analysis of Relevant Cancer Cells Apoptosis
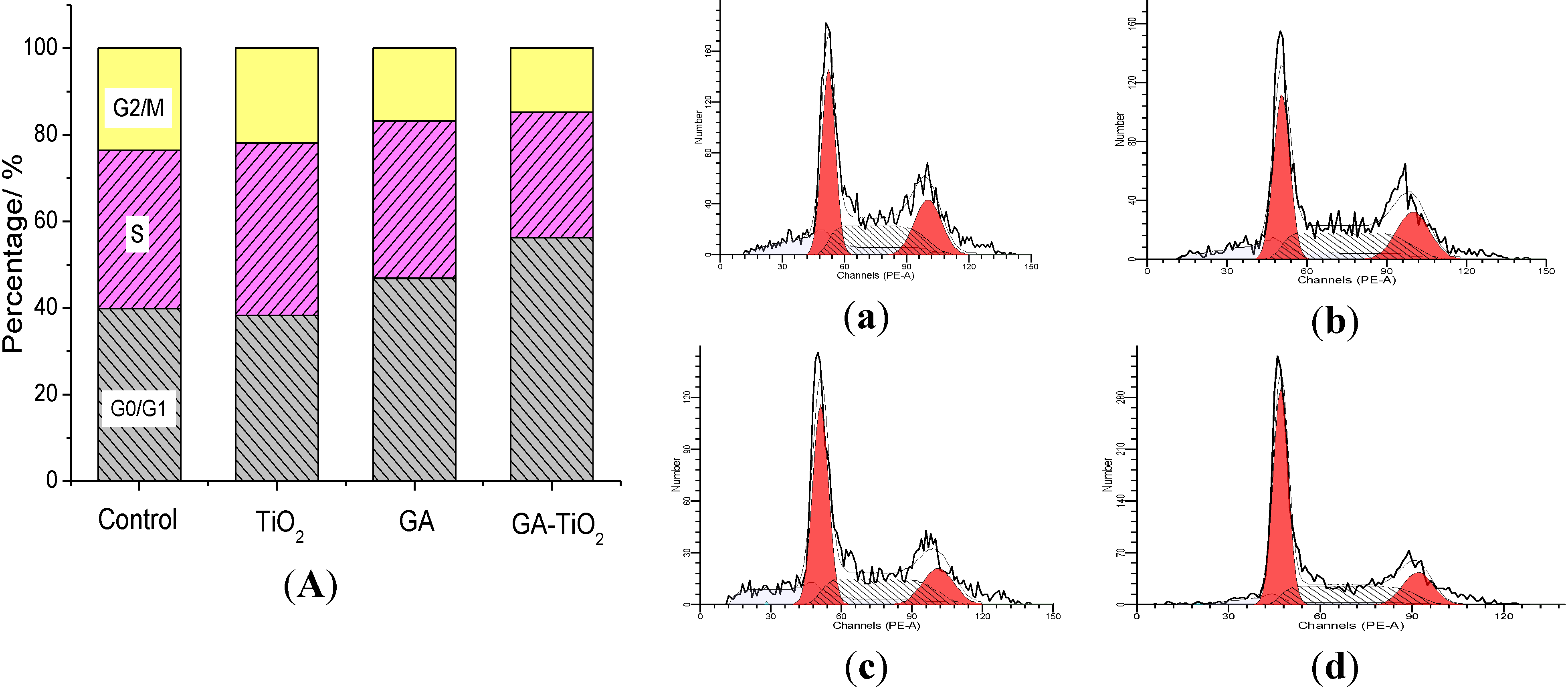
2.6. Study on the Cell Cycle Effect of GA and GA-TiO2 Nanocomposites
3. Experimental Section
3.1. Drugs and Reagents
3.2. Preparation and Characterization of TiO2 Nanofibers
3.3. Cell Culture
3.4. The Preparation of GA-TiO2 Nanocomposites and the Characterization of GA Loading and Release in vitro
Encapsulation efficiency = (amount of drug in drug-loaded nanofibers/initial amount of drug) × 100%.
3.5. MTT Assay of GA and GA-TiO2 Nanocomposites in the Absence/Presence of UV Irradiation
3.6. Cell Morphological Assessment
3.7. Apoptosis Assay of GA and GA-TiO2 Nanocomposites for HepG2 Cells
3.8. Cell Cycle Analysis of GA and GA-TiO2 Nanocomposites for HepG2 Cells
3.9. Statistics
4. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Klauson, D.; Budarnaja, O.; Beltran, I.C.; Krichevskaya, M.; Preis, S. Photocatalytic decomposition of humic acids in anoxic aqueous solutions producing hydrogen, oxygen and light hydrocarbons. Environ. Technol. 2014, 35, 2237–2243. [Google Scholar]
- Grabowska, E.; Diak, M.; Marchelek, M.; Zaleska, A. Decahedral TiO2 with exposed facets: Synthesis, properties, photoactivity and applications. Appl. Catal. B Environ. 2014, 156–157, 213–235. [Google Scholar]
- Mahoney, L.; Koodali, R.T. Versatility of evaporation-induced self-assembly (EISA) method for preparation of mesoporous TiO2 for energy and environmental applications. Materials 2014, 7, 2697–2746. [Google Scholar]
- Zhang, S.; Ma, H.M.; Yan, L.G.; Cao, W.; Yan, T.; Wei, Q.; Du, B. Copper-doped titanium dioxide nanoparticles as dual-functional labels for fabrication of electrochemical immunosensors. Biosens. Bioelectron. 2014, 59, 335–341. [Google Scholar]
- Zhang, H.; Shi, R.H.; Xie, A.J.; Li, J.C.; Chen, L.; Chen, P.; Li, S.K.; Huang, F.Z.; Shen, Y.H. Novel TiO2/PEGDA hybrid hydrogel prepared in situ on tumor cells for effective photodynamic therapy. ACS Appl. Mater. Int. 2013, 5, 12317–12322. [Google Scholar]
- Zhang, H.J.; Shan, Y.F.; Dong, L.J. A comparison of TiO2 and ZnO nanoparticles as photosensitizers in photodynamic therapy for cancer. J. Biomed. Nanotechnol. 2014, 10, 1450–1457. [Google Scholar]
- Yamaguchi, S.; Kobayashi, H.; Narita, T.; Kanehira, K.; Sonezaki, S.; Kudo, N.; Kubota, Y.; Terasaka, S.; Houkin, K. Sonodynamic therapy using water-dispersed TiO2-polyethylene glycol compound on glioma cells: Comparison of cytotoxic mechanism with photodynamic therapy. Ultrason. Sonochem. 2011, 18, 1197–1204. [Google Scholar]
- Zeng, L.Y.; Ren, W.Z.; Xiang, L.C.; Zheng, J.J.; Chen, B.; Wu, A.G. Multifunctional Fe3O4–TiO2 nanocomposites for magnetic resonance imaging and potential photodynamic therapy. Nanoscale 2013, 5, 2107–2113. [Google Scholar]
- Juzenas, P.; Kleinauskas, A.; Luo, P.G.; Sun, Y.P. Photoactivatable carbon nanodots for cancer therapy. Appl. Phys. Lett. 2013, 103, 063701. [Google Scholar]
- Venieri, D.; Fraggedaki, A.; Kostadima, M.; Chatzisymeon, E.; Binas, V.; Zachopoulos, A.; Kiriakidis, G.; Mantzavinos, D. Solar light and metal-doped TiO2 to eliminate water-transmitted bacterial pathogens: Photocatalyst characterization and disinfection performance. Appl. Catal. B Environ. 2014, 154, 93–101. [Google Scholar]
- Terriza, A.; Vilches-Pérez, J.; González-Caballero, J.L.; de la Orden, E.; Yubero, F.; Barranco, A.; Gonzalez-Elipe, A.R.; Vilches, J.; Salido, M. Osteoblasts interaction with PLGA membranes functionalized with titanium film nanolayer by PECVD. In vitro assessment of surface influence on cell adhesion during initial cell to material interaction. Materials 2014, 7, 1687–1708. [Google Scholar]
- Lorenzetti, M.; Pellicer, E.; Sort, J.; Baró, M.D.; Kovač, J.; Novak, S.; Kobe, S. Improvement to the corrosion resistance of Ti-based implants using hydrothermally synthesized nanostructured anatase coatings. Materials 2014, 7, 180–194. [Google Scholar]
- Yen, Y.C.; Chen, J.Z.; Lu, Y.J.; Gwo, S.; Lin, K.J. Chain-network anatase/TiO2 (B) thin film with improved photocatalytic efficiency. Nanotechnology 2014, 25. [Google Scholar] [CrossRef]
- Pablos, C.; Marugan, J.; van Grieken, R.; Adan, C.; Riquelme, A.; Palma, J. Correlation between photoelectrochemical behaviour and photoelectrocatalytic activity and scaling-up of P25-TiO2 electrodes. Electrochim. Acta 2014, 130, 261–270. [Google Scholar]
- Li, W.; Liu, C.; Zhou, Y.X.; Bai, Y.; Feng, X.; Yang, Z.H.; Lu, L.H.; Lu, X.H.; Chan, K.Y. Enhanced photocatalytic activity in anatase/TiO2(B) core-shell nanofiber. J. Phys. Chem. C 2008, 112, 20539–20545. [Google Scholar]
- Li, W.; Bai, Y.; Liu, W.J.; Liu, C.; Yang, Z.H.; Feng, X.; Lu, X.H.; Chan, K.Y. Single-crystalline and reactive facets exposed anatase TiO2 nanofibers with enhanced photocatalytic properties. J. Mater. Chem. 2011, 21, 6718–6724. [Google Scholar]
- Feng, X.H.; Zhang, S.K.; Lou, X. Controlling silica coating thickness on TiO2 nanoparticles for effective photodynamic therapy. Coll. Surf. B Biointerface 2013, 107, 220–226. [Google Scholar]
- Hu, Z.; Huang, Y.D.; Sun, S.F.; Guan, W.C.; Yao, Y.H.; Tang, P.Y.; Li, C.Y. Visible light driven photodynamic anticancer activity of graphene oxide/TiO2 hybrid. Carbon 2012, 50, 994–1004. [Google Scholar]
- Lv, X.Y.; Lu, W.; Zhang, X.L.; Lu, X.H.; Wang, X.M. The microbial fuel cell based on the nano MoO3/TiO2 whiskers. Nanosci. Nanotechnol. Lett. 2013, 5, 297–300. [Google Scholar]
- Zhuang, W.; Lu, L.H.; Wu, X.B.; Jin, W.; Meng, M.; Zhu, Y.D.; Lu, X.H. TiO2-B nanofibers with high thermal stability as improved anodes for lithium ion batteries. Electrochem. Commun. 2013, 27, 124–127. [Google Scholar]
- Zhang, H.J.; Wang, C.L.; Chen, B.A.; Wang, X.M. Daunorubicin-TiO2 nanocomposites as a “smart” pH-responsive drug delivery system. Int. J. Nanomed. 2012, 7, 235–242. [Google Scholar]
- Li, J.Y.; Wang, X.M.; Jiang, H.; Lu, X.H.; Zhu, Y.D.; Chen, B.A. New strategy of photodynamic treatment of TiO2 nanofibers combined with celastrol for HepG2 proliferation in vitro. Nanoscale 2011, 3, 3115–3122. [Google Scholar]
- Nie, F.F.; Zhang, X.N.; Qi, Q.; Yang, L.; Yang, Y.; Liu, W.; Lu, N.; Wu, Z.Q.; You, Q.D.; Guo, Q.L. Reactive oxygen species accumulation contributes to gambogic acid-induced apoptosis in human hepatoma SMMC-7721 cells. Toxicology 2009, 260, 60–67. [Google Scholar]
- Duan, D.Z.; Zhang, B.X.; Yao, J.; Liu, Y.P.; Sun, J.Y.; Ge, C.P.; Peng, S.J.; Fang, J.G. Gambogic acid induces apoptosis in hepatocellular carcinoma SMMC-7721 cells by targeting cytosolic thioredoxin reductase. Free Radical Bio. Med. 2014, 69, 15–25. [Google Scholar]
- Li, R.; Chen, Y.; Zeng, L.L.; Shu, W.X.; Zhao, F.; Wen, L.; Liu, Y. Gambogic acid induces G0/G1 arrest and apoptosis involving inhibition of SRC-3 and inactivation of Akt pathway in K562 leukemia cells. Toxicology 2009, 262, 98–105. [Google Scholar]
- Liu, N.N.; Huang, H.B.; Liu, S.T.; Li, X.F.; Yang, C.S.; Dou, Q.P.; Liu, J.B. Calcium channel blocker verapamil accelerates gambogic acid-induced cytotoxicity via enhancing proteasome inhibition and ROS generation. Toxicol. in vitro 2014, 28, 419–425. [Google Scholar]
- Li, X.F.; Liu, S.T.; Huang, H.B.; Liu, N.N.; Zhao, C.; Liao, S.Y.; Yang, C.S.; Liu, Y.R.; Zhao, C.G.; Li, S.J. Gambogic acid is a tissue-specific proteasome inhibitor in vitro and in vivo. Cell Rep. 2013, 3, 211–222. [Google Scholar]
- Qi, Q.; You, Q.D.; Gu, H.Y.; Zhao, L.; Liu, W.; Lu, N.; Guo, Q.L. Studies on the toxicity of gambogic acid in rats. J. Ethnopharmacol. 2008, 117, 433–438. [Google Scholar]
- Yang, L.J.; Chen, Y. New targets for the antitumor activity of gambogic acid in hematologic malignancies. Acta Pharmacol. Sin. 2013, 34, 191–198. [Google Scholar]
- Kessel, D.; Reiners, J.J. Enhanced efficacy of photodynamic therapy via a sequential targeting protocol. Photochem. Photobiol. 2014, 90, 889–895. [Google Scholar]
- Barar, J.; Omidi, Y. Surface modified multifunctional nanomedicines for simultaneous imaging and therapy of cancer. Bioimpacts 2014, 4, 3–14. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, J.; Wang, X.; Shao, Y.; Lu, X.; Chen, B. A Novel Exploration of a Combination of Gambogic Acid with TiO2 Nanofibers: The Photodynamic Effect for HepG2 Cell Proliferation. Materials 2014, 7, 6865-6878. https://doi.org/10.3390/ma7096865
Li J, Wang X, Shao Y, Lu X, Chen B. A Novel Exploration of a Combination of Gambogic Acid with TiO2 Nanofibers: The Photodynamic Effect for HepG2 Cell Proliferation. Materials. 2014; 7(9):6865-6878. https://doi.org/10.3390/ma7096865
Chicago/Turabian StyleLi, Jingyuan, Xuemei Wang, Yixiang Shao, Xiaohua Lu, and Baoan Chen. 2014. "A Novel Exploration of a Combination of Gambogic Acid with TiO2 Nanofibers: The Photodynamic Effect for HepG2 Cell Proliferation" Materials 7, no. 9: 6865-6878. https://doi.org/10.3390/ma7096865