Improvement of Interaction in a Composite Structure by Using a Sol-Gel Functional Coating on Carbon Fibers
Abstract
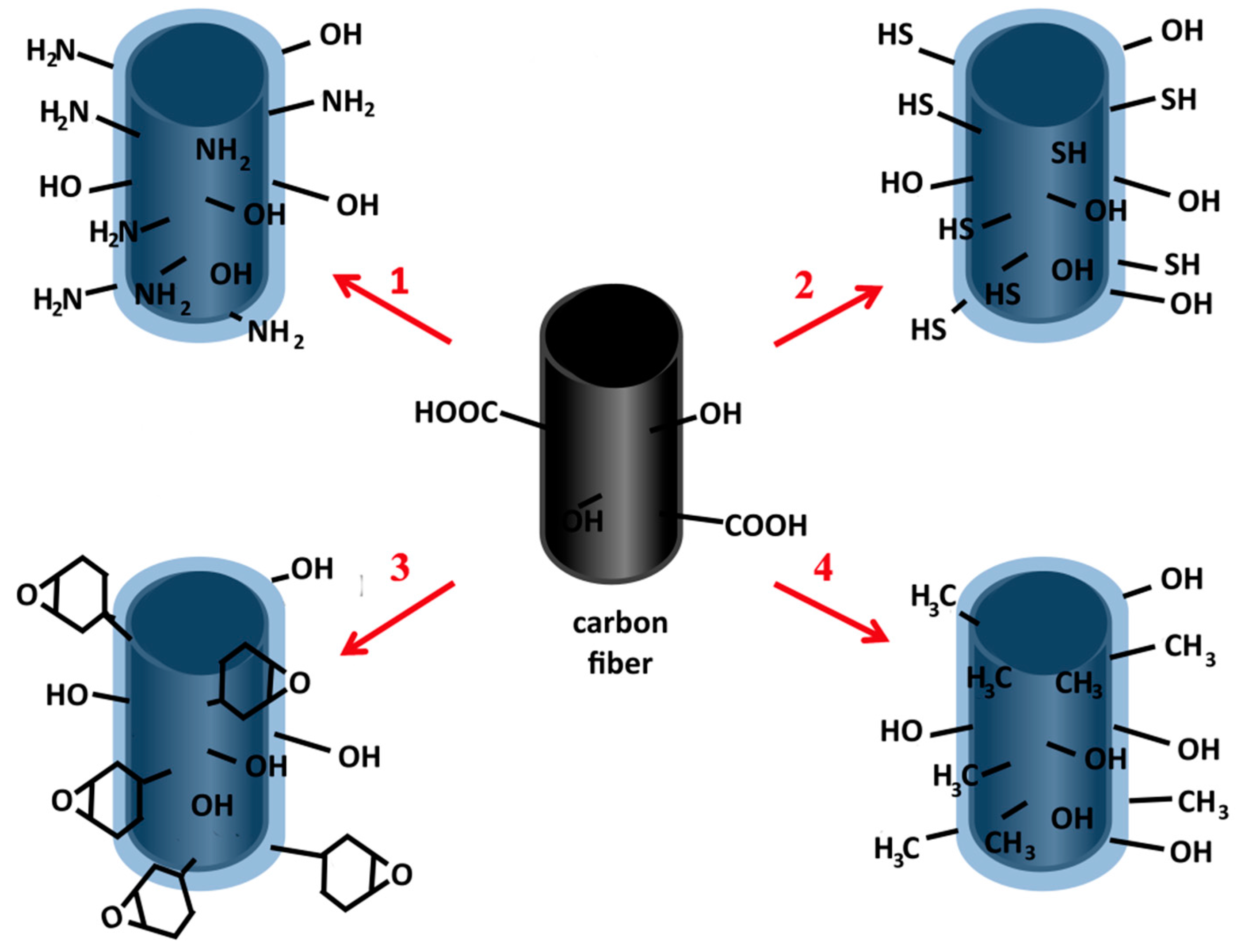
:1. Introduction
2. Results
2.1. SEM
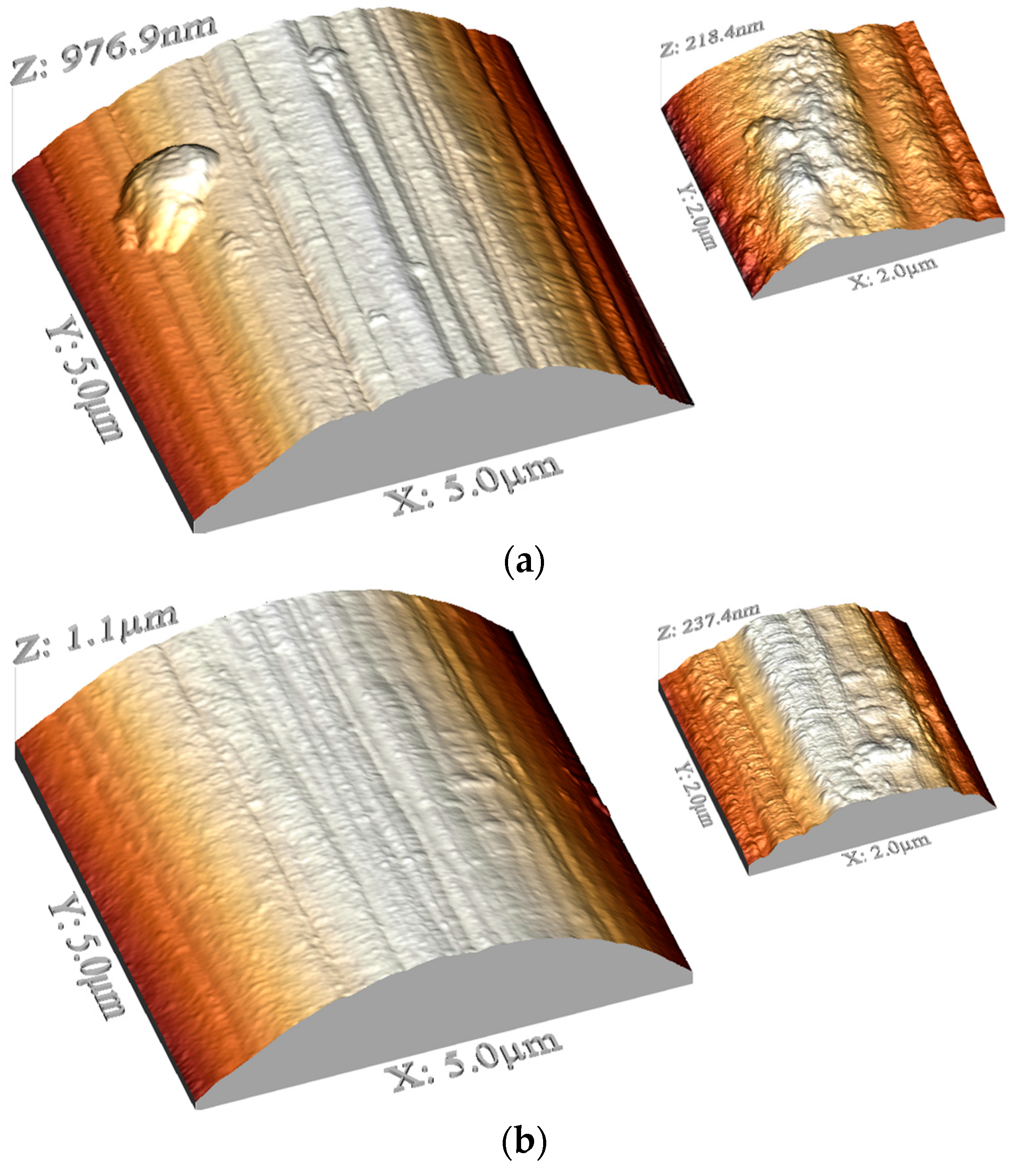
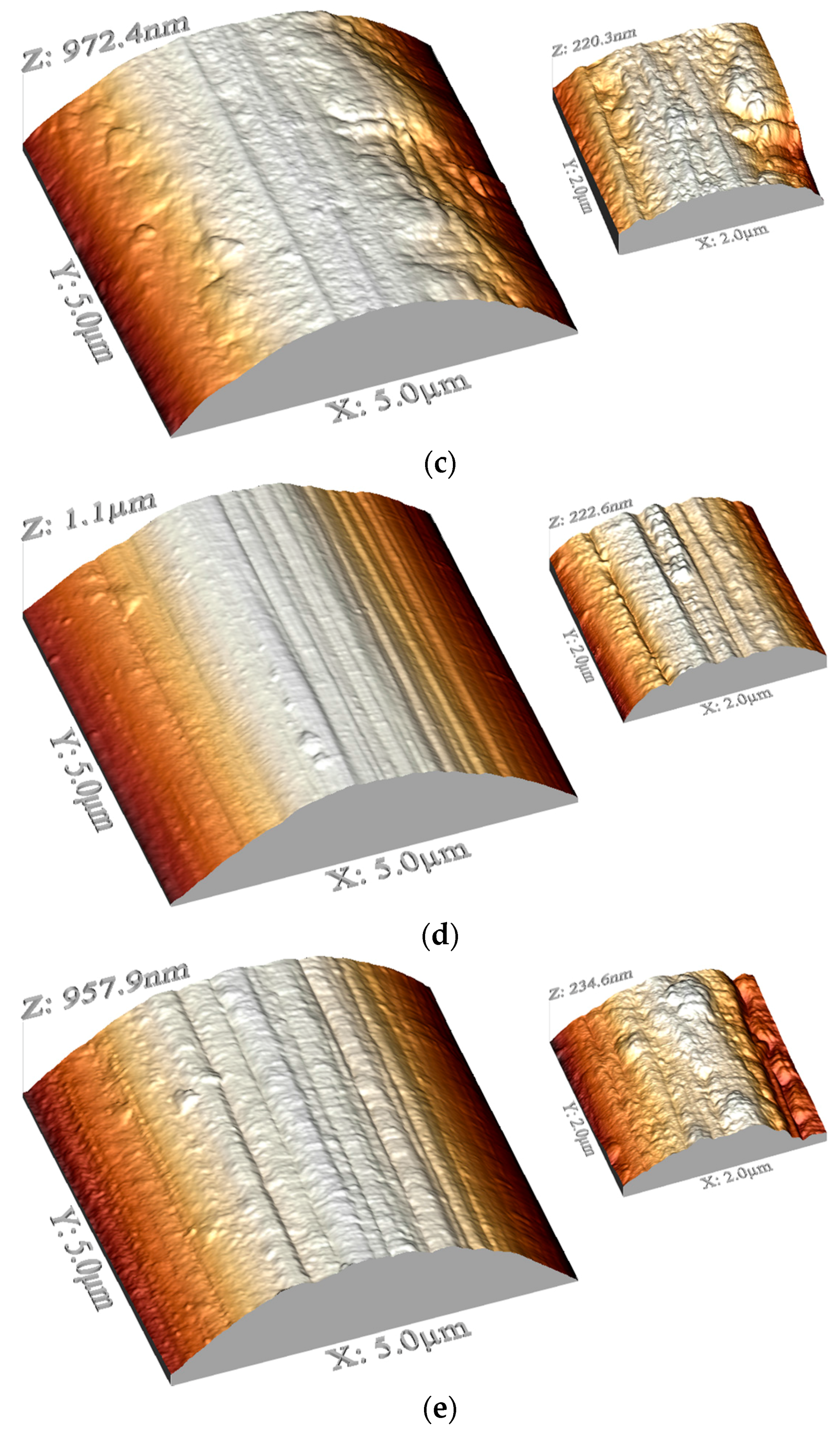
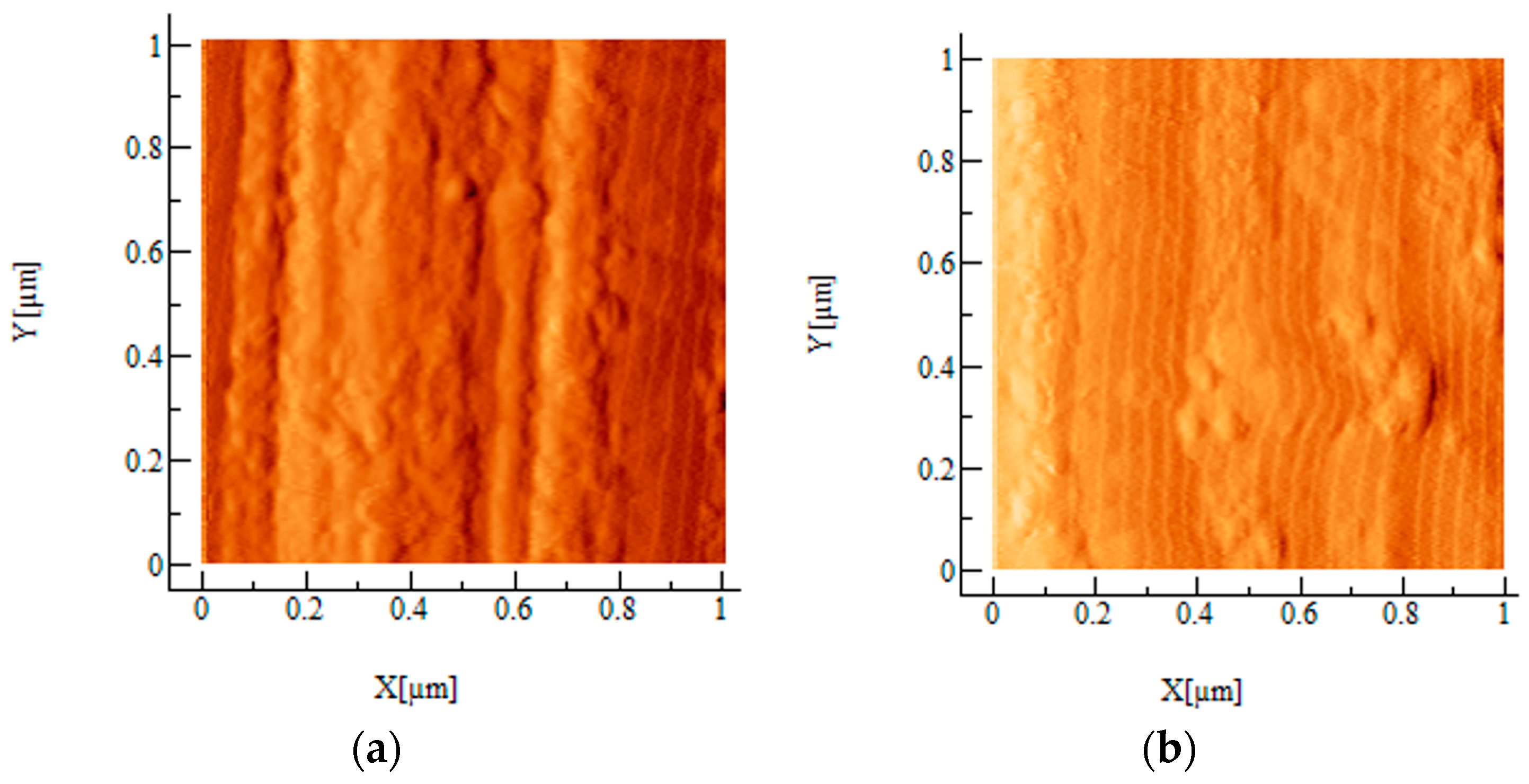
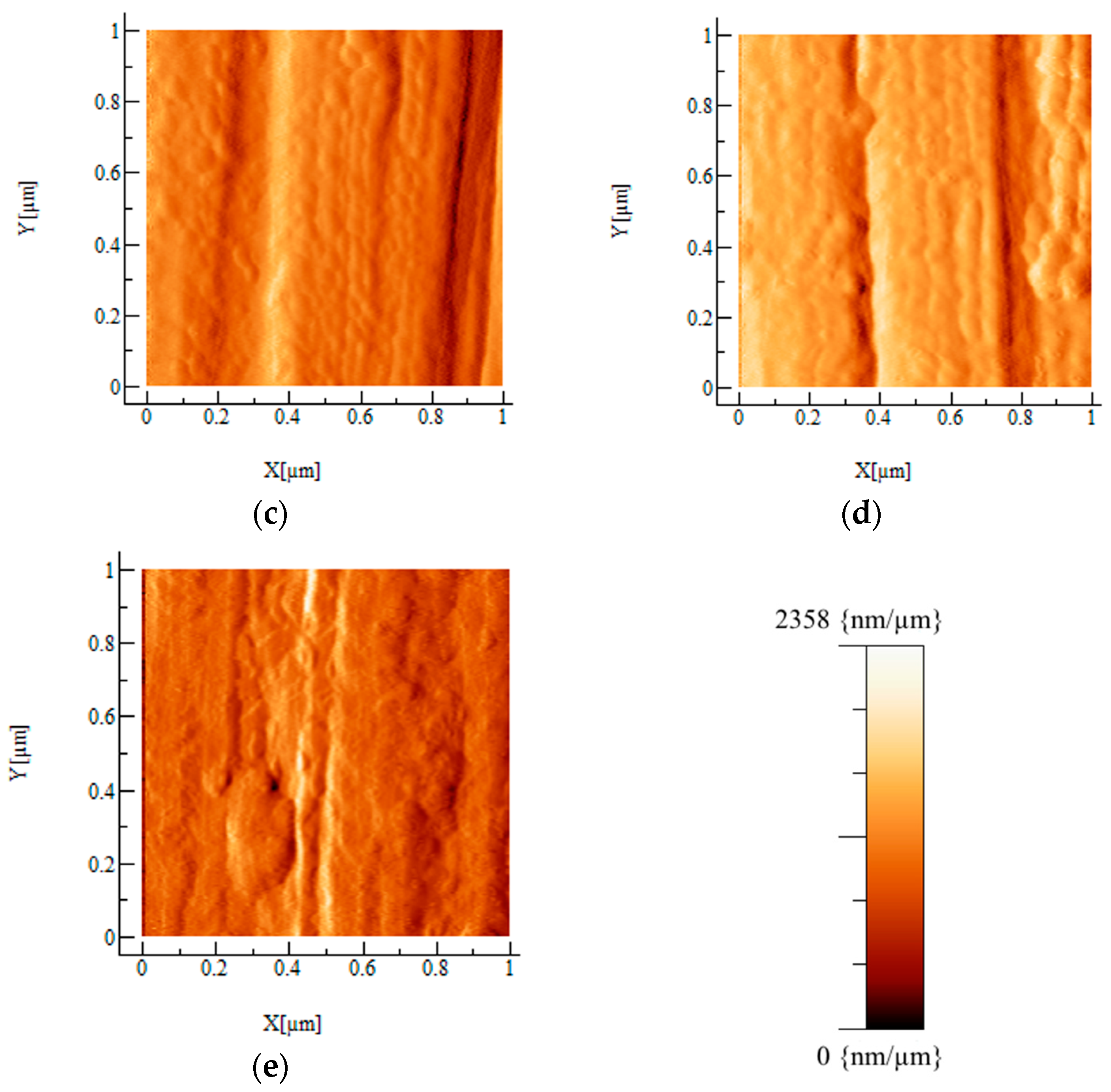
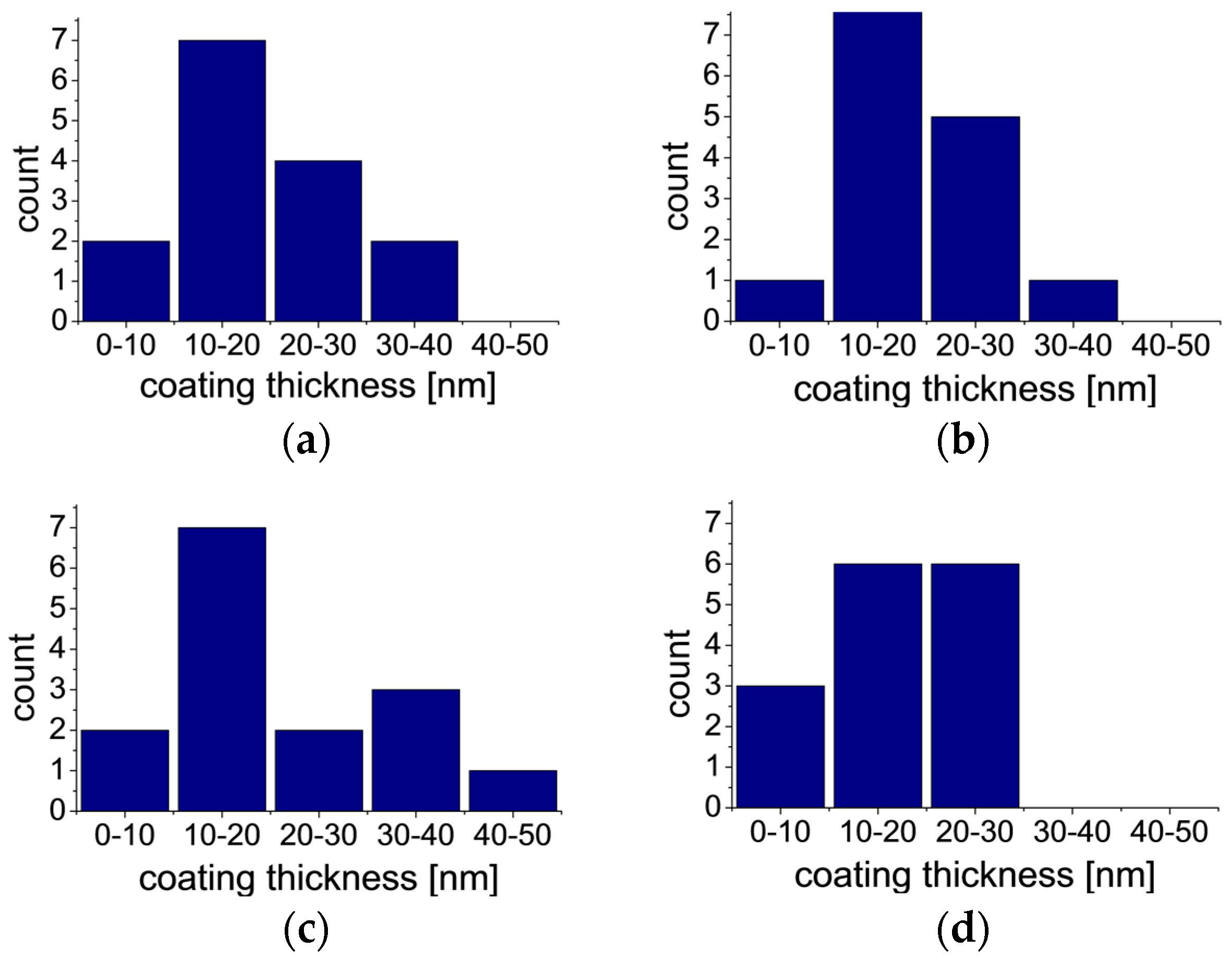
2.2. AFM
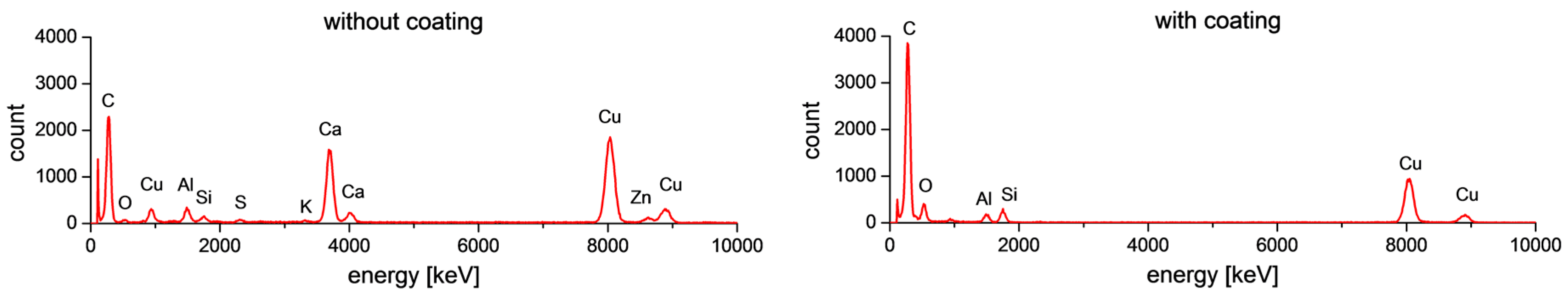
2.3. TEM and EDS
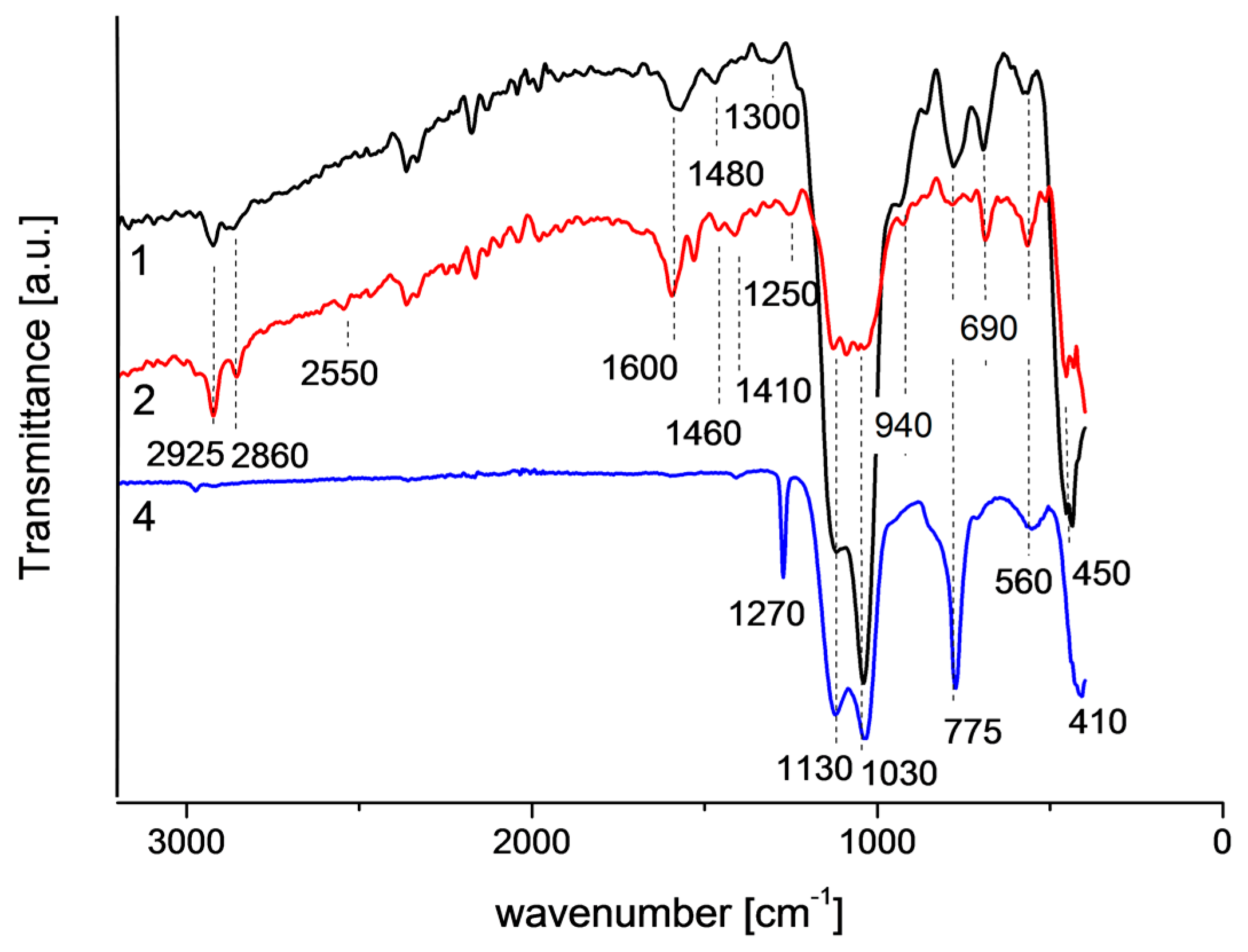
2.4. IR
2.5. Wettability
3. Discussion
4. Materials and Methods
4.1. Materials Preparation
4.2. SEM
4.3. AFM
4.4. TEM and EDS
4.5. IR
4.6. Wettability
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jean-Baptiste, D.; Chand, B.R. Carbon Fibers; CRC Press: Boca Raton, FL, USA, 1998. [Google Scholar]
- Dai, Z.; Shi, F.; Zhang, B.; Li, M.; Zhang, Z. Effect of sizing on carbon fiber surface properties and fibers/epoxy interfacial adhesion. Appl. Surf. Sci. 2011, 257, 6980–6985. [Google Scholar] [CrossRef]
- Dai, Z.; Zhang, B.; Shi, F.; Li, M.; Zhang, Z.; Gu, Y. Effect of heat treatment on carbon fiber surface properties and fibers/epoxy interfacial adhesion. Appl. Surf. Sci. 2011, 257, 8457–8461. [Google Scholar] [CrossRef]
- Brocks, T.; Cioffi, M.O.H.; Voorwald, H.J.C. Effect of fiber surface on flexural strength in carbon fabric reinforced epoxy composites. Appl. Surf. Sci. 2013, 274, 210–216. [Google Scholar] [CrossRef]
- Adams, D.F.; Bergmann, H.W.; Brunsch, K.; Dierendorf, R.J.; Fritzer, E.; Hasings, G.W.; Hayes, B.J.; Jager, H.; Kalnin, I.L.; Kim, C.Y.; et al. Carbon Fibers and Their Composites; Fritzer, E., Ed.; Springer: Berlin, Germany, 1985. [Google Scholar]
- Xie, J.; Xin, D.; Cao, H.; Wang, C.; Zhao, Y.; Yao, L.; Ji, F.; Qiu, Y. Improving carbon fiber adhesion to polyimide with atmospheric pressure plasma treatment. Surf. Coat. Technol. 2011, 206, 191–201. [Google Scholar] [CrossRef]
- Cao, H.; Huang, Y.; Zhang, Z.; Sun, J. Uniform modification of carbon fibers surface in 3-D fabrics using intermittent electrochemical treatment. Compos. Sci. Technol. 2005, 65, 1655–1662. [Google Scholar] [CrossRef]
- Wettmarshausen, S.; Friedrich, J.F.; Meyer-Plath, A.; Kalinka, G.; Hidde, G.; Weidner, S. Coating of carbon fibers with adhesion- promoting thin poly (acrylic acid) and poly (hydroxyethylmethacrylate) layers using electrospray ionization. J. Adhes. Sci. Technol. 2015, 29, 1628–1650. [Google Scholar] [CrossRef]
- Li, S.; Zhang, Y.M.; Zhou, Y.F. Preparation and characterization of sol-gel derived zirconia coated carbon fiber. Surf. Coat. Technol. 2012, 206, 4720–4724. [Google Scholar] [CrossRef]
- Tang, Y.; Deng, Y.; Zhang, K.; Liu, L.; Wu, Y.; Hu, W. Improvement of interface between Al and short carbon fibers by α-Al2O3 coatings deposited by sol-gel technology. Ceram. Int. 2008, 34, 1787–1790. [Google Scholar] [CrossRef]
- Wang, W.G.; Xiao, B.L.; Ma, Z.Y. Interfacial reaction and nanostructures in Mg matrix composites reinforced with carbon fibers modified by sol-gel method. Compos. Sci. Technol. 2013, 87, 69–76. [Google Scholar] [CrossRef]
- Malgras, V.; Ji, Q.; Kamachi, Y.; Mori, T.; Shieh, F.K.; Wu, K.C.W.; Ariga, K.; Yamauchi, Y. Templated Synthesis for Nanoarchitectured Porous Materials. Bull. Chem. Soc. Jpn. 2015, 88, 1171–1200. [Google Scholar] [CrossRef]
- Nisticò, R.; Scalarone, D.; Magnacca, G. Sol-gel chemistry, templating and spin-coating deposition: A combined approach to control in a simple way the porosity of inorganic thin films/coatings. Microporous Mesoporous Mater. 2017, 248, 18–29. [Google Scholar] [CrossRef]
- Feinle, A.; Elsaesser, M.S.; Hüsing, N.; Da’as, E.H.; Giannelis, E.P.; Wittstock, A.; Lee, J.R.I.; Biener, M.M.; Orme, C.A.; Kucheyev, S.O.; et al. Sol-gel synthesis of monolithic materials with hierarchical porosity. Chem. Soc. Rev. 2016, 45, 3377–3399. [Google Scholar] [CrossRef] [PubMed]
- Danks, A.E.; Hall, S.R.; Schnepp, Z.; Eastoe, J.; Grillo, I.; Shchukin, D.; Dieudonné, P.; Galarneau, A.; Vioux, A.; Park, J.H.; et al. The evolution of “sol-gel” chemistry as a technique for materials synthesis. Mater. Horiz. 2016, 3, 91–112. [Google Scholar] [CrossRef]
- Baccile, N.; Babonneau, F.; Thomas, B.; Coradin, T.; Brook, M.A.; Brennan, J.D.; Barbé, C.J.; Chang, S.; Park, J.G.; Hu, Y.F.; et al. Introducing ecodesign in silica sol-gel materials. J. Mater. Chem. 2009, 19, 8537. [Google Scholar] [CrossRef]
- Ismail, F.; Schoenleber, M.; Mansour, R.; Bastani, B.; Fielden, P.; Goddard, N.J.; Kagan, M.L.; Avnir, D. Strength of interactions between immobilized dye molecules and sol-gel matrices. Analyst 2011, 136, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Kappert, E.J.; Pavlenko, D.; Malzbender, J.; Nijmeijer, A.; Benes, N.E.; Tsai, P.A. Formation and prevention of fractures in sol-gel-derived thin films. Soft Matter 2015, 11, 882–888. [Google Scholar] [CrossRef] [PubMed]
- Birnie, D.P.; Kaz, D.M.; Taylor, D.J. Surface tension evolution during early stages of drying of sol-gel coatings. J. Sol-Gel Sci. Technol. 2009, 49, 233–237. [Google Scholar] [CrossRef]
- Aguiar, H.; Serra, J.; González, P.; León, B. Structural study of sol-gel silicate glasses by IR and Raman spectroscopies. J. Non-Cryst. Solids 2009, 355, 475–480. [Google Scholar] [CrossRef]
- Wu, Q. Preparation and conductibility of silica gels composited with undecatungstochromoferrous heteropoly acid. Mater. Chem. Phys. 2003. [Google Scholar] [CrossRef]
- Wright John, D.; Sommerdijk Nico, A.J.M. Sol-Gel Materials: Chemistry and Applications; CRC Press: Boca Raton, FL, USA, 2001. [Google Scholar]
- Innocenzi, P. Infrared spectroscopy of sol-gel derived silica-based films: A spectra-microstructure overview. J. Non-Cryst. Solids 2003, 316, 309–319. [Google Scholar] [CrossRef]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]
- Chernev, G.; Rangelova, N.; Djambazki, P.; Nenkova, S.; Salvado, I.; Fernandes, M.; Wu, A.; Kabaivanova, L. Sol-gel silica hybrid biomaterials for application in biodegradation of toxic compounds. J. Sol-Gel Sci. Technol. 2011. [Google Scholar] [CrossRef]
- Majoul, N.; Aouida, S.; Bessaïs, B. Progress of porous silicon APTES-functionalization by FTIR investigations. Appl. Surf. Sci. 2015, 331, 388–391. [Google Scholar] [CrossRef]
- Deng, C.; Jiang, J.; Liu, F.; Fang, L.; Wang, J.; Li, D.; Wu, J. Influence of graphene oxide coatings on carbon fiber by ultrasonically assisted electrophoretic deposition on its composite interfacial property. Surf. Coat. Technol. 2015, 272, 176–181. [Google Scholar] [CrossRef]
- Sarac, A.S.; Serantoni, M.; Tofail, S.A.M.; Henry, J.; Cunnane, V.; McMonagle, J.B. Characterisation of nanosize thin films of electrografted N-vinylcarbazole copolymers (P [NVCz-co-VBSA] and P [NVCz-co-3-MeTh]) onto carbon fibre. Appl. Surf. Sci. 2005, 243, 183–198. [Google Scholar] [CrossRef]
- Ma, Q.; Gu, Y.; Li, M.; Wang, S.; Zhang, Z. Effects of surface treating methods of high-strength carbon fibers on interfacial properties of epoxy resin matrix composite. Appl. Surf. Sci. 2016, 379, 199–205. [Google Scholar] [CrossRef]
- Olszówka-Myalska, A.; Botor-Probierz, A.; Swadźba, L.; Krzak-Roś, J. Technological coatings deposited on carbon fibres—An overview. Inżynieria Mater. 2009, 30, 163–168. (In Polish) [Google Scholar]
- Wang, J.; Lin, W.; Yan, X.; Wu, X.; Wu, F.; Yang, Y. Preparation and microstructure of Al2O3-SiO2-TiO2 coating on three-dimensional braided carbon fiber by sol-gel technology. Mater. Des. 2016, 89, 928–932. [Google Scholar] [CrossRef]
- Tiwari, S.; Bijwe, J.; Panier, S. Optimization of surface treatment to enhance fiber-matrix interface and performance of composites. Wear 2012, 274, 326–334. [Google Scholar] [CrossRef]
- Choi, W.K.; Kim, H.I.; Kang, S.J.; Lee, Y.S.; Han, J.H.; Kim, B.J. Mechanical interfacial adhesion of carbon fibers-reinforced polarized-polypropylene matrix composites: Effects of silane coupling agents. Carbon Lett. 2016, 17, 79–84. [Google Scholar] [CrossRef]
- Luo, Y.; Zhao, Y.; Duan, Y.; Du, S. Surface and wettability property analysis of CCF300 carbon fibers with different sizing or without sizing. Mater. Des. 2011, 32, 941–946. [Google Scholar] [CrossRef]
- Wang, J.; Hong, T.; Li, G.; Li, P. A combined process of coating and hybridizing for the fabrication of carbon fiber reinforced aluminum matrix composites. Compos. Part A Appl. Sci. Manuf. 1997, 28, 943–948. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, Y.; Wang, T. Surface analysis of plasma grafted carbon fiber. Appl. Surf. Sci. 2006, 253, 2885–2892. [Google Scholar] [CrossRef]
- Horcas, I.; Fernández, R.; Gómez-Rodríguez, J.M.; Colchero, J.; Gómez-Herrero, J.; Baro, A.M. WSXM: A software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 2007, 78, 13705. [Google Scholar] [CrossRef] [PubMed]



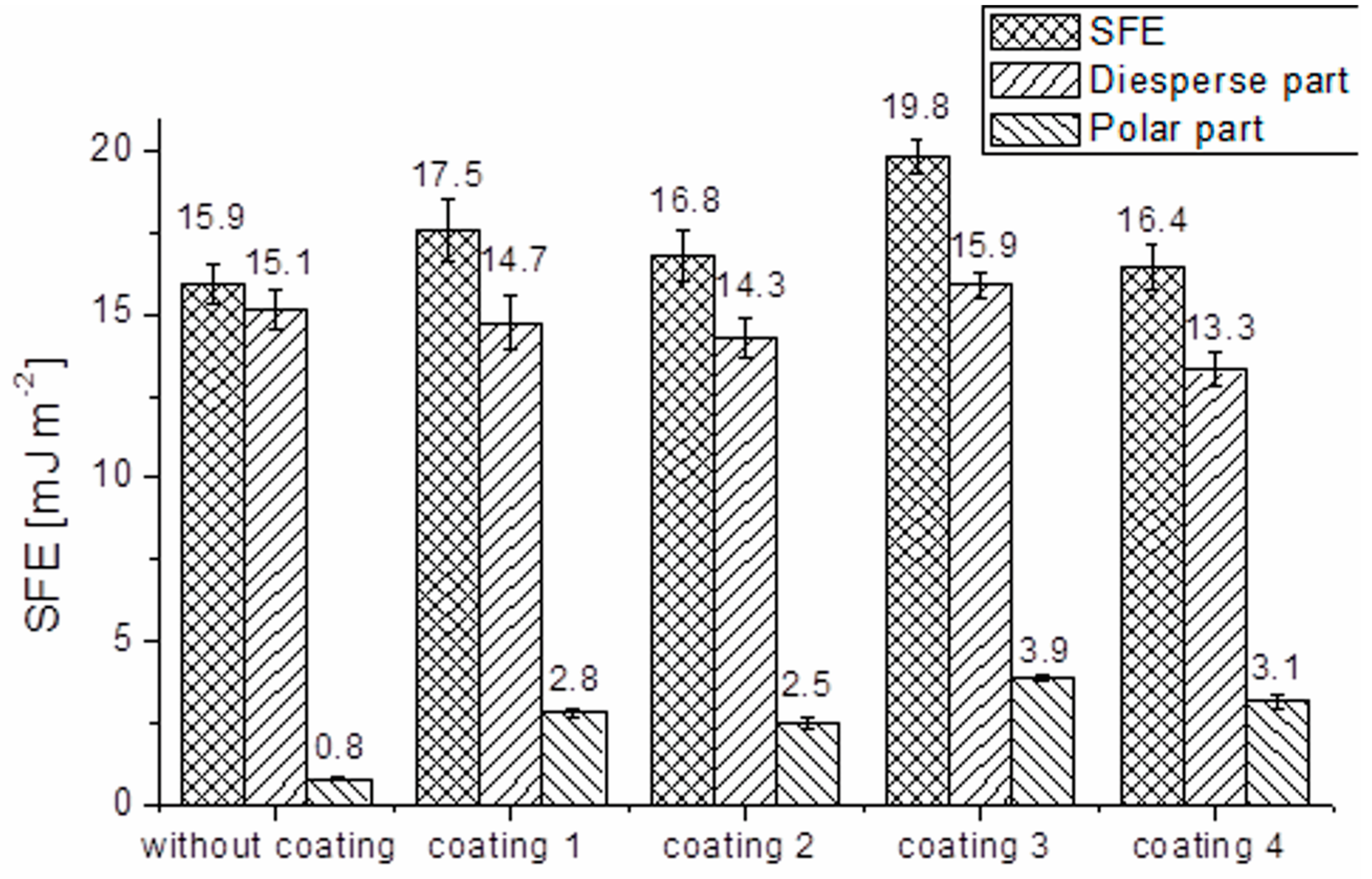
| Sample | % Polarity |
|---|---|
| without coating | 5.1 |
| coating 1 | 16.1 |
| coating 2 | 14.9 |
| coating 3 | 19.7 |
| coating 4 | 19.2 |
| Sol | Organically Functionalized Precursor | TMOS | Alcohol | HCl |
|---|---|---|---|---|
| 1 | 1 | 0.3 | 10.6 | 0.1 |
| 2 | 1 | 0.3 | 7.9 | 0.1 |
| 3 | 1 | 0.5 | 12.1 | 0.1 |
| 4 | 1 | 0.2 | 6.1 | 0.1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szczurek, A.; Barcikowski, M.; Leluk, K.; Babiarczuk, B.; Kaleta, J.; Krzak, J. Improvement of Interaction in a Composite Structure by Using a Sol-Gel Functional Coating on Carbon Fibers. Materials 2017, 10, 990. https://doi.org/10.3390/ma10090990
Szczurek A, Barcikowski M, Leluk K, Babiarczuk B, Kaleta J, Krzak J. Improvement of Interaction in a Composite Structure by Using a Sol-Gel Functional Coating on Carbon Fibers. Materials. 2017; 10(9):990. https://doi.org/10.3390/ma10090990
Chicago/Turabian StyleSzczurek, Anna, Michał Barcikowski, Karol Leluk, Bartosz Babiarczuk, Jerzy Kaleta, and Justyna Krzak. 2017. "Improvement of Interaction in a Composite Structure by Using a Sol-Gel Functional Coating on Carbon Fibers" Materials 10, no. 9: 990. https://doi.org/10.3390/ma10090990





