Improving the Mechanical Properties of Cu-15Ni-8Sn Alloys by Addition of Titanium
Abstract
:1. Introduction
2. Experimental Procedure
2.1. Materials and Processing
2.2. Microstructure Evaluation
2.3. Resistivity Measurement
2.4. Mechanical Properties Testing
3. Results and Discussion
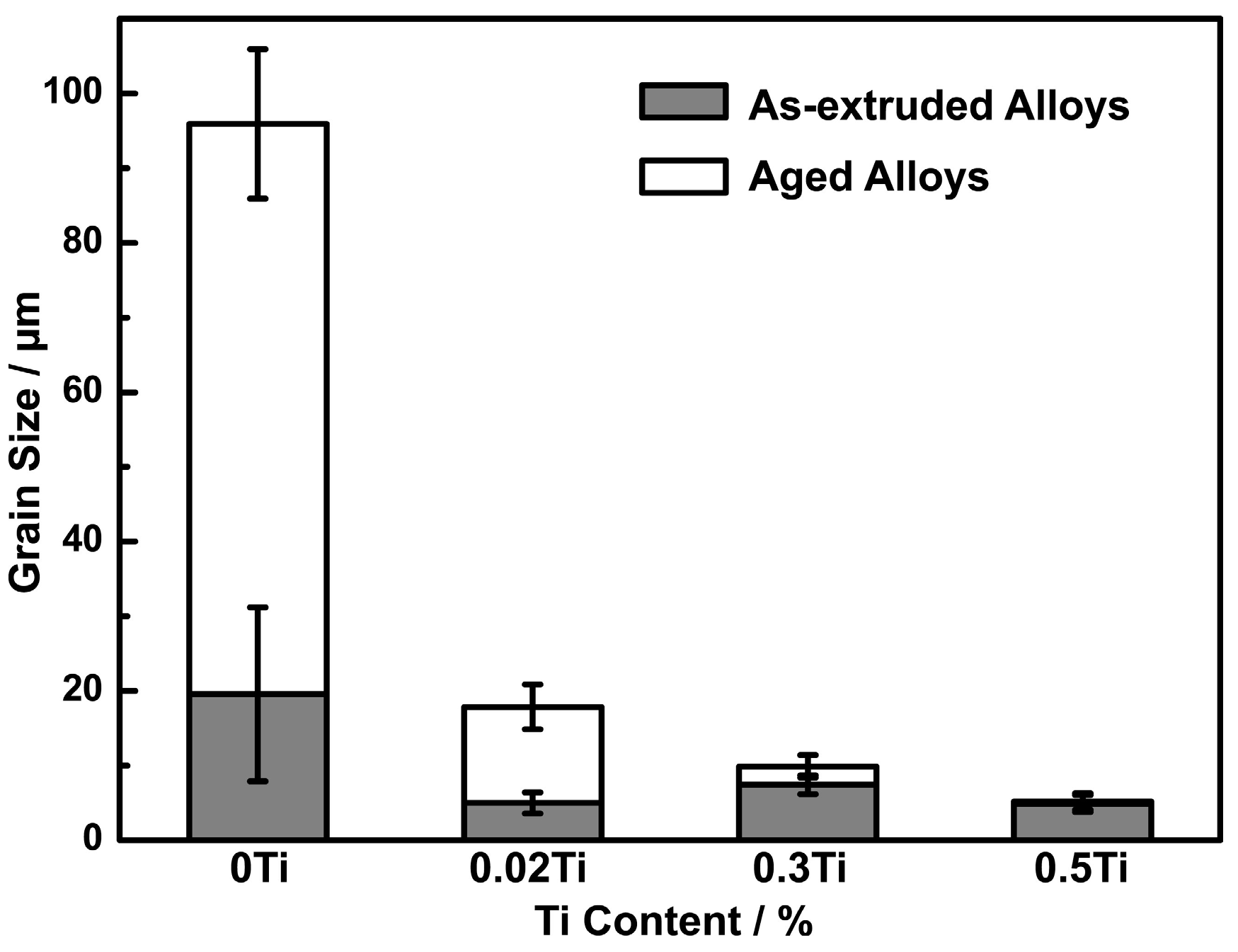
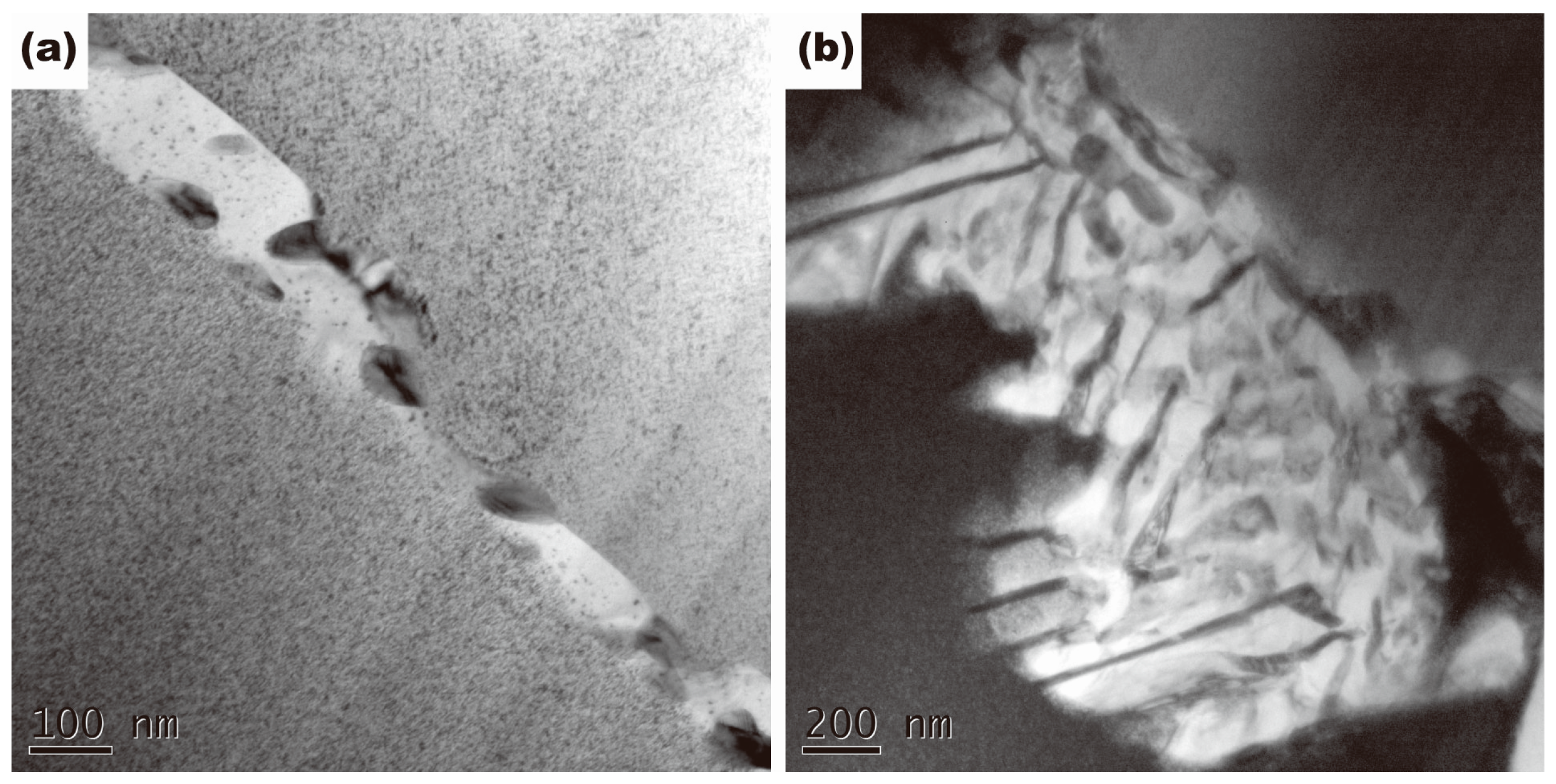
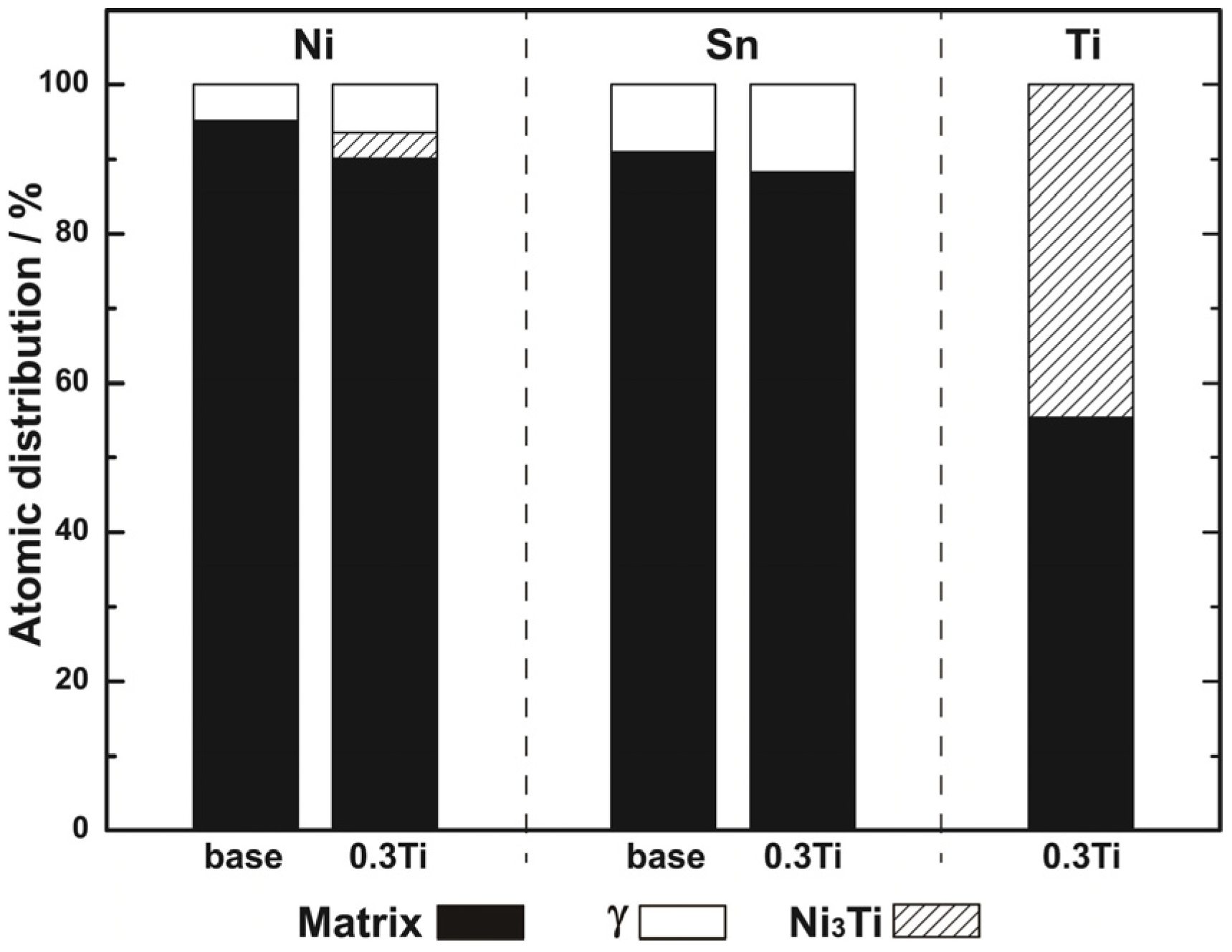
3.1. Microstructures
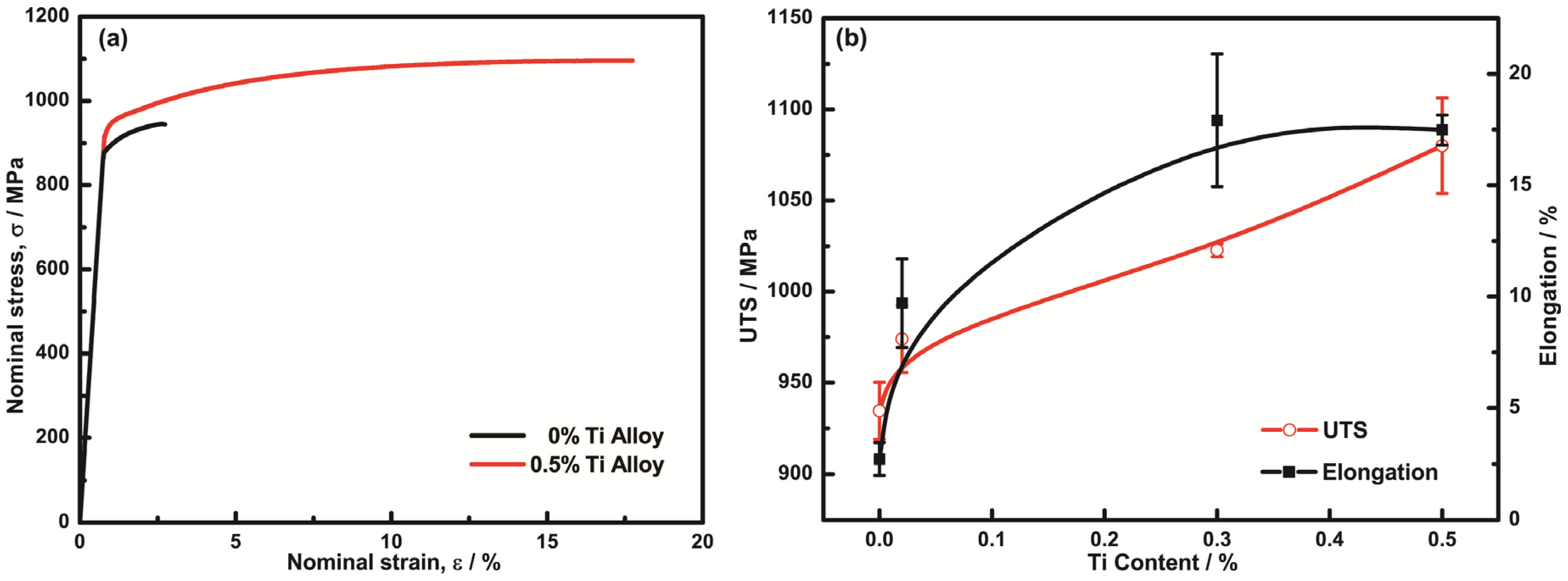
3.2. Mechanical Properties
4. Conclusions
- (1)
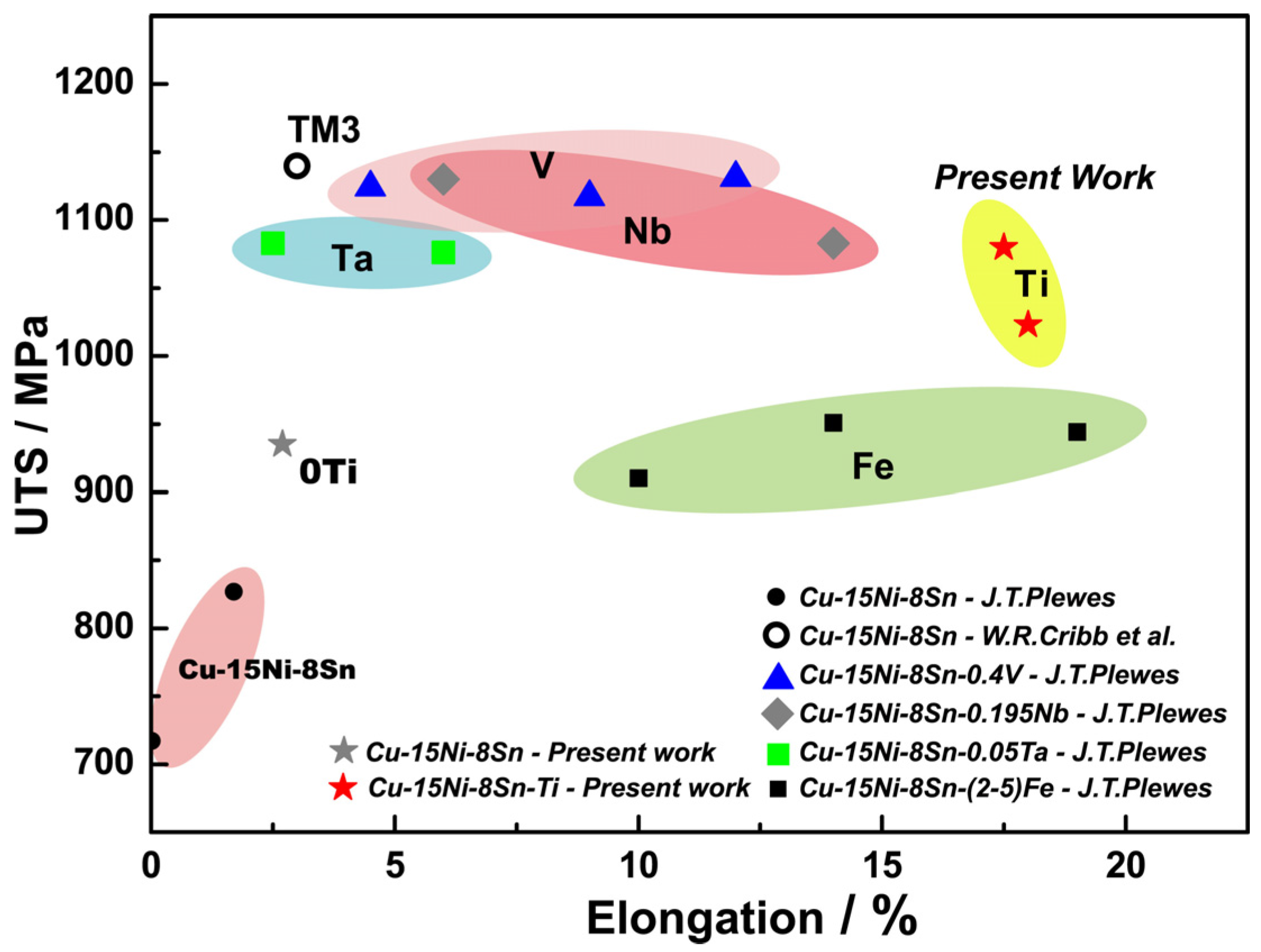
- Cu-15Ni-8Sn alloys with the addition of Ti possess an excellent combination of strength and ductility. Compared to the alloy without Ti, the tensile strength and the elongation of the alloy with 0.3% Ti increased from 935 MPa to 1024 MPa and from 2.7% to 17.9%, respectively.
- (2)
- The improvement of mechanical properties by the addition of Ti contributed to grain refinement and the suppression of discontinuous precipitation. By the addition of Ti, Ni3Ti phase was formed in the solidification process, which was undissolved in the following solution treatment. Ni3Ti phase had a pining effect on grain boundary migration in the process of solution treatment, and thus resulted in grain refinement. Ti addition also had a suppressive effect on discontinuous precipitation, owing to a reduction of nucleation sites of discontinuous precipitation.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shankar, K.V.; Sellamuthu, R. Determination on the effect of Tin content on microstructure hardness, optimum aging temperature and aging time for spinodal bronze alloys cast in metal mold. Int. J. Metalcast. 2016, 26, 1–6. [Google Scholar] [CrossRef]
- Kim, S.S.; Rhu, J.C.; Jung, Y.C.; Jung, Y.C.; Han, S.Z.; Kim, C.J. Aging characteristics of thermomechanically processed Cu-9Ni-6Sn alloy. Scr. Mater. 1999, 40, 1–6. [Google Scholar] [CrossRef]
- Plewes, J.T. High-strength Cu-Ni-Sn alloys by thermomechanical processing. Metall. Mater. Trans. A. 1975, 6A, 537–544. [Google Scholar] [CrossRef]
- Cribb, W.R. Copper spinodal alloys for aerospace. Adv. Mater. Process. 2006, 6, 44. [Google Scholar]
- Cribb, W.R.; Ratka, J.O. Copper spinodal alloys. Adv. Mater. Process. 2002, 12, 1–4. [Google Scholar]
- Kar, N.J. Copper-Based Spinodal Alloy Bearings. U.S. Patent 4,641,976, 10 February 1987. [Google Scholar]
- Caris, J.; Varadarajan, R.; Stephens, J.J., Jr.; Lewandowski, J.J. Microstructure effects on tension and fatigue behavior of Cu-15Ni-8Sn sheet. Mater. Sci. Eng. A 2008, 491, 137–146. [Google Scholar] [CrossRef]
- Plewes, J.T. Method for Producing Copper Based Spinodal Alloys. U.S. Patent 4,260,432, 7 April 1981. [Google Scholar]
- Plewes, J.T. Quaternary Spinodal Copper Alloys. U.S. Patent 4,052,204, 4 October 1977. [Google Scholar]
- Cribb, W.R.; Gedeon, M.J.; Grensing, F.C. Performance advances in Copper-Nickel-Tin spinodal alloys. Adv. Mater. Process. 2013, 9, 20–25. [Google Scholar]
- Manna, I.; Pabi, S.K.; Gust, W. Discontinuous reaction in solids. Int. Mater. Rev. 2001, 46, 53–91. [Google Scholar] [CrossRef]
- Kotval, P.S.; Hatwell, H. Discontinuous precipitation of M23C6 carbide in a Nickel-base superalloy. Trans. AIME 1969, 245, 1821–1823. [Google Scholar]
- Spooner, S.; Lefever, B.G. The effect of prior deformation on spinodal age hardening in Cu-15Ni-8Sn alloy. Metall. Mater. Trans. A 1980, 11A, 1085–1093. [Google Scholar] [CrossRef]
- Lefevre, B.G.; D’annessa, A.T.; Kalish, D. Age hardening in Cu-15Ni-8Sn alloy. Metall. Mater. Trans. A 1978, 9A, 577–586. [Google Scholar] [CrossRef]
- Miki, M.; Ogino, Y. Effect of Si addition on the cellular precipitation in a Cu-10Ni-8Sn alloy. Mater. Trans. JIM 1990, 31, 968–974. [Google Scholar] [CrossRef]
- Miki, M.; Ogino, Y. Influence of solution-treatment conditions on the cellular precipitation in Si-doped Cu-10Ni-8Sn alloy. Mater. Trans. JIM 1991, 32, 1135–1140. [Google Scholar] [CrossRef]
- Miki, M.; Ogino, Y. Effects of doped elements on the cellular precipitation in Cu-10Ni-8Sn alloy. Mater. Trans. JIM 1994, 35, 313–318. [Google Scholar] [CrossRef]
- Wang, Y.H.; Wang, M.P.; Hong, B.; Li, Z. Microstructure and properties of Cu-15Ni-8Sn-0.4Si alloy. Trans. Nonferrous Met. Soc. China 2003, 13, 1051–1055. [Google Scholar]
- Weng, Y.H.; Liu, Y.X.; Tan, W.; Zhang, W.W.; Luo, Z.Q. Influence of extrusion ratio on microstructure and properties of Cu-17Ni-3Al-X alloy. Appl. Mech. Mater. 2014, 477–478, 1307–1315. [Google Scholar] [CrossRef]
- He, W.X.; Wang, E.D.; Hu, L.X.; Yu, Y.; Sun, H.F. Effect of extrusion on microstructure and properties of a submicron crystalline Cu-5wt%Cr alloy. J. Mater. Process. Technol. 2008, 208, 205–210. [Google Scholar] [CrossRef]
- Zhao, J.C.; Notis, M.R. Spinodal decomposition, ordering transformation and discontinuous precipitation in a Cu-15Ni-8Sn alloy. Acta Mater. 1998, 46, 4203–4218. [Google Scholar] [CrossRef]
- Tu, K.N.; Turnbull, D. Morphology of cellular precipitation of tin from Lead-Tin bicrystals. Acta Metall. 1967, 15, 369–376 and 1317–1323. [Google Scholar] [CrossRef]
- Hornbogen, E. Systematics of the Cellular Precipitation Reactions. Metall. Trans. 1927, 3, 2717–2727. [Google Scholar] [CrossRef]
- Harmer, M.P.; Rohrer, G.S. Grain boundary complexion—Current status and future directions. Curr. Opin. Solid State Mater. Sci. 2016, 20, 225–336. [Google Scholar]
- Dillon, S.J.; Tang, M.; Cater, W.C.; Harmer, M.P. Complexion: A new concept for kinetic engineering in materials science. Acta Mater. 2007, 55, 6208–6218. [Google Scholar] [CrossRef]
- Dillon, S.J.; Tai, K.P.; Chen, S. The importance of grain boundary complexions in affecting physical properties of polycrystals. Curr. Opin. Solid State Mater. Sci. 2016, 20, 324–335. [Google Scholar] [CrossRef]
- Schumacher, O.; Marvel, C.J.; Kelly, M.N.; Cantwell, P.R.; Vinci, R.P.; Rickman, J.M.; Rohrer, G.S.; Harmer, M.P. Complexion time-temperature-transformation (TTT) diagrams: Opportunities and challenges. Curr. Opin. Solid State Mater. Sci. 2016, 20, 316–323. [Google Scholar] [CrossRef]
- Watanabe, C.; Takeshita, S.; Monzen, R. Effect of small addition of Ti on strength and microstructure of a Cu-Ni-Si alloy. Metall. Mater. Trans. A 2015, 46A, 2469–2475. [Google Scholar] [CrossRef]
- Basinski, Z.S.; Dugdale, J.S.; Howie, A. The electrical resistivity of dislocation. Philos. Mag. 1963, 8, 1989–1997. [Google Scholar] [CrossRef]
- Komatsu, S. Resistivity of copper alloys, its interpretation and application. J. JRICu 2002, 41, 1–9. [Google Scholar]
- Argon, A.S. Strengthening Mechanism in Crystal Plasticity; Oxford University Press: Oxford, UK, 2008; pp. 365–375. [Google Scholar]





| Alloy Designation | wt./% | |||
|---|---|---|---|---|
| Ni | Sn | Ti | Cu | |
| Cu-15Ni-8Sn | 15.03 | 8.12 | - | balance |
| Cu-15Ni-8Sn-0.02Ti | 15.03 | 8.14 | 0.02 | balance |
| Cu-15Ni-8Sn-0.3Ti | 15.23 | 8.36 | 0.32 | balance |
| Cu-15Ni-8Sn-0.5Ti | 14.96 | 8.02 | 0.53 | balance |
| at./% | ||||
|---|---|---|---|---|
| Cu | Ni | Sn | Ti | |
| A | 47.81 | 40.29 | 11.90 | - |
| B | 80.35 | 15.56 | 4.09 | - |
| C | 40.23 | 45.95 | 4.02 | 9.80 |
| D | 7.75 | 68.40 | 2.98 | 20.87 |
| Alloy | Electrical Conductivity (pct IACS 1) | ||
|---|---|---|---|
| E0 | E1 | E2 | |
| Base | 41.20 | 27.29 | - |
| 0.3Ti | 49.72 | 27.19 | 18.57 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.; Zhang, W.; Wang, Z.; Li, D.; Luo, Z.; Yang, C.; Zhang, D. Improving the Mechanical Properties of Cu-15Ni-8Sn Alloys by Addition of Titanium. Materials 2017, 10, 1038. https://doi.org/10.3390/ma10091038
Zhao C, Zhang W, Wang Z, Li D, Luo Z, Yang C, Zhang D. Improving the Mechanical Properties of Cu-15Ni-8Sn Alloys by Addition of Titanium. Materials. 2017; 10(9):1038. https://doi.org/10.3390/ma10091038
Chicago/Turabian StyleZhao, Chao, Weiwen Zhang, Zhi Wang, Daoxi Li, Zongqiang Luo, Chao Yang, and Datong Zhang. 2017. "Improving the Mechanical Properties of Cu-15Ni-8Sn Alloys by Addition of Titanium" Materials 10, no. 9: 1038. https://doi.org/10.3390/ma10091038





