3.1. Methane Production
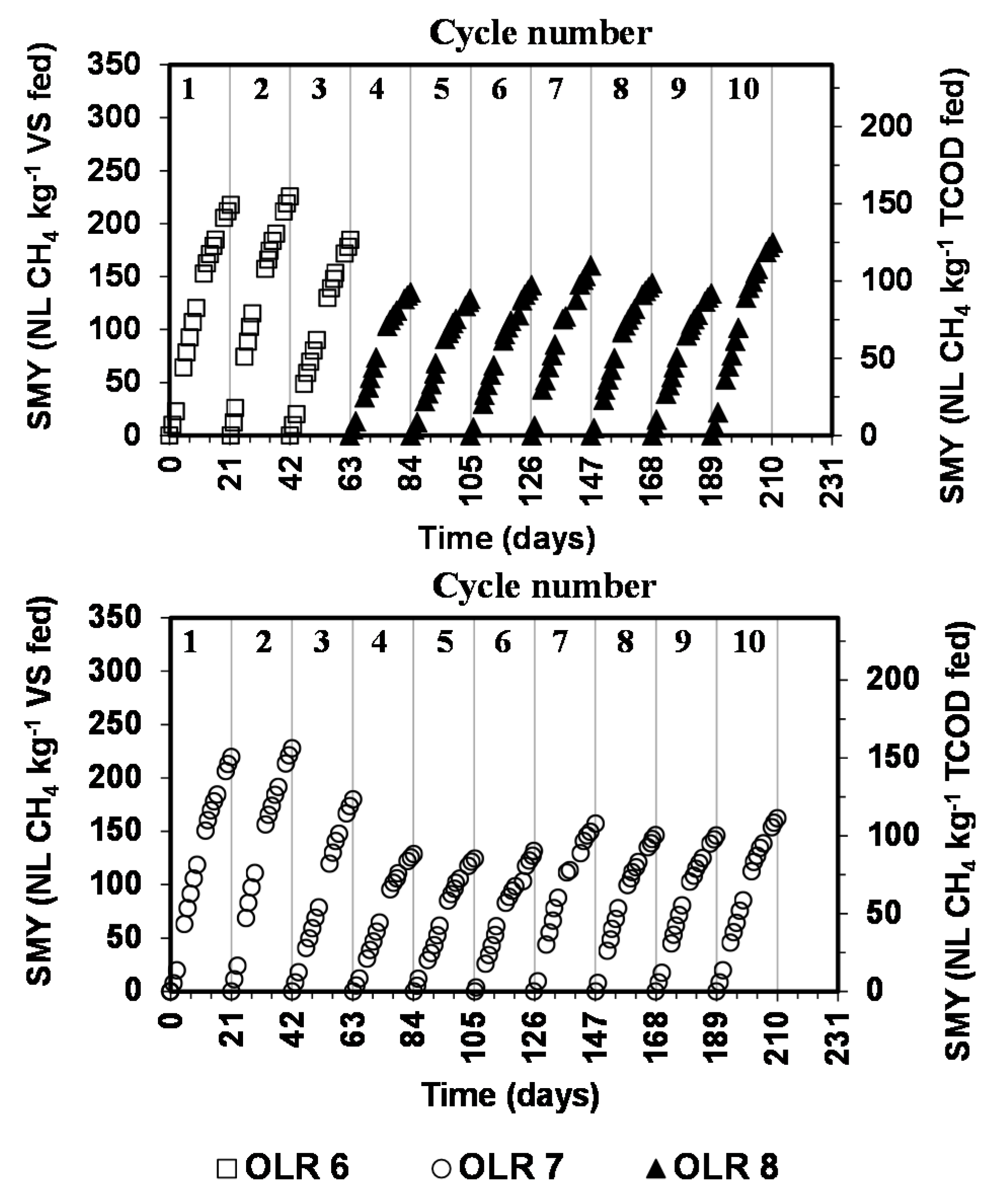
The operation of bioreactors was generally stable and biogas production proceeded steadily during the successive cycles. No inhibition or lag phase in biogas production has been observed. The profiles of methane production expressed as SMY (NL CH
4 kg
−1 VS fed) in the replicate bioreactors are shown in
Figure 2. The cumulative SMYs calculated during the successive cycles are given in
Table 5.
Figure 2.
Specific methane yield profiles for the psychrophilic anaerobic digestion of dairy cow feces. (Coefficient of variation between the duplicate bioreactors was less than 5%).
Figure 2.
Specific methane yield profiles for the psychrophilic anaerobic digestion of dairy cow feces. (Coefficient of variation between the duplicate bioreactors was less than 5%).
Table 5.
Rate and specific methane yield for the psychrophilic anaerobic digestion of cow feces (TS 11.0% to 16.5%).
Table 5.
Rate and specific methane yield for the psychrophilic anaerobic digestion of cow feces (TS 11.0% to 16.5%).
| Cycle | Organic Loading Rate (g TCOD kg−1 inoculum d−1) |
|---|
| 6.0 | 7.0 | 8.0 |
|---|
| SMY (NL CH4 kg−1 VS Fed) | CH4 Production Rate (NL CH4 kg−1 VS d−1) | SMY (NL CH4 kg−1 VS Fed) | CH4 Production Rate (NL CH4 kg−1 VS d−1) | SMY (NL CH4 kg−1 VS Fed) | CH4 Production Rate (NL CH4 kg−1 VS d−1) |
|---|
| 1 | 217.8 ± 6.0 a | 10.4 ± 1.1 | 219.6 ± 15.1 a | 10.5 ± 0.5 | – | – |
| 2 | 225.7 ± 1.2 a | 10.7 ± 1.4 | 227.9 ± 4.8 a | 10.9 ± 1.2 | – | – |
| 3 | 184.7 ± 0.5 a | 8.8 ± 0.4 | 179.9 ± 3.2 a | 8.6 ± 0.4 | – | – |
| 4 | – | – | 128.7 ± 3.4 a | 6.1 ± 0.5 | 135.1± 1.9 b | 6.4 ± 0.5 |
| 5 | – | – | 124.5 ± 1.4 a | 5.9 ± 0.5 | 128.9 ± 2.9 a | 6.1 ± 0.4 |
| 6 | – | – | 131.6 ± 3.8 a | 6.3 ± 0.4 | 141.9 ± 8.2 a | 6.8 ± 0.8 |
| 7 | – | – | 157.4 ± 0.4 a | 7.5 ± 0.9 | 160.4 ± 15.7 a | 7.6 ± 0.6 |
| 8 | – | – | 146.5 ± 4.1 a | 7.0 ± 1.0 | 143.7 ± 2.5 a | 6.8 ± 0.5 |
| 9 | – | – | 146.2 ± 3.4 a | 7.0 ± 0.8 | 134.0 ± 8.2 a | 6.4 ± 0.3 |
| 10 | – | – | 162.3 ± 2.4 a | 7.7 ± 0.4 | 182.2 ± 1.6 b | 8.7 ± 0.6 |
Two-tail t-test at 95% confidence level has been conducted on the values of SMY obtained in cycle 1–3 at OLR 6.0 and 7.0 g TCOD kg−1 inoculum d−1 where the same cow feces quality has been fed to the reactors operating at these two OLRs during each cycle. Similarly, two-tail t-test has also been conducted on the values of SMY obtained in cycle 4–10 at OLR 7.0 and 8.0 g TCOD kg−1 inoculum d−1 where the same cow feces quality has been fed to the reactors at operating these two OLRs during each cycle.
The SMYs obtained at OLR 6.0 and 7.0 g TCOD kg
−1 inoculum d
−1 during cycle 1, 2 and 3 (
Table 5), where the same cow manure has been fed to the bioreactors, are statistically similar to each other. The overall average of the SMY during the first 3 cycles was 209.4 ± 21.8 and 209.1 ± 25.6 NL CH
4 kg
−1 VS fed at OLR 6.0 and 7.0 g TCOD kg
−1 inoculum d
−1, respectively, and they are statistically similar.
Therefore, the variation in OLR between 6.0 and 7.0 did not influence the SMY. Similarly, the SMYs obtained at OLR 7.0 and 8.0 g TCOD kg−1 inoculum d−1 during cycle 5 to 9, where the same cow manure has been fed to the bioreactors operating at the these OLRs, are statistically similar to each other. The overall average of the SMY during the cycles 4 to 10 was 147.3 ± 14.8 and 149.5 ± 16.8 NL CH4 kg−1 VS fed at OLR 7.0 and 8.0 g TCOD kg−1 inoculum d−1, respectively, and they are statistically similar. Therefore, the variation in OLR between 7.0 and 8.0 did not influence the SMY. It appears that at this SIR, the inoculum is not overloaded and still has the ability to degrade substantial fraction of the biodegradable organics within the TCL of 21 days. However, further increase in OLR is expected to decrease the SMY until a maximum threshold OLR is reached when signs of instability are observed such as buildup of VFAs, pH drop, discontinuation of CH4 production. Notice that Tukey’s multiple comparisons test conducted at 95% confidence level indicated statistical differences in the quality of cow feces fed at OLR 7.0 and 8.0 g TCOD kg−1 inoculum d−1 (except for cycles 1 and 8–10 which were similar in terms of TCOD/VS ratio only). Therefore, variation in the SMY observed from cycle to cycle at the OLR applied was due to the variation in the quality (TCOD, VS, fiber content, and other biodegradable components) of cow feces fed.
Notice that at OLR of 7.0 g TCOD kg
−1 inoculum d
−1, the SMYs during the cycles 1, 2 and 3 were 219.6 ± 15.1, 227.9 ± 4.8, and 179.9 ± 3.2 NL CH
4 kg
−1 VS fed, respectively, because the cow feces fed had higher TCOD and VS compared to the feces used in the other cycles (4–10) as indicated in
Table 6. At the same OLR (7.0 g TCOD kg
−1 inoculum d
−1), the SMY during cycles 4 to 10 decreased to an average of 147.3 ± 14.8 NL CH
4 kg
−1 VS fed which is equivalent to around 66.6% of the average SMY obtained during cycles 1 to 3. Cow feces fed during the cycles 1, 2 and 3 had an average TCOD of 201.2 ± 16.7 g·kg
−1 feces and an average VS of 13.4% ± 0.8%, respectively, while cow feces fed during the cycles 4 to 10 had an average TCOD of 157.8 ± 11.6 g·kg
−1 and an average VS of 10.5% ± 0.93%, respectively (
Table 3). Moreover, because cow feces fed during cycle 1–3 had more VFAs than cow feces fed in cycles 4–10 (
Table 3) then the quantities of VFAs fed during cycle 1–3 were higher than those fed during cycle 4–10. The quality of cow feces has also been described in terms of mass of fiber fed, contents of cellulose, hemicellulose, and lignin, the (cellulose + hemicellulose)/lignin ratio and TCOD/VS ratio (
Table 6). The last two parameters (
i.e., (cellulose + hemicellulose)/lignin ratio and TCOD/VS ratio) were selected so that they provide standardized unbiased ratios to indicate the quality of cow feces.
Table 6.
Effects of quality of cow feces fed on specific methane yield.
Table 6.
Effects of quality of cow feces fed on specific methane yield.
| Cycle | SMY (NL CH4 kg−1 VS Fed) | TCOD/VS Ratio | Fiber Fed (kg) | (Cellulose + Hemicellulose)/ Lignin Ratio |
|---|
| 1 | 219.6 ± 15.1 a | 1.54 ± 0.00 a | 0.31 ± 0.00 a | 4.0 ± 0.00 a |
| 2–3 | 203.9 ± 33.9 a | 1.48 ± 0.00 b | 0.41 ± 0.00 b | 4.71 ± 0.00 b |
| 4–7 | 135.6 ± 14.9 b | 1.45 ± 0.02 c | 0.44 ± 0.02 c | 3.58 ± 0.23 c |
| 8–10 | 151.7 ± 9.2 b | 1.55 ± 0.01 a | 0.36 ± 0.01 d | 3.43 ± 0.00 c |
Generally, cow feces fed in cycles 1–3 had higher average contents of cellulose (25.51%) and less content of lignin (10.72%) in fiber compared to cow feces fed in cycles 4–10 (cellulose (24.16%) and lignin (12.98%)). This is reflected by (Cellulose + hemicellulose)/lignin ratio of 4.0 and 4.71 during cycles 1 and 2–3, respectively, compared to 3.58 and 3.43 during cycles 4–7 and 8–10, respectively. Lignin builds complexes with cellulose and hemicellulose, thus creates a physical barrier making it difficult for microbial enzymes to break down the organic compounds in short time. Therefore the higher the lignin quantity the harder is the degradation of cellulose and hemicellulose.
Although the comparative parameters were summarized in
Table 3 and
Table 6 they were not sufficient to quantitatively explain the reduction in the SMY. These parameters measure the quantity of organics but do not measure their biodegradability or quality. TCOD and VS tests do not distinguish between fiber, lipids, fat, proteins, amino acids,
etc. The content of these components which have not been measured might have been different and caused the difference in SMY between cycles 1–3 and 4–10. Notice that the theoretical methane potential per gram of VS is significantly greater for fat compared to proteins and carbohydrate (1014, 496, and 415 NL CH
4 kg
−1 VS, respectively) [
21]. These differences collectively caused that the SMY during cycles 1–3 was higher than that obtained in cycles 4–10.
The specific methane production rate obtained at OLR 6.0 and 7.0 g TCOD kg
−1 inoculum d
−1 during cycles 1, 2 and 3 (
Table 5) were similar to each other. The overall average of the specific methane production rate during the first 3 cycles was 10.0 ± 1.0 and 10.0 ± 1.2 NL CH
4 kg
−1 VS fed d
−1 at OLR 6.0 and 7.0 g TCOD kg
−1 inoculum d
−1, respectively. Similarly, the specific methane production rate obtained at OLR 7.0 and 8.0 g TCOD kg
−1 inoculum d
−1 during cycle 4 to 10 were similar to each other. The overall average of the specific methane production rate during the cycles (4 to 10) was 6.8 ± 0.7 and 7.0 ± 0.9 NL CH
4 kg
−1 VS fed d
−1 at OLR 7.0 and 8.0 g TCOD kg
−1 inoculum d
−1, respectively.
The data available in the accessible literature on the performance of psychrophilic high solids content anaerobic digestion (PDAD) is very scarce; therefore, the results have been compared to the performance of mesophilic and thermophilic DAD of cow manure (
Table 7). The average yield of 209.4 ± 21.8 NL CH
4 kg
−1 VS fed obtained at OLR of 6.0 g TCOD kg
−1 VS d
−1 (equivalent to 4.0 ± 0.1 g VS fed kg
−1·d
−1) in this study after 21 days of psychrophilic (20 °C) incubation is greater than the yield 161 NL CH
4 kg
−1 VS of dairy cattle feces fed at OLR of 1.4 g VS substrate kg
−1 VS inoculum reported by Møller
et al. [
22] at 35 °C and HRT of 40 days. Similarly, the SMYs of 157.8 ± 11.6 and 147.3 ± 14.8 NL CH
4 kg
−1 VS fed obtained in this study at OLR of 7.0 and 8.0 g TCOD kg
−1 VS d
−1 are within the range of the SMY 135 and 164 L CH
4 kg
−1 VS obtained during mesophilic (30 °C) anaerobic digestion of dairy cattle manure reported by Somayaji and Khanna [
23], and Shyam [
24] at HRT of 40 and 50 days, respectively.
However, the average SMYs (162.5 ± 38.6, and 140.7 ± 11.1 NL CH
4 kg
−1 VS fed) obtained at OLR of 7.0, and 8.0 g TCOD kg
−1 VS d
−1 (equivalent to 4.8 ± 0.2 and 5.7 ± 0.3 kg VS fed m
−3·d
−1, respectively) in this study are comparable to the yields reported by Møller
et al. [
22], Somayaji and Khanna [
23], and Shyam [
24]. For the same incubation time, Schafer
et al. [
25] reported a SMY of 85 L CH
4 kg
−1 VS from beef cattle manure in two-stage (hydrolysis-methanogenesis) reactor with OLR of 6 and 3 kg VS m
−3·d
−1 for hydrolysis and methanogenesis stages, respectively. The average SMY obtained in this study (209.4 ± 21.8 NL CH
4 kg
−1 VS fed) at OLR of 6.0 g TCOD kg
−1 inoculum d
−1 is less than those reported by Varel
et al. [
26] [240–280 L CH
4 kg
−1 VS] at temperatures between 35 and 65 °C, HRT of 18 days, and OLR of 3.3 kg VS m
−3·d
−1 (
Table 7). Compared to Varel
et al.’s results [
26] for operations at 35 to 65 °C, the current study demonstrated a potential technology which minimizes energy required for heating the bioreactor. Achieving a stable psychrophilic anaerobic digestion of undiluted cow manure at increased OLR (6.0 to 8.0 g TCOD kg
−1 inoculum d
−1) over long-term operation is a significant improvement with potential market in cold climate area.
The specific methane yields obtained in this study demonstrate that the novel PDAD of cow feces in sequence batch reactor is practically feasible and as efficient as mesophilic and thermophilic anaerobic digestion given that a well-acclimatized inoculum is developed and maintained. Since the results of this study showed that there was no difference in SMY obtained at OLR 6.0 to 8.0 g TCOD kg−1 inoculum d−1 then there is a potential to increase the OLR further. It is important to define the threshold of maximum OLR, which allows for using a maximum proportion of substrate and minimum proportion of inoculum compared to those used in this study without jeopardizing decreasing the SMY. This objective could be addressed in future research.
Table 7.
Specific methane yield (L CH4 kg−1 VS) of cow feces at different temperatures and total solids.
Table 7.
Specific methane yield (L CH4 kg−1 VS) of cow feces at different temperatures and total solids.
| Substrate | OLR (g VS kg−1 Inoculum d−1) unless Indicated otherwise | HRT (day) | TS (%) | Temperature (°C) | Reference |
|---|
| 15 | 20 | 30 | 35 | 40 | 50 | 55 | 60 | 65 |
|---|
| Dairy cow manure | 6.0 g TCOD L−1 | 21 | 11–16 | – | 209.4 ± 21.8 a | – | – | – | – | – | – | – | This study |
| Dairy cow manure | 7.0 g TCOD L−1 | 21 | 11–16 | – | 147.3 ± 14.8 a | – | – | – | – | – | – | – | This study |
| Dairy cow manure | 8.0 g TCOD L−1 | 21 | 11–16 | – | 149.5 ± 16.8 a | – | – | – | – | – | – | – | This study |
| Cow dung | 5.3 b | 84 | 13.1–14.7 | 174 | – | – | – | – | – | – | – | – | Zhu and Jha [27] |
| Cow dung | 10.7 b | 63 | 15.2 | – | – | – | 167 | – | – | 188 | – | – | Jha et al. [13] |
| Cow dung | 10.7 b | 63 | 7.7 | – | – | – | 194 | – | – | 226 | – | – | Jha et al. [13] |
| Cow dung (no mixing) | 18.6 b | 35 | 16.2 | – | – | – | 127 | – | – | – | – | – | Jha et al. [14] |
| Cow dung with mixing | 18.6 b | 35 | 16.2 | – | – | – | 150 | – | – | – | – | – | Jha et al. [14] |
| Feedlot manure | NR | 40 | 16.3 | – | – | – | 9.2 | – | – | – | – | – | Borhan et al. [28] |
| Cattle manure | 3.3 | 18 | – | – | – | – | 260 | 270 | 250 | 280 | 270 | 240 | Varel et al. [26] |
| Cattle manure | 3.0 | 15 | – | – | – | – | – | – | – | 202 | – | 165 | Ahring et al. [29] |
| Cattle manure | 2.0 | 20 | – | – | – | – | – | – | 260 | – | 230 | – | El-Mashad et al. [5] |
| Dairy manure | NR | 30 | – | – | – | – | 242.7 | – | – | – | – | – | Labatut et al. [30] |
| Cattle manure | NR | 40 | – | – | – | 135 | – | – | – | – | – | – | Somayaji and Khanna [23] |
| Cattle manure | NR | 50 | – | – | – | 164 | – | – | – | – | – | – | Shyam [24] |
| Cattle manure | NR | 100 | – | – | – | 230 | – | – | – | – | – | – | Shyam [24] |
| Dairy cattle manure | NR | 100 | – | – | – | – | 148 ± 41 | – | – | – | – | – | Møller et al. [22] |
| Cow manure | NR | 16 | – | – | – | – | 128 | – | – | – | – | – | Preeti Rao and Seenayya [31] |
| Cow manure | NR | 90 | – | – | – | – | 260 c | – | – | – | – | – | Guengoer-Demirici et al. [32] |
3.2. Volatile Fatty Acids (VFAs) Production
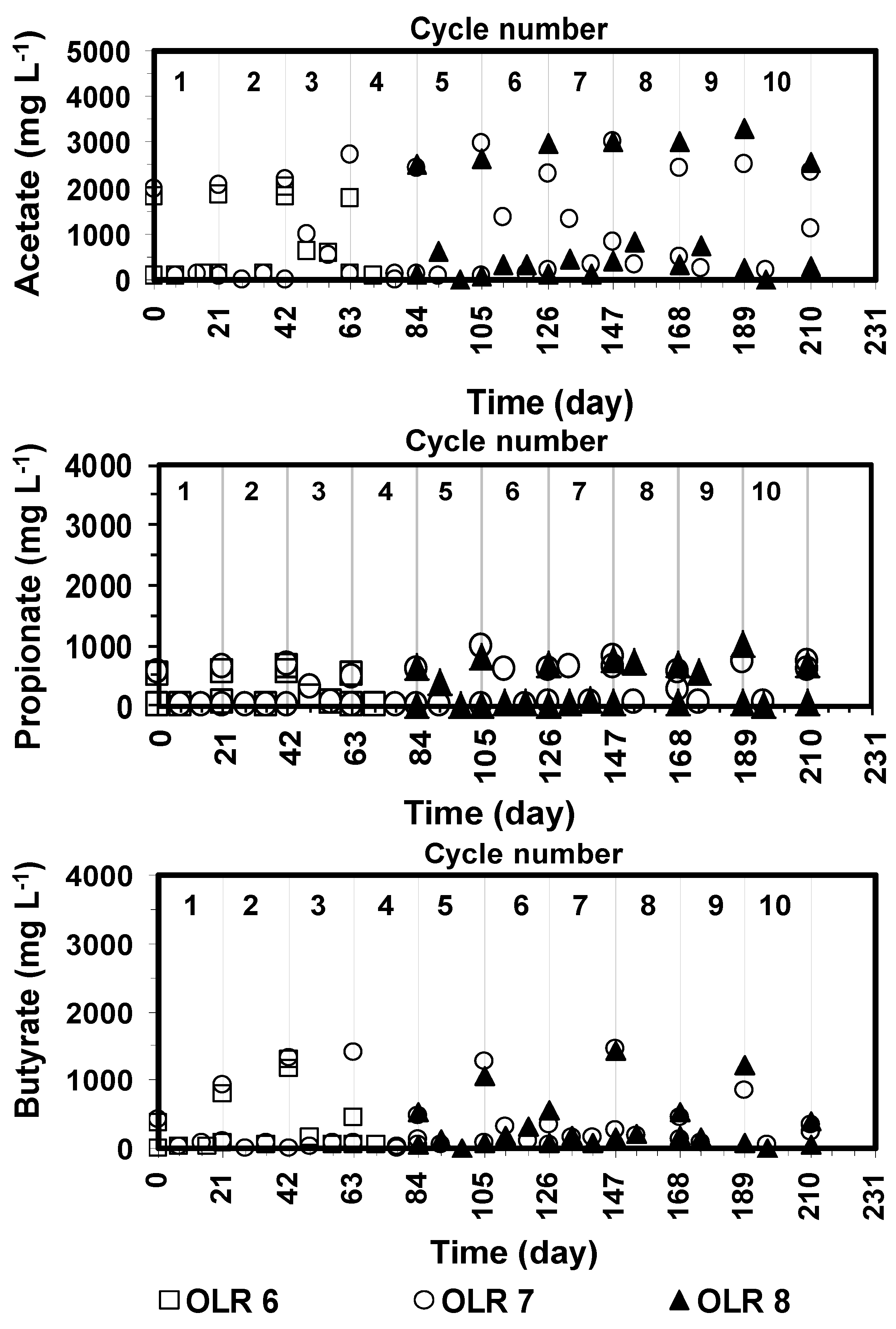
The profiles of acetic, propionic, and butyric acids concentrations on day 0, 7 and 21 are shown in
Figure 3. Samples at the beginning of each cycle (day 0) represents the ISM while those at the end of the treatment cycle (day 21) represent the digestate which has been used as inoculum for the next cycle. The average concentrations of iso-butyric, iso-valeric, valeric, and caproic acid were 60 ± 5, 58 ± 7, 28 ± 5, and 22 ± 6 mg·L
−1, respectively, throughout the successive cycles.
Generally, the profiles of VFAs produced during different cycles were almost identical. Throughout the successive cycles, acetic acid concentration peaked immediately after feeding to levels ranging between 2000 and 3500 mg·L
−1 but was consumed within a week in all bioreactors to levels of about 500 mg·L
−1 and to within 100 ± 50 mg·L
−1 after two weeks, as can be observed from
Figure 3, indicating that methanogenesis from acetate was not a rate limiting step. Similarly, propionic acid peaked to around 500 mg·L
−1 after feedings at OLR of 6.0 g TCOD kg
−1 inoculum d
−1 and to around 750–1000 mg·L
−1 after feedings at OLR of 7.0 and 8.0 g TCOD kg
−1·d
−1. However, it was consumed within a week to levels close to the detection limits of the instrument (25 ± 10 mg·L
−1).
Figure 3.
VFAs profiles during for the psychrophilic anaerobic digestion of dairy cow feces.
Figure 3.
VFAs profiles during for the psychrophilic anaerobic digestion of dairy cow feces.
The profile of propionic acid in the bioreactors was similar to that of acetic acid. In addition, butyric acid peaked to around 500–1500 mg·L−1 after feedings and was consumed within a week to levels within 250 mg·L−1. The concentrations of other volatile fatty acids (isobutyric-, iso-valeric-, valeric-, and caproic-acid) were between 100 and 250 mg·L−1 immediately after feeding and less than 50 mg·L−1 during the remaining time of the cycles.
Inhibitory effects of VFAs depend among other factors on pH, alkalinity, and type of culture [
33]. The VFAs toxicity increases with drop in pH. Salces [
34] reported that acetate and propionate concentrations of 2851 and 745 mg·L
−1 caused 50% inhibition in methane production at pH 7.0 while these concentrations increased to 8976 and 2358 mg·L
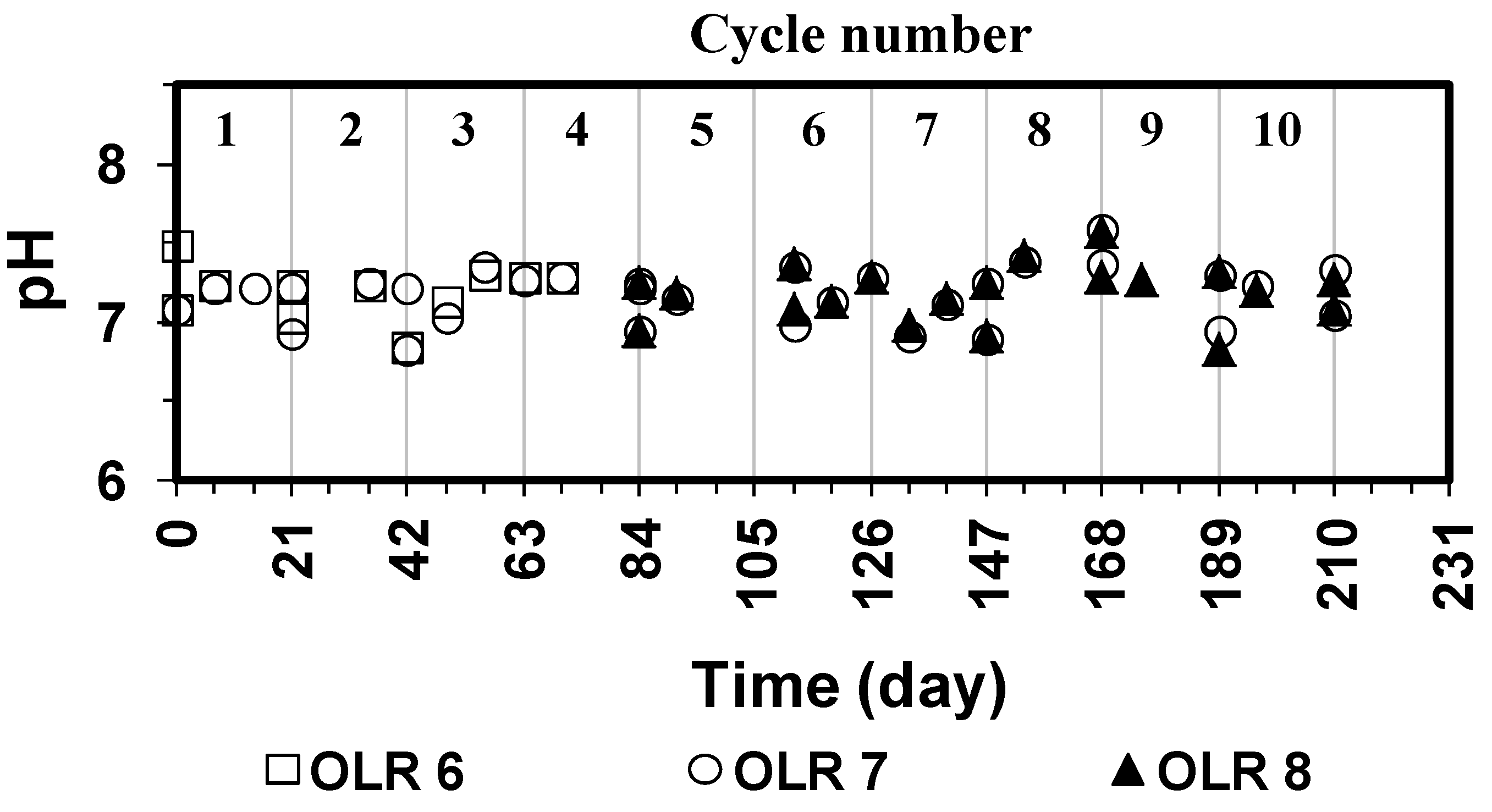
−1, respectively, at pH 7.5. The pH profile (
Figure 4) ranged between 6.8 at the beginning of the cycle (day 0) and 7.5 at the end of the cycle (day 21). The stability of pH was due to the sufficient buffering capacity of cow feces fed (alkalinity of 6900 to 9100 mg CaCO
3 kg
−1), that of the reactor content (8800 to 12600 mg CaCO
3 kg
−1) and absence of VFAs accumulation. The concentration of alkalinity in the reactor was generally in the same level or greater than that in the cow feces because methane producing reaction generates alkalinity as a by-product.
Figure 4.
Profile of pH during for the psychrophilic anaerobic digestion of dairy cow feces.
Figure 4.
Profile of pH during for the psychrophilic anaerobic digestion of dairy cow feces.
Concentrations of VFAs were always below the inhibitory levels (
Figure 3). Since no fluctuation has been observed in the rate of biogas production (
Figure 2) then there was no cause of inhibition whether due to VFAs accumulation, pH drop, ammonia inhibition or other causes. The profiles of the VFAs concentrations, the stable methane production and yield during the successive cycles indicate that acetogenic and methanogenic reactions proceeded fairly well.