An Improved CO2 Separation and Purification System Based on Cryogenic Separation and Distillation Theory
Abstract
: In this study, an improved CO2 separation and purification system is proposed based on in-depth analyses of cryogenic separation and distillation theory as well as the phase transition characteristics of gas mixtures containing CO2. Multi-stage compression, refrigeration, and separation are adopted to separate the majority of the CO2 from the gas mixture with relatively low energy penalty and high purity. Subsequently, the separated crude liquid CO2 is distilled under high pressure and near ambient temperature conditions so that low energy penalty purification is achieved. Simulation results indicate that the specific energy consumption for CO2 capture is only 0.425 MJ/kgCO2 with 99.9% CO2 purity for the product. Techno-economic analysis shows that the total plant investment is relatively low. Given its technical maturity and great potential in large-scale production, compared to conventional MEA and Selexol™ absorption methods, the cost of CO2 capture of the proposed system is reduced by 57.2% and 45.9%, respectively. The result of this study can serve as a novel approach to recovering CO2 from high CO2 concentration gas mixtures.1. Introduction
One of the most sophisticated challenges in environmental protection in the 21st century is global warming, which is caused by large amounts of greenhouse gas emissions, especially CO2. Measures must be taken to reduce CO2 emissions and consequently restrain global warming. From the perspective of energy utilization, carbon capture and storage (CCS) is considered to be one of the most significant methods in CO2 reduction [1], since it is reported that 90% of CO2 emissions are generated by the combustion of fossil fuels, which will be extensively used in the foreseeable future [2].
Currently, several primary CO2 capture and recovery methods are available: absorption (including chemical absorption and physical absorption), adsorption, membrane separation, and cryogenic separation [3–5]. Among these methods, chemical absorption can separate large amounts of high purity CO2 from low concentration flue gas, but high energy penalty and huge investments are expected [5–7]. Physical absorption is an effective approach to recovering low purity CO2 with low energy penalty, but additional energy is needed for sequent compression because the separated CO2 is in the gas state [7–9]. Both absorption methods draw extensive attention because of their high technical maturity [5–9]. Adsorption and membrane separation are recognized as promising CO2 capture methods despite inevitable problems such as low processing ability and high investment because of their operational feasibility and low separation energy penalty [10–16].
Cryogenic separation is a physical process that separates CO2 under extremely low temperature. It enables direct production of liquid CO2 at a low pressure, so that the liquid CO2 can be stored or sequestered via liquid pumping instead of compression of gaseous CO2 to a very high pressure, thereby saving on compression energy [17–20]. During the cryogenic separation process, the components of gas mixtures are separated by a series of compression, refrigeration, and separation steps. Since all these steps are highly mature technologies in the chemical industry, their operation and design feasibility can be guaranteed [20–22]. The cryogenic separation process requires no chemical agent, hence avoiding secondary pollution [17–22]. As far as industrial application is concerned, gas mixtures are usually composed of CO2 and other gases, the boiling points of which are relatively low. These gases include H2, N2, O2, Ar, and CH4. These impurities lower the phase transition temperature of CO2, which can even drop to under –80 °C. In this case, the refrigeration energy penalty increases substantially, and CO2 frost formation becomes highly possible, thereby threatening equipment safety [23]. Attention should thus be paid to raising the phase transition temperature of CO2 to improve the cryogenic separation method and consequently avoid facility freezing problems and high energy penalty [24–27].
Recently, many studies concerning cryogenic CO2 separation methods have been conducted. For instance, Besong et al. [28] proposed a cryogenic liquefaction system whose mainstay is formed by compressor and flash unit, so the energy penalty decreases due to sufficient recovery of cold energy. Song et al. [29] developed a novel CO2 capture process based on a Stirling cooler, whereby CO2 is separated in liquid state after continuous cooling down by three Stirling coolers. Jana [30] researched the integration and optimization of a CO2 capture system, and discussed the influences of several parameters on system performance. Based on the phase transition mechanism and the principle of energy cascade utilization, in a previous work we presented a novel system that simultaneously fulfills CO2 separation and compression by adopting multi-stage compression and separation. Compared with conventional CO2 capture methods, this novel system shows superior performance with CO2-H2 mixture and reduces the CO2 recovery energy penalty by 65% and 15%, respectively [31].
Interestingly, the studies mentioned above mainly focus on achieving high CO2 capture rates and low recovery energy penalties, whereas little attention is paid to the purity of the captured CO2. In fact, CO2 purity in the product separated by the cryogenic separation method might be relatively low. For example, when applying the cryogenic separation method to separate CO2 from CO2-N2-O2-Ar mixtures, the impurity content in the separated liquid can be as high as 2% to 5%; at this level, the CO2 purity cannot satisfy the requirements of most industrial applications, as well as transport and storage [1,32,33].
In the present work, we propose an improved CO2 separation and purification system that can separate the majority of the CO2 in liquid state from the mixed gases with relatively low energy penalty via multi-stage compression, refrigeration, and separation. Furthermore, by introducing high pressure and near ambient temperature distillation into the improved system, CO2 purity in the final product reaches 99.9%.
2. Proposal of the Cryogenic Separation Method
2.1. Phase Transition Characteristics of Mixed Gases Containing CO2
In our previous works, the phase transition characteristics of CO2-H2 mixture (common in the syngas generated by shift reaction) were analyzed. Results indicate that CO2 separation ratio is determined by two critical factors: the initial CO2 concentration and the initial pressure of the gas mixture [31]. In the present study, we analyze the CO2-N2-O2-Ar mixture, which is common in oxy-fuel combustion.
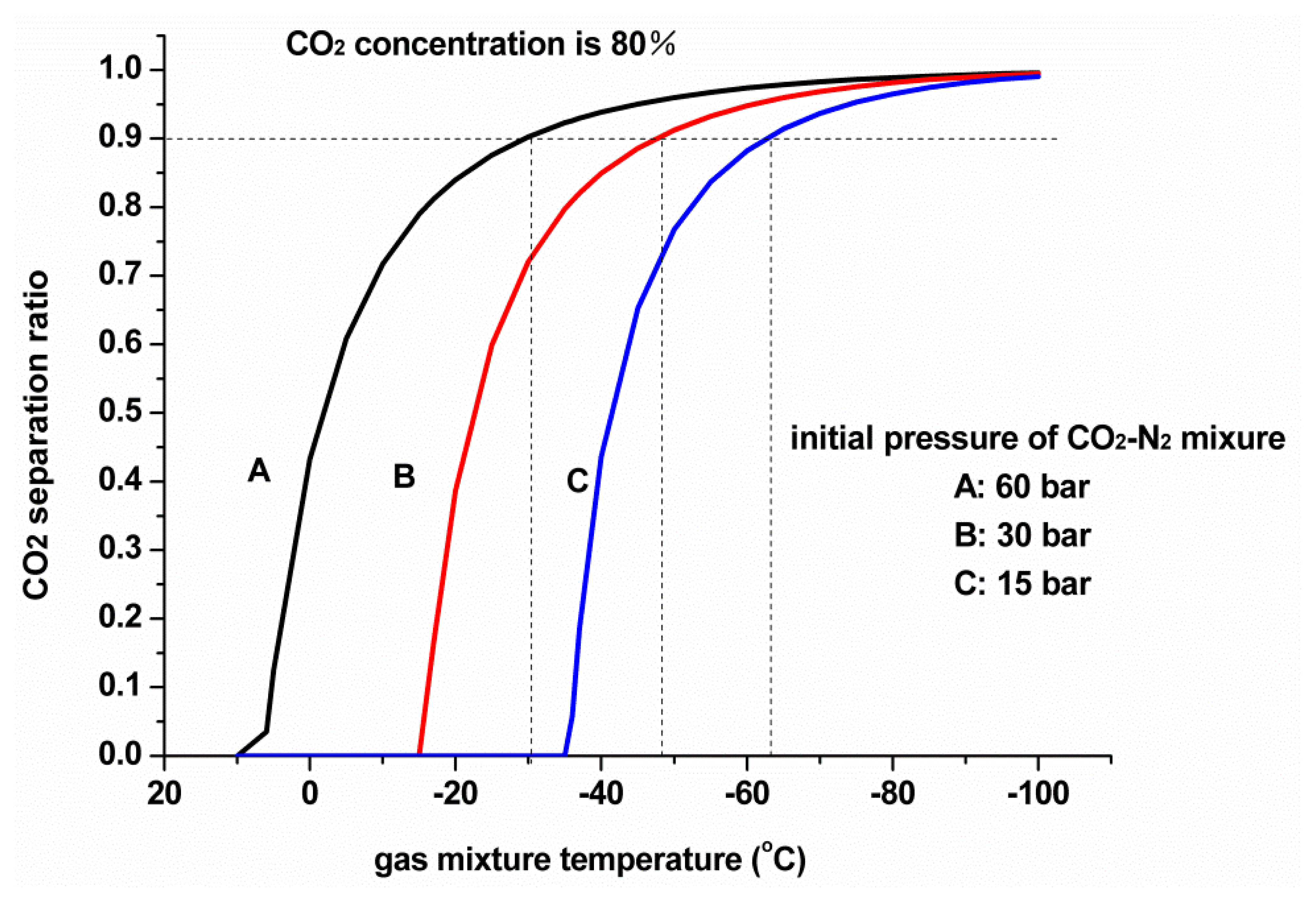
Figure 1 presents the relationship between the CO2 separation ratio and the temperature of CO2-N2 mixtures under different initial pressures, at an initial CO2 concentration of 80%.
The CO2 separation ratio increases as the initial pressure rises. Under the initial pressures of 15, 30, and 60 bar, to separate 90% CO2 from the gas mixture, the temperature must be dropped to approximately –63 °C, –48 °C, and –30 °C, respectively, so increasing the initial pressure is an effective approach for improving the performance of the cryogenic separation method. Especially after the gas mixture enters the cryogenic CO2 separation unit, the CO2 concentration in the gas mixture continuously declines with CO2 condensation. If the total pressure of the gas mixture could be increased at this moment, then CO2 partial pressure will also increase, which is very important in maintaining the liquefaction temperature of CO2 at a high level.
2.2. CO2 Purity Characteristics of the Cryogenic Separation Method
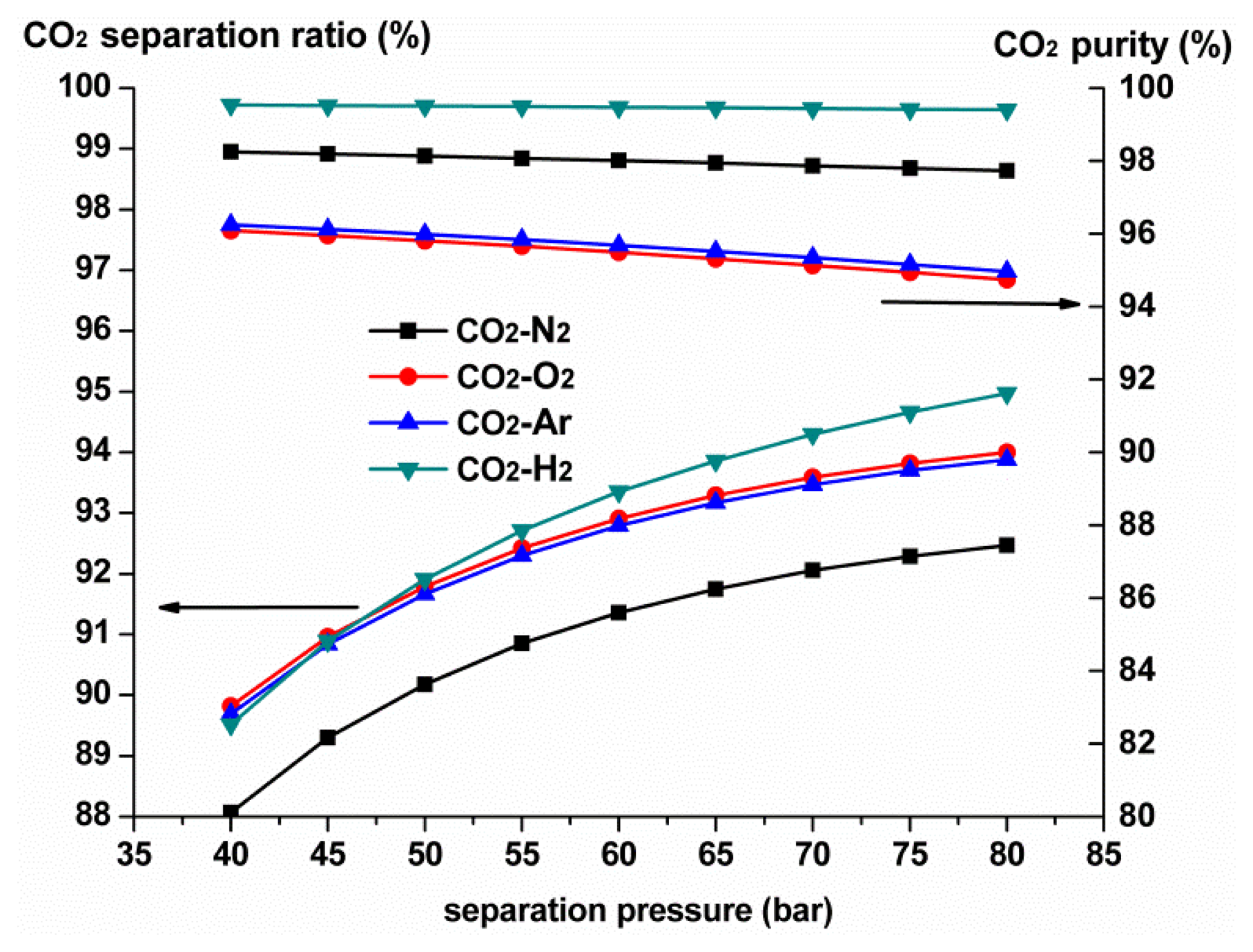
Generally, a small amount of impurities always dissolve in the liquid CO2 separated under high pressure, and the higher separation pressure, the larger the amount of impurities [28]. Figure 2 shows the variation in CO2 purity and separation ratio under different separation pressures with four kinds of typical impurity compositions, at the initial CO2 concentration of 80%. The following conclusions can be drawn based on Figure 2. On the one hand, the CO2 separation ratio constantly increases with the increment of separation pressure, whereas the CO2 purity in the product decreases. On the other hand, different impurity compositions have different effects on the CO2 purity in the product. At the same separation pressure of 60 bar and initial CO2 concentration of 80%, the CO2 purity in the product of the CO2-H2 mixture is 99.47%, for the CO2-N2 mixture it's 98.01%, whereas for CO2-O2 and CO2-Ar mixtures, it sharply reduces to 95.5% and 95.69%, respectively. This is because there exist significant differences in the physical properties of the different impurity gases, which affect the thermodynamic properties such as dew and bubble points, heat capacity, enthalpy and entropy of the CO2 mixture, so the operating conditions and separation performance of the purification process will thus vary accordingly, resulting in different CO2 purity in the product [27]. Generally, if the physical properties of the impurity gas are distinguished from those of the CO2 (H2 for example), it is easier to separate them by high pressure cryogenic separation [31]. However, for gas mixtures consisting of CO2, N2, O2, and Ar, the CO2 purity in the product attained by high pressure cryogenic separation is too low to satisfy the requirements of most industrial applications as well as transport and storage. Further purification measures should thus be considered.
3. Distillation Mechanism and Feasibility Analysis
3.1. Distillation Mechanism
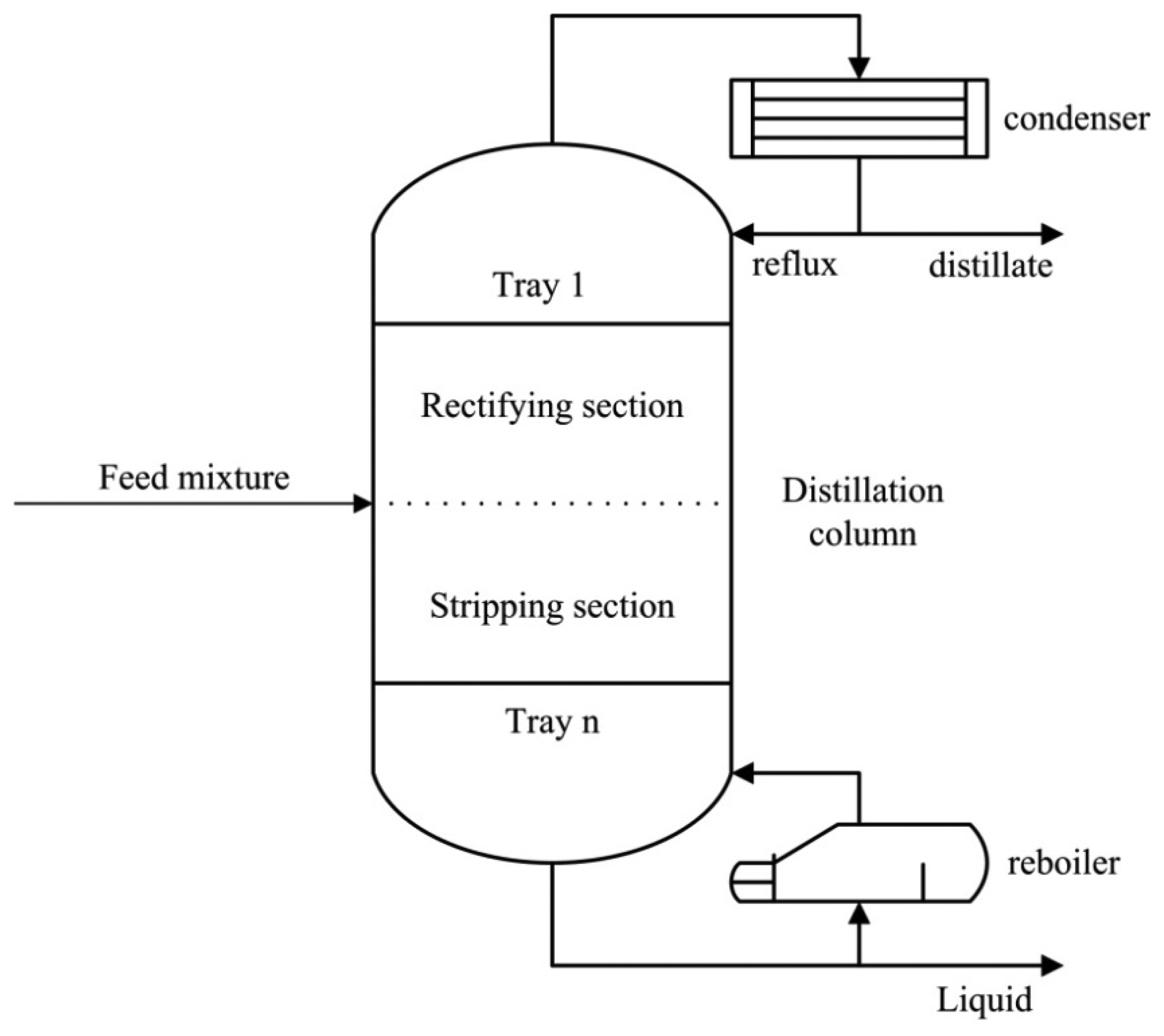
Distillation, which is the workhorse of chemical process industries, is widely used because of its high technical maturity [34,35]. It separates gas or liquid mixtures via consecutive partial vaporization and condensation in a distillation column. Figure 3 illustrates a simplified layout of the conventional distillation process. A feed mixture enters the column from the intermediate section. After condensing by the condenser installed on top of the column, part of the condensed liquid is refluxed, while the rest is discharged as distillate. Generally, the feed entrance divides the distillation column into two sections. The upper section is called the rectifying section, where the rising steam passes through the trays and comes in contact with the refluxed liquid to realize the material transfer and densification of volatile components [36]. Underneath the entrance is the stripping section, where the steam is heated by the reboiler located at the bottom of the column. Energy and material transfer proceeds as long as the heated steam is in countercurrent contact with the descending liquid, thus resulting in the accumulation of involatile components at the bottom.
3.2. Feasibility Analysis of Purifying CO2 Mixture by Conventional Distillation
Certain conditions must be met when using conventional distillation to purify a mixture. In general, the basic condition lies in the difference in the boiling points of different components, the larger the difference, the easier to separate. In the meantime, operating pressure directly affects the performance of low temperature distillation. High pressure maintains the mixture completely in its critical state, thus lowering the possibility of separation. On the contrary, if the operating pressure is too low, then a large amount of refrigeration energy is required to maintain a low temperature at the top of the column.
Another fundamental condition of separating a mixture by conventional distillation is that it does not form azeotropes. In the temperature-composition diagram of an azeotrope, the vapor curve is tangent to the liquidus, this point of tangency is called the azeotropic point. Neither partial vaporization nor partial condensation can change the chemical composition of an azeotropic mixture at boiling point. That is, conventional distillation is not suitable for purifying azeotropic mixturee near their boiling point.
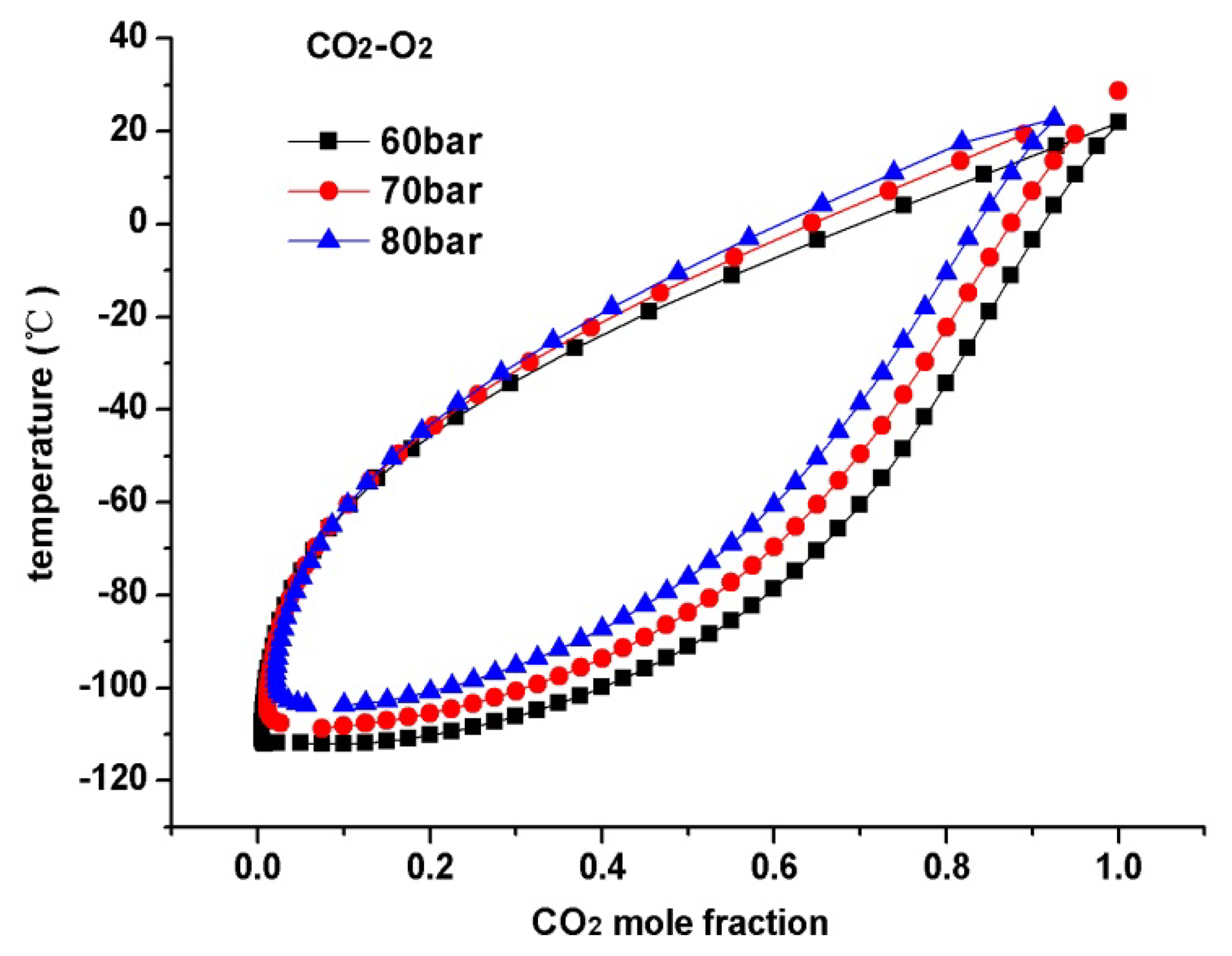
Figures 4, 5, and 6 present the temperature-composition diagrams of CO2-N2, CO2-O2, and CO2-Ar mixtures, respectively. The following conclusions can be drawn based on the figures: (1) The differences in the boiling points of CO2 and other impurities (i.e., N2, O2, and Ar) are still very large, even under high pressure; (2) For CO2-N2, CO2-O2, and CO2-Ar mixtures, no azeotropic point is found under high pressure conditions, hence, purifying a CO2 mixture consisting of impurities such as N2, O2, and Ar via conventional distillation is feasible. The distillation process can also be conducted under high pressure and near ambient temperature conditions, which ensures a low energy penalty.
4. Proposal and Performance Analysis of the Improved Separation and Purification System
4.1. Schematic Diagram of the Improved Separation and Purification System
Based on the analysis above, an improved CO2 separation and purification system is proposed. The whole system is made up of two subsystems: the cryogenic separation subsystem and the distillation subsystem. According to the traditional cryogenic separation method, the liquefaction temperature increases by improving the initial pressure of the mixed gases. The separation ratio could also be maintained at a high level by multi-stage separation and compression. In the distillation subsystem, crude product is distillated under high pressure and near ambient temperature conditions. Figure 7 shows the schematic diagram of this improved system.
An initial dehydration of the mixed gases is performed before they are fed into the proposed system: by cooling down to near ambient temperature, the majority of H2O is condensed and can be extracted out afterwards, while the rest is absorbed by a high-efficiency adsorbent (e.g., molecular sieve) [37]. As illustrated in Figure 7, when the dehydrated mixed gases (Stream 1 or S1) undergo the cryogenic separation and liquefaction processes, they are first compressed to an appropriate pressure (S2) by compressor 1 (C1). After cooling by the separation product, they would be cooled to a lower temperature by the external cold energy (S3). At this point, a part of the CO2 is liquefied from the mixed gases. Using a gas-liquid separator (Sep1), we can separate the CO2 from the mixture (S4) and pressurize it with a pump (P1). Then, part of the cold energy of the separated CO2 (S5) is recovered back to the system by a heat exchanger (H1) with the mixed gases (S2) and leaves the system (S6). The abovementioned steps comprise the first stage of the process. If the mixed gases (S7) from the first stage could not satisfy the separation requirement, they are then separated in the second or the third stages. The processes of the next two stages are similar to the first one. In the cryogenic separation subsystem, three-stage separation and liquefaction are employed. When most of the CO2 is separated, the purge gas (S20) leaves the system after its cold energy is recycled by a heat exchanger (H5).
The crude liquid CO2 (S21) separated from the cryogenic separation subsystem is further purified in the distillation subsystem to improve its CO2 purity. Before distillation, it is adjusted by a pressure regulating valve (V1) and a heat exchanger (H7). Temperatures on top and at the bottom of the distillation column (R) are precisely regulated within the range of –20 °C to 20 °C and –10 °C to 30 °C, respectively. After adjustment by the pressure regulating valve (V2) and heat exchanger (H8), the CO2 product with high purity (S25) is finally obtained. V1, V2, H7, and H8 can realize pressure and temperature adjustments to a small extent, thereby ensuring that the distillation process proceeds even in abnormal working conditions, such as start and stop. However, these adjustments are not necessarily needed in normal working conditions.
4.2. Simulations and Results Analysis
In this study, process simulation is conducted by ASPEN PLUS™. The thermodynamic properties of the mixed gases are calculated by the PRMHV2 equations, because the prediction of the PRMHV2 equation can reflect the corresponding change trend of the mixture system when the initial parameters change, especially for nonpolar gas systems. The compressor and pump efficiencies are assumed to be 0.8, and the smallest temperature difference of the low-temperature heat exchanger is set at 2 °C.
Table 1 illustrates the main streams corresponding to Figure 7. As can be seen, after multi-stage compression, refrigeration, and separation, 92% of the CO2 can be separated from the mixed gases in liquid state. The CO2 concentrations of the crude liquid reaches 96.9%, at a pressure of 80 bar (S21). After distillation and adjustments in parameters, the CO2 concentration in the final product is greatly improved to 99.9%, with the pressure decreasing to 60 bar (S23), which is suitable for most industrial applications as well as transport and storage.
The analysis data of the energy penalty for CO2 recovery, along with some other performance parameters are summarized in Table 2. Note that the results and analysis of Table 2 are valid exclusively for the proposed system, which could be considered as polishing process instead of an intact CO2 capture system, since the energy consumption of obtaining high CO2 concentration is not taken into account here.
The proposed system clearly has excellent performance. The CO2 recovery ratio is 90.04% with 99.9% CO2 purity in the product, the energy penalty for the cryogenic separation subsystem is 29.77 MW, out of which C1, C2, and C3 consume 11.40, 2.21 and 0.72 MW, respectively; the total energy consumption for refrigeration is 18.34 MW (13.75, 3.41 and 1.18 MW for H2, H4 and H6); the total energy consumption for pumps is 0.519 MW (0.44, 0.07 and 0.009 MW for P1, P2 and P3, respectively), with 3.42 MW recovered by expansion; and the energy consumption of distillation is only 2.61 MW. In summary, the total energy penalty for this improved system is 32.38 MW, and the specific energy consumption for CO2 capture is only 0.425 MJ/kgCO2.
The excellent performance of the proposed system can be attributed to its delicate process design, which is associated with highly mature technologies. The process and structural characteristics of the improved system are listed below:
- (1)
Compression, refrigeration, and cryogenic separation are carried out several times in the system. Despite the fact that CO2 concentration decreases continuously with CO2 condensation, it can be improved by the increasing of the initial pressure, in order to maintain CO2 liquefaction temperature at a high level. This condition in turn lowers the energy penalty for the cryogenic separation subsystem.
- (2)
The distillation process is conducted under high pressure and near ambient temperature conditions. It can take full advantage of the large differences between the physical properties of the CO2 and its impurities. It also connects perfectly with the cryogenic separation subsystem because the crude liquid CO2 are under the same conditions. Consequently, the specific energy consumption for CO2 capture could be as low as 0.425 MJ/kgCO2.
- (3)
As a result of the distillation process, the CO2 purity in the product increases dramatically and finally meets the requirements for transport and storage. Note that higher CO2 purity can be expected with simple parameter improvements, such as an increase in the number of distillation trays or an enhancement of the stripping rate. The final CO2 product obtained by the proposed system then becomes available to special industries (e.g., food industry), thus enhancing its additional value.
5. Techno-Economic Analysis of the Proposed System
5.1. Component Overnight Cost Estimation
Given that our proposed system is similar to the cryogenic air separation unit (ASU), the reference data for component overnight cost estimation are gathered from the literature on ASU to ensure the calculation's accuracy and validity [38–41]. The calculation methodology employed to estimate the component overnight costs follows the method used by Holt and Kreutz in studies comparing alternative IGCC systems based on a series of EPRI-sponsored studies. The present work applies the overnight cost, which includes installation investment, balance of plant, general facilities costs, engineering fees, and contingencies [42,43]. Detailed reference data are listed in Table 3.
In general, the overnight component cost is the function of its own size. The overnight cost of a specific component can be obtained by the following equation:
5.2. Total Plant Investment
Total plant investment (TPI) is calculated as follows: TPI = total overnight cost (TOC) + interest during construction (IDC) [43]. According to Equation (1) and detailed parameters, overnight costs of major plant components are presented in Table 4. Notably, equipment made in China is generally much cheaper than that made in Western countries, essentially because of the low labor cost in China, as presented in literature [44–46].
The main economic analysis assumptions employed in this work are: (1) The lifespan of the proposed system is assumed to be 20 years with annual working hours set at 6000 h/year [47]; (2) IDC is taken as 12.3% of TOC based on a four-year construction schedule with equal annual payments and a real discount rate (k) of 10%/year; (3) The annual operation and maintenance cost (O&M) takes over 4% of TPI; (4) CO2 transport and storage is charged for 5$/ton, no extra carbon emission tax is attached.
The summary of the TPI calculation is shown in Table 4. TOC is 29.872 M$ when major components and necessary auxiliaries such as pipelines and valves are considered. IDC is 3.674 M$. The TPI of the proposed system is 33.546 M$, and the annual O&M cost is 1.342 M$.
Table 5 presents a brief performance comparison of several CO2 recovery processes, including MEA absorption, Selexol™ absorption, and the proposed system. The techno-economic data of the MEA and Selexol™ absorption processes are collected from the IPCC report and related literature. The cost of CO2 capture of the proposed system is calculated using the following equation:
According to the previous calculation assumptions, CRF is equal to 0.117, whereas the total capture process investment and annual O&M cost are calculated based on Tables 2, 3, 4 and 5.
As shown in Table 5, the specific capture process investment of the improved system is only 0.440 M$/(kg·s−1), and its cost of CO2 capture is 10.28 $/tCO2. As for the MEA and Selexol™ absorption methods, the specific capture process investments are 1.178 M$/(kg·s−1) and 0.835 M$/(kg·s−1), respectively, whereas their costs of CO2 capture increase to 24 $/tCO2 and 19 $/tCO2, respectively. Which means compared to conventional MEA and Selexol™ absorption methods, the cost of CO2 capture of the proposed system reduces by 57.2% and 45.9%, respectively.
Note that the cost data found in related literature varies widely due to different estimation methods, design requirements, construction materials, and national conditions. Different recovery processes are applicable to various flue gas compositions, as revealed in Table 5. Hence, the improved system is not necessarily much better than or able to replace conventional absorption processes. We try to demonstrate in this study that if the initial CO2 concentration of the gas mixture is relatively high (e.g., oxy-fuel combustion or pre-combustion capture), then the proposed system provides a feasible and competitive approach to CO2 capture with respect to thermodynamic and economic performance. Briefly, performance of the proposed system in combination with oxy-fuel combustion is evaluated. The amount of oxygen needed for oxy-fuel combustion is roughly 65.4–75.7 kg/s according to the law of conservation of mass, the energy consumption and additional investment of air separation unit are about 39–44 MW and 39–42 M$ with reference to related bibliography [44,50,51]. As a result, the total energy penalty for CO2 capture will increase from 0.425 MJ/kgCO2 to 0.937–1.003 MJ/kgCO2, specific capture process investment will increase from 0.440 M$/(kg·s−1) to 0.952–0.992 M$/(kg·s−1), and cost of CO2 capture will rise from 10.28 $/tCO2 to approximately 18.32–18.60 $/tCO2.
6. Discussion
6.1. Influences of Initial Pressure and Initial Concentration on the CO2 Capture Energy Penalty
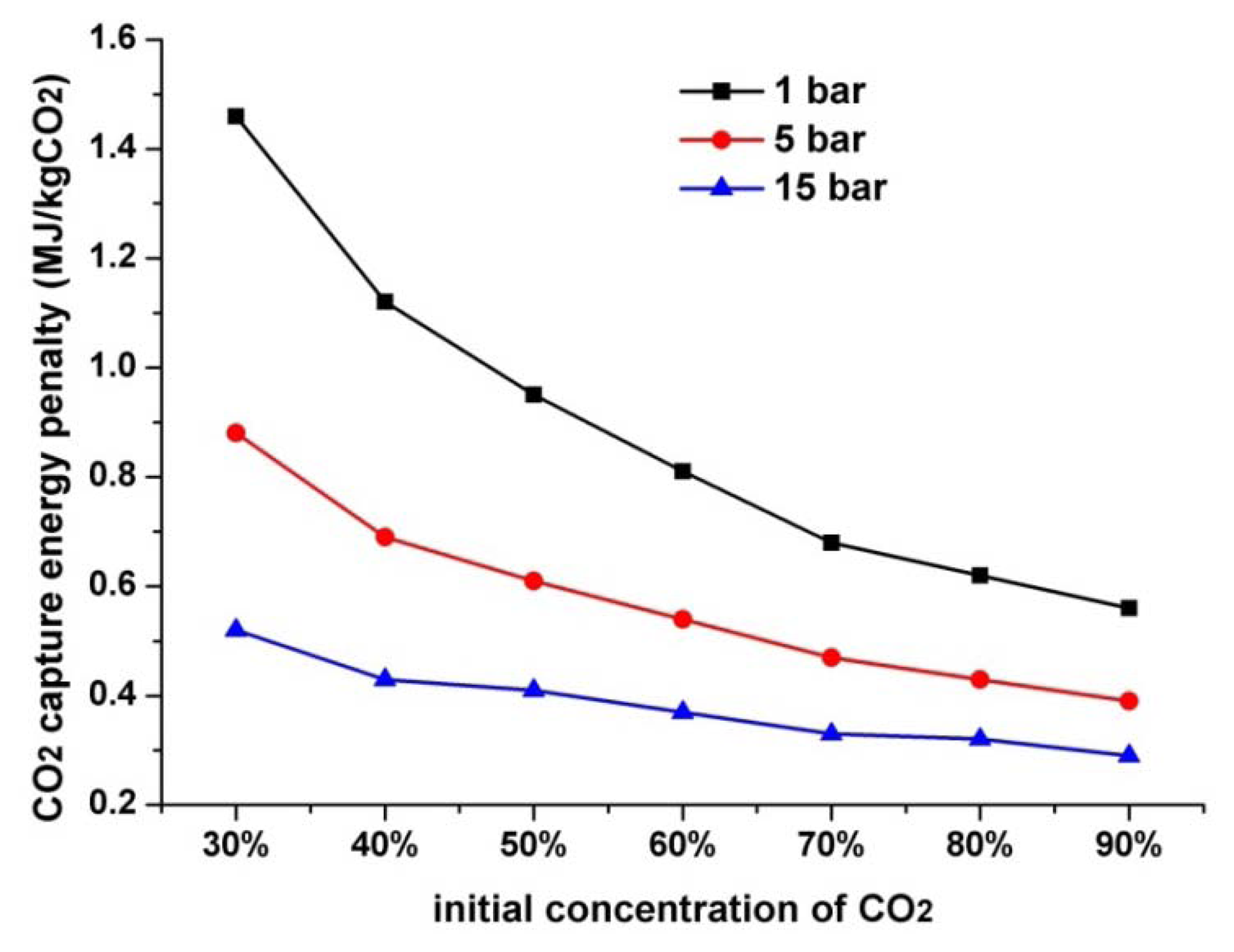
The initial pressure and initial concentration of the mixed gases have a great influence on the performance of the proposed system. Figure 8 presents the relationship between the CO2 capture energy penalty against its initial pressure and concentration.
As shown in the curves, the energy penalty for CO2 capturing unit greatly decreases with the increase in the initial pressure. In the proposed system, the mixed gases must first be compressed into a relatively high pressure to keep the liquefaction temperature at a high level, thus compression work of the first stage is relatively high and could consume over 30% to 50% of the total energy penalty. If the initial pressure of the mixed gases is relatively high at the beginning, lots of compression work could be saved for the first stage. The result is a decrease in the CO2 capture energy penalty.
The CO2 capture energy penalty also decreases substantially due to the increase of initial CO2 concentration. As shown in Figure 8, the CO2 capture energy penalty at an initial concentration of 60% increases by approximately 50% compared with that at an initial concentration of 80% in a fixed initial pressure. This value increases by approximately 150% when the initial concentration is 40%. This condition is due to in low initial CO2 concentration, large refrigeration work is required to deal with the low liquefaction temperature. If the initial CO2 concentration is enhanced, the CO2 capture energy penalty will decrease significantly. In summary, the proposed system has superior performance in recovering CO2 from mixed gases with high initial CO2 concentration and initial pressure.
6.2. CO2 Purity Comparison before and after Distillation
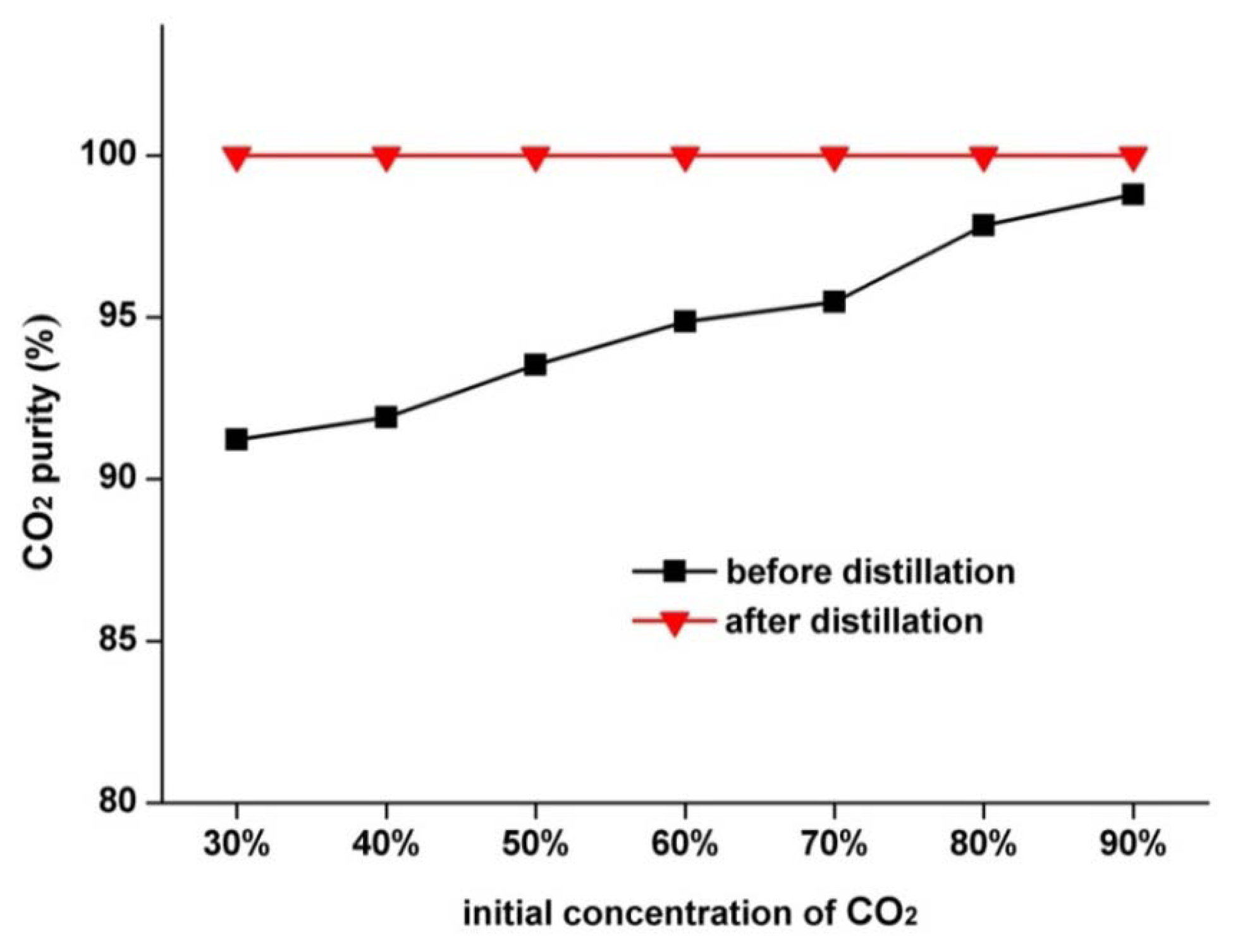
If the initial CO2 concentration in the CO2-N2 mixture changes, the CO2 purity in the final product obtained through the cryogenic separation method varies. Figure 9 provides the relationship between CO2 purity and initial concentration of CO2 before and after distillation. The CO2 purity in the product is relatively low before distillation, although it is improved as the initial CO2 concentration increases. Specifically, CO2 purity without distillation is only 92% at an initial concentration of 30% and reaches only 98.78% at an initial CO2 concentration of 90%. By contrast, the CO2 purity in the product is constantly above 99.9% after distillation regardless of the initial CO2 concentration. At this level, the CO2 purity perfectly meets the requirements for most industrial applications as well as transport and storage. The distillation process can significantly improve the CO2 purity in the product, thus proving that it is an effective and necessary purification method for separating CO2-N2 mixture.
6.3. Analysis of the CO2 Purity in the Product with Different Initial Compositions
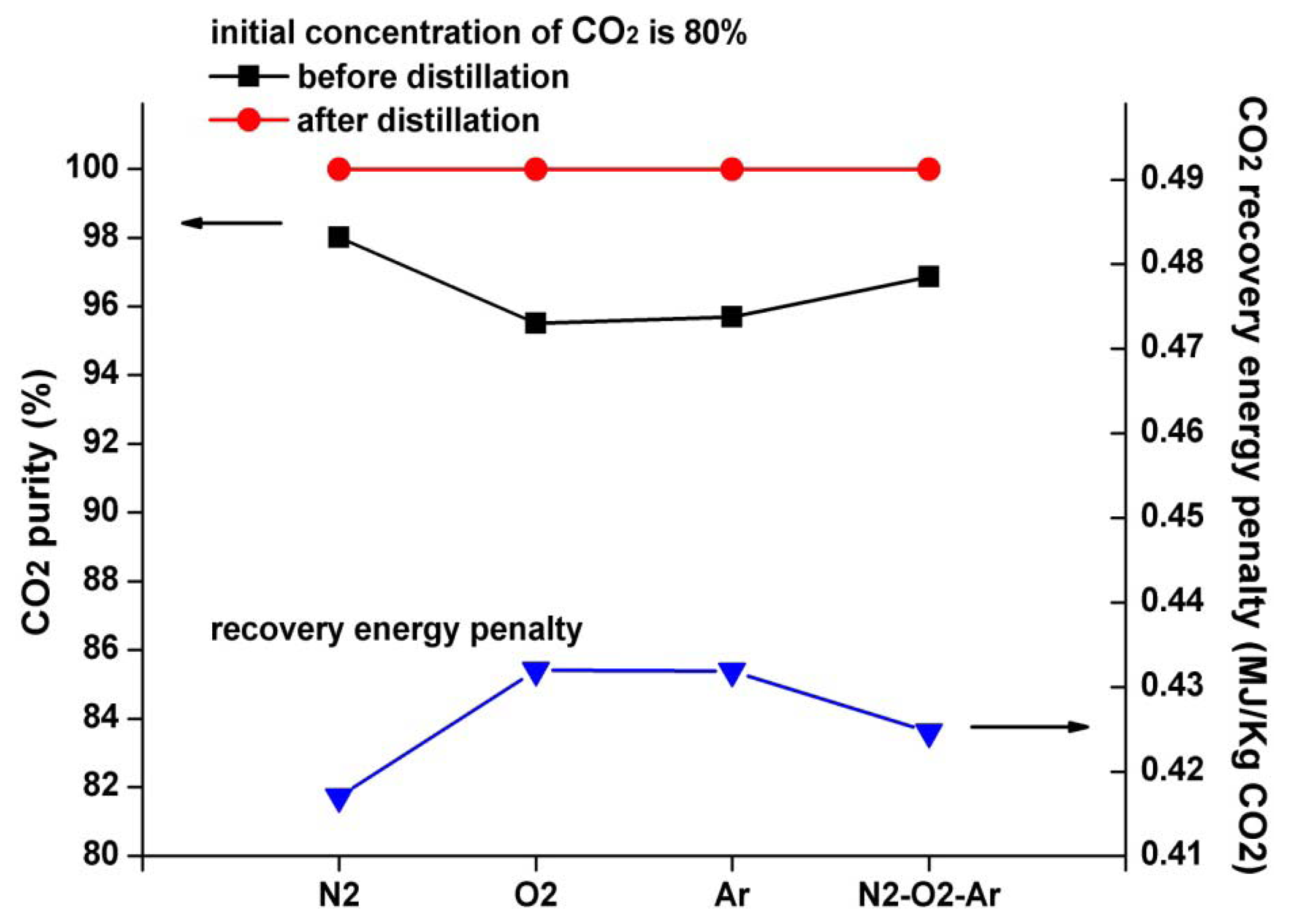
Figure 10 shows the influences of different initial compositions on CO2 purity and CO2 recovery energy penalty. Supposing the initial CO2 concentration of the mixed gases is 80%, four kinds of typical initial compositions are discussed: N2, O2, Ar, and N2-O2-Ar. The concentrations of these components are equally set at 20%. For N2-O2-Ar, the concentration of each component is 10%, 5%, and 5%, respectively. As can be seen, before distillation, the CO2 purity is greatly affected by the change in initial composition. For N2, O2, Ar, and N2-O2-Ar, their CO2 purities without distillation are only 98.01%, 95.5%, 95.69%, and 96.86%, respectively. After distillation, the CO2 purity increases to more than 99.9% for all circumstances. The recovery energy penalty fluctuates within the range of 5% when the initial composition varies, which demonstrates that the proposed system presents excellent performance for various initial compositions.
7. Conclusions
Based on an in-depth analyses of cryogenic separation and distillation theory as well as the phase transition characteristics of gas mixtures containing CO2, this study presents an improved CO2 separation and purification system. According to the theoretical analysis, case simulations, and regularity analysis discussed above, the following conclusions are drawn:
- (1)
By adopting multi-stage compression, refrigeration, and separation, the resulting improved cryogenic separation subsystem could separate the majority of CO2 from gas mixtures with relatively low energy penalty and could fully recover the cold energy of the separation product.
- (2)
Considering the large difference between the physical properties of CO2 and other impurities, the distillation process is conducted under high pressure and near ambient temperature conditions. Consequently, the CO2 purity in the product significantly increases to more than 99.9%, whereas the energy penalty for distillation is rather low. This condition finally realizes the low energy penalty of purification.
- (3)
The cost of CO2 capture of the proposed system is much lower than those of conventional absorption methods, because it mainly adopts common equipment which are widely utilized and highly mature in the chemical industry (e.g., compressors, heat exchangers, and pumps). Besides, this equipment can operate effectively for a long term under comparatively mild working condition as there is no serious corrosion or secondary pollution problems. Consequently, the TPI and annual O&M could be maintained at low levels.
- (4)
The proposed system has superior performance in recovering CO2 from mixed gases with high initial CO2 concentration. Note that the high initial pressure of mixed gases contributes to lowering the CO2 recovery energy penalty. Furthermore, the analysis proves that the proposed system can efficiently recover CO2 from mixed gases, regardless of initial compositions as the CO2 purity in the product could be as high as 99.9% under various circumstances.
Acknowledgments
This study was supported by the National Nature Science Fund of China (No. 51025624), National Key Technology R&D Program of China (2012BAC24B01), the 111 Project (B12034), and the Fundamental Research Funds for the Central Universities (2014ZD04).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Working Group III of the Intergovernmental Panel on Climate Change (IPCC). In IPCC Special Report on Carbon Dioxide Capture and Storage; Cambridge University Press: Cambridge, UK, 2005.
- Working Group III of the Intergovernmental Panel on Climate Change (IPCC). In IPCC's Fourth Assessment Report (AR4): Mitigation of Climate Change; Cambridge University Press: Cambridge, UK, 2007.
- Ma'mun, S.; Svendsen, H.F.; Hoff, K.A.; Juliussen, O. Selection of new absorbents for carbon dioxide capture. Energy Convers. Manag. 2007, 48, 251–258. [Google Scholar]
- Saha, A.K.; Biswas, A.K.; Bandyopadhyayc, S.S. Absorption of CO2 in a sterically hindered amine: Modeling absorption in a mechanically agitated contactor. Sep. Purif. Technol. 1999, 15, 101–112. [Google Scholar]
- Mandal, B.P.; Guha, M.; Biswas, A.K.; Bandyopadhyayc, S.S. Removal of carbon dioxide by absorption in mixed amines: Modeling of absorption in aqueous MDEA/MEA and AMP/MEA solutions. Chem. Eng. Sci. 2001, 56, 6217–6224. [Google Scholar]
- Alie, C.; Backham, L.; Croiset, E.; Douglas, P.L. Simulation of CO2 capture using MEA scrubbing: A flowsheet decomposition method. Energy Convers. Manag. 2005, 46, 475–487. [Google Scholar]
- Dorctor, R.D.; Molburg, J.C.; Thimmapuram, P.R. Gasification Combined Cycle: Carbon Dioxide Recovery, Transport, and Disposal; Argonne National Laboratory: ANL/ESD-24; Argonne, IL, USA, 1994. [Google Scholar]
- Dorctor, R.D.; Molburg, J.C.; Thimmapuram, P.R. KRW Oxygen-blown Gasification Combined Cycle: Carbon Dioxide Recovery, Transport, and Disposal; ANL/ESD-34; Argonne National Laboratory: Argonne, IL, USA, 1996. [Google Scholar]
- Zhang, J.; Webley, P.A.; Xiao, P. Effect of process parameters on power requirements of vacuum swing adsorption technology for CO2 capture from flue gas. Energy Convers. Manag. 2008, 49, 346–356. [Google Scholar]
- Na, B.K.; Koo, K.K.; Eum, H.M. CO2 Recovery from flue gas by PSA process using activated carbon. Korean J. Chem. Eng. 2001, 18, 220–227. [Google Scholar]
- Siriwardane, R.V.; Shen, M.S.; Fisher, E.P. Adsorption of CO2 on molecular sieves and activated carbon. Energy Fuels 2001, 15, 279–284. [Google Scholar]
- Siriwardane, R.V.; Shen, M.S.; Fisher, E.P. Adsorption of CO2, N2, and O2 on natural zeolites. Energy Fuels 2003, 17, 571–576. [Google Scholar]
- Gomes, V.G.; Yee, K.W.K. Pressure swing adsorption for carbon dioxide sequestration from exhaust gases. Sep. Purif. Technol. 2002, 28, 161–171. [Google Scholar]
- Yan, S.P.; Fang, M.X.; Zhang, W.F.; Zhong, W.L.; Luo, Z.Y.; Cen, K.F. Comparative analysis of CO2 separation from flue gas by membrane gas absorption technology and chemical absorption technology in China. Energy Convers. Manag. 2008, 49, 3188–3197. [Google Scholar]
- Powell, C.E.; Qiao, G.G. Polymeric CO2/N2 gas separation membranes for the capture of carbon dioxide from power plant flue gases. J. Membr. Sci. 2006, 279, 1–49. [Google Scholar]
- Feron, P.H.M.; Jansen, A.E. CO2 separation with polyolefin membrane contactors and dedicated absorption liquids: Performances and prospects. Sep. Purif. Technol. 2002, 27, 231–242. [Google Scholar]
- Pierce, W.F.; Riemer, P.; William, G.O. International perspectives and the results of carbon dioxide capture disposal and utilisation studies. Energy Convers. Manag. 1995, 36, 813–818. [Google Scholar]
- Wang, B.Q. Process Mechanism and System Synthesis for CO2Capture in IGCC System; Chinese Academy of Sciences: Beijing, China, 2004. [Google Scholar]
- Wang, B.Q.; Jin, H.G.; Han, W. IGCC system with integration of CO2recovery and the cryogenic energy in air separation unit. Proceedings of ASME Turbo Expo, Vienna, Austria, 14–17 June 2004. GT-2004-53723.
- Wang, B.Q.; Jin, H.G. A novel IGCC system with H2/O2cycle and CO2recovery by ASU cryogenic energy. Proceedings of the 7th International Conference on Greenhouse Gas Control Technologies, Vancouver, BC, Canada, 5–9 September 2004.
- Deng, S.; Jin, H.; Cai, R. Novel cogeneration power system with LNG cryogenic exergy utilization. Energy 2004, 29, 497–512. [Google Scholar]
- Zhang, N.; Lior, N. A novel near-zero CO2 emission thermal cycle with LNG cryogenic exergy utilization. Energy 2006, 31, 1666–1679. [Google Scholar]
- Song, C.F.; Kitamura, Y.; Li, S.H. Evaluation of Stirling cooler system for cryogenic CO2 capture. Appl. Energy 2012, 98, 491–501. [Google Scholar]
- Kanniche, M.; Gros-Bonnivarda, R.; Jauda, P.; Valle-Marcosa, J.; Amannb, J.M.; Boualloub, C. Pre-combustion, post-combustion and oxy-combustion in thermal power plant for CO2 capture. Appl. Therm. Eng. 2010, 30, 53–62. [Google Scholar]
- Zanganeh, K.E.; Shafeen, A. A novel process integration, optimization and design approach for large-scale implementation of oxy-fired coal power plants with CO2 capture. Greenh. Gas Control 2007, 1, 47–54. [Google Scholar]
- Zanganeh, K.E.; Shafeen, A.; Salvador, C. CO2 capture and development of an advanced pilot-scale cryogenic separation and compression unit. Energy Procedia 2009, 1, 247–252. [Google Scholar]
- Li, H.; Yan, J.; Yan, J.; Anheden, M. Impurity impacts on the purification process in oxy-fuel combustion based CO2 capture and storage system. Appl. Energy 2009, 86, 220–213. [Google Scholar]
- Besong, M.T.; Maroto-Valer, M.M.; Finn, A.J. Study of design parameters affecting the performance of CO2 purification units in oxy-fuel combustion. Int. J. Greenh. Gas Control 2013, 12, 441–449. [Google Scholar]
- Song, C.F.; Kitamura, Y.; Li, S.H.; Jiang, W.Z. Analysis of CO2 frost formation properties in cryogenic capture process. Int. J. Greenh. Gas Control 2013, 13, 26–33. [Google Scholar]
- Jana, A.K. Heat integrated distillation operation. Appl. Energy 2010, 87, 1477–1494. [Google Scholar]
- Xu, G.; Li, L.; Yang, Y.P.; Tian, L.H.; Liu, T.; Zhang, K. A novel CO2 cryogenic liquefaction and separation system. Energy 2012, 42, 522–529. [Google Scholar]
- Xu, G.; Jin, H.G.; Yang, Y.P.; Duan, L.Q.; Han, W.; Gao, L. A novel coal-based hydrogen production system with low CO2 emissions. ASME J. Eng. Gas Turbines Power 2010, 132, 031701–031709. [Google Scholar]
- Xu, G.; Yang, Y.P.; Duan, L.Q.; Wang, N. A novel integration method for CO2 separation and compression. J. Eng. Thermophys. 2010, 31, 1643–1646, in Chinese. [Google Scholar]
- Humphrey, J.L.; Siebert, A.F. Separation technologies: An opportunity for energy savings. Chem. Eng. Prog. 1992, 88, 80–92. [Google Scholar]
- Engelien, H.K.; Skogestad, S. Selecting appropriate control variables for a heat integrated distillation system with prefractionator. Comput. Chem. Eng. 2004, 28, 683–691. [Google Scholar]
- Al-Muslim, H.; Dincer, I. Thermodynamic analysis of crude oil distillation systems. Int. J. Energy Res. 2005, 29, 637–655. [Google Scholar]
- Toftegaard, M.B.; Brix, J.; Jensen, P.A.; Glarborg, P.; Jensen, A.D. Oxy-fuel combustion of solid fuels. Prog. Energy Combust. Sci 2010, 36, 581–625. [Google Scholar]
- Agahi, R. GE Power Systems; Rotoflow, Inc.: Gardena, CA, USA; December; 2002; personal communication. [Google Scholar]
- Lozza, G.; Chiesa, P. CO2sequestration techniques for IGCC and natural gas power plants: A comparative estimation of their thermodynamic and economic performance. Proceedings of International Conference on Clean Coal Technologies for our Future, Chia Laguna, Italy, 21–23 October 2002.
- El-Enin, S.A.A.; Attia, N.K.; El-Ibiari, N.N.; El-Diwani, G.I.; El-Khatib, K.M. In-situ transesterification of rapeseed and cost indicators for biodiesel production. xRenew. Sustain. Energy Rev. 2013, 18, 471–477. [Google Scholar]
- Haas, M.J.; McAloon, A.J.; Yee, W.C.; Foglia, T.A. A process model to estimate biodiesel production costs. Bioresour. Technol. 2006, 97, 671–678. [Google Scholar]
- Holt, N. IGCC Power Plants—EPRI Design and Cost Studies. Proceedings of EPRI/GTC Gasification Technologies Conference, San Francisco, CA, USA, 6 October 1998.
- Kreutz, T.; Williams, R.; Consonni, S.; Chiesa, P. Co-production of hydrogen, electricity and CO2 from coal with commercially ready technology. Part B: Economic analysis. Int. J. Hydrog. Energy 2005, 30, 769–784. [Google Scholar]
- Huang, B.; Xu, S.S.; Gao, S.H.; Liu, L.B.; Tao, J.Y.; Niu, H.W.; Cai, M.; Cheng, J. Industrial test and techno-economic analysis of CO2 capture in Huaneng Beijing coal-fired power station. Appl. Energy 2010, 87, 3347–3354. [Google Scholar]
- Zhao, M.; Minett, A.I.; Harris, A.T. A review of techno-economic models for the retrofitting of conventional pulverised-coal power plants for post-combustion capture (PCC) of CO2. Energy Environ. Sci. 2013, 6, 25–40. [Google Scholar]
- Huang, B.; Xu, S.S.; Gao, S.H.; Liu, L.B.; Tao, J.Y.; Niu, H.W.; Cai, M.; Cheng, J. Industrial test of CO2 capture in Huaneng Beijing coal-fired power station. Proc. CSEE 2009, 29, 14–20, In Chinese. [Google Scholar]
- Nam, H.; Lee, T.; Lee, J.; Lee, J.; Chung, H. Design of carrier-based offshore CCS system: Plant location and fleet assignment. Int. J. Greenh. Gas Control 2013, 12, 220–230. [Google Scholar]
- Abu-Zahra, M.R.M.; Niederer, J.P.M.; Feron, P.H.M.; Versteeg, G.F. CO2 capture from power plants Part II. A parametric study of the economical performance based on mono-ethanolamine. Int. J. Greenh. Gas Control 2007, 1, 135–142. [Google Scholar]
- Parsons, E.L.; Shelton, W.W.; Lyons, J.L. Advanced Fossil Power Systems Comparison Study, Final Report Prepared for NETL; National Energy Technology Laboratory: Morgantown, WV, USA, 2012. [Google Scholar]
- Xiong, J.; Zhao, H.B.; Zheng, C.G.; Liu, Z.H.; Zeng, L.D.; Liu, H.; Qiu, J.R. An economic feasibility study of O2/CO2 recycle combustion technology based on existing coal-fired power plants in China. Fuel 2009, 88, 1135–1142. [Google Scholar]
- Doukelis, A.; Vorrias, I.; Grammelis, P.; Kakaras, E.; Whitehouse, M.; Riley, G. Partial O2-fired coal power plant with post-combustion CO2 capture: A retrofitting option for CO2 capture ready plants. Fuel 2009, 88, 2428–2436. [Google Scholar]



| Flow | Temperature (°C) | Pressure (bar) | Mass Flow (kg/s) | Mole Fraction (%) | |||
|---|---|---|---|---|---|---|---|
| CO2 | N2 | O2 | Ar | ||||
| S1 | 30.0 | 5 | 100.00 | 80.0 | 10.0 | 5.0 | 5.0 |
| S2 | 30.0 | 21 | 100.00 | 80.0 | 10.0 | 5.0 | 5.0 |
| S3 | −26.5 | 21 | 100.00 | 80.0 | 10.0 | 5.0 | 5.0 |
| S4 | −35.0 | 21 | 100.00 | 80.0 | 10.0 | 5.0 | 5.0 |
| S5 | −29.7 | 80 | 62.93 | 98.5 | 0.4 | 0.5 | 0.6 |
| S6 | 8.3 | 80 | 62.93 | 98.0 | 0.6 | 0.7 | 0.7 |
| S7 | −40.0 | 21 | 37.07 | 53.2 | 23.9 | 11.4 | 11.5 |
| S8 | 10.4 | 38 | 37.07 | 53.2 | 23.9 | 11.4 | 11.5 |
| S9 | −25.0 | 38 | 37.07 | 53.2 | 23.9 | 11.4 | 11.5 |
| S10 | −40.0 | 38 | 37.07 | 53.2 | 23.9 | 11.4 | 11.5 |
| S11 | −31.0 | 80 | 13.35 | 93.8 | 2.1 | 2.1 | 2.0 |
| S12 | 3.4 | 80 | 13.35 | 93.8 | 2.1 | 2.1 | 2.0 |
| S13 | −40.0 | 38 | 23.74 | 34.2 | 34.1 | 15.8 | 15.9 |
| S14 | −0.7 | 60 | 23.74 | 34.2 | 34.1 | 15.8 | 15.9 |
| S15 | −31.0 | 60 | 23.74 | 34.2 | 34.1 | 15.8 | 15.9 |
| S16 | −35.0 | 60 | 23.74 | 34.2 | 34.1 | 15.8 | 15.9 |
| S17 | −36.1 | 80 | 3.53 | 88.0 | 4.3 | 3.9 | 3.8 |
| S18 | −3.3 | 80 | 3.53 | 88.0 | 4.3 | 3.9 | 3.8 |
| S19 | −40.0 | 60 | 20.19 | 26.5 | 38.4 | 17.5 | 17.6 |
| S20 | −3.3 | 60 | 20.19 | 26.5 | 38.4 | 17.5 | 17.6 |
| S21 | 7.3 | 80 | 79.81 | 96.9 | 1.1 | 1.1 | 1.0 |
| S22 | 30.0 | 80 | 79.81 | 96.9 | 1.1 | 1.1 | 1.0 |
| S23 | 22.5 | 60 | 76.18 | 99.9 | 9 ppm | 48 ppm | 27 ppm |
| S24 | −10.8 | 60 | 3.62 | 40.5 | 20.1 | 20.0 | 19.3 |
| Items | Value | Unit |
|---|---|---|
| Mass flux of mixed gases fed to the system | 100 | kg/s |
| Mole fraction of CO2 fed to the system | 80 | % |
| Mass flux of CO2 fed to the system | 84.62 | kg/s |
| Mass flux of captured CO2 | 76.18 | kg/s |
| CO2 purity in product | 99.9 | % |
| CO2 recovery ratio | 90.04 | % |
| Energy penalty for cryogenic separation subsystem | 29.77 | MW |
| Energy consumption for distillation subsystem | 2.61 | MW |
| Total energy penalty for improved system | 32.38 | MW |
| Specific energy consumption for CO2 capture | 0.425 | MJ/kgCO2 |
| Component | Scaling parameter | C0 (M$) | S0 | f | n d | Notes |
|---|---|---|---|---|---|---|
| Compressor | Compression power | 6.3 | 10 MWe | 0.67 | 1 | a |
| Heat exchanger | MAF coal input (LHV) | 39.8 | 1377 MWth | 0.67 | 1 | a |
| Separator | Inlet flow rate | 0.5 | 71250 ton/year | 0.67 | 1 | b |
| Distillation column | Inlet flow rate | 0.12 | 17600 ton/year | 0.67 | 1 | c |
| Pump | Outlet pressure | 0.093 | 80 bar | 0.67 | 1 | b |
aCosts taken from Agahi [38] and Lozza and Chiesa [39];bGas-liquid separator is applied here; costs taken from El-Enin [40];cData taken from Haas [41];dn = 1 for all components in the proposed system.
| Overnight costs of plant components (M$) | Value | |
|---|---|---|
| C1 | 3.295 | |
| C2 | 1.767 | |
| C3 | 1.061 | |
| Heat Exchangers (H1–H8) | 8.800 | |
| Sep1 | 3.747 | |
| Sep2 | 1.923 | |
| Sep3 | 1.425 | |
| Pumps (P1–P3) | 0.279 | |
| Distillation Column (R) | 3.825 | |
| Pipeline | 2.500 e | |
| Auxiliaries (i.e., valves) | 1.250 f | |
| TOC | 29.872 | |
| IDC | 3.674 | |
| TPI | 33.546 | |
| Annual O&M | 1.342 | |
eOvernight costs for pipeline and auxiliaries are estimated to be approximately 8% and 4% of TOC, respectively.fOvernight costs for pipeline and auxiliaries are estimated to be approximately 8% and 4% of TOC, respectively.
| Items | Improved separation and purification system | MEA absorption process g | Selexol™ absorption process h |
|---|---|---|---|
| Mole fractions of flue gas | |||
| CO2 (%) | 80.00 | 13.30 | 29.14 |
| N2 (%) | 10.00 | 68.12 | 2.37 |
| O2 (%) | 5.00 | 3.81 | 0.00 |
| Ar (%) | 5.00 | 3.50 | 0.43 |
| H2O (%) | –– | 11.25 | 26.38 |
| H2 (%) | –– | –– | 40.13 |
| Other (%) | –– | 0.02 | 1.55 |
| Techno-economic indicators | |||
| Mass flux of captured CO2 (kg/s) | 76.18 | 113.33 | 66.83 |
| CO2 recovery ratio (%) | 90.04 | 90.0 | 87 |
| CO2 purity in product (%) | 99.9 | 98 | 95 |
| Total energy penalty (MW) | 32.38 | 441.9 | 62 |
| Energy penalty for recovering unit CO2 (MJ/kgCO2) | 0.425 | 3.9 | 0.928 |
| Total capture process investment (M$) | 33.546 | 133.470 | 55.8 |
| Specific capture process investment (M$/(kg·s−1)) | 0.440 | 1.178 | 0.835 |
| Cost of CO2 capture ($/tCO2) | 10.28 | 24 | 19 |
gData taken from Abu-Zahra [48] and the IPCC report (2007) [2];hData taken from the IPCC report (2007) [2] and NETL (2002) [49].
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, G.; Liang, F.; Yang, Y.; Hu, Y.; Zhang, K.; Liu, W. An Improved CO2 Separation and Purification System Based on Cryogenic Separation and Distillation Theory. Energies 2014, 7, 3484-3502. https://doi.org/10.3390/en7053484
Xu G, Liang F, Yang Y, Hu Y, Zhang K, Liu W. An Improved CO2 Separation and Purification System Based on Cryogenic Separation and Distillation Theory. Energies. 2014; 7(5):3484-3502. https://doi.org/10.3390/en7053484
Chicago/Turabian StyleXu, Gang, Feifei Liang, Yongping Yang, Yue Hu, Kai Zhang, and Wenyi Liu. 2014. "An Improved CO2 Separation and Purification System Based on Cryogenic Separation and Distillation Theory" Energies 7, no. 5: 3484-3502. https://doi.org/10.3390/en7053484




