Characteristics of CO2 Hydrate Formation and Dissociation in Glass Beads and Silica Gel
Abstract
:1. Introduction
2. Experimental
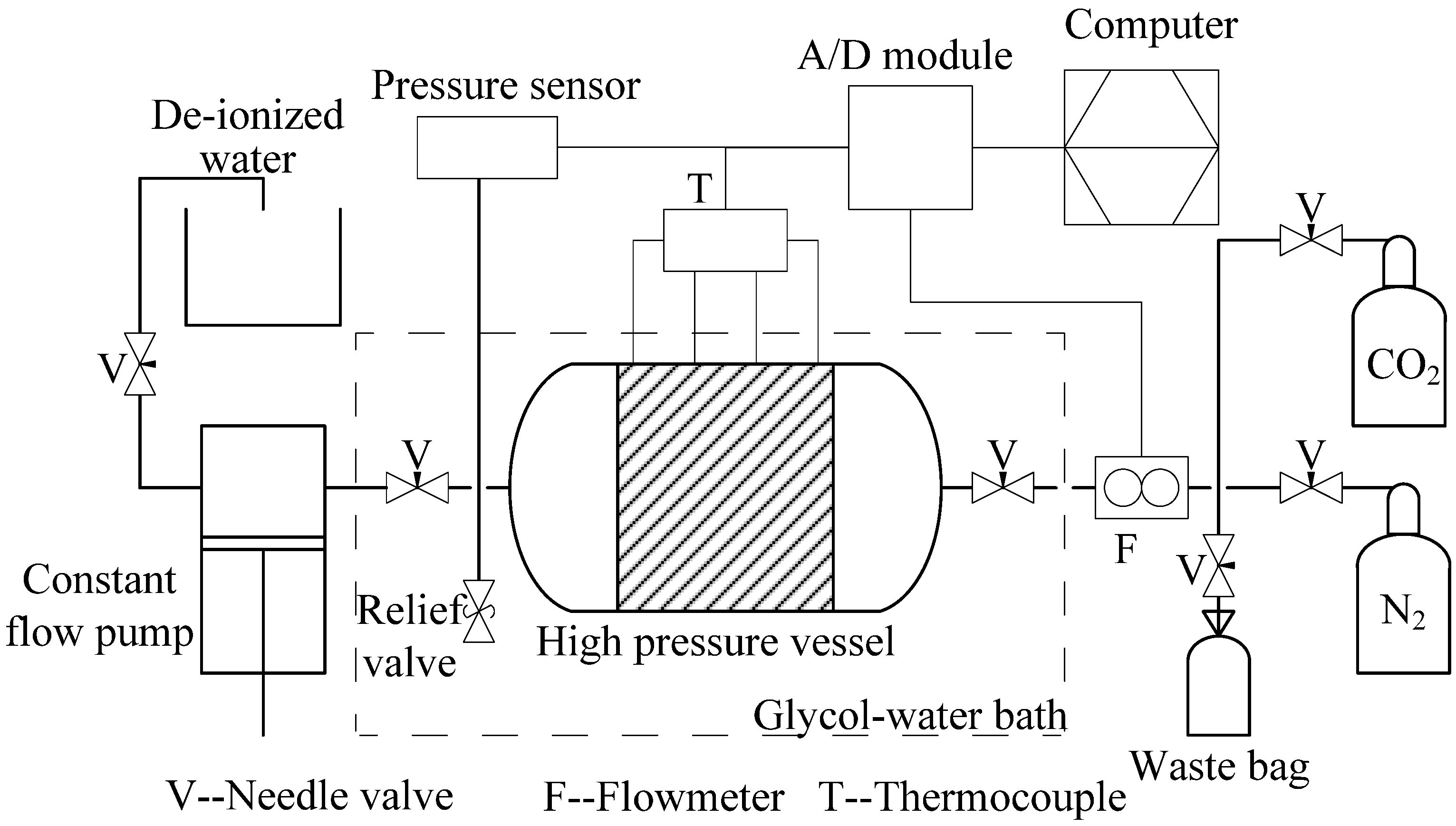
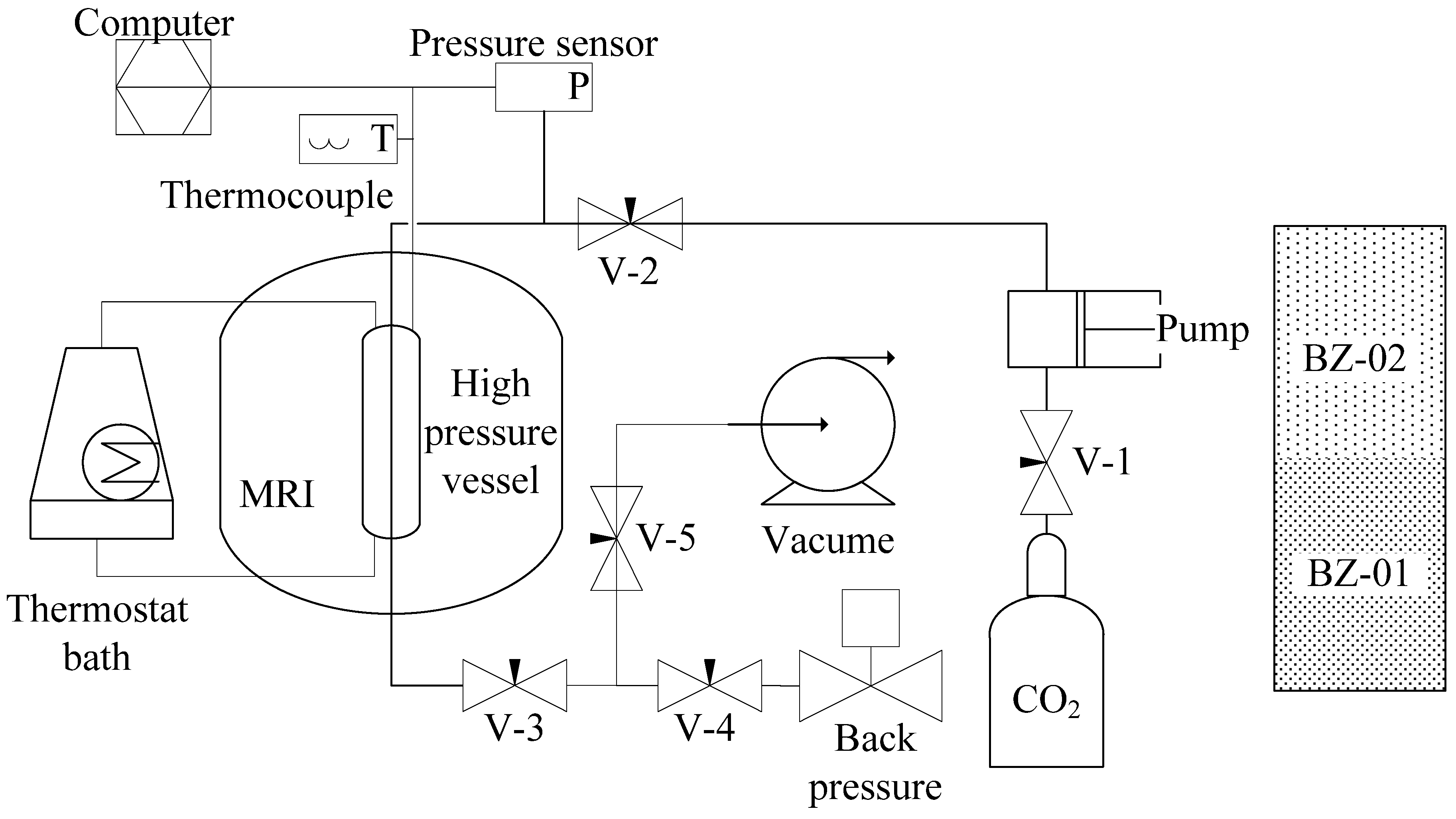
2.1. Experimental Apparatus and Materials
| Material | Purity/Composition | Particle size | Supplier |
|---|---|---|---|
| CO2 | 99.9% | ― | Dalian Guangming special gas Co., Ltd., China |
| BZ-01 | Soda glass | 0.105–0.125 mm | As-One Co., Ltd., Japan |
| BZ-02 | Soda glass | 0.177–0.250 mm | As-One Co., Ltd., Japan |
| BZ-04 | Soda glass | 0.350–0.500 mm | As-One Co., Ltd., Japan |
| Mix glass beads | Soda glass and clay | 0.125–1.0 mm | Self-made |
| Silica gel | Silica gel | 0.42–0.84 mm | Anhui Liangchen Silicon Material Co., Ltd., China (mean particle pore size 8.0–10 nm) |
2.2. Experimental Procedures
3. Results and Discussion
3.1. CO2 Hydrate Formation and Dissociation Process

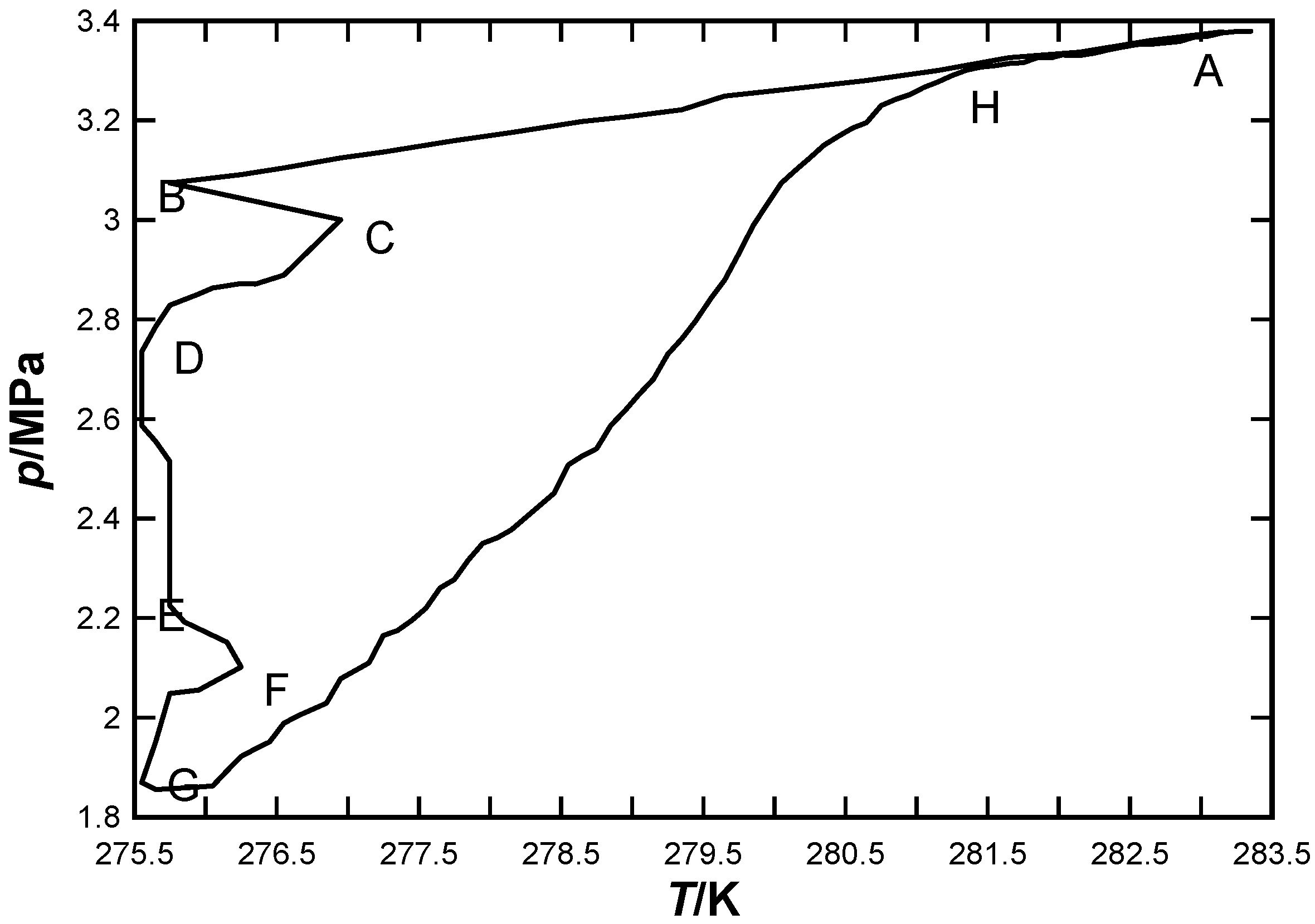
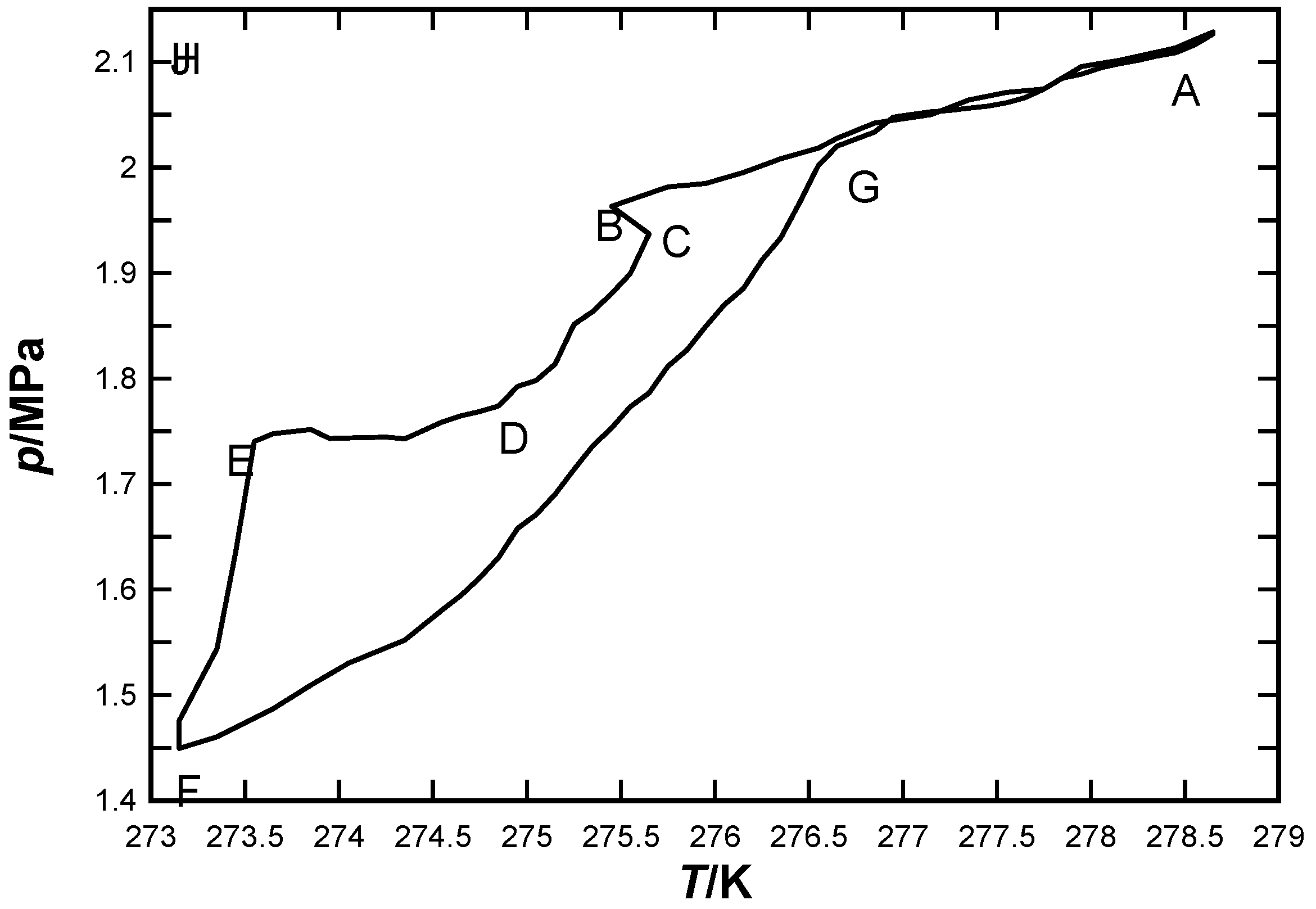
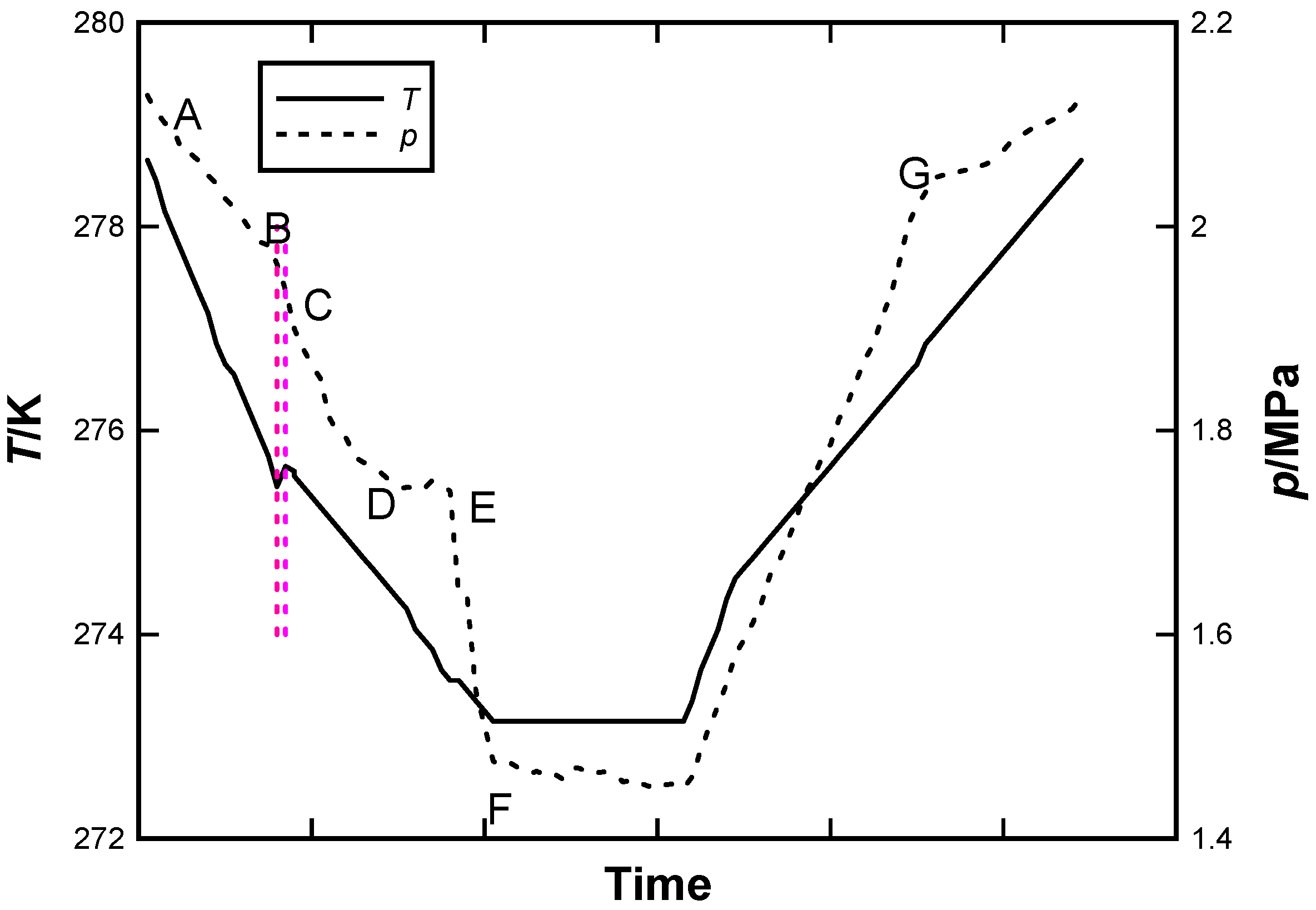
3.2. Determination of CO2 Hydrate Priority Formation Site Using MRI

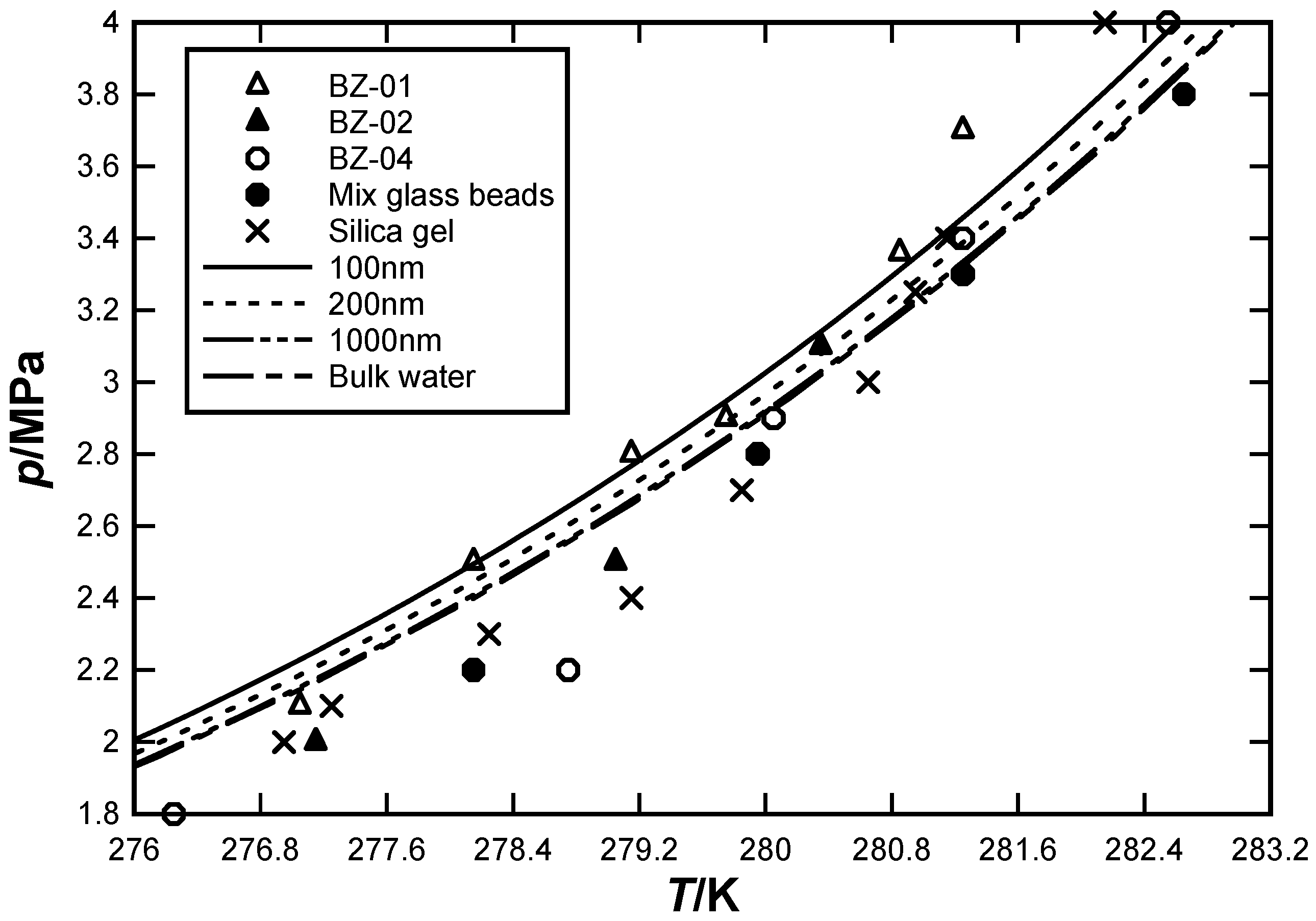
3.3. Effects of Pores Size on CO2 Hydrate Equilibrium Condition

3.4. Prediction of Hydrate Equilibrium Condition by the Improved Model
| Porous medium | T range/K | P range/MPa | Np | AADT (%) | AADP (%) |
|---|---|---|---|---|---|
| BZ-01 | 273.1–281.8 | 1.6–3.6 | 10 | 0.14 | 4.10 |
| BZ-02 | 274.8–282.0 | 1.5–4.1 | 5 | 0.32 | 9.29 |
| BZ-04 * | 276.2–282.8 | 1.8–4.0 | 5 | 0.21 | 5.36 |
| BZ-04 ** | 276.2–282.8 | 1.8–4.0 | 5 | 0.30 | 6.83 |
| Mix glass beads * | 275.2–282.7 | 1.5–3.8 | 5 | 0.20 | 8.78 |
| Mix glass beads ** | 275.2–282.7 | 1.5–3.8 | 5 | 0.24 | 9.38 |
| Silica gel * | 274.8–282.2 | 1.6–4.0 | 10 | 0.20 | 5.82 |
| Silica gel ** | 274.8–282.2 | 1.6–4.0 | 10 | 0.19 | 5.78 |
4. Conclusions
Acknowledgments
References
- Ji, Y.H.; Ji, X.Y.; Feng, X.; Liu, C.; Lv, L.H.; Lu, X.H. Progress in the study on the phase equilibria of the CO2-H2O and CO2-H2O-NaCl systems. Chin. J. Chem. Eng. 2007, 15, 439–448. [Google Scholar] [CrossRef]
- Bachu, S. Sequestration of CO2 in geological media: Criteria and approach for site selection in response to climate change. Energy Convers. Manag. 2000, 41, 953–970. [Google Scholar] [CrossRef]
- Unander, F. Energy Technology Perspectives: Scenarios and Strategies to 2050; Technical Report; International Energy Agency: Paris, France, 2008. [Google Scholar]
- Sloan, E.D. Clathrate Hydrates of Natural Gases, 2nd ed.; Marcel Dekker Inc.: New York, NY, USA, 1998. [Google Scholar]
- Yang, H.Q.; Xu, Z.H.; Fan, M.H.; Gupta, R.; Slimane, R.B.; Bland, A.E.; Wright, I. Progress in carbon dioxide separation and capture: A review. J. Environ. Sci. 2008, 20, 14–27. [Google Scholar] [CrossRef]
- Brewer, P.G.; Orr, F.M., Jr.; Friederich, G.; Kvenvolden, K.A.; Orange, D.L. Gas hydrate formation in the deep sea: In situ experiments with controlled release of methane, natural gas and carbon dioxide. Energy Fuels 1998, 12, 183–188. [Google Scholar] [CrossRef]
- Wendland, M.; Hasse, H.; Maurer, G. Experimental pressure-temperature data on three- and four-phase equilibria of fluid, hydrate and ice phases in the system carbon dioxide water. J. Chem. Eng. Data 1999, 44, 901–906. [Google Scholar] [CrossRef]
- Yang, S.O.; Yang, I.M.; Kim, Y.S.; Lee, C.S. Measurement and prediction of phase equilibria for water + CO2 mixture in hydrate forming conditions. Fluid Phase Equilib. 2000, 175, 75–89. [Google Scholar] [CrossRef]
- Englezos, P.; Hall, S. Phase equilibrium data on carbon dioxide hydrate in the presence of electrolytes, water soluble polymers and montmorillonite. Can. J. Chem. Eng. 1994, 72, 887–893. [Google Scholar] [CrossRef]
- Breland, E.; Englezos, P. Equilibrium hydrate formation data for carbon dioxide in aqueous glycerol solutions. J. Chem. Eng. Data 1996, 41, 11–13. [Google Scholar] [CrossRef]
- Dholabhai, P.D.; Scott Parent, J.; Raj Bishnoi, P. Carbon dioxide hydrate equilibrium conditions in aqueous solutions containing electrolytes and methanol using a new apparatus. Ind. Eng. Chem. Res. 1996, 35, 819–823. [Google Scholar] [CrossRef]
- Dholabhai, P.D.; Scott Parent, J.; Raj Bishnoi, P. Equilibrium conditions for hydrate formation from binary mixtures of methane and carbon dioxide in the presence of electrolytes, methanol and ethylene glycol. Fluid Phase Equilib. 1997, 41, 235–246. [Google Scholar] [CrossRef]
- Kang, S.P.; Chun, M.K.; Lee, H. Phase equilibria of methane and carbon dioxide hydrates in the aqueous MgCl2 solutions. Fluid Phase Equilib. 1998, 147, 229–238. [Google Scholar] [CrossRef]
- Mohammadi, A.H.; Afzal, W.; Richon, D. Gas hydrates of methane, ethane, propane and carbon dioxide in the presence of single NaCl, KCl and CaCl2 aqueous solutions: Experimental measurements and predictions of dissociation conditions. J. Chem. Thermodyn. 2008, 40, 1693–1697. [Google Scholar] [CrossRef]
- Handa, Y.P.; Stupin, D.Y. Thermodynamic properties and dissociation characteristics of methane and propane hydrates in 70-Å-radius silica gel pores. J. Phys. Chem. 1992, 96, 8599–8603. [Google Scholar] [CrossRef]
- Uchida, T.; Ebinuma, T.; Ishizaki, T. Dissociation condition measurements of methane hydrate in confined small pores of porous glass. J. Phys. Chem. B 1999, 103, 3659–3662. [Google Scholar] [CrossRef]
- Clennell, M.B.; Hovland, M.; Booth, J.S.; Henry, P.; Winters, W.J. Formation of natural gas hydrates in marine sediments 1: Conceptual model of gas hydrate growth conditioned by host sediment properties. J. Geophys. Res. 1999, 104, 22985–23003. [Google Scholar] [CrossRef]
- Henry, P.; Thomas, M.; Clennell, M.B. Formation of natural gas hydrates in marine sediments 2: Thermodynamic calculations of stability conditions in porous sediments. J. Geophys. Res. 1999, 104, 23005–23022. [Google Scholar] [CrossRef]
- Zatsepina, O.Y.; Buffet, B.A. Nucleation of CO2 hydrate in porous medium. Fluid Phase Equilib. 2002, 200, 263–275. [Google Scholar] [CrossRef]
- Smith, D.; Wilder, J.; Seshadri, K. Thermodynamics of carbon dioxide hydrate formation in media with broad pore-size distributions. Environ. Sci. Technol. 2002, 36, 5192–5198. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.; Llamedo, M.; Tohidi, B.; Burgass, R.W. Experimental measurement of methane and carbon dioxide clathrate hydrate equilibria in mesoporous silica. J. Phys. Chem. B. 2003, 107, 3507–3514. [Google Scholar] [CrossRef]
- Kumar, A. Formation and Dissociation of Gas Hydrates in Porous Media. Master Thesis, University of Calgary, Calgary, Canada, 2005. [Google Scholar]
- Turner, D.J.; Cherry, R.S.; Sloan, E.D. Sensitivity of methane hydrate phase equilibria to sediment pore size. Fluid Phase Equilib. 2005, 228–229, 505–510. [Google Scholar] [CrossRef]
- Aladko, E.Y.; Dyadin, Y.A.; Fenelonov, V.B.; Larionov, E.G.; Manakov, A.Y.; Mel’gunov, M.S.; Zhurko, F.V. Formation and decomposition of ethane, propane, and carbon dioxide hydrates in silica gel mesopores under high pressure. J. Phys. Chem. B 2006, 110, 19717–19725. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.C.; Yang, M.J.; Liu, Y.; Li, Q.P. Influence of ions on phase equilibrium of methane hydrate. CIESC J. 2009, 60, 1362–1366. [Google Scholar]
- Yang, M.J.; Song, Y.C.; Liu, Y.; Chen, Y.J.; Li, Q.P. Influence of pore size, salinity and gas composition upon the hydrate formation conditions. Chin. J. Chem. Eng. 2010, 18, 292–296. [Google Scholar] [CrossRef]
- Yang, M.J.; Song, Y.C.; Zhao, Y.C.; Liu, Y.; Jiang, L.L.; Li, Q.P. MRI measurements of CO2 hydrate dissociation rate in a porous medium. Magn. Reson. Imaging 2011, 29, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Wilder, J.W.; Seshadri, K.; Smith, D.H. Modeling hydrate formation in media with broad pore size distributions. Langmuir 2001, 17, 6729–6735. [Google Scholar] [CrossRef]
- Clarke, M.A.; Darvish, M.P.; Bishnoi, P.R. A method to predict equilibrium conditions of gas hydrate formation in porous media. Ind. Eng. Chem. Res. 1999, 38, 2485–2490. [Google Scholar] [CrossRef]
- Song, Y.C.; Yang, M.J.; Chen, Y.J.; Li, Q.P. An improved model for predicting hydrate phase equilibrium in marine sediment environment. J. Nat. Gas Chem. 2010, 19, 241–245. [Google Scholar] [CrossRef]
- Van der Waals, J.H.; Platteeuw, J.C. Clathrate solution. Adv. Chem. Phys. 1959, 2, 1–57. [Google Scholar]
- Nasrifar, K. A model for prediction of gas hydrate formation conditions in aqueous solutions containing electrolytes and/or alcohol. J. Chem. Thermodyn. 2001, 33, 999–1014. [Google Scholar] [CrossRef]
- Mei, D.H.; Liao, J.; Yang, J.T.; Guo, T.M. Prediction of equilibrium hydrate formation conditions in electrolyte aqueous solutions. Acta Petrolei Sinica 1998, 14, 86–93. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, M.; Song, Y.; Ruan, X.; Liu, Y.; Zhao, J.; Li, Q. Characteristics of CO2 Hydrate Formation and Dissociation in Glass Beads and Silica Gel. Energies 2012, 5, 925-937. https://doi.org/10.3390/en5040925
Yang M, Song Y, Ruan X, Liu Y, Zhao J, Li Q. Characteristics of CO2 Hydrate Formation and Dissociation in Glass Beads and Silica Gel. Energies. 2012; 5(4):925-937. https://doi.org/10.3390/en5040925
Chicago/Turabian StyleYang, Mingjun, Yongchen Song, Xuke Ruan, Yu Liu, Jiafei Zhao, and Qingping Li. 2012. "Characteristics of CO2 Hydrate Formation and Dissociation in Glass Beads and Silica Gel" Energies 5, no. 4: 925-937. https://doi.org/10.3390/en5040925




