Alcohol Consumption and Oxidative DNA Damage
Abstract
:1. Introduction
2. 8-Oxoguanine
3. 8-Oxoguanine Repair System
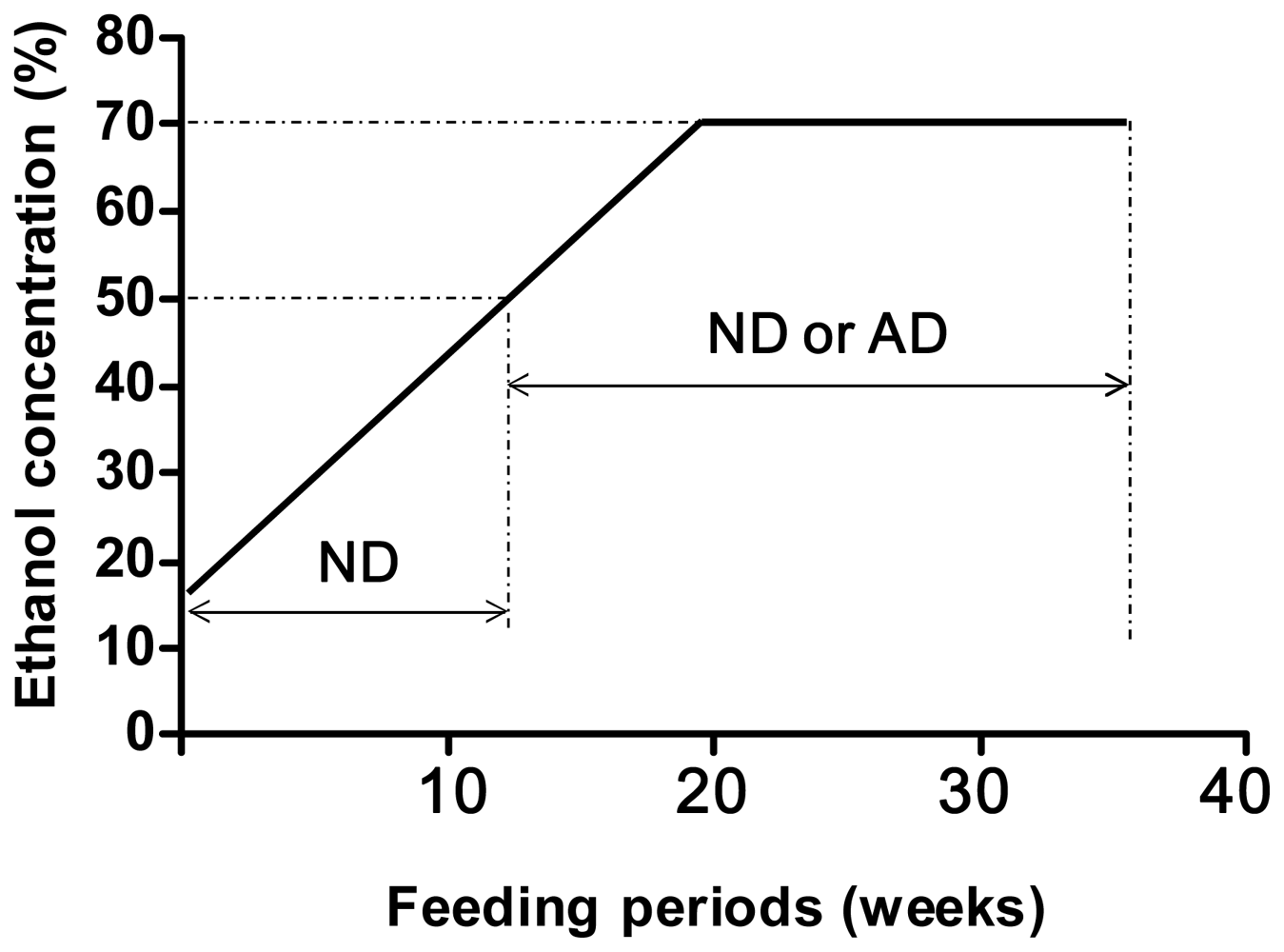
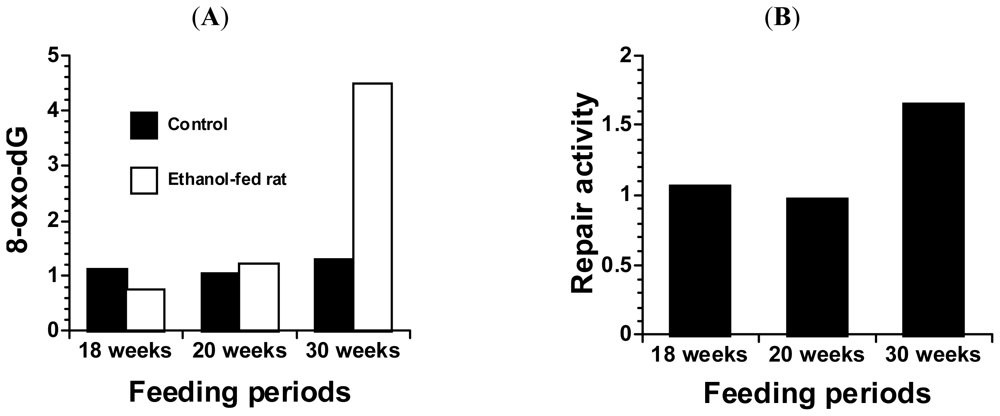
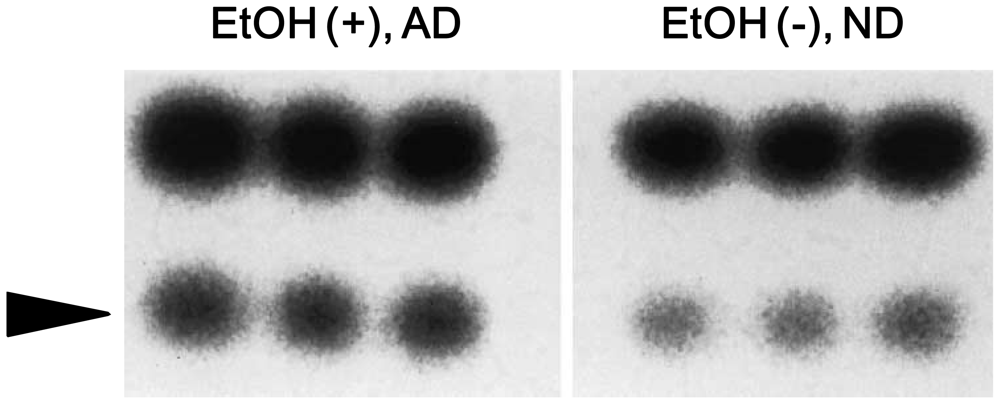
4. Alcohol and 8-Oxoguanine in Liver DNA
5. Alcohol and 8-Oxoguanine in Esophageal DNA
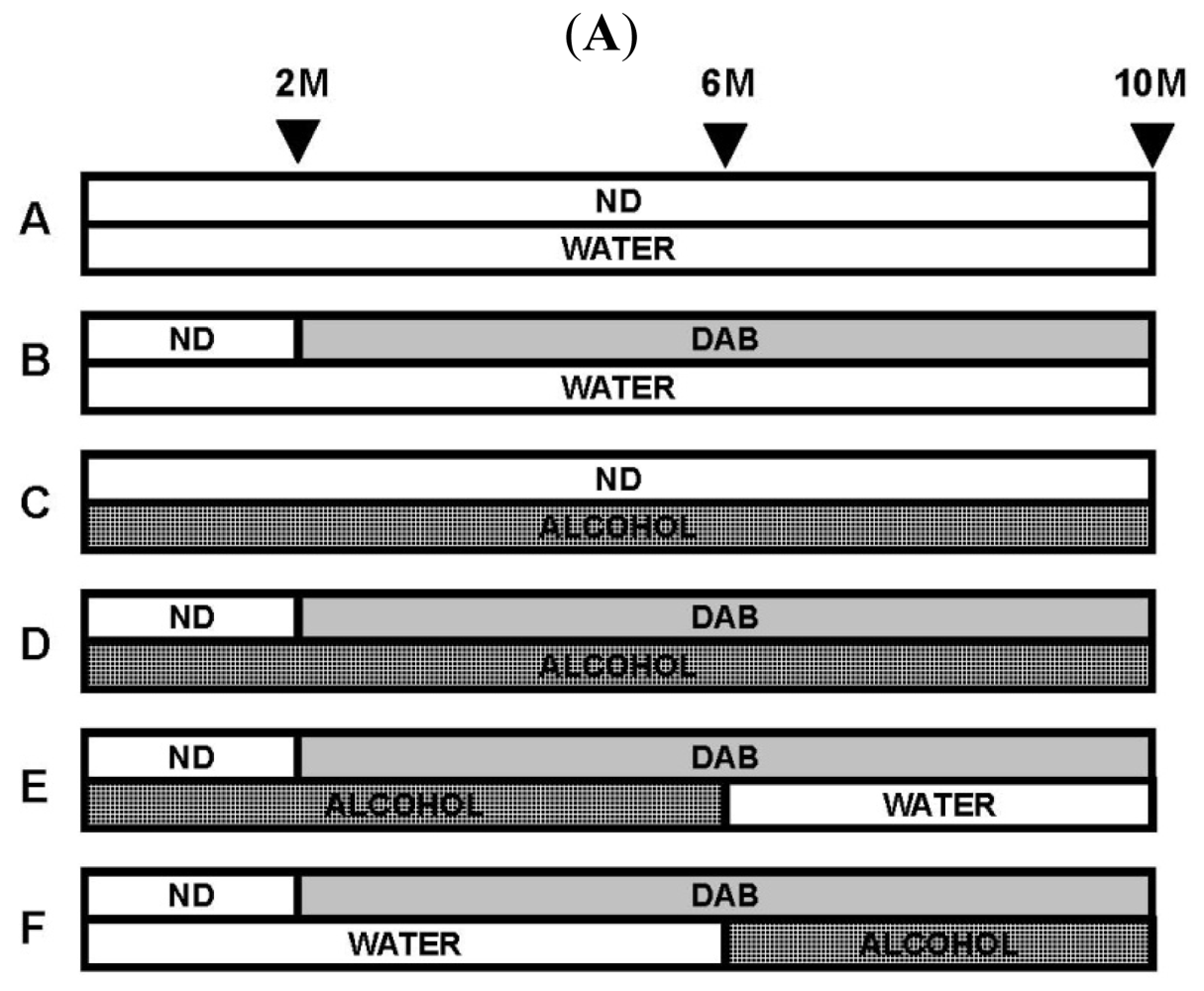
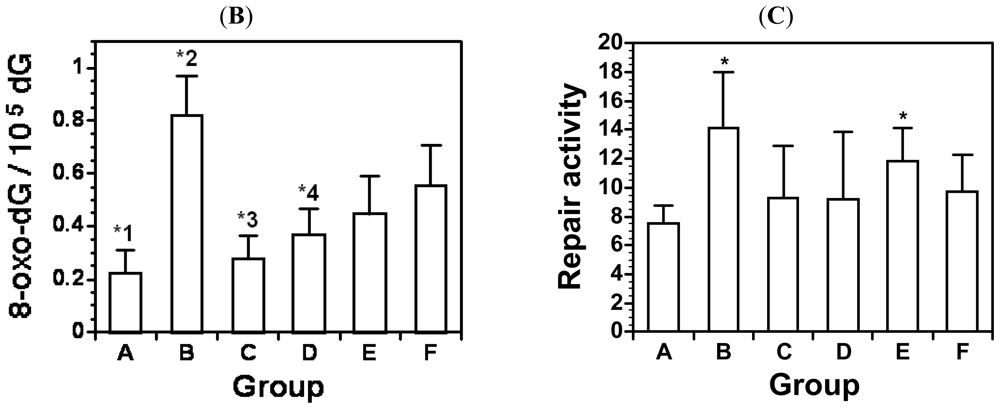
6. Chronic Alcohol Consumption Prevents 8-Oxoguanine Accumulation in the Livers of Carcinogen-Treated Mice
7. Conclusions
Acknowledgements
References
- Smith-Warner, SA; Spiegelman, D; Yaun, SS; van den Brandt, PA; Folsom, AR; Goldbohm, A; Graham, S; Holmberg, L; Howe, GR; Marshall, JR; et al. Alcohol and breast cancer in women: A pooled analysis of cohort studies. JAMA 1998, 279, 535–540. [Google Scholar]
- Stolzenberg-Solomon, RZ; Chang, SC; Leitzmann, MF; Johnson, KA; Johnson, C; Buys, SS; Hoover, RN; Ziegler, RG. Folate intake, alcohol use, and postmenopausal breast cancer risk in the prostate, lung, colorectal, and ovarian cancer screening trial. Am. J. Clin. Nutr 2006, 83, 895–904. [Google Scholar]
- Wu, M; Zhao, JK; Hu, XS; Wang, PH; Qin, Y; Lu, YC; Yang, J; Liu, AM; Wu, DL; Zhang, ZF; et al. Association of smoking, alcohol drinking and dietary factors with esophageal cancer in high- and low-risk areas of Jiangsu Province, China. World J. Gastroenterol 2006, 12, 1686–1693. [Google Scholar]
- Klatsky, AL; Armstrong, MA; Friedman, GD. The relations of alcoholic beverage use to colon and rectal cancer. Am. J. Epidemiol 1998, 128, 1007–1015. [Google Scholar]
- Ho, JW; Lam, TH; Tse, CW; Chiu, LK; Lam, HS; Leung, PF; Ng, KC; Ho, SY; Woo, J; Leung, SS; et al. Smoking, drinking and colorectal cancer in Hong Kong Chinese: A case control study. Int. J. Cancer 2004, 109, 587–597. [Google Scholar]
- Hirano, T; Homma, Y; Kasai, H. Formation of 8-Hydroxyguanine in DNA by Aging and Oxidative Stress. In Oxidative Stress and Aging; Cutler, RG, Packer, L, Bertran, J, Mori, A, Eds.; Birkhauser Verlag: Basel, Switzerland, 1995; pp. 69–76. [Google Scholar]
- Kasai, H; Nishimura, S. Hydroxylation of deoxyguanosine at the C-8 position by ascorbic acid and other reducing agents. Nucleic Acids Res 1984, 12, 2137–2145. [Google Scholar]
- Hirano, H; Hirano, T; Hirata, T; Tamura, M; Yamaura, T; Hamada, T. Experimental liver fibrosis induced in rats receiving high doses of alcohol and alternating between regular and vitamin-depleted diets. Experientia 1996, 52, 710–715. [Google Scholar]
- Asami, S; Hirano, T; Yamaguchi, R; Tsurudome, Y; Itoh, H; Kasai, H. Increase in 8-hydroxyguanine and its repair activity in the esophagi of rats given long-term ethanol and nutrition-deficient diet. Jpn. J. Cancer Res 2000, 91, 973–978. [Google Scholar]
- Hirano, T; Sakai, A; Ootsuyama, Y; Kasai, H. Chronic alcohol consumption prevents 8-hydroxyguanine accumulation in 3’-methyl-4-dimethylaminoazobenzene-treated mouse liver. Biochem. Biophys. Res. Commun 2009, 387, 316–320. [Google Scholar]
- Floyd, RA; Watson, JJ; Wong, PK; Altmiller, DH; Rickard, RC. Hydroxyl free radical adduct of deoxyguanosine: Sensitive detection and mechanisms of formation. Free Radical Res. Commun 1986, 1, 163–172. [Google Scholar]
- Marnett, LJ. Oxyradicals and DNA damage. Carcinogenesis 2000, 21, 361–370. [Google Scholar]
- Hirano, T; Yamaguchi, Y; Kasai, H. Inhibition of 8-hydroxyguanine repair in testes after administration of cadmium chloride to GSH-depleted rats. Toxicol. Appl. Pharmacol 1997, 147, 9–14. [Google Scholar]
- Tsurudome, Y; Hirano, T; Yamato, H; Tanaka, I; Sagai, M; Hirano, H; Nagata, N; Itoh, H; Kasai, H. Changes in levels of 8-hydroxyguanine in DNA, its repair and OGG1 mRNA in rat lungs after intratracheal administration of diesel exhaust particles. Carcinogenesis 1999, 20, 1573–1576. [Google Scholar]
- Hirano, T; Higashi, K; Sakai, A; Tsurudome, Y; Ootsuyama, Y; Kido, R; Kasai, H. Analyses of oxidative DNA damage and its repair activity in the livers of 3’-methyl-4-dimethylaminoazobenzene-treated rodents. Jpn. J. Cancer Res 2000, 91, 681–685. [Google Scholar]
- Mei, N; Tamae, K; Kunugita, N; Hirano, T; Kasai, H. Analysis of 8-hydroxydeoxyguanosine 5’-monophosphate (8-OH-dGMP) as a reliable marker of cellular oxidative DNA damage after gamma-irradiation. Environ. Mol. Mutagen 2003, 41, 332–338. [Google Scholar]
- Hirano, T; Yamaguchi, Y; Hirano, H; Kasai, H. Age-associated change of 8-hydroxyguanine repair activity in cultured human fibroblasts. Biochem. Biophys. Res. Commun 1995, 214, 1157–1162. [Google Scholar]
- Hirano, T; Yamaguchi, R; Asami, S; Iwamoto, N; Kasai, H. 8-Hydroxyguanine levels in nuclear DNA and its repair activity in rat organs associated with age. J. Gerontol 1996, A51, B303–B307. [Google Scholar]
- Tsurudome, Y; Hirano, T; Hirata, K; Higure, A; Nagata, N; Takahashi, K; Itoh, H; Kasai, H. Age associated increase of 8-hydroxydeoxyguanosine in human colorectal tissue DNA. J. Gerontol 2001, 56A, B483–B485. [Google Scholar]
- Asami, S; Hirano, T; Yamaguchi, R; Tomioka, Y; Itoh, H; Kasai, H. Increase of a type of oxidative DNA damage, 8-hydroxyguanine, and its repair activity in human leukocytes by cigarette smoking. Cancer Res 1996, 56, 2546–2549. [Google Scholar]
- Asami, S; Hirano, T; Yamaguchi, R; Tsurudome, Y; Itoh, H; Kasai, H. Effects of forced and spontaneous exercise on 8-hydroxydeoxyguanosine levels in rat organs. Biochem. Biophys. Res. Commun 1998, 243, 678–682. [Google Scholar]
- Cheng, KC; Cahill, DS; Kasai, H; Nishimura, S; Loeb, LA. 8-Hydroxyguanine, an abundant form of oxidative DNA damage, causes G→T and A→C substitutions. J. Biol. Chem 1992, 267, 166–172. [Google Scholar]
- Basu, AK; Loechler, EL; Leadon, SA; Essigmann, JM. Genetic effects of thymine glycol: Site-specific mutagenesis and molecular modeling studies. Proc. Natl. Acad. Sci. USA 1989, 86, 7677–7681. [Google Scholar]
- Yamaguchi, R; Hirano, T; Asami, S; Chung, KM; Sugita, A; Kasai, H. Increased 8-hydroxyguanine levels in DNA and its repair activity in rat kidney after administration of a renal carcinogen, ferric nitrilotriacetate. Carcinogenesis 1996, 17, 2419–2422. [Google Scholar]
- Mei, N; Kunugita, N; Hirano, T; Kasai, H. Acute arsenite-induced 8-hydroxyguanine is associated with inhibition of repair activity in cultured human cells. Biochem. Biophys. Res. Commun 2002, 297, 924–930. [Google Scholar]
- Hoeijimakers, JH. Genome maintenance mechanisms for preventing cancer. Nature 2001, 411, 366–374. [Google Scholar]
- Wood, ML; Dizdarogle, M; Gajewski, E; Essigmann, JM. Mechanistic studies of ionizing radiation and oxidative mutagenesis: Genetic effects of a single 8-hydroxyguanine (7-hydro-8-oxoguanine) residue inserted at a unique site in a viral genome. Biochemistry 1990, 29, 7024–7032. [Google Scholar]
- Shibutani, S; Takeshita, M; Grollman, AP. Insertion of specific bases during DNA synthesis past the oxidation-damaged base 8-oxodG. Nature 1991, 349, 431–434. [Google Scholar]
- Kamiya, H; Miura, K; Ishikawa, H; Inoue, H; Nishimura, S; Ohtsuka, E. c-Ha-ras containing 8-hydroxyguanine at codon 12 induces point mutations at the modified and adjacent positions. Cancer Res 1992, 52, 3483–3485. [Google Scholar]
- Kasai, H; Crain, PF; Kuchino, Y; Nishimura, S; Ootsuyama, A; Tanooka, H. Formation of 8-hydroxyguanine moiety in cellular DNA by agents producing oxygen radicals and evidence for its repair. Carcinogenesis 1986, 11, 1849–1851. [Google Scholar]
- Boiteux, S; O’Connor, TR; Lederer, F; Gouyette, A; Laval, J. Homogeneous Escherichia coli FPG protein. J. Biol. Chem 1990, 265, 3916–3922. [Google Scholar]
- Auffret van der Kemp, P; Thomas, D; Barbey, R; De Oliveira, R; Boiteux, S. Cloning and expression in Escherichia coli of the OGG1 gene of Saccharomyces cerevisiae, which codes for a DNA glycosylase that excises 7,8-dihydro-8-oxoguanine and 2,6-diamino-4-hydroxy-5-N-methyl-formamidopyrimidine. Proc. Natl. Acad. Sci. USA 1996, 93, 5197–5202. [Google Scholar]
- Nash, HM; Bruner, SD; Schärer, OD; Kawate, T; Addona, TA; Sponner, E; Lane, WS; Verdine, GI. Cloning of a yeast 8-oxoguanine DNA glycosylase reveals the existence of a base-excision DNA-repair protein superfamily. Curr. Biol 1996, 6, 968–980. [Google Scholar]
- Rosenquist, TA; Zharkov, DO; Grollman, AP. Cloning and characterization of a mammalian 8-oxoguanine DNA glycosylase. Proc. Natl. Acad. Sci. USA 1997, 94, 7429–7434. [Google Scholar]
- Radicella, JP; Dherin, C; Desmaze, C; Fox, MS; Boiteux, S. Cloning and characterization of hOGG1, a human homolog of OGG1 gene of Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1997, 94, 8010–8015. [Google Scholar]
- Lu, R; Nash, HM; Verdine, GL. A mammalian DNA repair enzyme that excises oxidatively damaged guanines maps to a locus frequently lost in lung cancer. Curr. Biol 1997, 7, 397–407. [Google Scholar]
- Roldán-Arjona, T; Wei, YF; Carter, KC; Klungland, A; Anselmino, C; Wang, RP; Augustus, M; Lindahl, T. Molecular cloning and functional expression of a human cDNA encoding the antimutator enzyme 8-hydroxyguanine-DNA glycosylase. Proc. Natl. Acad. Sci. USA 1997, 94, 8016–8015. [Google Scholar]
- Bjoräs, M; Luna, L; Johnson, B; Hoff, E; Haug, T; Rongnes, T; Seeberg, E. Opposite base-dependent reactions of a human base excision repair enzyme on DNA containing 7,8-dihydro-8-oxoguanine and abasic sites. EMBO J 1997, 16, 6314–6322. [Google Scholar]
- Arai, K; Morishita, K; Shinmura, K; Kohno, T; Kim, SR; Nohmi, T; Taniwaki, M; Ohwada, S; Yokota, J. Cloning of a human homolog of the yeast OGG1 gene that is involved in the repair of oxidative DNA damage. Oncogene 1997, 14, 2857–2861. [Google Scholar]
- Aburatani, H; Hippo, Y; Ishida, T; Takashima, R; Matsuba, C; Kodama, T; Takao, M; Yasui, A; Yamamoto, K; Asano, M; et al. Cloning and characterization of mammalian 8-hydroxy- guanine-specific DNA glycosylase/apurinic, a pyrimidinic lyase, a functional mutM homologue. Cancer Res 1997, 57, 2151–2156. [Google Scholar]
- Tani, M; Shinmura, K; Kohno, T; Shiroishi, T; Wakana, S; Kim, SR; Nohmi, T; Kasai, H; Takenoshita, S; Nagamachi, Y; et al. Genomic structure and chromosomal localization of the mouse Ogg1 gene that is involved in the repair of 8-hydroxyguanine in DNA damage. Mamm. Genome 1989, 9, 32–37. [Google Scholar]
- Lehman, TA; Bennett, WP; Metcalf, RA; Welsh, JA; Ecker, J; Modali, RV; Ullrich, S; Romano, JW; Appella, E; Testa, JR; et al. p53 mutations, ras mutations and p53-heat shock 70 protein complexes in human lung carcinoma cell lines. Cancer Res 1991, 51, 4090–4096. [Google Scholar]
- Iggo, R; Gatter, K; Bartek, J; Lane, D; Harris, AL. Increased expression of mutant forms of p53 oncogene in primary lung cancer. Lancet 1990, 335, 675–679. [Google Scholar]
- Takahashi, T; Nau, MM; Chiba, I; Birrer, MJ; Rosenberg, RK; Vinocour, M; Levitt, M; Pass, H; Gazdar, AF; Minna, JD. P53: A frequent target for genetic abnormalities in lung cancer. Science 1989, 246, 491–494. [Google Scholar]
- D’Amico, D; Carbone, D; Mitsudomi, T; Nau, M; Fedorko, J; Russell, E; Johnson, B; Buchhagen, D; Bodner, S; Phelps, R. High frequency of somatically acquired p53 mutations in small-cell lung cancer cell lines and tumors. Oncogene 1992, 7, 339–346. [Google Scholar]
- Hussain, SP; Harris, CC. Molecular epidemiology of human cancer: Contribution of mutation spectra studies of tumor suppressor genes. Cancer Res 1998, 58, 4023–4037. [Google Scholar]
- Chevillard, S; Radicella, JP; Levalois, C; Lebeau, J; Poupon, MF; Oudard, S; Dutrillaux, B; Boiteux, S. Mutations in OGG1, a gene involved in the repair of oxidative DNA damage, are found in human lung and kidney tumours. Oncogene 1998, 16, 3083–3086. [Google Scholar]
- Sugimura, H; Kohno, T; Wakai, K; Nagura, K; Genka, K; Igarashi, H; Morris, BJ; Baba, S; Ohno, Y; Gao, C; et al. hOGG1 Ser326Cys polymorphism and lung cancer susceptibility. Cancer Epidemiol. Biomarkers Prev 1999, 8, 669–674. [Google Scholar]
- Wikman, H; Risch, A; Klimek, F; Schmezer, P; Spiegelhalder, B; Dienemann, H; Kayser, K; Schulz, V; Drings, P; Bartsch, H. hOGG1 polymorphism and loss of heterozygosity (LOH): Significance for lung cancer susceptibility in a Caucasian population. Int. J. Cancer 2000, 88, 932–937. [Google Scholar]
- Jensen, A; Calvayrac, G; Karahalil, B; Bohr, VA; Stevnsner, T. Mammalian 8-oxoguanine DNA glycosylase 1 incises 8-oxoadenine opposite cytosine in nuclei and mitochondria, while a different glycosylase incises 8-oxoadenine opposite guanine in nuclei. J. Biol. Chem 2003, 278, 19541–19548. [Google Scholar]
- Hirano, T. Repair system of 7,8-dihydro-8-oxoguanine as a defense line against carcinogenesis. J. Radiat. Res 2008, 49, 329–340. [Google Scholar]
- Lu, AL; Fawcett, WP. Characterization of the recombinant MutY homolog, an adenine DNA glycosylase, from yeast Schizosaccharomyces pombe. J. Biol. Chem 1998, 273, 25098–25105. [Google Scholar]
- Kakuma, T; Nishida, J; Tsuzuki, T; Sekiguchi, M. Mouse MTH1 protein with 8-oxo-7,8-dihydro-2’-deoxyguanosine 5’-triphosphatase activity that prevents transversion mutation. J. Biol. Chem 1995, 270, 25942–25948. [Google Scholar]
- Labuc, GE; Blunck, JM. Metabolic activation of the hepatocarcinogen 3’-methyl-4-dimethyl-aminoazobenzene by a rat liver cell-free-system. Biochem. Pharmacol 1979, 28, 2367–2373. [Google Scholar]
- Tarpley, WG; Miller, JA; Miller, EC. Adducts from the reaction of N-benzoyloxy-N-methyl-4-aminoazobenzene with deoxyguanosine or DNA in vitro and from hepatic DNA of mice treated with N-methyl- or N,N-dimethyl-4-aminoazobenzene. Cancer Res 1980, 40, 2493–2499. [Google Scholar]
- Tullis, DL; Dooley, KL; Miller, DW; Baetcke, KP; Kadlubar, FF. Characterization and properties of the DNA adducts formed from N-methyl-4-aminoazobenzene in rats during a carcinogenic treatment regimen. Carcinogenesis 1987, 8, 577–583. [Google Scholar]
- Duchesne, J; Goutier, R; Maréchal, R; van de Vorst, A. Free radical evolution in rat liver following the ingestion of DAB. Phys. Med. Biol 1975, 20, 305–309. [Google Scholar]
 : 8-oxo-Gua.
: 8-oxo-Gua.
 : 8-oxo-Gua.
: 8-oxo-Gua.
| Ingredient | Normal diet | Autoclaved diet |
|---|---|---|
| Vitamin B1 | 1.73 mg | 0.35 mg (20%) |
| Vitamin C | 19.0 mg | 9.0 mg (47%) |
| Retinol | 0.36 mg | 0.19 mg (53%) |
| Vitamin B12 | 4.9 μg | 2.6 μg (53%) |
| Inositol | 1.63 mg | 109 mg (67%) |
| Pantoic acid | 2.85 mg | 2.21 mg (78%) |
| Vitamin B6 | 1.26 mg | 0.99 mg (79%) |
| Niacin | 15.9 mg | 14.0 mg (88%) |
| Vitamin B2 | 1.37 mg | 1.21 mg (88%) |
| Vitamin E | 11.0 mg | 10.0 mg (91%) |
| Folic acid | 0.15 mg | 0.15 mg (100%) |
| Biotin | 47.8 μg | 48.1 μg (101%) |
| Choline | 250 mg | 260 mg (104%) |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hirano, T. Alcohol Consumption and Oxidative DNA Damage. Int. J. Environ. Res. Public Health 2011, 8, 2895-2906. https://doi.org/10.3390/ijerph8072895
Hirano T. Alcohol Consumption and Oxidative DNA Damage. International Journal of Environmental Research and Public Health. 2011; 8(7):2895-2906. https://doi.org/10.3390/ijerph8072895
Chicago/Turabian StyleHirano, Takeshi. 2011. "Alcohol Consumption and Oxidative DNA Damage" International Journal of Environmental Research and Public Health 8, no. 7: 2895-2906. https://doi.org/10.3390/ijerph8072895




