Human Health Risk Assessment of Pharmaceuticals in Water: Issues and Challenges Ahead
Abstract
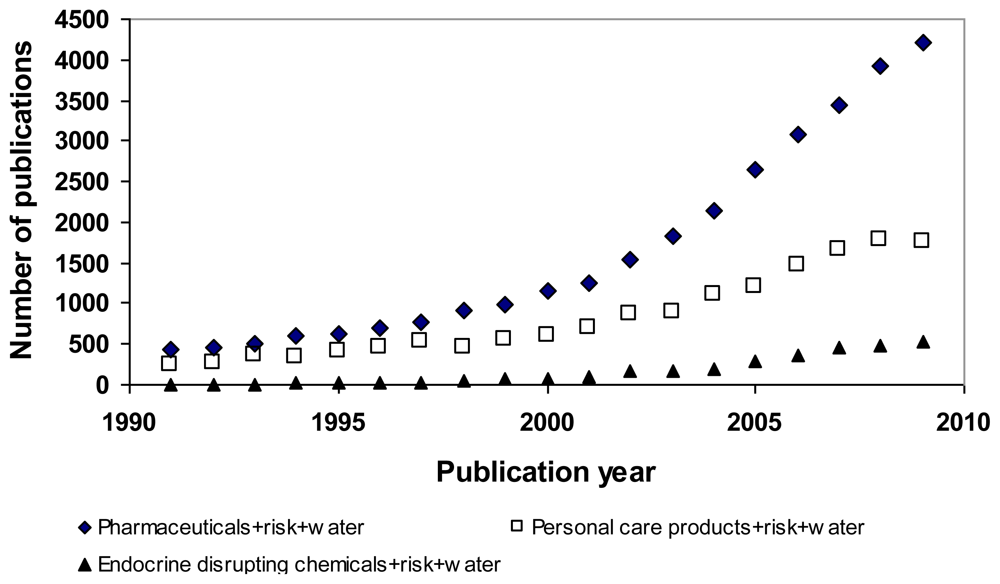
:1. Introduction
2. Identification of Existing Issues
2.1. Hazard Identification
2.1.1. Measured versus predicted pharmaceutical concentration
2.1.2. Pharmaceuticals-of-concern
2.2. Exposure Assessment
2.2.1. Exposure routes
2.3. Dose-Response Relationship
2.3.1. Uncertainty factors
2.3.2. Endpoints
2.3.3. Sensitive subpopulation
2.3.4. Mixture effects
3. Conclusions
Acknowledgements
References and Notes
- Benotti, MJ; Trenholm, BA; Vanderford, BJ; Holady, JC; Stanford, BD; Snyder, SA. Pharmaceuticals and endocrine disrupting compounds in U.S. drinking water. Environ. Sci. Technol 2009, 43, 597–603. [Google Scholar]
- Jones, OA; Lester, JN; Voulvoulis, N. Pharmaceuticals: a threat to drinking water? Trends Biotechnol 2005, 23, 163–167. [Google Scholar]
- Schwab, RW; Hayes, EP; Fiori, JM; Mastrocco, FJ; Roden, NM; Cragin, D; Meyerhoff, RD; D’Aco, VJ; Anderson, PD. Human pharmaceuticals in US surface waters: A human health risk assessment. Regul. Toxicol. Pharmacol 2005, 42, 296–312. [Google Scholar]
- Stackelberg, PE; Gibs, J; Furlong, ET; Meyer, MT; Zaugg, SD; Lippincott, RL. Efficiency of conventional drinking-water-treatment processes in removal of pharmaceuticals and other organic compounds. Sci. Total Environ 2007, 377, 255–272. [Google Scholar]
- Xagoraraki, I; Kuo, D. Heggenhougen, K, Quah, S, Eds.; Water pollution: Emerging contaminants associated with drinking water. In International Encyclopedia of Public Health; Academic Press: San Diego, CA, USA, 2008; pp. 539–550. [Google Scholar]
- Bercu, JP; Parke, NJ; Fiori, JM; Meyerhoff, RD. Human health risk assessments for three neuropharmaceutical compounds in surface waters. Regul. Toxicol. Pharmacol 2008, 50, 420–427. [Google Scholar]
- Christensen, FM. Pharmaceuticals in the environment—A human risk? Regul. Toxicol. Pharmacol 1998, 28, 212–221. [Google Scholar]
- Cunningham, VL; Binks, SP; Olson, MJ. Human health risk assessment from the presence of human pharmaceuticals in the aquatic environment. Regul. Toxicol. Pharmacol 2009, 53, 39–45. [Google Scholar]
- Cunningham, VL; Perino, C; D’Aco, VJ; Hartmann, A; Bechter, R. Human health risk assessment of carbamazepine in surface waters of North America and Europe. Regul. Toxicol. Pharmacol 2010, 56, 343–351. [Google Scholar]
- Kumar, A; Xagoraraki, I. Human health risk assessment of pharmaceuticals in water: An uncertainty analysis for meprobamate, carbamazepine, and phenytoin. Regul. Toxicol. Pharmacol 2010a, 57, 146–156. [Google Scholar]
- Schulman, LJ; Sargent, EV; Naumann, BD; Faria, EC; Dolan, DG; Wargo, JP. A human health risk assessment of pharmaceuticals in the aquatic environment. Hum. Eco. Risk Assess 2002, 8, 657–680. [Google Scholar]
- Webb, S; Ternes, T; Gibert, M; Olejniczak, K. Indirect human exposure to pharmaceuticals via drinking water. Toxicol. Lett 2003, 142, 157–167. [Google Scholar]
- Donn, J; Mendoza, M; Pritchard, J. AP: Drugs Found in Drinking Water. Available online: http://www.usatoday.co/news/nation/2008-03-10-drugs-tap-water_N.htm (accessed on 26 February 2009).
- Larsson, DGJ; de Pedro, C; Paxeus, N. Effluent from drug manufactures contains extremely high levels of pharmaceuticals. J. Hazard Mater 2007, 148, 751–755. [Google Scholar]
- Rodriguez-Mozaz, S; Weinberg, HS. Meeting report: Pharmaceuticals in water-an interdisciplinary approach to a public health challenge. Environ. Health Perspect 2010, 118, 1016–1020. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Available online: http://www.epa.gov/risk/health-risk.htm (accessed on 26 February 2009).
- Rowney, NC; Johnson, AC; Williams, RJ. Cytotoxic drugs in drinking water: A prediction and risk assessment exercise for the Thames catchment in the United Kingdom. Environ. Toxicol. Chem 2009, 28, 2722–2743. [Google Scholar]
- Snyder, SA. Occurrence, treatment, and toxicological relevance of EDCs and pharmaceuticals in water. Ozone: Sci. Eng 2008, 30, 65–69. [Google Scholar]
- Watts, C; Maycock, D; Crane, M; Fawell, J; Goslan, E. Desk Based Review of Current Knowledge on Pharmaceuticals in Drinking Water and Estimation of Potential Levels; Final Report to Defra Project Code: CSA 7184/WT02046/DWI70/2/213; 2007. [Google Scholar]
- Johnson, AC; Jurgens, MD; Williams, RJ; Kummerer, K; Kortenkamp, A; Sumpter, JP. Do cytotoxic chemotherapy drugs discharged into rivers pose a risk to the environment and human health? An overview and UK case study. J. Hydrol 2008, 348, 167–175. [Google Scholar]
- Crider, KS; Cleves, MA; Reefhuis, J; Berry, RJ; Hobbs, CA; Hu, DJ. Antibacterial medication use during pregnancy and risk of birth defects. Arch. Pediatr. Adolesc. Med 2009, 163, 978–985. [Google Scholar]
- Schriks, M; Heringa, MB; van der Kooi, MME; de Voogt, P; van Wezel, AP. Toxicological relevance of emerging contaminants for drinking water quality. Water Res 2009, 44, 61–476. [Google Scholar]
- Kolpin, DW; Furlong, ET; Meyer, MT; Thurman, EM; Zaugg, SD; Barber, LB; Buxton, HT. Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999–2000: A national reconnaissance. Environ. Sci. Technol 2002, 36, 1202–1211. [Google Scholar]
- Anderson, PD; D’Aco, VJ; Shanahan, P; Chapra, SC; Buzby, ME; Cunningham, VL; Duplessie, BM; Hayes, EP; Mastrocco, FJ; Parke, NJ; Rader, JC; Samuelian, JH; Schwab, BW. Screening analysis of human pharmaceutical compounds in U.S. surface waters. Environ. Sci. Technol 2004, 38, 838–849. [Google Scholar]
- Feijtel, T; Boeije, G; Matthies, M; Young, A; Morris, G; Gandolfi, C; Hansen, B; Fox, K; Holt, M; Koch, V; Schroder, R; Cassani, G; Schowanek, D; Rosenblom, J; Niessen, H. Development of a geography-referenced regional exposure assessment tool for European rivers—GREAT-ER. Contribution to GREAT-ER #1. Chemosphere 1997, 34, 2351–2373. [Google Scholar]
- Dorne, JLCM; Ragas, AMJ; Frampton, GK; Spurgeon, DS; Lewis, DF. Trends in human risk assessment of pharmaceuticals. Anal. Bioanal. Chem 2007, 387, 1167–1172. [Google Scholar]
- Li, D; Yang, M; Hu, J; Ren, L; Zhang, Y; Li, K. Determination and fate of oxytetracycline and related compounds in oxytetracycline production wastewater and the receiving river. Environ. Toxicol. Chem 2008, 27, 80–86. [Google Scholar]
- Phillips, PJ; Smith, SG; Kolpin, DW; Zuagg, SD; Buxton, HT; Furlong, ET; Esposito, K; Stinson, B. Pharmaceutical formulation facilities as sources of opioids and other pharmaceuticals to wastewater-treatment-plant effluents. Environ. Sci. Technol 2010, 44, 4910–4916. [Google Scholar]
- United States Environmental Protection Agency (USEPA), Guidelines for Exposure Assessment; EPA 600-Z-92-001; USEPA: Washington, DC, USA, 1992.
- United States Environmental Protection Agency (USEPA), Methodology for Deriving Ambient Water Quality Criteria for the Protection of Human Health; EPA-822-B-00-004; USEPA: Washington, DC, USA, 2000.
- United States Environmental Protection Agency (USEPA). Human Health Exposure Assessment. Available online: http://epa.gov/region08/r8risk/hh_exposure.html (accessed on 14 April 2009).
- Barnes, DG; Dourson, M. Reference dose (RfD): Description and use in health risk assessments. Regul. Toxicol. Pharmacol 1988, 8, 471–486. [Google Scholar]
- Cleuvers, M. Mixture toxicity of the anti-inflammatory drugs diclofenac, ibuprofen, naproxen, and acetylsalicylic acid. Ecotoxicol. Environ. Saf 2004, 59, 309–315. [Google Scholar]
- United States Environmental Protection Agency (USEPA) , Guidelines for the Health Risk Assessment of Chemical Mixtures; EPA-630-R-98-002; USEPA: Washington, DC, USA, 1986.
- Silva, E; Rajapakse, N; Kortenkamp, A. Something from “Nothing”—Eight weak estrogenic chemicals combined at concentrations below NOECs produce significant mixture effects. Environ. Sci. Technol 2002, 36, 1751–1756. [Google Scholar]
- Pomati, F; Orlandi, C; Clerici, M; Luciani, F; Zuccato, E. Effects and interactions in an environmentally relevant mixture of pharmaceuticals. Toxicol. Sci 2008, 102, 129–137. [Google Scholar]
- Kumar, A; Xagoraraki, I. Pharmaceuticals, personal care products and endocrine-disrupting chemicals in U.S. surface and finished drinking waters: A proposed ranking system. Sci. Total Environ 2010b, 408, 5972–5989. [Google Scholar]
| Reference (Country) | Hazard Identification | Exposure Assessment | Dose-response Relationship | Risk Characterization and Conclusions |
|---|---|---|---|---|
| Christensen [7] (Denmark) (Academia) | 17α-ethinylestradiol (EE2), phenoxymethylpenicillin (Pen V), Cyclophosphamide (CP) | Intake of drinking water, leaf crops, root crops, fishes, dairy products, and inhalation of air; Only one subpopulation type (adults: 70 kg body weight) | For EE2: Higher intake from fish than other medium; For PenV and CP: Higher intakes from drinking water than other medium For PenV-at least 10 international units of penicillin (5.9 μg penicillin) is required to trigger a mode of action. For CP-toxic endpoint is genotoxicity (i.e., a genotoxic carcinogen) No consideration for mixture effect | Negligible human risks; For the case of PenV-considerations are required for sensitive population which may react with few molecules; More risk assessment studies for other veterinary drugs |
| Schulman et al. [11] (Europe) (Academia) | Acetylsalicylic acid, clofibrate, cyclophosphamide, indomethacin | Ingestion of water and consumption of fish Water ingestion rate: 2 L/d (adults); Fish ingestion rate: 0.0178 kg/d general population and sports anglers, 0.039 kg/d subsistence fishers; Body weights: 70 kg (adults) and 30 kg(children) | No health risks | |
| Webb et al. [12] (Europe) (Academia and Industry) | Acetyl salicylic acid, clofibric acid, ibuprofen, gemfibrozil, fenoprofen, ketoprofen, diclofenac, fenofibric acid, bezafibrate, indometacine, salicylic acid, atenolol, sotalol, salbutamol, terbutalin, fenoterol, nadolol, metropolol, celiprolol, carazolol, clenbuterol, phenazone, ifosfamide, cyclophosphamide, carbamazepine, pentoxifylline, clofibrate, phenazone, dimethlaminophenazon, ifosfamide, cyclophosphamide, carbamazepine, pentoxifylline, diazepam, fenofibrate, etofibrate, clarithromycin, dehydrato-erythromycin, roxithromycin, sulfamethazine, sulfamethoxazole, trimethoprim, chloramphenicol, chloroteracycline, doxycycline, tetracycline, cloxacillin, dicloxacillin, methicillin, nafcillin, oxacillin, benzylpenicillin, phenoxymethylpenicillin, iopamidol, iopromide, ioxithalamic acid, iothalamic acid, diatrizoate, 17α-Ethinylestradiol | Ingestion of water Water ingestion rate: 2 L/d (adults); | Comparisons of daily intake values (or dose equivalent for exposures for 70 years) with minimum daily therapeutic dose; No consideration for mixture effect | Low possibility of health risks |
| Schwab et al. [3] (U.S.A.) (Industry) | Acetaminophen, ablution, cimetidine, ciprofloxacin, codeine, dehydronifedipine, digoxigenin, digoxin, diltiazem, doxycycline, enalaprilat, erithromycin-H2O, fluoxetine, gemfibrozil, ibuprofen, lincomycin, metformin, norfloxacin, oxytetracycline, paroxetine metabolite, ranitidine, sulfamethoxazole, sulfathiazole, tetracycline, trimethoprim, warfarin | Ingestion of surface water and consumption of fish Water ingestion rate: 2 L/d (adults) and 1 L/d (children); Fish ingestion rate: 0.0175 kg/d (adults) and 0.0065 kg/d (children); Both subpopulation types with body weights = 70 kg (adults) and 14 kg (children); also inclusion of elderly and infirm where pharmacologic effect is undesirable Non-volatile active pharmaceutical ingredients => inhalation exposure is neglected; Similarly, dermal exposure pathways is assumed to contribute smaller risk compared to incidental ingestion risks | Use of both measured and predicted environmental concentrations and comparison of these values with calculated predicted-no-effect concentration (PNEC) values (separately for consumption of drinking water, fish, and for simultaneous consumption of both drinking water and fish) No consideration for mixture effect | No appreciable risks to humans Due to smaller values of bioconcentration factors, fish consumption is unlikely to be major exposure pathway |
| Watts et al. [19] (U.K.) (Industry and Academia) | 396 pharmaceuticals and 11 illicit drugs | Ingestion of surface water Water ingestion rate: 1 L/d (adults) (no considerations for sensitive subpopulation) Calculation of surface water pharmaceutical concentration using the European Medicines Agency (EMA) models | Comparison of minimum therapeutic dose (MTD) with estimated exposure dose Quantitative consideration for mixture effect | No human health risks (margin-of-safety observed >1,000 for most of the drugs except for some drugs where ratios were observed to be below 100, when combination of 19 NSADI were used with MTD value. |
| Bercu et al. [6] (U.S.A.) (Industry) | Atomoxetine, duloxetine, olanzapine (neuropharmaceutical compounds) | Ingestion of surface water and consumption of fish Water ingestion rate: 2 L/d (adults) and 1 L/d (children) Fish ingestion rate: 0.0175 kg/d (adults) and 0.013 kg/d (children) Body weights: 70 kg (adults) and 30 kg(children) Calculation of surface water pharmaceutical concentration using the USFDA (1998) and PhFATE surface water models | Determination of lowest relevant effect dose using preclinical and clinical studies and its use for calculation of ADI; Use of human study for estimation of point-of-departure (POD) if effect is same from both animal and human studies and use of animal study with uncertainty factors for estimation of POD. Use of default uncertainty factors in the case of insufficient data for extrapolation purposes and where applicable, use of clinical trial data (i.e., chemical- specific-adjustment factors) accounting human variability Calculation of predicted no- effect concentration (PNEC) in water, assuming that ADI could be delivered through drinking water and by eating fish No consideration for mixture effect | No appreciable health risks |
| Snyder [18] (U.S.A.) (Government) | Atenolol, atorvastatin, carbamazepine, diazepam, diclofenac, enalapril, fluoxetin, gemfibrozil, meprobamate, naproxen, phenytoin, risperidone, simvastatin, sulfamethoxazole, triclosan, trimethoprim | No health risks | ||
| Johnson et al. [20] (UK) (Academia) | Cytotoxic chemotherapy drugs (5-fluorouracil, cyclophosphamide, epirubicin/doxorubicin) | Ingestion of water Water ingestion rate: 1.5 L/d (adults) Body weight: 70 kg (adults) | Prediction of concentration for 5-fluorouracil and comparison with 10 ng/L trigger cytotoxic concentration value for humans; and comparison with PNEC value for aquatic fauna No consideration for mixture effect | No appreciable health risks |
| Rowney et al. [17] (UK) (Academia) | Three cytotoxic drug groups: alkylating agents (oxaliplatin, temozolomide, cisplatin, carboplatin, cyclophosphamide), antimetabolites (gemcitabine, fludarabine, fluorouracil-a metabolite to the prodrug capecitabine), and anthracycline antibiotics (epirubicin, doxorubicin). | Calculation of predicted environmental concentration using information, such as drug use, excretion rate and removal in sewage treatment plant and dissipation (assumed to be negligible) and dilution considerations in stream water Ingestion of raw stream water (using predicted drug concentrations for mean and 90th percentile flow conditions); Water ingestion rate: 2 L/d (adults) | Comparison of PEC estimates with threshold-of-toxicological- concern (TTC) and no-significant-risk-levels (NSRLs). No consideration for mixture effects | No health risks |
| Cunningham et al. [8] (U.S.A.) (Industry) | 44 active pharmaceutical ingredients marketed by GlaxoSmithKline | Ingestion of water and consumption of fish Water ingestion rate: 1.5 L/d (adults) and 1 L/d (children); Fish ingestion rate = 0.0065 kg/d Both subpopulation types with body weights = 70 kg (adults) and 14 kg (children) | Use of both measured and predicted environmental concentrations and their comparisons with PNEC levels; Use of threshold of toxicological concern No consideration for mixture effect | No appreciable health risks |
| Crider et al. [21] (U.S.A.) (Academia) | Penicillins, erythromycins, cephalosporins, sulfonamides, quinolones, tetracyclines, aminoglycosides, and nitrofurantoins | Exposure : Reported maternal use of these antibiotics (1 month before the pregnancy through the end of the first trimester); A retrospective case-control study | Calculated Odds ratio and measured association of antibiotic use and selected birth defects No consideration for mixture effect | Penicillins erythromycins, cephalosporins: No association with birth defects. Sulfonamides, nitrofurantoins: Association with several birth defects |
| Schriks et al. [22] (Netherlands) (Academia) | 50 chemicals in surface, groundwater, and drinking water | Water ingestion rate: 2 L/d (adults) Adult subpopulation with 70 kg body weight | Compared environmental concentration with threshold of toxicological concern For chemicals in surface water and groundwater, a benchmark quotient of 0.2 is used as chemicals are removed in drinking water treatment plants also. No consideration for mixture effect | No appreciable health concern |
| Cunningham et al. [9] (U.S.A.) (Industry) | Carbamazepine and its metabolites (carbamazepine diol and carbamazepine N-glucuronide) | Ingestion of water and consumption of fish Water ingestion rate: 1.5 L/d (adults) and 1 L/d (children); Fish ingestion rate = 0.0065 kg/d Both subpopulation types with body weights = 70 kg (adults) and 14 kg (children) (consideration for sensitive subpopulation) | Use of both measured and predicted environmental concentrations and their comparisons with PNEC levels; Use of threshold of toxicological concern No consideration for mixture effect | No appreciable health risks |
| Kumar and Xagoraraki [10] (U.S.A.)/(Academia) | Carbamazepine, phenytoin, meprobamate in U.S. surface water and finished drinking water | Ingestion of water and consumption of fish during recreational activity and direct consumption of finished drinking water Water ingestion rate: 0.1 L/d for surface water and 2 L/d for finished drinking water; Fish ingestion rate = 0.020 kg/d Both subpopulation types with body weights = 70 kg (adults in 18–75 years age group) and children of 1–10 years age group with body weight (kg) = 8 + 2 × Age) | Use of sub-population specific toxic endpoints; Use of calculated therapeutic- and toxicity- based acceptable daily intake values Consideration for mixture effect qualitatively (No reported interactions of meprobamate with phenytoin and carbamazepine at therapeutic concentration levels; Reported interaction of carbamazepine with phenytoin (decrease in pharmaceutical concentration due to presence of other pharmaceutical compound) | No appreciable health concern |
| Less than 90% removal | More than 90% removal | |
|---|---|---|
| Conventional treatment | ||
| Name of chemicals | 4-nonylphenol; 7-Acetyl-1,1,3,4,4,6-Hexamethyl-1,2,3,4- tetrahydronaphthalene (AHTN); bisphenol A; codeine; dehydronifedipine; Diethoxyoctylphenol (OP2EO); methylbenzyldene camphor; sulfathiazole; tri(2-butoxyethyl) phosphate; triclosan; triethylcitrate; Tris (2-chloroethyl)phosphate (TCEP) | 17β-estradiol; acetaminophen; atorvastatin; benzophenone-3; carbamazepine; carbaryl; clofibric acid; diazepam; diazinon; diclofenac; erythromycin; estrone; fluoxetine; gemfibrozil; HHCB; lincomycin; metolachlor; naproxen; progesterone; sulfamethoxazole; trimethoprim |
| Advanced treatment | ||
| Name of chemicals | 4-nonylphenol; triclosan; TCEP | Atenolol; atrazine; bezafibrate; carbamazepine; clofibric acid; cotinine; diclofenac; estrone; gemfibrozil; ibuprofen; linuron; meprobamate; metolachlor; N,N-diethyltoluamide (DEET); naproxen; phenytoin; progesterone; sulfamethoxazole |
| Pharmaceutical name | Webb et al. [12] (assumed body weight = 60 kg) | Schwab et al. [3] | Schulman et al. [11] (assumed body weight = 60 kg) | Christensen [7] |
|---|---|---|---|---|
| Acetylsalicylic acid | 8.3 μg/kg/d (30 mg/day therapeutic dose as anticoagulation therapy) | Not applicable | 16.67 μg/kg/d | Not applicable |
| Cyclophosphamide | 16.67 μg/kg/d (1 mg/d based on immunobullous skin disorders) | Not applicable | 0.017 μg/kg/d (1 μg/d based on no-significant-risk-level) | 0.01 μg/d (for rat) |
| Doxycycline | 3 μg/kg/d (100 mg/day therapeutic dose based on bacterial infection) | 30 μg/kg/d (value established from WHO representing antimicrobial sensitivity of human intestinal microflora) | Not applicable | Not applicable |
| Tetracycline | 3 μg/kg/d (1,000 mg/day therapeutic dose based on bacterial infection) | 30 μg/kg/d (value established from WHO representing antimicrobial sensitivity of human intestinal microflora) | Not applicable | Not applicable |
| Oxytetracycline | 3 μg/kg/d (1,000 mg/day therapeutic dose based on bacterial infection) | 30 μg/kg/d (value established from WHO representing antimicrobial sensitivity of human intestinal microflora) | Not applicable | Not applicable |
| 17α-ethinylestradiol | 0.167 μg/kg/d (0.010 mg/d therapeutic dose based on menopausal symptoms) | Not applicable | Not applicable | 6 μg/d (prepubescent boys) |
| Phenoxymethyl-penicillin | 16666 μg/kg/d (1,000 mg/day therapeutic dose based on bacterial infection) | Not applicable | Not applicable | 5.9 μg/d |
| Clofibrate | 8333 μg/kg/d (500 mg/d therapeutic dose based on hyperlipoproteinaemia) | Not applicable | 278 μg/kg/d | Not applicable |
| Reference | Component chemicals of mixture | Testing approach | Observed mixture effects |
|---|---|---|---|
| Silva et al. [34] | Eight chemicals of environmental relevance: 2′,3′,4′,5′-tetrachloro-4-biphenylol, 2′,5′-dichloro-4biphenylol, 4′-chloro-4-biphenylol, genistein, 2,4-dihydroxybenzophenone, benzyl-4-hydroxyparabene, bisphenol A, resorcinol monobenzoate | Recombinant yeast estrogen screen (YES) | There were substantial mixture effects even though each chemical was present at levels well below its NOEC and EC01. |
| Cleuvers [33] | Diclofenac, ibuprofen, naproxen, acetylsalicylic acid | Acute Daphnia and algal tests | Toxicity of the mixture was considerable, even at concentrations at which the single substances showed no or only very slight effects. |
| Pomati et al. [35] | A mixture of 13 different drugs at environmentally relevant concentrations: atenolol, bezafibrate, carbamazepine, ciprofloxacin, cyclophosphamide, furosemide, hydrochlorothiazide, furosemide, hydrochlorothiazide, ibuprofen, lincomycin, ofloxacin, ranitidine, salbutamol, sulfamethoxazole | in vitro cytotoxicity in Escherichia coli, human embryonic HEK293, and estrogen-responsive OVCAR3 tumor cells | (1) Drugs could interact and behave as chemosensitizers, with joint effects representing a statistically significant element of mixture toxicity. (2) Effects and interactions were concentration dependent. |
| Issue | Issue description | Research needs/Suggestions |
|---|---|---|
| Measured versus predicted pharmaceutical concentration | Very few predictive models for pharmaceutical concentrations have been validated [8,9,24,25]; It is difficult to model low-detected pharmaceuticals. | Validate models using measured concentrations ; Conduct uncertainty analysis of risk estimates to address issue of low detection. |
| Pharmaceuticals-of-concern | The list of both parent compounds and metabolites is consistently increasing [12,25,28] and it becomes difficult to conduct QPhRA for all detected compounds. | Update pharmaceuticals list and integrate prioritization approach with the QPhRA framework [36]. |
| Pharmaceuticals needing special attention | Therapeutic dose-based POD estimates might not represent effects of anti-neoplastics, antibiotics, bioaccumulative, allergens, and metabolites on different subpopulations [7,8,9,3,12]. | Consider final effects of these pharmaceuticals on different receptors during estimation of POD and conduct group-specific QPhRA for these pharmaceuticals. |
| Source water versus finished drinking water | Use of source water pharmaceutical concentration for risk estimation as a conservative approach for exposures to pharmaceutical from finished drinking water [6,8,25,12]. | Conduct water source-specific QPhRA; Use source water pharmaceutical concentration as finished drinking water pharmaceutical if data on pharmaceutical concentration in finished drinking water is missing. |
| Exposure route | Assumed dominance of oral ingestion route compared to other indirect ingestion- or inhalation-related exposure routes [2,3,12]. | Conduct pharmaceutical class-specific comprehensive QPhRA studies using all exposure routes for a given receptor. |
| Values uncertainty factors (UFs) | Uncertainty exists due to different choices of values of UFs [3,6,12]. | Use chemical-specific adjustment factors (CSAFs) [6,26]; Use default UF values only if CSAFs are not available. Conduct long-term toxicity studies or combination of experiment-simulation based studies to predict long-term toxicity using short-term toxicity data to address the issue of uncertainty related to short-term/long-term extrapolation. |
| Sensitive subpopulation | For some pharmaceuticals that are developed for just one gender or age class, the therapeutic dose for the target population may not be the appropriate point-of-departure (POD) for calculating estimates of ADI for non-targeted population (i.e., pregnant women, elderly, children) | Use subpopulation-specific POD values [3,6,8,9,11,12]; Use uncertainty factor equal to 10 only in the absence of subpopulation-related endpoints information. |
| Mixture effects | Co-occurrence of different pharmaceuticals in water may affect risk estimates. | Discuss all assumptions involved during QPhRA for mixture of pharmaceuticals [33]. Conduct more toxicity studies to develop mixture effects-related uncertainty factors. |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kumar, A.; Chang, B.; Xagoraraki, I. Human Health Risk Assessment of Pharmaceuticals in Water: Issues and Challenges Ahead. Int. J. Environ. Res. Public Health 2010, 7, 3929-3953. https://doi.org/10.3390/ijerph7113929
Kumar A, Chang B, Xagoraraki I. Human Health Risk Assessment of Pharmaceuticals in Water: Issues and Challenges Ahead. International Journal of Environmental Research and Public Health. 2010; 7(11):3929-3953. https://doi.org/10.3390/ijerph7113929
Chicago/Turabian StyleKumar, Arun, Biao Chang, and Irene Xagoraraki. 2010. "Human Health Risk Assessment of Pharmaceuticals in Water: Issues and Challenges Ahead" International Journal of Environmental Research and Public Health 7, no. 11: 3929-3953. https://doi.org/10.3390/ijerph7113929
APA StyleKumar, A., Chang, B., & Xagoraraki, I. (2010). Human Health Risk Assessment of Pharmaceuticals in Water: Issues and Challenges Ahead. International Journal of Environmental Research and Public Health, 7(11), 3929-3953. https://doi.org/10.3390/ijerph7113929




