Biodegradation of the High Explosive Hexanitrohexaazaiso-wurtzitane (CL-20)
Abstract
:1. Introduction
2. Experimental Section
2.1. Reagents and Chemicals
2.2. Analytical Methods
2.3. Direct Treatment of CL-20 with Activated Sludge
2.4. Treatment of Radiolabeled [14C]-CL-20 Hydrolysates with Activated Sludge
2.5. Biotransformation of CL-20 by Phanerochaete Chrysosporium
3. Results and Discussion
3.1. Treatment of CL-20 and its Hydrolysates with Aerobic Sludge
3.1.1. Direct Treatment of CL-20 with Activated Sludge
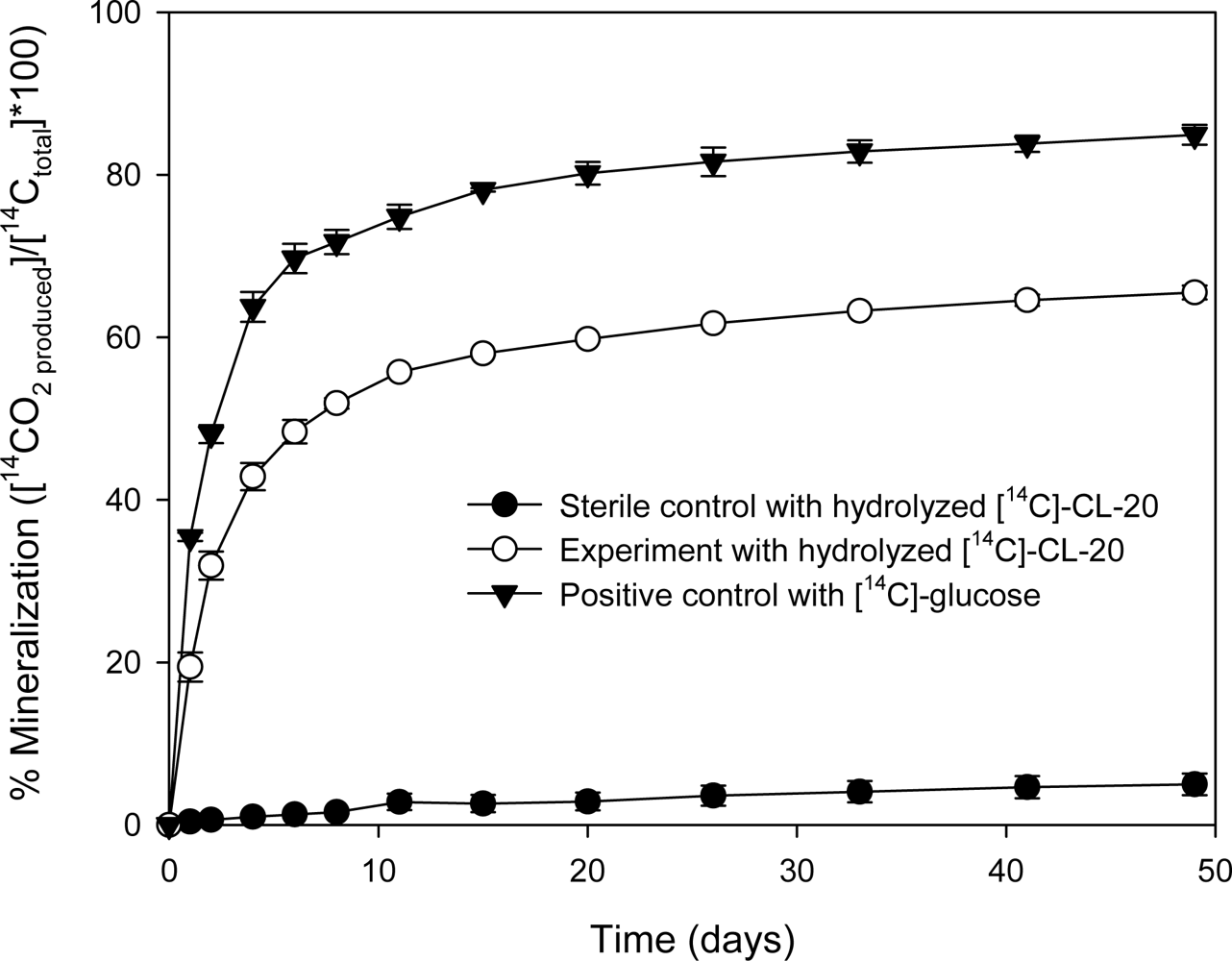
3.1.2. Treatment of Radiolabeled [14C]-CL-20 Hydrolysates with Aerobic Sludge
3.2. Biotransformation of CL-20 by Phanerochaete Chrysosporium
3.2.1. Biodegradation Experiments with Growing Cultures of Phanerochaete chrysosporium
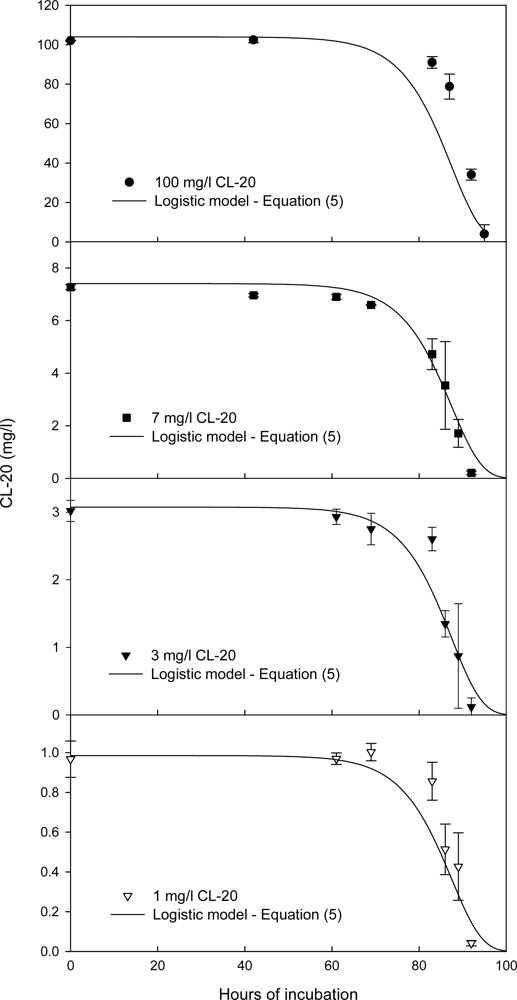
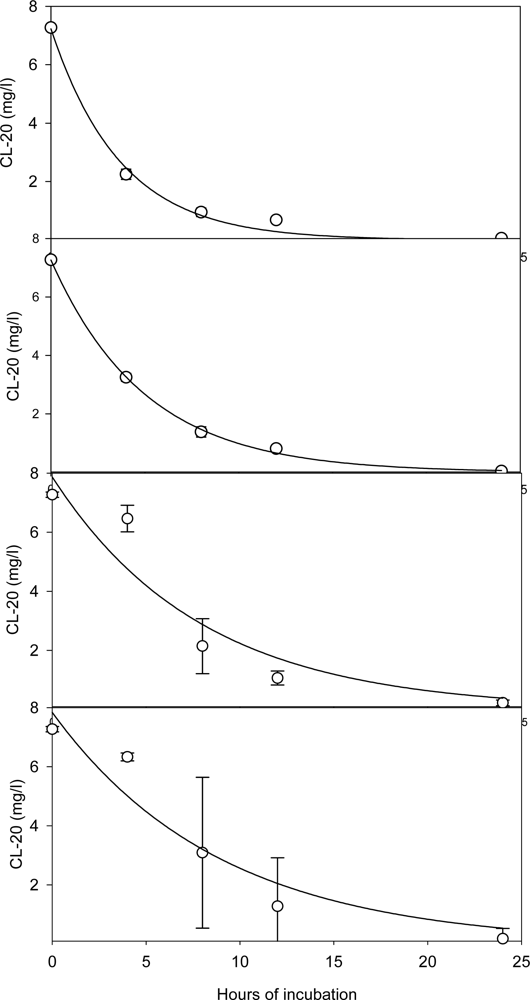
Effect of the Initial CL-20 Concentration on Biodegradation
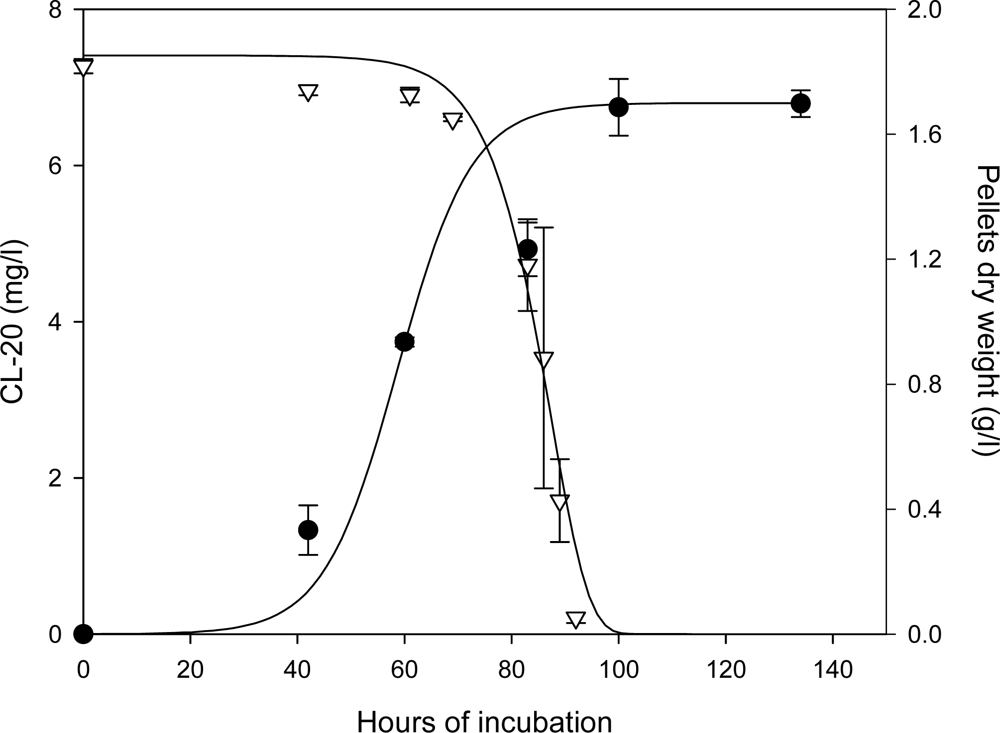
Kinetics of Fungal Growth and CL-20 Degradation
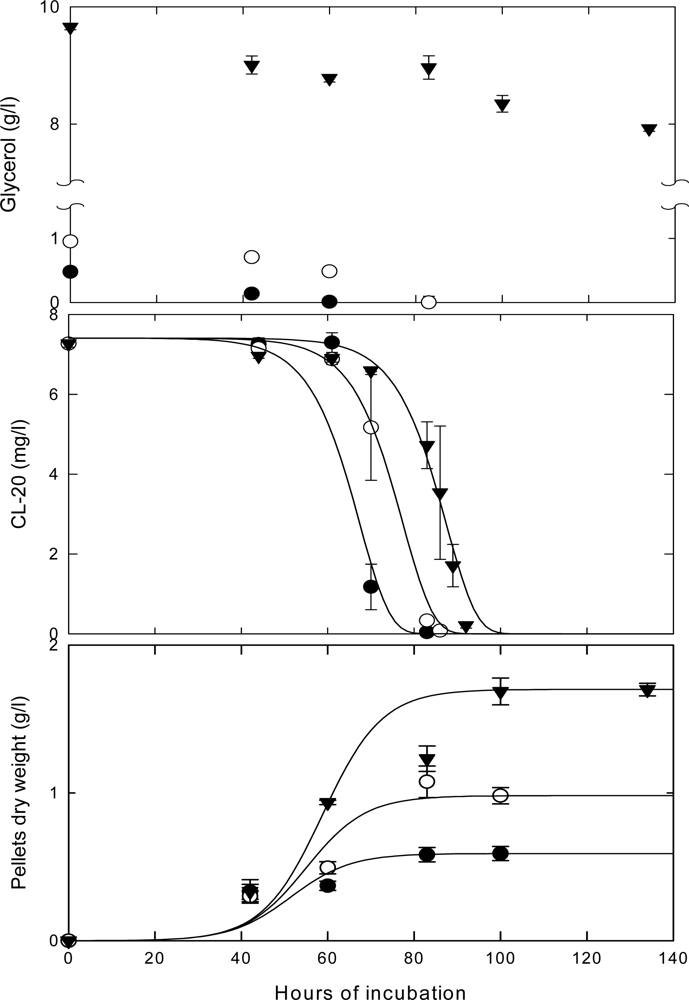
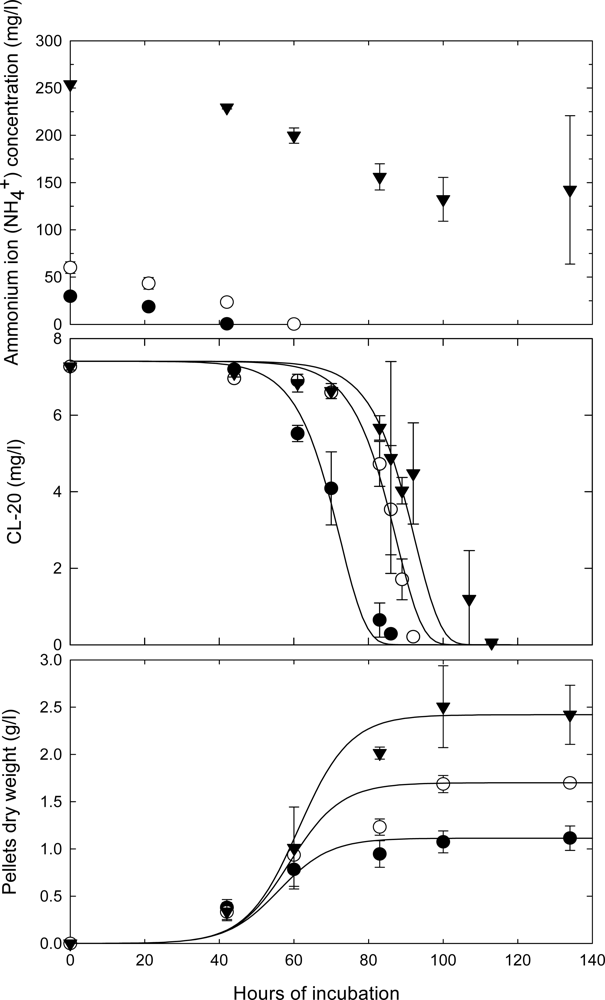
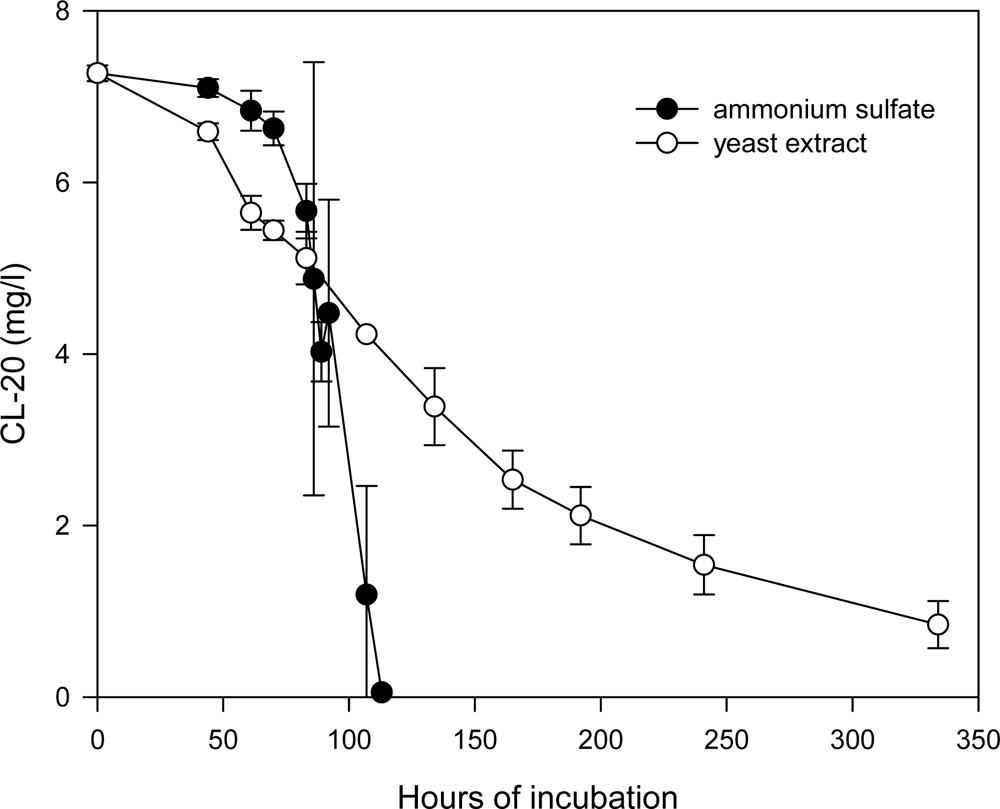
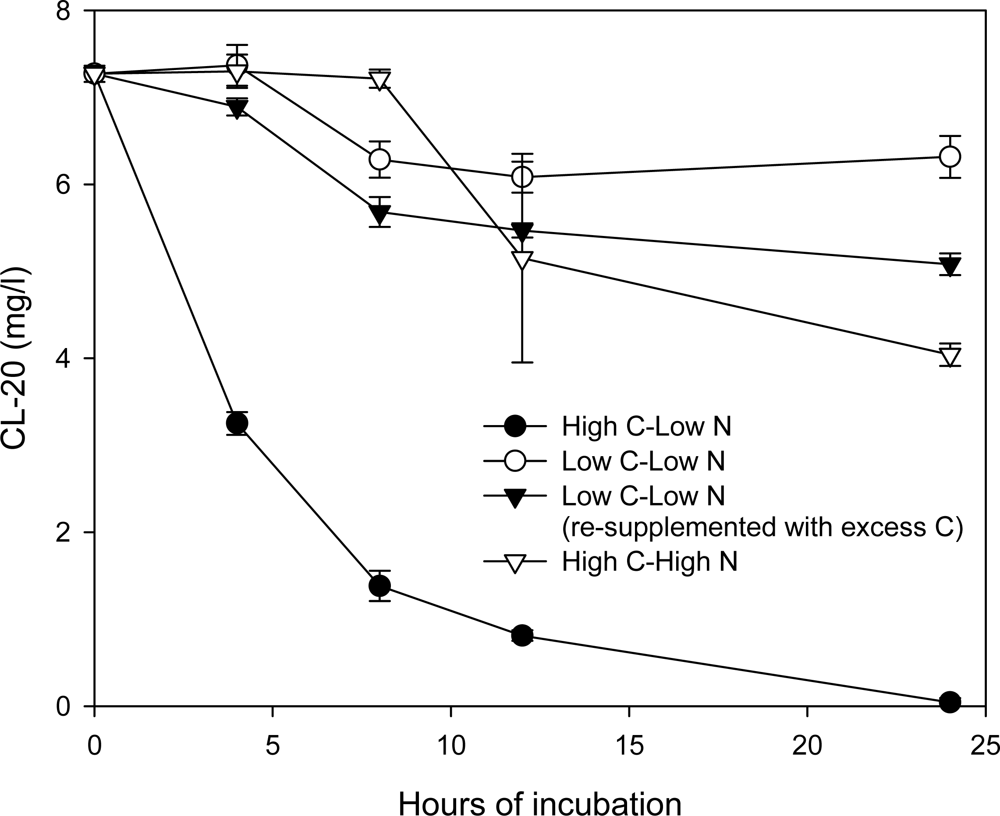
Effects of Nutrient Types and Levels on Biodegradation
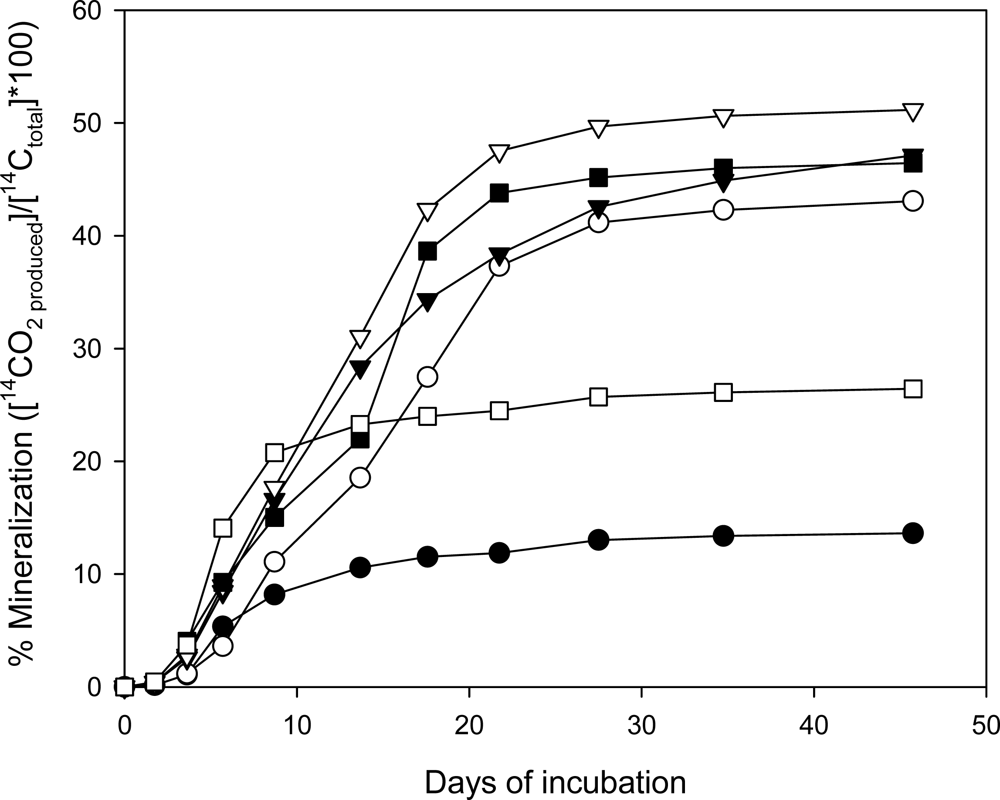
Mineralization of [14C]-CL-20 by Phanerochaete Chrysosporium
3.2.2. Biodegradation Experiments with Pre-Grown Cultures of Phanerochaete chrysosporium
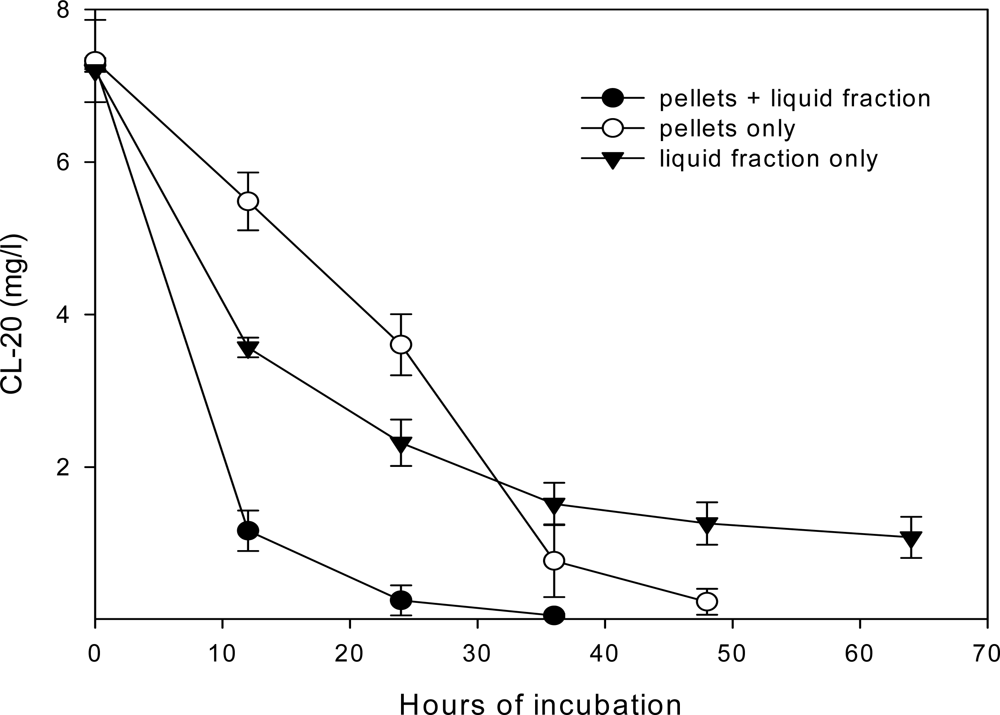
Roles of Mycelial Mass and Extracellular Fluid in Biodegradation
Effect of Nutrient Levels on Biodegradation
Effect of Culture Age on Biodegradation
4. Conclusions
Acknowledgments
References and Notes
- Nielsen, AT. Caged polynitramine compound. U.S. Patent 5693794, 1997.
- Geetha, M; Nair, UR; Sarwade, DB; Gore, GM; Asthana, SN; Singh, H. Studies on CL-20: the most powerful high energy material. J. Therm. Anal. Calorim 2003, 73, 913–922. [Google Scholar]
- Giles, J. Green explosives: collateral damage. Nature 2004, 427, 580–581. [Google Scholar]
- Talmage, SS; Opresko, DM; Maxwell, CJ; Welsh, CJE; Cretella, FM; Reno, PH; Daniel, FB. Nitroaromatic munition compounds: environmental effects and screening values. Rev. Environ. Contam. Toxicol 1999, 161, 1–156. [Google Scholar]
- Gong, P; Sunahara, GI; Rocheleau, S; Dodard, SG; Robidoux, PY; Hawari, J. Preliminary ecotoxicological characterization of a new energetic substance, CL-20. Chemosphere 2004, 56, 653–658. [Google Scholar]
- Robidoux, PY; Sunahara, GI; Savard, K; Berthelot, Y; Dodard, S; Martel, M; Gong, P; Hawari, J. Acute and chronic toxicity of the new explosive CL-20 to the earthworm (Eisenia andrei) exposed to amended natural soils. Environ. Toxicol. Chem 2004, 23, 1026–1034. [Google Scholar]
- Shen, CF; Guiot, SR; Thiboutot, S; Ampleman, G; Hawari, J. Fate of explosives and their metabolites in bioslurry treatment processes. Biodegradation 1998, 8, 339–347. [Google Scholar]
- Bell, BA; Burrows, WD; Carrazza, JA. Pilot scale testing of a semi continuous activated sludge treatment system for RDX/HMX wastewater; Contractor Report ARAED-CR-87018; U.S. Army Armament Research, Development and Engineering Center: Picatinny Arsenal, NJ, USA, 1987. [Google Scholar]
- Fournier, D; Monteil-Rivera, F; Halasz, A; Bhatt, M; Hawari, J. Degradation of CL-20 by white-rot fungi. Chemosphere 2006, 63, 175–181. [Google Scholar]
- Trott, S; Nishino, SF; Hawari, J; Spain, JC. Biodegradation of the nitramine explosive CL-20. Appl. Environ. Microbiol 2003, 69, 1871–1874. [Google Scholar]
- Bhushan, B; Paquet, L; Spain, JC; Hawari, J. Biotransformation of 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane (CL-20) by denitrifying pseudomonas sp. strain FA1. Appl. Environ. Microbiol 2003, 69, 5216–5221. [Google Scholar]
- Bhushan, B; Halasz, A; Thiboutot, S; Ampleman, G; Hawari, J. Chemotaxis-mediated biodegradation of cyclic nitramine explosives RDX, HMX, and CL-20 by Clostridium sp. EDB2. Biochem. Biophys. Res. Commun 2004, 316, 816–821. [Google Scholar]
- Bhushan, B; Halasz, A; Hawari, J. Nitroreductase catalyzed biotransformation of CL-20. Biochem. Biophys. Res. Commun 2004, 322, 271–276. [Google Scholar]
- Pal, N; Christodoulatos, C. Fungal degradation of 2,4-dinitrotoluene and nitroglycerin in batch and fixed-film bioreactors. J. Energ. Mater 1995, 13, 259–282. [Google Scholar]
- Hawari, J; Halasz, A; Beaudet, S; Paquet, L; Ampleman, G; Thiboutot, S. Biotransformation of 2,4,6-trinitrotoluene with Phanerochaete chrysosporium in agitated cultures at pH 4.5. Appl. Environ. Microbiol 1999, 65, 2977–2986. [Google Scholar]
- Sheramata, TW; Hawari, J. Mineralization of RDX by the white rot fungus Phanerochaete chrysosporium to carbon dioxide and nitrous oxide. Environ. Sci. Technol 2000, 34, 3384–3388. [Google Scholar]
- Fournier, D; Halasz, A; Thiboutot, S; Ampleman, G; Manno, D; Hawari, J. Biodegradation of octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine (HMX) by Phanerochaete chrysosporium: new insight into the degradation pathway. Environ. Sci. Technol 2004, 38, 4130–4133. [Google Scholar]
- Barr, DP; Aust, SD. Pollutant degradation by white rot fungi. Rev. Environ. Contam. Toxicol 1994, 138, 49–72. [Google Scholar]
- Karakaya, P; Sidhoum, M; Christodoulatos, C; Nicolich, S; Balas, W. Aqueous solubility and alkaline hydrolysis of the novel high explosive hexanitrohexaazaisowurtzitane (CL-20). J. Hazard. Mater 2005, 120, 183–191. [Google Scholar]
- Balakrishnan, VK; Halazs, A; Hawari, J. Alkaline hydrolysis of the cyclic nitramine explosives RDX, HMX, and CL-20: New insights into degradation pathways obtained by the observation of novel intermediates. Environ. Sci. Technol 2003, 37, 1838–1843. [Google Scholar]
- Zoh, KD; Daniels, JI; Knezovich, JP; Stenstrom, MK. Treatment of hydrolysates of the high explosives hexahydro-1,3,5-trinitro-1,3,5-triazine and octahydro-1,3,5-tetranitro-1,3,5,7-tetrazocine using biological denitrification. Water Environ. Res 1999, 71, 148–155. [Google Scholar]
- Zoh, KD; Stenstrom, MK. Application of a membrane bioreactor for treating explosives process wastewater. Water Res 2002, 36, 1018–1024. [Google Scholar]
- Jenkins, TF; Leggett, DC; Grant, CL. Reversed-phase high-performance liquid chromatographic determination of nitroorganics in munitions wastewater. Anal. Chem 1986, 58, 170–175. [Google Scholar]
- Monteil-Rivera, F; Paquet, L; Deschamps, S; Balakrishnan, VK; Beaulieu, C; Hawari, J. Physico-chemical measurements of CL-20 for environmental applications, comparison with RDX and HMX. J. Chromatogr. A 2004, 1025, 125–132. [Google Scholar]
- Pavlov, J; Christodoulatos, C; Nicolich, S; Balas, W; Koutsospyros, A; Sidhoum, M. Hydrolysis of Hexanitrohexaazaisowurtzitane (CL-20). J. Energ. Mater 2007, 25, 1–18. [Google Scholar]
- Balakrishnan, VK; Monteil-Rivera, F; Gautier, MA; Hawari, J. Sorption and stability of the polycyclic nitramine explosive CL-20 in soil. J. Environ. Qual 2004, 33, 1362–1368. [Google Scholar]
- Stahl, JD; Van Aken, B; Cameron, MD; Aust, SD. Hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) biodegradation in liquid and solid-state matrices by Phanerochaete chrysosporium. Bioremed. J 2001, 5, 13–25. [Google Scholar]
- Schmidt, SK; Simkins, S; Alexander, M. Models for the kinetics of biodegradation of organic compounds not supporting growth. Appl. Environ. Microbiol 1985, 50, 323–331. [Google Scholar]
- Alexander, M. Biodegradation and Bioremediation, 2nd Ed ed; Academic Press: London, UK, 1999. [Google Scholar]
- Fritsche, W; Scheibner, K; Herre, A; Hofrichter, M. Fungal degradation of explosives: TNT and related nitroaromatic compounds. In Biodegradation of Nitroaromatic Compounds and Explosives; Spain, JC, Hughes, JB, Knackmuss, H-J, Eds.; CRC Press: Boca Raton, FL, USA, 2000; pp. 213–237. [Google Scholar]
- Kapich, AN; Prior, BA; Botha, A; Galkin, S; Lundell, T; Hatakka, A. Effect of lignocellulose-containing substrates on production of ligninolytic peroxidases in submerged cultures of Phanerochaete chrysosporium ME-446. Enzyme Microb. Technol 2004, 34, 187–195. [Google Scholar]
- Vahabzadeh, F; Mehranian, M; Saatari, AR. Color removal ability of Phanerochaete chrysosporium in relation to lignin peroxidase and manganese peroxidase produced in molasses wastewater. World J. Microbiol Biotechnol 2004, 20, 859–864. [Google Scholar]
- Dutta, SK; Jackson, MM; Hou, LH; Powell, D; Tatem, HE. Non-ligninolytic TNT mineralization in contaminated soil by Phanerochaete chrysosporium. Bioremed. J 1998, 2, 97–103. [Google Scholar]
- Dosoretz, CG; Rothschild, N; Hadar, Y. Overproduction of lignin peroxidase by Phanerochaete chrysosporium (BKM- F-1767) under nonlimiting nutrient conditions. Appl. Environ. Microbiol 1993, 59, 1919–1926. [Google Scholar]
- Wu, F; Ozaki, H; Terashima, Y; Imada, T; Ohkouchi, Y. Activities of ligninolytic enzymes of the white rot fungus, Phanerochaete chrysosporium and its recalcitrant substance degradability. Water Sci. Technol 1996, 34, 69–78. [Google Scholar]
| CL-20 concentration (mg/L) | Dry biomass (g/L) |
|---|---|
| 0 | 1.506 (0.211)* |
| 1 | 1.704 (0.021) |
| 3 | 1.534 (0.123) |
| 7 | 1.556 (0.066) |
| 100 | 1.569 (0.147) |
| 500 | 1.574 (0.123) |
| Glycerol (g/L) | Ammonium Sulfate (g/L) | Yeast Extract (g/L) | |
|---|---|---|---|
| low C-low N | 0.5 | 0.2 | |
| low C-low N | 1 | 0.2 | |
| high C-low N | 10 | 0.2 | |
| high C-low N | 10 | 0.1 | |
| high C-high N | 10 | 1 | |
| high C-high N | 10 | 1 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Karakaya, P.; Christodoulatos, C.; Koutsospyros, A.; Balas, W.; Nicolich, S.; Sidhoum, M. Biodegradation of the High Explosive Hexanitrohexaazaiso-wurtzitane (CL-20). Int. J. Environ. Res. Public Health 2009, 6, 1371-1392. https://doi.org/10.3390/ijerph6041371
Karakaya P, Christodoulatos C, Koutsospyros A, Balas W, Nicolich S, Sidhoum M. Biodegradation of the High Explosive Hexanitrohexaazaiso-wurtzitane (CL-20). International Journal of Environmental Research and Public Health. 2009; 6(4):1371-1392. https://doi.org/10.3390/ijerph6041371
Chicago/Turabian StyleKarakaya, Pelin, Christos Christodoulatos, Agamemnon Koutsospyros, Wendy Balas, Steve Nicolich, and Mohammed Sidhoum. 2009. "Biodegradation of the High Explosive Hexanitrohexaazaiso-wurtzitane (CL-20)" International Journal of Environmental Research and Public Health 6, no. 4: 1371-1392. https://doi.org/10.3390/ijerph6041371




