Arsenic Resistance and Prevalence of Arsenic Resistance Genes in Campylobacter jejuni and Campylobacter coli Isolated from Retail Meats
Abstract
:1. Introduction
2. Experimental Section
2.1. Bacterial Isolates
2.2. Arsenic Resistance Screening
| Arsenicals | Conc. 1 (µg/mL) | Conc. 2 (µg/mL) | Conc. 3 (µg/mL) | Conc. 4 (µg/mL) | Conc. 5 (µg/mL) | Conc. 6 (µg/mL) | Conc. 7 (µg/mL) | Conc. 8 (µg/mL) | Conc. 9 (µg/mL) | Conc. 10 (µg/mL) |
|---|---|---|---|---|---|---|---|---|---|---|
| Arsanilic Acid | 4 | 8 | 16 | 32 | 64 | 128 | 256 | 512 | 1,024 | 2,048 |
| Arsenate | 16 | 32 | 64 | 128 | 256 | 512 | 1,024 | 2,048 | 4,096 | 8,192 |
| Arsenite | 4 | 8 | 16 | 32 | 64 | 128 | 256 | 512 | 1,024 | 2,048 |
| Roxarsone | 4 | 8 | 16 | 32 | 64 | 128 | 256 | 512 | 1,024 | 2,048 |
2.3. DNA Extraction
2.4. PCR for Arsenic Genes
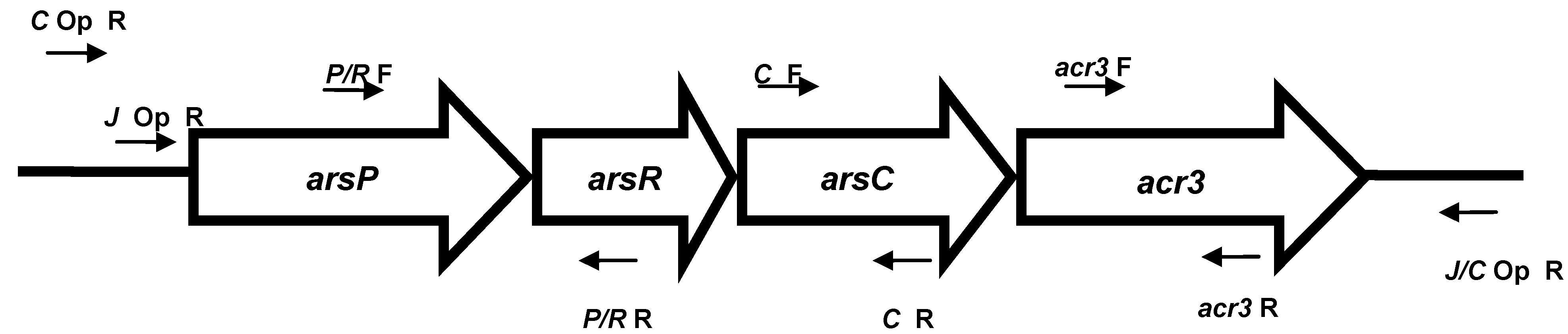
3. Results and Discussion
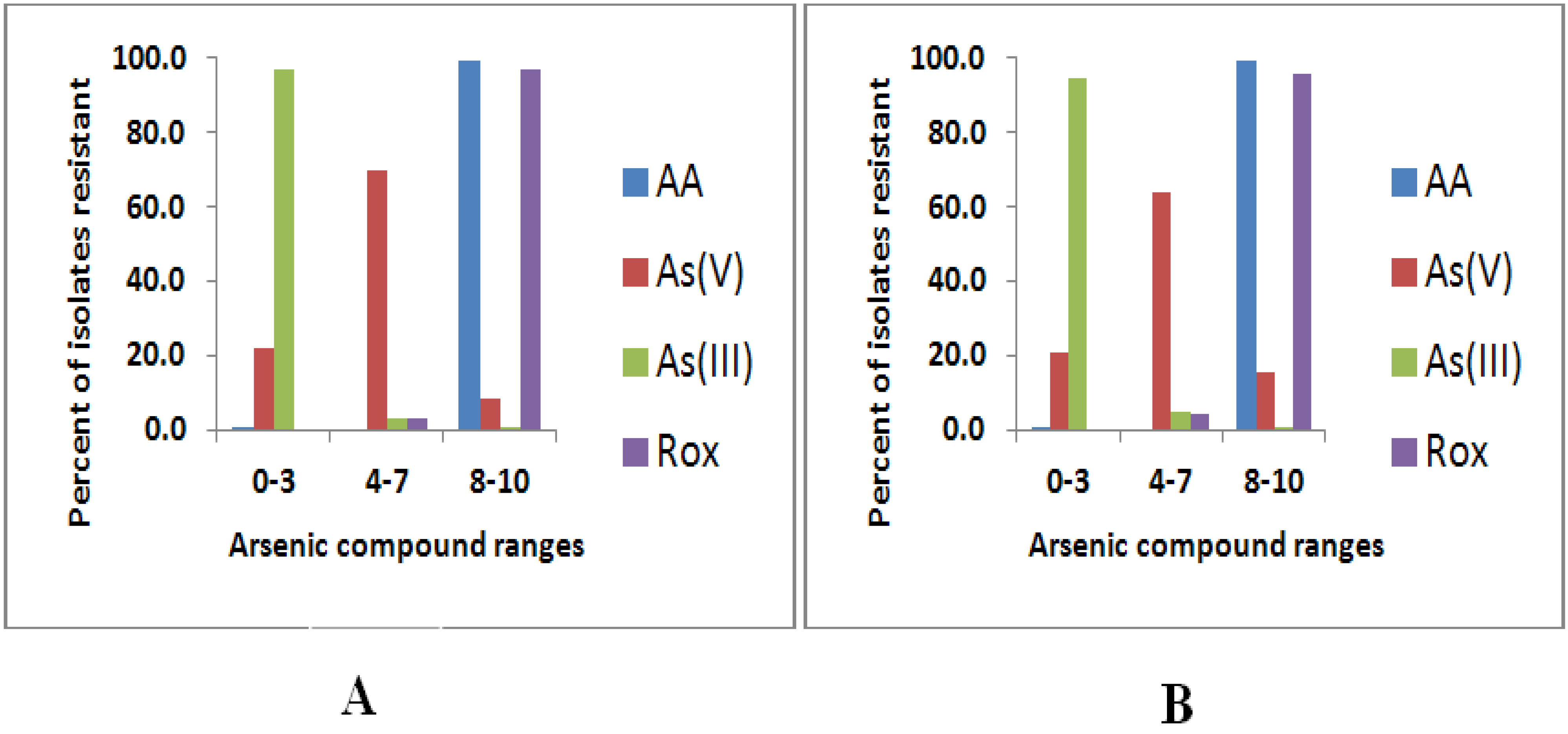
3.1. Arsenic Resistance Screening
| Campylobacter coli | Campylobacter jejuni | |||||||
|---|---|---|---|---|---|---|---|---|
| Conc. | AA (%) | As(V) (%) | As(III) (%) | Rox (%) | AA (%) | As(V) (%) | As(III) (%) | Rox (%) |
| 0–3 | 1/271 (0.4) | 56/271 (20.7) | 256/271 (94.5) | 0/271 (0.0) | 1/281 (0.4) | 61/281 (21.7) | 272/281 (96.8) | 0/281 (0.0) |
| 4–7 | 0/271 (0.0) | 173/271 (63.8) | 13/271 (4.8) | 12/271 (4.4) | 0/281 (0.0) | 196/281 (69.8) | 8/281 (2.8) | 9/281 (3.2) |
| 8–10 | 270/271 (99.6) | 42/271 (15.5) | 2/271 (0.7) | 259/271 (95.6) | 280/281 (99.6) | 24/281 (8.5) | 1/281 (0.4) | 272/281 (96.8) |
3.2. Arsenic Resistance Gene Screening
| Genes | Campylobacter jejuni | Campylobacter coli | ||
|---|---|---|---|---|
| Isolates | % | Isolates | % | |
| arsR | 108/114 | 94.7 | 109/109 | 100 |
| arsP | 108/114 | 94.7 | 109/109 | 100 |
| arsC | 17/114 | 14.9 | 105/109 | 96.3 |
| acr3 | 20/114 | 17.5 | 104/109 | 95.4 |
| arsB | 112/114 | 98 | 0/109 | 0 |

| Source | arsP *np/n (%) | arsR *np/n (%) | arsC *np/n (%) | acr3 *np/n (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C. jejuni | C. coli | Total | C. jejuni | C. coli | Total | C. jejuni | C. coli | Total | C. jejuni | C. coli | Total | |
| Beef livers | 13/13 (100) | 26/26 (100) | 39/39 (100) | 13/13 (100) | 26/26 (100) | 39/39 (100) | 0/13 (0) | 26/26 (100) | 26/39 (66.7) | 0/13 (0) | 26/26 (100) | 26/39 (66.7) |
| Chicken | 34/40 (85) | 9/9 (100) | 43/49 (87.8) | 34/40 (85) | 9/9 (100) | 43/49 (87.8) | 3/40 (7.5) | 9/9 (100) | 12/49 (24.5) | 6/40 (15) | 8/9 (88.9) | 14/49 (28.6) |
| Chicken gizzards | 10/10 (100) | 3/3 (100) | 13/13 (100) | 10/10 (100) | 3/3 (100) | 13/13 (100) | 6/10 (60) | 3/3 (100) | 9/13 (69.2) | 6/10 (60) | 3/3 (100) | 9/13 (69.2) |
| Chicken livers | 46/46 (100) | 67/67 (100) | 113/113 (100) | 46/46 (100) | 67/67 (100) | 113/113 (100) | 5/46 (10.9) | 64/67 (95.5) | 69/113 (61.1) | 5/46 (10.9) | 64/67 (95.5) | 69/113 (61.1) |
| Pork | 0/0 (0) | 2/2 (100) | 2/2 (100) | 0/0 (0) | 2/2 (100) | 2/2 (100) | 0/0 (0) | 2/2 (100) | 2/2 (100) | 0/0 (0) | 2/2 (100) | 2/2 (100) |
| Turkey | 5/5 (100) | 2/2 (100) | 7/7 (100) | 5/5 (100) | 2/2 (100) | 7/7 (100) | 3/5 (60) | 1/2 (50) | 4/7 (57.1) | 3/5 (60) | 1/2 (50) | 4/7 (57.1) |
3.3. Correlation between Genotype and Arsenic Resistance
4. Conclusions

Acknowledgments
Conflict of Interest
References
- Lasky, T.; Sun, W.; Kadry, A.; Hoffman, M.K. Mean total arsenic concentrations in chicken 1989–2000 and estimated exposures for consumers of chicken. Environ. Health Perspect. 2004, 112, 18–21. [Google Scholar]
- Wallinga, D. Playing Chicken: Avoiding Arsenic in Your Meat. The Institute of Agriculture and Trade Policy: Minneapolis, MN, USA, 2006. Available online: http://www.iatp.org/documents/playing-chicken-avoiding-arsenic-in-your-meat (accessed on 5 May 2010).
- Food and Drug Administration (FDA). 3-Nitro (Roxarsone) and Chicken. 2011. Available online: http://www.fda.gov/AnimalVeterinary/SafetyHealth/ProductSafetyInformation/ucm257540.htm (accessed on 29 August 2011).
- Food and Drug Administration (FDA). FDA: Pfizer will Voluntarily Suspend Sale of Animal Drug 3-Nitro. 2011. Available online: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm258342.htm (accessed on 8 June 2011).
- Kowalski, L.M.; Reid, W.M. Effects of roxarsone on pigmentation and coccidiosis in broilers. Poult. Sci. 2011, 54, 1544–1549. [Google Scholar] [CrossRef]
- Sapkota, A.R.; Lefferts, L.Y.; McKenzie, S.; Walker, P. What do we feed to food-production animals? A review of animal feed ingredients and their potential impacts on human health. Environ. Health Perspect. 2007, 115, 663–670. [Google Scholar] [CrossRef]
- Chapman, H.D.; Johnson, Z.B. Use of antibiotics and roxarsone in broiler chickens in the USA: Analysis for the years 1995 to 2000. Poult. Sci. 2002, 81, 356–364. [Google Scholar]
- Waldroup, P.W.; Johnson, Z.B.; Helwig, H.M.; Ramsey, B.E.; Spencer, G.K. The response of broiler chickens to 3-nitro-4hydroxyphrnylarsonic acid (roxarson) in a series of broiler feeding trials. Nutr. Rep. Int. 1984, 30, 1079–1088. [Google Scholar]
- Li, C.; Wang, X.; Wang, G.; Wu, C.; Li, N. Genome-wide expression analysis of roxarson-stimulated growth of broiler chickens (Gallus gallus). Comp. Biochem. Physiol. 2011, 6, 264–270. [Google Scholar] [CrossRef]
- Garbarino, J.R.; Bednar, A.J.; Rutherford, D.W.; Beyer, R.S.; Wershaw, R.L. Environmental fate of roxartsone in poultry litter 1. Degradation of roxarsone during composting. Environ. Sci. Technol. 2003, 37, 1509–1514. [Google Scholar] [CrossRef]
- Wang, L.; Jeon, B.; Sahin, O.; Zhang, Q. Identification of an arsenic resistance and arsenic-sensing system in Campylobacter jejuni. Appl. Environ. Microbiol. 2009, 75, 5064–5073. [Google Scholar] [CrossRef]
- Nakajima, T.; Hayashi, K.; Nagatomi, R.; Matsubara, K.; Moore, J.E.; Millar, B.C.; Matsuda, M. Molecular identification of an arsenic four-gene operon in Campylobacter lari. Folia Microbiol. 2012, 58, 253–260. [Google Scholar]
- Shen, Z.; Han, J.; Wang, Y.; Sahin, O.; Zhang, Q. The contribution of ArsB to arsenic resistance in Campylobacter jejuni. PLoS One 2013, 8. [Google Scholar] [CrossRef]
- Sapkota, A.R.; Price, L.B.; Silbergeld, E.K.; Schwab, K.J. Arsenic resistance in Campylobacter spp. isolated from retail poultry products. Appl. Environ. Microbiol. 2006, 72, 3069–3071. [Google Scholar] [CrossRef]
- Ordonez, E.; Letek, M.; Valbeuna, N.; Gil, J.A.; Mateos, L.M. Analysis of genes involved in arsenic resistance in Corynebacterium glutamicum ATCC 13032. Appl. Environ. Microbiol. 2005, 71, 6206–6215. [Google Scholar] [CrossRef]
- Ryan, D.; Colleran, E. Arsenical resistance in the IncHI2 plasmids. Plasmid 2002, 47, 234–240. [Google Scholar] [CrossRef]
- Gotz, F.; Zabielski, J.; Philipson, L.; Lindberg, M. DNA homology between the arsenate resistance plasmid pSX267 from Staphylococcus xylosus and the penicillinase plasmid pI258 from Staphylococcus aureus. Plasmid 1983, 9, 126–137. [Google Scholar] [CrossRef]
- Cai, J.; Salmon, K.; DuBow, M.S. A chromosomal ars operon homologue of Pseudomonas aeruginosa confers increased resistance to arsenic and antimony in Escherichia coli. Microbiology 1998, 144, 2705–2713. [Google Scholar] [CrossRef]
- Butcher, B.G.; Deane, S.M.; Rawlings, D.E. The chromosomal arsenic resistance genes of Thiobacillus ferrooxidans have an unusual arrangement and confer increased arsenic and antimony resistance to Escherichia coli. Appl. Environ. Microbiol. 2000, 66, 1826–1833. [Google Scholar] [CrossRef]
- Sato, T.; Kobayashi, Y. The ars operon in the skin element of Bacillus subtilis confers resistance to arsenate and arsenite. J. Bacteriol. 1998, 180, 1655–1661. [Google Scholar]
- Noormohamed, A.; Fakhr, M.K. Incidence and antimicrobial resistance profiling of Campylobacter in retail chicken livers and gizzards. Foodborne Pathog. Dis. 2012, 9, 617–624. [Google Scholar] [CrossRef]
- Noormohamed, A.; Fakhr, M.K. A higher prevalence rate of Campylobacter in retail beef livers compared to other beef and pork meat cuts. Int. J. Environ. Res. Public Health. 2013, 10, 2058–2068. [Google Scholar] [CrossRef]
- Marmur, J. A procedure for the isolation of deoxyribonucleic acid from microorganisms. J. Mol. Biol. 1961, 3, 208–218. [Google Scholar] [CrossRef]
- Thibodeau, A.; Fravalo, P.; Garneau, P.; Masson, L.; Laurent-lewandowski, S.; Quessy, S.; Harel, J.; letellier, A. Distribution of colonization and antimicrobial resistance genes in Campylobacter jejuni isolated from chicken. Foodborne Pathog. Dis. 2013, 10, 382–391. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Noormohamed, A.; Fakhr, M.K. Arsenic Resistance and Prevalence of Arsenic Resistance Genes in Campylobacter jejuni and Campylobacter coli Isolated from Retail Meats. Int. J. Environ. Res. Public Health 2013, 10, 3453-3464. https://doi.org/10.3390/ijerph10083453
Noormohamed A, Fakhr MK. Arsenic Resistance and Prevalence of Arsenic Resistance Genes in Campylobacter jejuni and Campylobacter coli Isolated from Retail Meats. International Journal of Environmental Research and Public Health. 2013; 10(8):3453-3464. https://doi.org/10.3390/ijerph10083453
Chicago/Turabian StyleNoormohamed, Aneesa, and Mohamed K. Fakhr. 2013. "Arsenic Resistance and Prevalence of Arsenic Resistance Genes in Campylobacter jejuni and Campylobacter coli Isolated from Retail Meats" International Journal of Environmental Research and Public Health 10, no. 8: 3453-3464. https://doi.org/10.3390/ijerph10083453




