Detection of β-Lactamase Residues in Milk by Sandwich ELISA
Abstract
:1. Introduction
2. Experimental Section
2.1. Chemicals and Materials
2.2. Solutions
2.3. Antibodies and Conjugated Antibodies
2.4. Sandwich ELISA
2.5. Indirect ELISA
2.6. Pairwise Interaction Analysis
2.7. Selection of Optimum mAb Pairs for β-Lactamase Detection
2.8. Assessment of Cross-Reactivity of the Developed Sandwich ELSA
2.9. Sample Preparation
2.10. Analysis of β-Lactamase-Spiked Milk by Sandwich ELISA
3. Results and Discussion
3.1. Production of Anti-β-Lactamase mAbs
3.2. Pairwise Interaction Analysis of mAbs
| D mAb | Capture mAb | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | |
| 1-HRP | 12.29 | 2.07 | 3.48 | 7.30 | 7.77 | 5.72 | 7.25 | 2.00 | 9.85 | 8.29 | 2.59 | 3.56 | 1.84 | 1.18 | |
| 2-HRP | 11.88 | 2.36 | 15.28 | 6.41 | 5.24 | 6.05 | 5.96 | 4.47 | 3.09 | 3.74 | 3.72 | 4.78 | 3.26 | 1.43 | |
| 3-HRP | 3.57 | 1.99 | 4.21 | 1.19 | 1.38 | 1.11 | 1.41 | 1.20 | 2.16 | 1.92 | 1.07 | 1.34 | 1.27 | 0.92 | |
| 4-HRP | 5.95 | 14.70 | 4.51 | 12.56 | 12.81 | 10.60 | 10.73 | 2.78 | 13.09 | 9.11 | 3.84 | 4.69 | 1.93 | 0.91 | |
| 5-HRP | 9.70 | 4.77 | 1.38 | 11.35 | 3.01 | 1.96 | 2.75 | 1.86 | 5.13 | 2.52 | 1.37 | 2.29 | 2.10 | 1.19 | |
| 6-HRP | 5.18 | 1.60 | 1.34 | 5.73 | 2.16 | 2.11 | 1.78 | 1.60 | 1.17 | 1.42 | 1.37 | 1.72 | 1.67 | 1.10 | |
| 7-HRP | 3.01 | 2.26 | 1.00 | 6.20 | 1.29 | 1.59 | 1.76 | 1.45 | 2.75 | 2.09 | 1.06 | 1.25 | 1.33 | 0.89 | |
| 8-HRP | 3.97 | 1.69 | 1.38 | 7.37 | 2.66 | 2.39 | 2.98 | 0.99 | 1.71 | 2.17 | 1.24 | 2.35 | 1.95 | 1.18 | |
| 9-HRP | 0.96 | 1.82 | 0.89 | 0.90 | 1.27 | 1.33 | 1.19 | 1.14 | 2.14 | 1.10 | 0.77 | 1.01 | 1.01 | 1.01 | |
| 10-HRP | 11.41 | 5.16 | 2.41 | 13.10 | 4.93 | 5.23 | 5.80 | 4.95 | 3.65 | 3.97 | 3.80 | 5.06 | 3.71 | 1.79 | |
| 11-HRP | 10.56 | 5.09 | 2.53 | 15.55 | 6.10 | 4.81 | 6.35 | 5.06 | 3.81 | 2.76 | 4.47 | 4.22 | 3.25 | 1.58 | |
| 12-HRP | 2.34 | 2.73 | 1.06 | 2.97 | 1.99 | 2.24 | 1.64 | 1.48 | 1.07 | 3.45 | 2.03 | 1.54 | 1.27 | 0.91 | |
| 13-HRP | 3.13 | 4.64 | 1.21 | 4.66 | 2.25 | 2.57 | 2.00 | 2.78 | 1.47 | 3.93 | 2.88 | 1.77 | 1.49 | 1.31 | |
| 14-HRP | 3.86 | 9.41 | 2.19 | 3.63 | 4.79 | 6.10 | 5.33 | 5.11 | 1.74 | 10.96 | 7.80 | 2.75 | 1.13 | 0.92 | |
| 15-HRP | 1.63 | 2.83 | 1.19 | 1.19 | 1.93 | 1.77 | 1.59 | 1.50 | 1.08 | 4.32 | 2.11 | 1.01 | 3.95 | 1.67 | |
3.3. Selection of an Optimum mAb Pair for β-Lactamase Detection
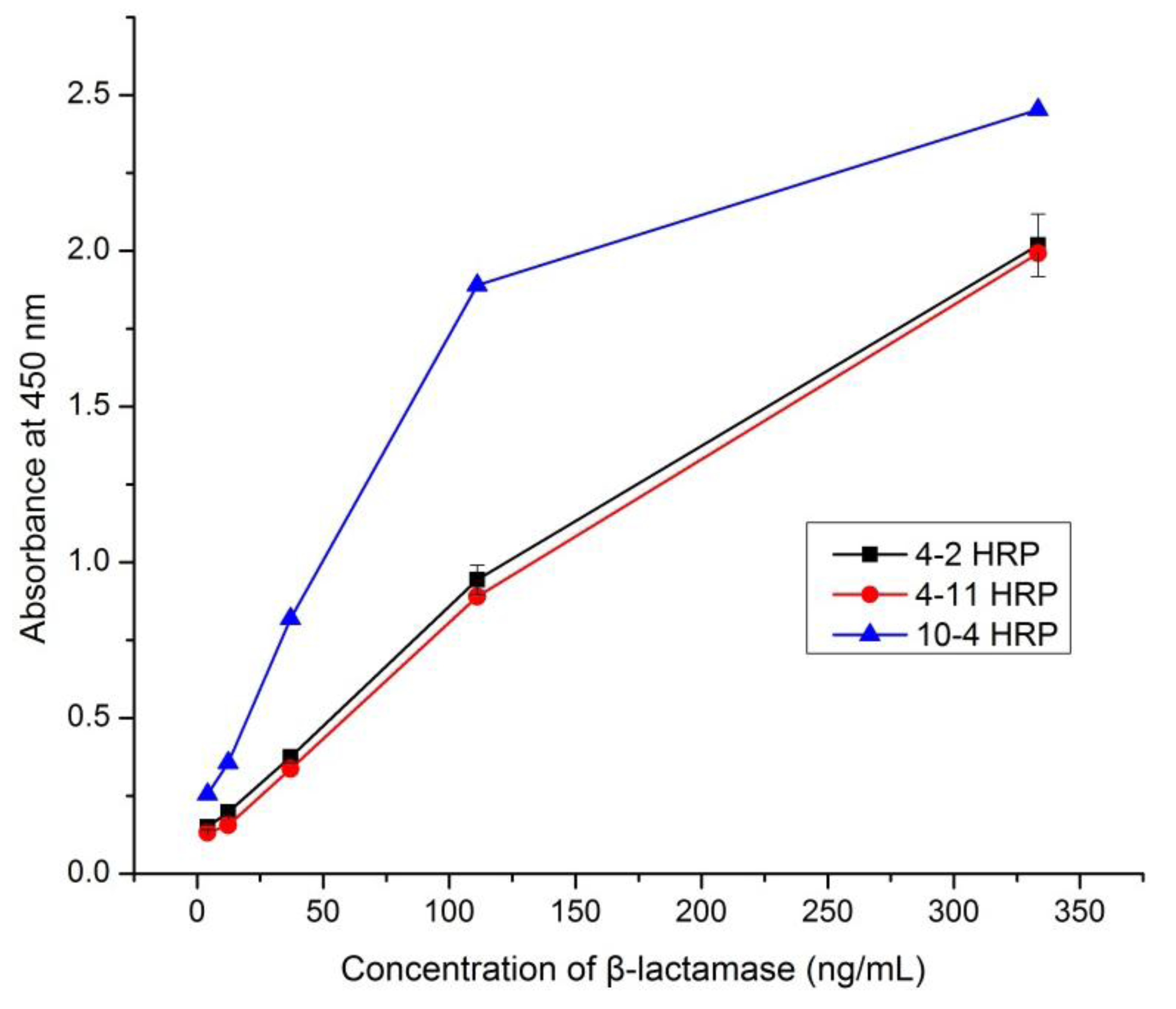
3.4. Development of mAb-Based Sandwich ELISA for the Detection of β-Lactamase

3.5. Method Specificity
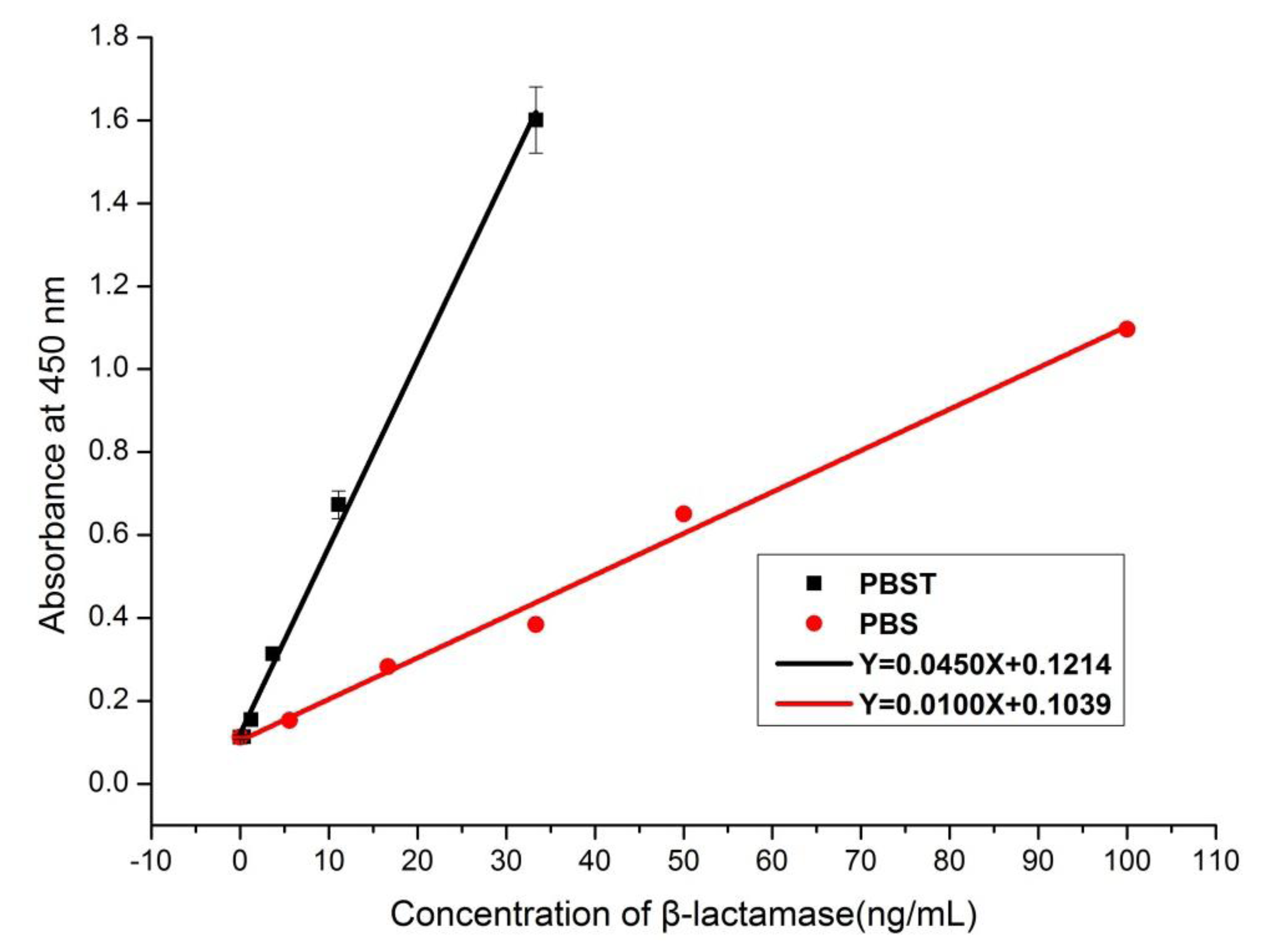
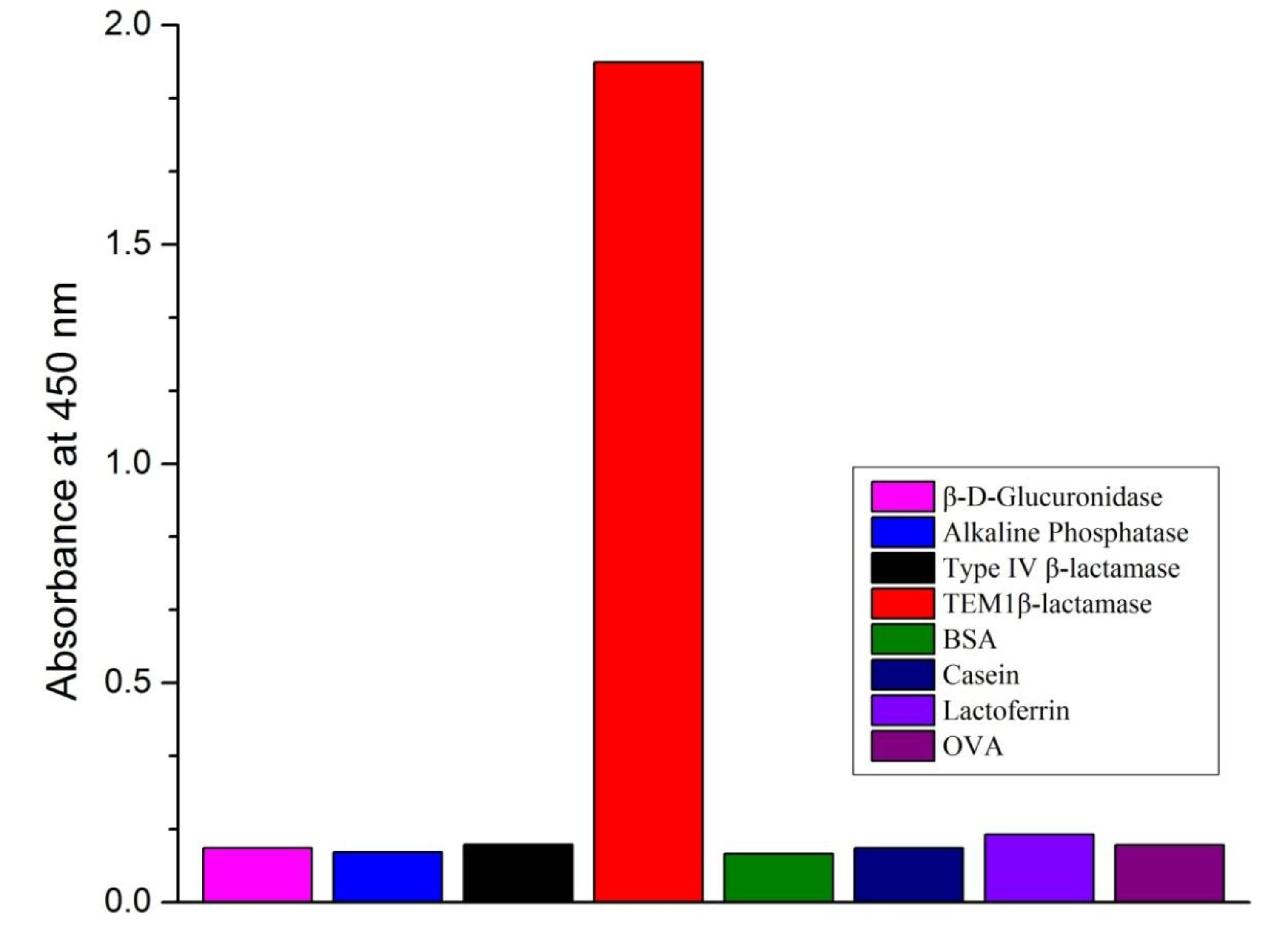
3.6. Recovery and Validation Tests
| Sparked level (ng/mL) | Intra-assay (n = 6) | Inter-assay (n = 6) | ||||
|---|---|---|---|---|---|---|
| Mean ± SD | Recoveries | CV (%) | Mean ±SD | Recoveries | CV (%) | |
| (ng/mL) | (%) | (ng/mL) | (%) | |||
| 40 | 40.086 ± 2.963 | 100.21 | 7.38 | 38.886 ± 5.035 | 97.22 | 12.96 |
| 10 | 10.313 ± 0.6422 | 103.13 | 6.21 | 9.682 ± 1.326 | 96.82 | 13.74 |
4. Conclusions
Acknowledgements
References
- Goldman, E. Antibiotic abuse in animal agriculture: Exacerbating drug resistance in human pathogens. Hum. Ecol. Risk Assess. 2004, 10, 121–134. [Google Scholar] [CrossRef]
- Khan, S.A.; Feroz, F.; Noor, R. Study of extended-spectrum β-lactamase-producing bacteria from urinary tract infections in Bangladesh. Tzu Chi Med. J. 2013, 25, 39–42. [Google Scholar] [CrossRef]
- Korycka-Dahl, M.; Richardson, T.; Bradley, R.L., Jr. Use of microbial β-lactamase to destroy penicillin added to milk. J. Dairy Sci. 1985, 68, 1910–1916. [Google Scholar] [CrossRef]
- Luvsansharav, U.O.; Hirai, I.; Nakata, A.; Imura, K.; Yamauchi, K.; Niki, M.; Komalamisra, C.; Kusolsuk, T.; Yamamoto, Y. Prevalence of and risk factors associated with faecal carriage of CTX-M beta-lactamase-producing Enterobacteriaceae in rural Thai communities. J. Antimicrob. Chemother. 2012, 67, 1769–1774. [Google Scholar] [CrossRef]
- Shea, K.M. Antibiotic resistance: What is the impact of agricultural uses of antibiotics on children’s health? Pediatrics 2003, 112, 253–258. [Google Scholar]
- Bush, K. Characterization of beta-lactamases. Antimicrob. Agents Chemother. 1989, 33, 259–263. [Google Scholar] [CrossRef]
- Cui, S.H.; Li, J.Y.; Hu, C.Q.; Jin, S.H.; Ma, Y. Development of a method for the detection of beta-lactamases in milk samples. J. Aoac Int. 2007, 90, 1128–1132. [Google Scholar]
- Doi, Y.; Paterson, D.L. Detection of plasmid-mediated class C beta-lactamases. Int. J. Infect. Dis. 2007, 11, 191–197. [Google Scholar] [CrossRef]
- O’Callaghan, C.H.; Morris, A.; Kirby, S.M.; Shingler, A.H. Novel method for detection of β-Lactamases by using a chromogenic cephalosporin substrate. Antimicrob. Agents Chemother. 1972, 1, 283–288. [Google Scholar] [CrossRef]
- Perez-Perez, F.J.; Hanson, N.D. Detection of plasmid-mediated AmpC β-lactamase genes in clinical isolates by using multiplex PCR. J. Clin. Microbiol. 2002, 40, 2153–2162. [Google Scholar] [CrossRef]
- Robberts, F.J.; Kohner, P.C.; Patel, R. Unreliable extended-spectrum β-lactamase detection in the presence of plasmid-mediated AmpC in Escherichia coli clinical isolates. J. Clin. Microbiol. 2009, 47, 358–361. [Google Scholar] [CrossRef]
- Ruppe, E.; Bidet, P.; Verdet, C.; Arlet, G.; Bingen, E. First detection of the Ambler class C 1 AmpC beta-lactamase in Citrobacter freundii by a new, simple double-disk synergy test. J. Clin. Microbiol. 2006, 44, 4204–4207. [Google Scholar] [CrossRef]
- Sanguinetti, M.; Posteraro, B.; Spanu, T.; Ciccaglione, D.; Romano, L.; Fiori, B.; Nicoletti, G.; Zanetti, S.; Fadda, G. Characterization of clinical isolates of Enterobacteriaceae from Italy by the BD phoenix extended-spectrum β-lactamase detection method. J. Clin. Microbiol. 2003, 41, 1463–1468. [Google Scholar] [CrossRef]
- Selepak, S.T.; Witebsky, F.G. beta-Lactamase detection in nine staphylococcal species. J. Clin. Microbiol. 1984, 20, 1200–1201. [Google Scholar]
- Walsh, T.R.; Bolmstrom, A.; Qwarnstrom, A.; Gales, A. Evaluation of a new etest for detecting metallo-β-lactamases in routine clinical testing. J. Clin. Microbiol. 2002, 40, 2755–2759. [Google Scholar] [CrossRef]
- Sun, H.W.; Li, H.; Zhang, J.X.; Zhou, Z. Qualitative analysis of active beta-lactamases in milk samples by rapid resolution liquid chromatography-tandem mass spectrometry. Chinese J. Anal. Chem. 2010, 38, 1203–1205. [Google Scholar]
- Xu, Z.; Wang, H.Y.; Huang, S.X.; Wei, Y.L.; Yao, S.J.; Guo, Y.L. Determination of beta-lactamase residues in milk using matrix-assisted laser desorption/ionization fourier transform mass spectrometry. Anal. Chem. 2010, 82, 2113–2118. [Google Scholar] [CrossRef]
- Deng, X.F.; Liu, L.Q.; Ma, W.W.; Xu, C.L.; Wang, L.B.; Kuang, H. Development and validation of a sandwich ELISA for quantification of peanut agglutinin (PNA) in foods. Food Agr. Immunol. 2012, 23, 265–272. [Google Scholar] [CrossRef]
- Feng, M.; Yong, Q.; Wang, W.; Kuang, H.; Wang, L.; Xu, C. Development of a monoclonal antibody-based ELISA to detect Escherichia coli O157:H7. Food Agr. Immunol. 2012. [Google Scholar] [CrossRef]
- Kuang, H.; Wang, W.; Xu, L.; Ma, W.; Liu, L.; Wang, L.; Xu, C. Monoclonal antibody-based sandwich ELISA for the detection of staphylococcal enterotoxin A. Int. J. Environ. Res. Public Health 2013, 10, 1598–1608. [Google Scholar]
- Peng, J.; Meng, X.; Deng, X.; Zhu, J.; Kuang, H.; Xu, C. Development of a monoclonal antibody-based sandwich ELISA for the detection of ovalbumin in foods. Food Agr. Immunol 2013. [Google Scholar] [CrossRef]
- Morin, C.J.; Patel, P.C.; Levesque, R.C.; Letarte, R. Monoclonal antibodies to TEM-1 plasmid-mediated beta-lactamase. Antimicrob. Agent. Chemother. 1987, 31, 1761–1767. [Google Scholar] [CrossRef]
- Hujer, A.M.; Page, M.G.P.; Helfand, M.S.; Yeiser, B.; Bonomo, R.A. Development of a sensitive and specific enzyme-linked immunosorbent assay for detecting and quantifying CMY-2 and SHV β-lactamases. J. Clin. Microbiol. 2002, 40, 1947–1957. [Google Scholar]
- Bauernfeind, A.; Stemplinger, I.; Jungwirth, R.; Ernst, S.; Casellas, J.M. Sequences of beta-lactamase genes encoding CTX-M-1 (MEN-1) and CTX-M-2 and relationship of their amino acid sequences with those of other beta-lactamases. Antimicrob. Agent. Chemother. 1996, 40, 509–513. [Google Scholar]
- Hujer, A.M.; Keslar, K.S.; Dietenberger, N.J.; Bethel, C.R.; Endimiani, A.; Bonomo, R.A. Detection of SHV beta-lactamases in Gram-negative bacilli using fluorescein-labeled antibodies. BMC Microbiol 2009, 9, 46. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, W.; Liu, L.; Xu, L.; Ma, W.; Kuang, H.; Xu, C. Detection of β-Lactamase Residues in Milk by Sandwich ELISA. Int. J. Environ. Res. Public Health 2013, 10, 2688-2698. https://doi.org/10.3390/ijerph10072688
Wang W, Liu L, Xu L, Ma W, Kuang H, Xu C. Detection of β-Lactamase Residues in Milk by Sandwich ELISA. International Journal of Environmental Research and Public Health. 2013; 10(7):2688-2698. https://doi.org/10.3390/ijerph10072688
Chicago/Turabian StyleWang, Wenbin, Liqiang Liu, Liguang Xu, Wei Ma, Hua Kuang, and Chuanlai Xu. 2013. "Detection of β-Lactamase Residues in Milk by Sandwich ELISA" International Journal of Environmental Research and Public Health 10, no. 7: 2688-2698. https://doi.org/10.3390/ijerph10072688




