Surveillance and Correlation of Antibiotic Consumption and Resistance of Acinetobacter baumannii complex in a Tertiary Care Hospital in Northeast China, 2003–2011
Abstract
:1. Introduction
2. Experimental Section
2.1. Hospital Setting and Definitions
2.2. Bacterial Isolates
2.3. Antimicrobial Susceptibility Testing
2.4. Antimicrobial Utilization
2.5. Statistical Analysis
3. Results and Discussion
3.1. Bacterial Isolates
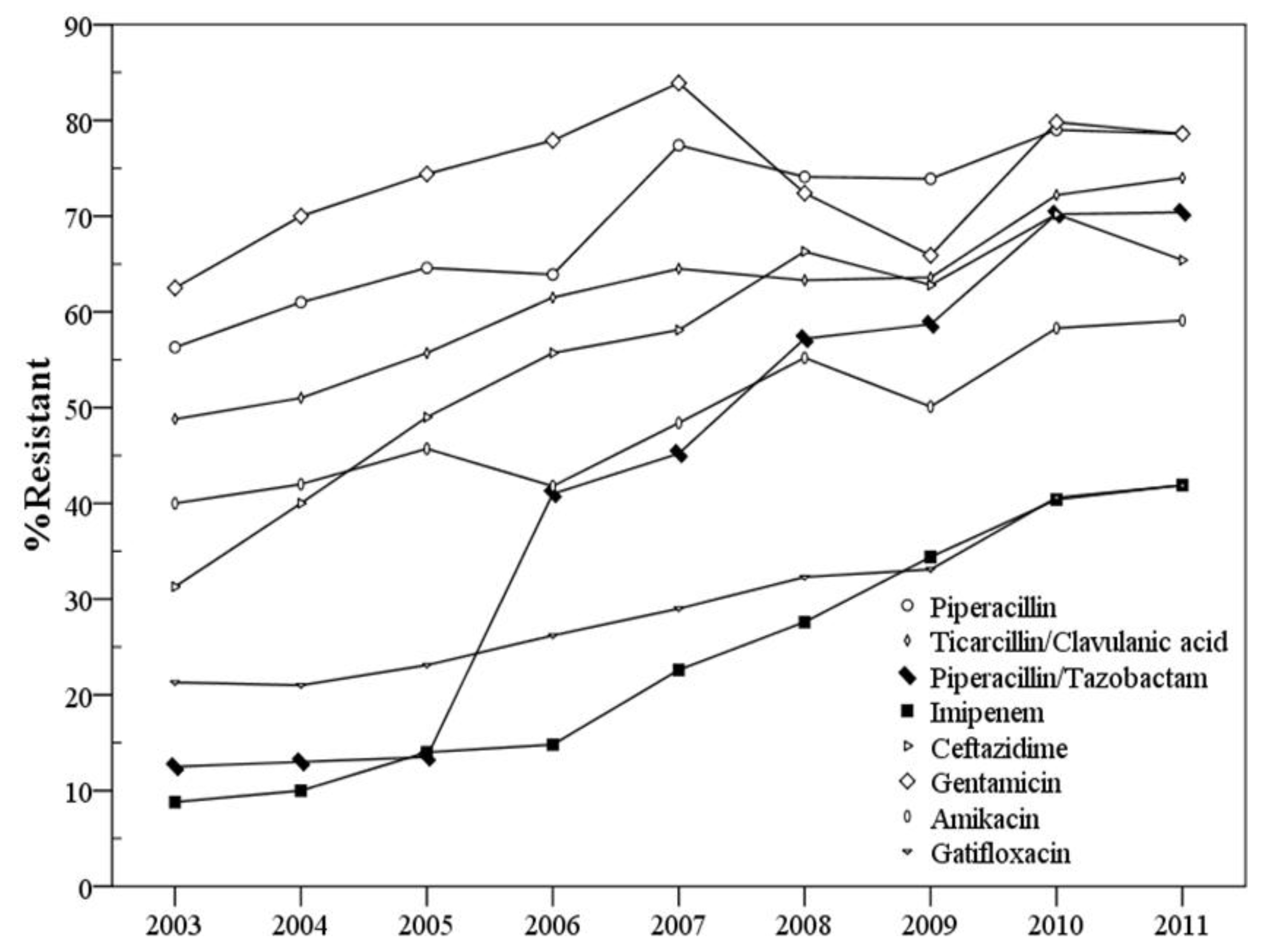
3.2. Changes in Resistance to Different Antimicrobial Agents over the Years
| Strata | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | Total | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | ||
| Patient location | |||||||||||||||||||||
| ICU | 25 | 31.3 | 32 | 32.0 | 39 | 36.8 | 47 | 38.5 | 60 | 38.7 | 115 | 38.7 | 155 | 40.1 | 179 | 40.4 | 312 | 39.2 | 964 | 38.8 | |
| Non-ICU | 55 | 68.8 | 68 | 68.0 | 67 | 63.2 | 75 | 61.5 | 95 | 61.3 | 182 | 61.3 | 232 | 59.9 | 264 | 59.6 | 483 | 60.7 | 1521 | 61.2 | |
| Specimen type | |||||||||||||||||||||
| Respiratory | 52 | 65.0 | 65 | 65.0 | 68 | 64.2 | 79 | 64.8 | 100 | 64.5 | 194 | 65.3 | 258 | 66.7 | 287 | 64.8 | 515 | 64.8 | 1618 | 65.1 | |
| Secretions and pus | 15 | 18.8 | 18 | 18.0 | 20 | 18.9 | 23 | 18.9 | 28 | 18.1 | 56 | 18.9 | 70 | 18.1 | 89 | 20.1 | 146 | 18.4 | 465 | 18.7 | |
| Urine | 5 | 6.3 | 5 | 5.0 | 6 | 5.7 | 7 | 5.7 | 9 | 5.8 | 16 | 5.4 | 19 | 4.9 | 22 | 5.0 | 48 | 6.0 | 137 | 5.5 | |
| Blood | 4 | 5.0 | 5 | 5.0 | 5 | 4.7 | 6 | 4.9 | 9 | 5.8 | 13 | 4.4 | 18 | 4.7 | 19 | 4.3 | 36 | 4.5 | 115 | 4.6 | |
| Pleural fluid and abdominal fluid | 2 | 2.5 | 4 | 4.0 | 5 | 4.7 | 5 | 4.1 | 6 | 3.9 | 14 | 4.7 | 17 | 4.4 | 18 | 4.1 | 33 | 4.2 | 104 | 4.2 | |
| Others | 2 | 2.5 | 3 | 3.0 | 2 | 1.9 | 2 | 1.6 | 3 | 1.9 | 4 | 1.3 | 5 | 1.3 | 8 | 1.8 | 17 | 2.1 | 46 | 1.9 | |
| Total | 80 | 100 | 106 | 122 | 155 | 297 | 387 | 443 | 795 | 2485 | |||||||||||
| Antimicrobial agents | Resistance rate (%) by year | 2003 to 2006 | 2006 to 2011 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2003 (n = 80) | 2004 (n = 100) | 2005 (n = 106) | 2006 (n = 122) | 2007 (n = 155) | 2008 (n = 297) | 2009 (n = 387) | 2010 (n = 443) | 2011 (n = 795) | p | OR | 95% CI | p | OR | 95% CI | |
| Piperacillin | 56.3 | 61.0 | 64.6 | 63.9 | 77.4 | 74.1 | 73.9 | 79.0 | 78.6 | 0.274 | 1.379 | 0.775–2.453 | 0.001 | 2.074 | 1.381–3.114 |
| Ticarcillin/Clavulanic acid | 48.8 | 51.0 | 55.7 | 61.5 | 64.5 | 63.3 | 63.6 | 72.2 | 74.0 | 0.075 | 1.678 | 0.949–2.967 | 0.004 | 1.780 | 1.196–2.649 |
| Piperacillin/Tazobactam | 12.5 | 13.0 | 13.5 | 41.0 | 45.2 | 57.2 | 58.7 | 70.2 | 70.4 | 0.001 | 4.861 | 2.286–10.337 | 0.001 | 3.431 | 2.319–5.077 |
| Cefoperazone/Sulbactam | 13.8 | 13.0 | 13.2 | 20.5 | 43.9 | 27.9 | 40.1 | 40.6 | 44.7 | 0.223 | 1.617 | 0.746–3.504 | 0.001 | 3.130 | 1.974–4.965 |
| Ampicillin/Sulbactam | 37.5 | 39.0 | 44.4 | 61.0 | 71.0 | 69.0 | 63.6 | 73.4 | 75.6 | 0.001 | 2.569 | 1.438–4.591 | 0.001 | 2.009 | 1.350–2.991 |
| Imipenem | 8.8 | 10.0 | 14.0 | 14.8 | 22.6 | 27.6 | 34.4 | 40.4 | 41.9 | 0.210 | 1.805 | 0.717–4.542 | 0.001 | 4.165 | 2.476–7.003 |
| Meropenem | 7.5 | 9.0 | 9.6 | 15.6 | 23.2 | 27.3 | 39.5 | 39.5 | 40.3 | 0.095 | 2.275 | 0.867–5.973 | 0.001 | 3.652 | 2.194–6.079 |
| Ceftazidime | 31.3 | 40.0 | 49.0 | 55.7 | 58.1 | 66.3 | 62.8 | 70.2 | 65.4 | 0.001 | 2.770 | 1.532–5.011 | 0.039 | 1.502 | 1.021–2.209 |
| Cefotaxime | 45.0 | 50.0 | 56.6 | 59.0 | 63.9 | 68.7 | 71.1 | 74.5 | 76.7 | 0.052 | 1.760 | 0.996–3.110 | 0.001 | 2.290 | 1.540–3.404 |
| Ceftriaxone | 48.8 | 51.0 | 59.6 | 60.7 | 65.2 | 71.7 | 70.8 | 75.0 | 76.7 | 0.096 | 1.621 | 0.917–2.864 | 0.001 | 2.139 | 1.435–3.187 |
| Cefepime | 28.8 | 30.0 | 34.0 | 52.5 | 58.1 | 68.0 | 61.2 | 70.2 | 66.0 | 0.001 | 2.735 | 1.500–4.986 | 0.004 | 1.762 | 1.200–2.588 |
| Gentamicin | 62.5 | 70.0 | 74.4 | 77.9 | 83.9 | 72.4 | 65.9 | 79.8 | 78.6 | 0.030 | 2.000 | 1.071–3.734 | 0.678 | 1.103 | 0.695–1.751 |
| Amikacin | 40.0 | 42.0 | 45.7 | 41.8 | 48.4 | 55.2 | 50.1 | 58.3 | 59.1 | 0.799 | 1.077 | 0.607–1.913 | 0.001 | 2.013 | 1.368–2.963 |
| Ciprofloxacin | 28.8 | 31.0 | 33.3 | 37.7 | 45.2 | 55.2 | 46.5 | 60.4 | 59.7 | 0.190 | 1.500 | 0.817–2.752 | 0.001 | 2.452 | 1.656–3.632 |
| Levofloxacin | 19.0 | 19.0 | 22.6 | 31.1 | 39.4 | 47.1 | 36.4 | 53.5 | 56.6 | 0.052 | 1.960 | 0.993–3.868 | 0.001 | 2.883 | 1.917–4.336 |
| Gatifloxacin | 21.3 | 21.0 | 23.1 | 26.2 | 29.0 | 32.3 | 33.1 | 40.6 | 41.9 | 0.420 | 1.318 | 0.674–2.576 | 0.001 | 2.027 | 1.322–3.108 |
| Trimethoprim/Sulfamethoxazole | 47.5 | 49.0 | 51.9 | 64.8 | 70.2 | 70.4 | 64.9 | 74.4 | 77.4 | 0.016 | 2.031 | 1.143–3.608 | 0.003 | 1.860 | 1.238–2.794 |
| Antimicrobial agents | Penicillins | β-lactam/β-lactamase inhibitors | Cephalosporins | Carbapenems | Aminoglycosides | Fluoroquinolones | ||
|---|---|---|---|---|---|---|---|---|
| Antimicrobial consumption (DDDs/1,000 patients/day) by quarter | 2003 | 1st quarter | 1.8 | 73.7 | 139.8 | 3.1 | 44.1 | 46.7 |
| 2nd quarter | 1.9 | 76.6 | 143.7 | 3.2 | 44.1 | 43.4 | ||
| 3rd quarter | 1.9 | 75.9 | 141.6 | 3.2 | 43.8 | 45.1 | ||
| 4th quarter | 1.8 | 75.1 | 139.1 | 3.4 | 43.8 | 44.1 | ||
| 2004 | 1st quarter | 1.7 | 146.2 | 123.6 | 4.9 | 44.8 | 49.9 | |
| 2nd quarter | 1.7 | 147.5 | 117.8 | 5.1 | 44.1 | 53.1 | ||
| 3rd quarter | 1.6 | 159.5 | 120.1 | 5.1 | 43.9 | 51.1 | ||
| 4th quarter | 1.6 | 169.3 | 112.3 | 5.3 | 44.8 | 52.3 | ||
| 2005 | 1st quarter | 1.6 | 217.9 | 59.3 | 6.9 | 45.3 | 63.8 | |
| 2nd quarter | 1.7 | 289.3 | 53.1 | 7.6 | 44.8 | 65.7 | ||
| 3rd quarter | 1.7 | 300.9 | 50.1 | 8.1 | 43.9 | 69.7 | ||
| 4th quarter | 1.8 | 278.9 | 48.2 | 8.2 | 44.6 | 68.6 | ||
| 2006 | 1st quarter | 1.5 | 180.5 | 92.4 | 8.5 | 43.8 | 50.3 | |
| 2nd quarter | 1.2 | 170.5 | 103.8 | 8.8 | 42.3 | 49.8 | ||
| 3rd quarter | 1.3 | 161.2 | 120.4 | 9.1 | 42.1 | 50.6 | ||
| 4th quarter | 1.5 | 160.7 | 129.5 | 9.9 | 43.2 | 49.1 | ||
| 2007 | 1st quarter | 2.1 | 120.3 | 147.8 | 10.9 | 39.7 | 41.8 | |
| 2nd quarter | 2.7 | 112.2 | 159.3 | 11.9 | 40.9 | 42.1 | ||
| 3rd quarter | 2.9 | 101.1 | 172.6 | 12.6 | 41.6 | 39.1 | ||
| 4th quarter | 2.9 | 91.2 | 165.1 | 13.1 | 40.6 | 39.5 | ||
| 2008 | 1st quarter | 3.9 | 59.8 | 151.7 | 13.9 | 35.6 | 36.3 | |
| 2nd quarter | 4.4 | 60.1 | 140.7 | 14.1 | 33.4 | 37.2 | ||
| 3rd quarter | 4.9 | 52.1 | 136.7 | 14.5 | 30.9 | 31.4 | ||
| 4th quarter | 4.8 | 54.2 | 130.9 | 15.1 | 31.3 | 31.3 | ||
| 2009 | 1st quarter | 3.1 | 80.3 | 189.9 | 23.9 | 38.3 | 43.2 | |
| 2nd quarter | 3.0 | 76.9 | 198.8 | 26.6 | 36.7 | 43.6 | ||
| 3rd quarter | 2.1 | 79.4 | 195.4 | 25.4 | 35.6 | 43.6 | ||
| 4th quarter | 2.4 | 75.1 | 190.1 | 25.0 | 38.1 | 42.9 | ||
| 2010 | 1st quarter | 0.8 | 59.3 | 146.7 | 27.5 | 69.8 | 30.3 | |
| 2nd quarter | 0.4 | 56.7 | 151.2 | 28.5 | 77.7 | 27.4 | ||
| 3rd quarter | 0.4 | 52.1 | 140.1 | 28.9 | 78.8 | 31.2 | ||
| 4th quarter | 0.3 | 53.9 | 138.5 | 28.1 | 73.6 | 27.1 | ||
| 2011 | 1st quarter | 15.2 | 69.6 | 157.3 | 29.9 | 25.5 | 40.8 | |
| 2nd quarter | 16.1 | 71.9 | 149.4 | 29.1 | 25.1 | 46.3 | ||
| 3rd quarter | 16.7 | 74.2 | 159.2 | 30.4 | 24.1 | 45.9 | ||
| 4th quarter | 15.8 | 76.1 | 145.2 | 29.9 | 23.9 | 44.2 | ||
| Time-series analysis model | β | 0.199 | −0.015 | −0.021 | 1.661 | −0.001 | −0.010 | |
| p | 0.473 | 0.546 | 0.561 | 0.001 | 0.987 | 0.936 | ||
3.3. Association of Hospital Antimicrobial Usage and the Rates of CRAB
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Peleg, A.Y.; Seifert, H.; Paterson, D.L. Acinetobacter baumannii: Emergence of a successful pathogen. Clin. Microbiol. Rev. 2008, 21, 538–582. [Google Scholar] [CrossRef]
- Bergogne-Berezin, E. The increasing significance of outbreaks of Acinetobacter spp.: The need for control and new agents. J. Hosp. Infect. 1995, 30 (Suppl.), 441–452. [Google Scholar] [CrossRef]
- Bou, G.; Cervero, G.; Dominguez, M.A.; Quereda, C.; Martinez-Beltran, J. Characterization of a nosocomial outbreak caused by a multiresistant Acinetobacter baumannii strain with a carbapenem-hydrolyzing enzyme: High-level carbapenem resistance in A. baumannii is not due solely to the presence of beta-lactamases. J. Clin. Microbiol. 2000, 38, 3299–3305. [Google Scholar]
- Quale, J.; Bratu, S.; Landman, D.; Heddurshetti, R. Molecular epidemiology and mechanisms of carbapenem resistance in Acinetobacter baumannii endemic in New York City. Clin. Infect. Dis. 2003, 37, 214–220. [Google Scholar] [CrossRef]
- Dijkshoorn, L.; Nemec, A.; Seifert, H. An increasing threat in hospitals: Multidrug-resistant Acinetobacter baumannii. Nat. Rev. Microbiol. 2007, 5, 939–951. [Google Scholar] [CrossRef]
- Unal, S.; Garcia-Rodriguez, J.A. Activity of meropenem and comparators against Pseudomonas aeruginosa and Acinetobacter spp. isolated in the MYSTIC Program, 2002–2004. Diagn. Microbiol. Infect. Dis. 2005, 53, 265–271. [Google Scholar]
- Perez, F.; Hujer, A.M.; Hujer, K.M.; Decker, B.K.; Rather, P.N.; Bonomo, R.A. Global challenge of multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2007, 51, 3471–3484. [Google Scholar] [CrossRef]
- Su, C.H.; Wang, J.T.; Hsiung, C.A.; Chien, L.J.; Chi, C.L.; Yu, H.T.; Chang, F.Y.; Chang, S.C. Increase of carbapenem-resistant Acinetobacter baumannii infection in acute care hospitals in Taiwan: Association with hospital antimicrobial usage. PLoS ONE 2012, 7, e37788. [Google Scholar] [CrossRef]
- Arda, B.; Sipahi, O.R.; Yamazhan, T.; Tasbakan, M.; Pullukcu, H.; Tunger, A.; Buke, C.; Ulusoy, S. Short-term effect of antibiotic control policy on the usage patterns and cost of antimicrobials, mortality, nosocomial infection rates and antibacterial resistance. J. Infect. 2007, 55, 41–48. [Google Scholar] [CrossRef]
- Burke, J.P. Antibiotic resistance—Squeezing the balloon? JAMA 1998, 280, 1270–1271. [Google Scholar] [CrossRef]
- Molstad, S.; Erntell, M.; Hanberger, H.; Melander, E.; Norman, C.; Skoog, G.; Lundborg, C.S.; Soderstrom, A.; Torell, E.; Cars, O. Sustained reduction of antibiotic use and low bacterial resistance: 10-year follow-up of the Swedish Strama programme. Lancet Infect. Dis. 2008, 8, 125–132. [Google Scholar]
- Vander Stichele, R.H.; Elseviers, M.M.; Ferech, M.; Blot, S.; Goossens, H. Hospital consumption of antibiotics in 15 European countries: Results of the ESAC Retrospective Data Collection (1997–2002). J. Antimicrob. Chemother. 2006, 58, 159–167. [Google Scholar] [CrossRef]
- Jawad, A.; Heritage, J.; Snelling, A.M.; Gascoyne-Binzi, D.M.; Hawkey, P.M. Influence of relative humidity and suspending menstrua on survival of Acinetobacter spp. on dry surfaces. J. Clin. Microbiol. 1996, 34, 2881–2887. [Google Scholar]
- Fournier, P.E.; Richet, H. The epidemiology and control of Acinetobacter baumannii in health care facilities. Clin. Infect. Dis. 2006, 42, 692–699. [Google Scholar] [CrossRef]
- Seifert, H.; Dijkshoorn, L.; Gerner-Smidt, P.; Pelzer, N.; Tjernberg, I.; Vaneechoutte, M. Distribution of Acinetobacter species on human skin: Comparison of phenotypic and genotypic identification methods. J. Clin. Microbiol. 1997, 35, 2819–2825. [Google Scholar]
- Manchanda, V.; Sanchaita, S.; Singh, N. Multidrug resistant acinetobacter. J. Glob. Infect. Dis. 2010, 2, 291–304. [Google Scholar] [CrossRef]
- Maragakis, L.L.; Perl, T.M. Acinetobacter baumannii: Epidemiology, antimicrobial resistance, and treatment options. Clin. Infect. Dis. 2008, 46, 1254–1263. [Google Scholar] [CrossRef]
- Peleg, A.Y.; Paterson, D.L. Multidrug-resistant Acinetobacter: A threat to the antibiotic era. Intern. Med. J. 2006, 36, 479–482. [Google Scholar] [CrossRef]
- Tsai, H.T.; Wang, J.T.; Chen, C.J.; Chang, S.C. Association between antibiotic usage and subsequent colonization or infection of extensive drug-resistant Acinetobacter baumannii: A matched case-control study in intensive care units. Diagn. Microbiol. Infect. Dis. 2008, 62, 298–305. [Google Scholar] [CrossRef]
- Goel, N.; Wattal, C.; Oberoi, J.K.; Raveendran, R.; Datta, S.; Prasad, K.J. Trend analysis of antimicrobial consumption and development of resistance in non-fermenters in a tertiary care hospital in Delhi, India. J. Antimicrob. Chemother. 2011, 66, 1625–1630. [Google Scholar] [CrossRef]
- Song, W.; Cao, J.; Mei, Y.L. Correlation between cephamycin consumption and the incidence of antimicrobial resistance in Acinetobacter baumannii at a university hospital in China from 2001 to 2009. Int. J. Clin. Pharmacol. Ther. 2011, 49, 765–771. [Google Scholar]
- Falagas, M.E.; Kopterides, P. Risk factors for the isolation of multi-drug-resistant Acinetobacter baumannii and Pseudomonas aeruginosa: A systematic review of the literature. J. Hosp. Infect. 2006, 64, 7–15. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, J.; Sun, Z.; Li, Y.; Zhou, Q. Surveillance and Correlation of Antibiotic Consumption and Resistance of Acinetobacter baumannii complex in a Tertiary Care Hospital in Northeast China, 2003–2011. Int. J. Environ. Res. Public Health 2013, 10, 1462-1473. https://doi.org/10.3390/ijerph10041462
Xu J, Sun Z, Li Y, Zhou Q. Surveillance and Correlation of Antibiotic Consumption and Resistance of Acinetobacter baumannii complex in a Tertiary Care Hospital in Northeast China, 2003–2011. International Journal of Environmental Research and Public Health. 2013; 10(4):1462-1473. https://doi.org/10.3390/ijerph10041462
Chicago/Turabian StyleXu, Jiancheng, Zhihui Sun, Yanyan Li, and Qi Zhou. 2013. "Surveillance and Correlation of Antibiotic Consumption and Resistance of Acinetobacter baumannii complex in a Tertiary Care Hospital in Northeast China, 2003–2011" International Journal of Environmental Research and Public Health 10, no. 4: 1462-1473. https://doi.org/10.3390/ijerph10041462




