Tobacco Smoke Exposure during Childhood: Effect on Cochlear Physiology
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
- control group (n = 85) having no history of hearing impairment; having no learning disabilities; having no language disorders; not exposed to passive smoking during childhood (urinary cotinine < 5.0 ng/mL);
- tobacco smoke exposure (TSE) group (n = 60): exposed to passive smoking during childhood (urinary cotinine ≥ 5.0 ng/mL).
2.2. Procedures
2.2.1. Indirect Cochlear Physiology Evaluation
2.2.2. Nicotine Metabolite Measurements—Cotinine
2.3. Data Analysis
Measurements and Statistical Analysis
3. Results and Discussion
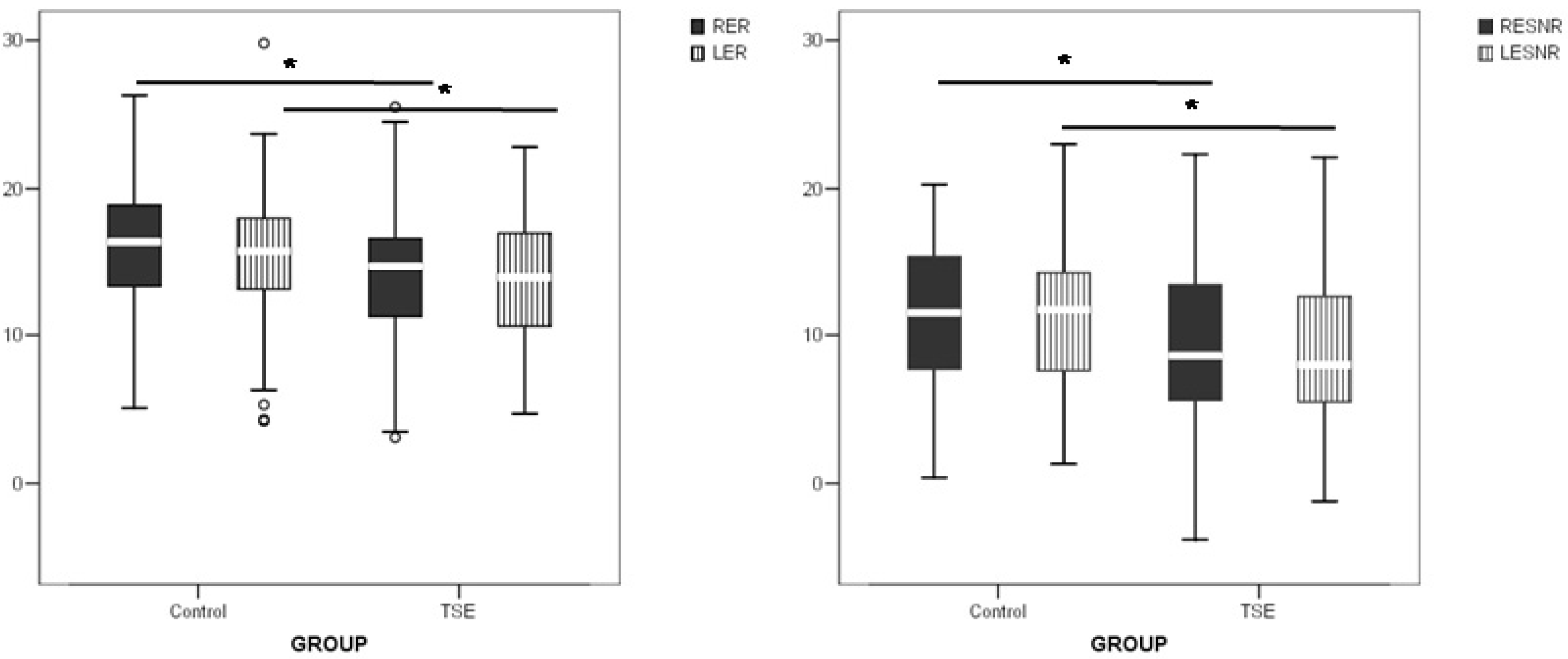

4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Levy, D.; Almeida, L.M.; Szklo, A. The Brazil simsmoke policy simulation model: The effect of strong tobacco control policies on smoking prevalence and smoking-attributable deaths in a middle income nation. PLoS Med. 2012, 9, e1001336. [Google Scholar] [CrossRef]
- Brazil. Tabagismo. Portal Saúde. Available online: http://portal.saude.gov.br/portal/saude/area.cfm?id_area=1446 (accessed on 4 October 2013).
- Hill, S.C.; Liang, L. Smoking in the home and children’s health. Tob. Control 2008, 17, 32–37. [Google Scholar] [CrossRef]
- Weitzman, M.; Govil, N.; Liu, Y.H.; Lalwani, A.K. Maternal prenatal smoking and hearing loss among adolescents. JAMA Otolaryngol. Head Neck Surg. 2013, 20, 1–8. [Google Scholar] [CrossRef]
- Ahn, J.H.; Joo, H.S.; Suh, J.K.; Kim, H.; So, H.S.; Chung, J.W. Effects of cigarette smoking on hearing recovery from noise-induced temporary hearing threshold shifts in mice. Otol. Neurotol. 2011, 32, 926–932. [Google Scholar] [CrossRef]
- Fechter, L.D.; Young, J.S.; Carlisle, L. Potentiation of noise induced threshold shifts and hair cell loss by carbon monoxide. Hear. Res. 1988, 34, 39–48. [Google Scholar] [CrossRef]
- Dhar, S.; Hall, J.W. Otoacoustic Emissions Principles, Procedures and Protocols; Plural Publishing: San Diego, FL, USA, 2012. [Google Scholar]
- Kemp, D.T.; Chum, R. Properties of the generator of stimulated acoustic emissions. Hear. Res. 1980, 2, 213–232. [Google Scholar] [CrossRef]
- Durante, A.S.; Ibidi, S.M.; Lotufo, J.P.; Carvallo, R.M.M. Maternal smoking during pregnancy: Impact on otoacoustic emissions in neonates. Int. J. Pediatr. Otorhinolaryngol. 2011, 75, 1093–1098. [Google Scholar] [CrossRef]
- Korres, S.; Riga, M.; Balatsouras, D.; Papadakis, C.; Kanellos, P.; Ferekidis, E. Influence of smoking on developing cochlea. Does smoking during pregnancy affect the amplitudes of transient evoked otoacoustic emissions in newborns? Int. J. Pediatr. Otorhinolaryngol. 2007, 71, 781–786. [Google Scholar] [CrossRef]
- Katbamna, B.; Klutz, N.; Pudrith, C.; Lavery, J.P.; Ide, C.F. Prenatal smoke exposure: Effects on infant auditory system and placental gene expression. Neurotoxicol. Teratol. 2013, 38, 61–71. [Google Scholar]
- Peck, J.D.; Neas, B.; Robledo, C.; Saffer, E.; Beebe, K.; Wild, R.A. Intrauterine tobacco exposure may alter auditory brainstem responses in newborns. Acta Obstet. Gynecol. Scand. 2010, 89, 592–596. [Google Scholar] [CrossRef]
- Kable, J.A.; Coles, C.D.; Lynch, M.E.; Carroll, J. The impact of maternal smoking on fast auditory brainstem responses. Neurotoxicol. Teratol. 2009, 31, 216–224. [Google Scholar] [CrossRef]
- Balhara, Y.P.; Jain, R. A receiver operated curve-based evaluation of change in sensitivity and specificity of cotinine urinalysis for detecting active tobacco use. J. Cancer Res. Ther. 2013, 9, 84–89. [Google Scholar] [CrossRef]
- Lando, H.A.; Hipple, B.; Muramoto, M.; Klein, J.; Prokhorov, A.; Ossip-Klein, D.; Winickoff, J.P. Tobacco is a global pediatric concern. Bull. World Health Org. 2010, 88((1)), 2. [Google Scholar] [CrossRef]
- Csákányi, Z.; Czinner, A.; Spangler, J.; Rogers, T.; Katona, G. Relationship of environmental tobacco smoke to otitis media (OM) in children. Int. J. Pediatr. Otorhinolaryngol. 2012, 76, 989–993. [Google Scholar] [CrossRef]
- Mohammadi, S.; Mazhari, M.M.; Mehrparvar, A.H.; Attarchi, M.S. Effect of simultaneous exposure to occupational noise and cigarette smoke on binaural hearing impairment. Noise Health 2010, 12, 187–190. [Google Scholar] [CrossRef]
- Vinay. Effect of smoking on transient evoked otoacoustic emissions and contralateral suppression. Auris Nasus Larynx 2010, 37, 299–302. [Google Scholar] [CrossRef]
- Paschoal, C.P.; Azevedo, M.F. Cigarette smoking as a risk factor for auditory problems. Braz. J. Otorhinolaryngol. 2009, 75, 893–902. [Google Scholar] [CrossRef]
- Jerger, J. Smoking and otoacoustic emissions. J. Am. Acad. Audiol. 2007, 18, 641. [Google Scholar] [CrossRef]
- Negley, C.; Katbamna, B.; Crumpton, T.; Lawson, G.D. Effects of cigarette smoking on distortion product otoacoustic emissions. J. Am. Acad. Audiol. 2007, 18, 665–674. [Google Scholar] [CrossRef]
- Torre, P.; Dreisbach, L.E.; Kopke, R.; Jackson, R.; Balough, B. Risk factors for distortion product otoacoustic emissions in young men with normal hearing. J. Am. Acad. Audiol. 2007, 18, 749–759. [Google Scholar] [CrossRef]
- Fabry, D.A.; Davila, E.P.; Arheart, K.L.; Serdar, B.; Dietz, N.A.; Bandiera, F.C.; Lee, D.J. Secondhand smoke exposure and the risk of hearing loss. Tob. Control 2011, 20, 82–85. [Google Scholar] [CrossRef]
- Cruickshanks, K.; Klein, R.; Klein, B.; Wiley, T.; Nondahl, D.; Tweed, T. Cigarette smoking and hearing loss: The epidemiology of hearing loss study. JAMA 1998, 279, 1715–1719. [Google Scholar] [CrossRef]
- Rahu, K.; Rahu, M.; Pullmann, H.; Allik, J. Effect of birth weight, maternal education and prenatal smoking on offspring intelligence at school age. Early Hum. Dev. 2010, 86, 493–497. [Google Scholar] [CrossRef]
- McCartney, J.S.; Fried, P.A.; Watkinson, B. Central auditory processing in school-age children prenatally exposed to cigarette smoke. Neurotoxicol. Teratol. 1994, 16, 269–276. [Google Scholar] [CrossRef]
- Sun, W.; Hansen, A.; Zhang, L.; Lu, J.; Stolzberg, D.; Kraus, K.S. Neonatal nicotine exposure impairs development of auditory temporal processing. Hear. Res. 2008, 245, 58–64. [Google Scholar] [CrossRef]
- Motlagh, M.G.; Sukhodolsky, D.G.; Landeros-Weisenberger, A.; Katsovich, L.; Thompson, N.; Scahill, L.; King, R.A.; Peterson, B.S.; Schultz, R.T.; Leckman, J.F. Adverse effects of heavy prenatal maternal smoking on attentional control in children with ADHD. J. Atten. Disord. 2011, 15, 593–603. [Google Scholar] [CrossRef]
- Key, A.P.; Ferguson, M.; Molfese, D.L.; Peach, K.; Lehman, C.; Molfese, V.J. Smoking during pregnancy affects speech-processing ability in newborn infants. Environ. Health Perspect 2007, 115, 623–629. [Google Scholar]
- Veiga, P.V.; Wilder, R.P. Maternal smoking during pregnancy and birthweight: A propensity score matching approach. Matern Child. Health J. 2008, 12, 194–203. [Google Scholar] [CrossRef]
- Nigg, J.T.; Breslau, N. Prenatal smoking exposure, low birth weight, and disruptive behavior disorders. J. Am. Acad. Child. Adolesc. Psychiatr. 2007, 46, 362–369. [Google Scholar] [CrossRef]
- Braun, J.; Kahn, R.; Froehlich, T.; Auinger, P.; Lanphear, B. Exposures to environmental toxicants and attention deficit hyperactivity disorder in U.S. children. Environ. Health Perspect 2006, 114, 1904–1909. [Google Scholar]
- Herrmann, M.; King, K.; Weitzman, M. Prenatal tobacco smoke and postnatal secondhand smoke exposure and child neurodevelopment. Curr. Opin. Pediatr. 2008, 20, 184–190. [Google Scholar] [CrossRef]
- Warren, C.W.; Jones, N.R.; Peruga, A.; Chauvin, J.; Baptiste, J.; Costa de Silva, V.; el Awa, F.; Tsouros, A.; Rahman, K.; Fishburn, B.; et al. Global youth tobacco surveillance, 2000–2007. MMWR Surveill. Summ. 2008, 57, 1–21. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Durante, A.S.; Pucci, B.; Gudayol, N.; Massa, B.; Gameiro, M.; Lopes, C. Tobacco Smoke Exposure during Childhood: Effect on Cochlear Physiology. Int. J. Environ. Res. Public Health 2013, 10, 5257-5265. https://doi.org/10.3390/ijerph10115257
Durante AS, Pucci B, Gudayol N, Massa B, Gameiro M, Lopes C. Tobacco Smoke Exposure during Childhood: Effect on Cochlear Physiology. International Journal of Environmental Research and Public Health. 2013; 10(11):5257-5265. https://doi.org/10.3390/ijerph10115257
Chicago/Turabian StyleDurante, Alessandra S., Beatriz Pucci, Nicolly Gudayol, Beatriz Massa, Marcella Gameiro, and Cristiane Lopes. 2013. "Tobacco Smoke Exposure during Childhood: Effect on Cochlear Physiology" International Journal of Environmental Research and Public Health 10, no. 11: 5257-5265. https://doi.org/10.3390/ijerph10115257



