European Surveillance for West Nile Virus in Mosquito Populations
Abstract
:1. Introduction
2. Survey by Country
2.1. Italy
| Mosquito species | 2008 | 2009 | 2010 | 2011 | 2012 | Total |
|---|---|---|---|---|---|---|
| Aedes albopictus | 46 | 317 | 1,053 | 1,449 | 1,179 | 4,044 |
| Aedes sp. | 18 | 2 | 1 | 32 | 53 | |
| Aedes vexans | 2 | 3 | 16 | 16 | 40 | 77 |
| Anopheles algerensis | 1 | 0 | 1 | |||
| Anopheles claviger | 1 | 3 | 16 | 16 | 13 | 49 |
| Anopheles hyrcanus | 6 | 6 | ||||
| Anopheles maculipennis s.l. | 228 | 658 | 755 | 2,675 | 661 | 4,977 |
| Anopheles plumbeus | 63 | 8 | 54 | 213 | 2 | 340 |
| Anopheles sp. | 12 | 15 | 16 | 9 | 10 | 62 |
| Anopheles superpictus | 3 | 4 | 7 | |||
| Coquillettidia richiardii | 4 | 5 | 10 | 60 | 16 | 95 |
| Culex brumpti | 1 | 3 | 0 | 4 | ||
| Culex hortensis | 1 | 1 | 31 | 38 | 5 | 76 |
| Culex impudicus | 7 | 1 | 0 | 8 | ||
| Culex laticinctus | 6 | 0 | 6 | |||
| Culex mimeticus | 1 | 1 | 1 | 0 | 3 | |
| Culex modestus | 2 | 12 | 2 | 22 | 0 | 38 |
| Culex pipiens s.l. | 5,004 | 12,487 | 6,933 | 12,635 | 15,448 | 52,507 |
| Culex sp. | 45 | 135 | 233 | 1,426 | 1,048 | 2,887 |
| Culex territans | 1 | 6 | 3 | 10 | ||
| Culex theileri | 14 | 422 | 142 | 15 | 593 | |
| Culex univittatus | 68 | 126 | 2 | 0 | 196 | |
| Culiseta annulata | 78 | 25 | 158 | 250 | 132 | 643 |
| Culiseta litorea | 18 | 0 | 18 | |||
| Culiseta longiareolata | 55 | 56 | 278 | 392 | 112 | 893 |
| Culiseta sp. | 4 | 5 | 1 | 0 | 10 | |
| Culiseta subochrea | 1 | 0 | 1 | |||
| Ochlerotatus atropalpus | 1 | 0 | 1 | |||
| Ochlerotatus caspius | 1,561 | 1,775 | 1,586 | 1,673 | 1,603 | 8,198 |
| Ochlerotatus communis | 214 | 1 | 48 | 109 | 45 | 417 |
| Ochlerotatus detritus | 430 | 10 | 177 | 42 | 659 | |
| Ochlerotatus dorsalis | 1 | 3 | 0 | 4 | ||
| Ochlerotatus echinus | 2 | 0 | 2 | |||
| Ochlerotatus geniculatus | 5 | 1 | 6 | 12 | ||
| Ochlerotatus rusticus | 72 | 1 | 1 | 1 | 0 | 75 |
| Ochlerotatus sp. | 280 | 198 | 10 | 644 | 233 | 1365 |
| Ochlerotatus zammiti | 187 | 0 | 187 | |||
| Uranotaenia unguiculata | 7 | 6 | 9 | 5 | 7 | 34 |
| Total | 8,343 | 15,806 | 11,960 | 21,787 | 20,662 | 78,558 |
| Mosquito species | Number of positive/tested pools | |||||
|---|---|---|---|---|---|---|
| 2008 | 2009 | 2010 | 2011 | 2012 | Total | |
| Aedes albopictus | 0/96 | 0/183 | 0/230 | 0/689 | 0/511 | 0/1,709 |
| Aedes koreicus | 0/3 | 0/3 | ||||
| Aedes sp. | 0/19 | 0/35 | 0/1 | 0/5 | 0/11 | 0/71 |
| Aedes vexans | 0/73 | 0/122 | 0/297 | 0/301 | 0/204 | 0/997 |
| Anopheles claviger | 0/6 | 0/16 | 0/10 | 0/32 | ||
| Anopheles hyrcanus | 0/2 | 0/2 | ||||
| Anopheles maculipennis s.l. | 0/52 | 0/113 | 0/147 | 0/466 | 0/239 | 0/1,017 |
| Anopheles plumbeus | 0/2 | 0/16 | 0/32 | 0/83 | 0/23 | 0/156 |
| Anopheles sp. | 0/4 | 0/3 | 0/4 | 0/4 | 0/15 | |
| Anopheles superpictus | 0/1 | 0/1 | ||||
| Coquillettidia richiardii | 0/0 | 0/1 | 0/4 | 0/59 | 0/15 | 0/79 |
| Culex brumpti | 0/2 | 0/2 | ||||
| Culex hortensis | 0/1 | 0/2 | 0/10 | 0/3 | 0/16 | |
| Culex impudicus | 0/2 | 0/1 | 0/3 | |||
| Culex mimeticus | 0/1 | 0/1 | ||||
| Culex modestus | 0/13 | 0/95 | 0/55 | 1/57 | 0/64 | 1/284 |
| Culex pipiens | 5/510 | 27/1,898 | 13/5,539 | 8/4,568 | 13/3,357 | 66/15,872 |
| Culex sp. | 0/27 | 0/3 | 0/14 | 0/132 | 0/147 | 0/323 |
| Culex territans | 0/4 | 0/6 | 0/10 | |||
| Culex theileri | 0/17 | 0/30 | 0/11 | 0/58 | ||
| Culex univittatus | 0/46 | 0/2 | 0/48 | |||
| Culiseta annulata | 0/10 | 0/18 | 0/48 | 0/134 | 0/62 | 0/272 |
| Culiseta longiareolata | 0/1 | 0/16 | 0/107 | 0/48 | 0/172 | |
| Ochlerotatus geniculatus | 0/16 | 0/7 | 0/9 | 0/8 | 0/2 | 0/36 |
| Ochlerotatus punctor | 0/1 | 0/1 | ||||
| Ochlerotatus rusticus | 0/1 | 0/1 | 0/2 | |||
| Ochlerotatus sp. | 0/2 | 0/1 | 0/37 | 0/45 | 0/85 | |
| Uranotaenia unguiculata | 0/2 | 0/2 | ||||
| Total | 9/1,222 | 27/3,050 | 13/7,219 | 9/7,768 | 13/5,609 | 71/24,852 |
| Mosquito species | Number of positive/tested pools | ||||
|---|---|---|---|---|---|
| 2009 | 2010 | 2011 | 2012 | Total | |
| Aedes albopictus | 2/175 | 2/144 | 6/675 | 52/12 | 12/1,506 |
| Aedes sp. | 0/35 | 0/0 | 0/5 | 0/11 | 0/51 |
| Aedes koreicus | 0/3 | 0/3 | |||
| Aedes vexans | 122 | 0/288 | 0/301 | 0/204 | 0/915 |
| Anopheles claviger | 0/0 | 0/0 | 0/15 | 0/10 | 0/25 |
| Anopheles hyrcanus | 0/0 | 0/0 | 0/0 | 0/2 | 0/2 |
| Anopheles maculipennis s.l. | 0/99 | 0/55 | 1/442 | 238 | 1/834 |
| Anopheles plumbeus | 0/16 | 0/6 | 0/78 | 0/21 | 0/121 |
| Anopheles sp. | 0/0 | 0/1 | 0/4 | 0/4 | 0/9 |
| Anopheles superpictus | 0/0 | 0/0 | 0/0 | 0/1 | 0/1 |
| Coquillettidia richiardii | 0/1 | 0/4 | 0/56 | 0/15 | 0/76 |
| Culex brumpti | 0/0 | 0/0 | 0/2 | 0/0 | 0/2 |
| Culex hortensis | 0/0 | 0/0 | 0/8 | 0/3 | 0/11 |
| Culex modestus | 0/95 | 0/53 | 0/52 | 0/64 | 0/264 |
| Culex pipiens | 63/1,836 | 112/5,138 | 105/4,442 | 112/3,356 | 392/14,772 |
| Culex sp. | 0/3 | 0/1 | 0/125 | 1/146 | 1/275 |
| Culex territans | 0/0 | 0/0 | 0/4 | 0/6 | 0/10 |
| Culex theileri | 0/0 | 0/4 | 0/23 | 0/11 | 0/38 |
| Culex univittatus | 0/1 | 0/0 | 0/1 | 0/0 | 0/2 |
| Culiseta annulata | 0/18 | 0/2 | 1/101 | 0/62 | 1/183 |
| Culiseta longiareolata | 0/0 | 0/3 | 0/95 | 0/48 | 0/146 |
| Culiseta sp. | 0/0 | 0/1 | 0/1 | 0/0 | 0/2 |
| Culiseta subochrea | 0/0 | 0/0 | 0/1 | 0/0 | 0/1 |
| Ochlerotatus annulipes | 0/9 | 0/1 | 0/10 | ||
| Ochlerotatus berlandi | 0/1 | 0/1 | |||
| Ochlerotatus caspius | 0/519 | 1/594 | 1/953 | 3/804 | 5/2,870 |
| Ochlerotatus cinereus | 0/8 | 0/6 | 0/8 | 0/0 | 0/22 |
| Ochlerotatus communis | 0/0 | 0/0 | 0/16 | 0/12 | 0/28 |
| Ochlerotatus detritus | 0/9 | 0/2 | 1/15 | 0/21 | 1/47 |
| Ochlerotatus dorsalis | 0/1 | 0/0 | 0/0 | 0/0 | 0/1 |
| Ochlerotatus geniculatus | 0/7 | 0/5 | 0/8 | 0/2 | 0/22 |
| Ocherotatus punctor | 0/0 | 0/1 | 0/0 | 0/0 | 0/1 |
| Ochlerotatus rusticus | 0/0 | 0/0 | 0/1 | 0/0 | 0/1 |
| Ochlerotatus sp. | 0/4 | 0/0 | 0/34 | 0/45 | 0/83 |
| Uranotaenia unguiculata | 0/0 | 0/0 | 0/0 | 0/2 | 0/2 |
| Total | 65/2,949 | 115/6,308 | 115/7,475 | 118/5,605 | 413/22,337 |

2.2. Greece
2.3. Spain
| Mosquito species | Pools | Mosquitoes | WNV + pools | USUV + pools |
|---|---|---|---|---|
| Aedes albopictus | 28 | 62 | - | - |
| Aedes vexans | 42 | 433 | - | - |
| Anopheles algeriensis | 59 | 241 | - | - |
| Anopheles atroparvus | 644 | 6,520 | - | - |
| Anopheles claviger | 2 | 2 | - | - |
| Anopheles hyrcanus | 1 | 1 | - | - |
| Anopheles maculipennis | 1 | 236 | - | - |
| Anopheles plumbeus | 5 | 12 | - | - |
| Anopheles sp. | 9 | 89 | - | - |
| Coquillettidia richiardii | 62 | 147 | - | - |
| Culex modestus | 1,181 | 21,426 | - | - |
| Culex perexiguus | 527 | 7,366 | 7 | 1 |
| Culex pipiens | 3,763 | 55,469 | 1 | 1 |
| Culex sp. | 69 | 551 | - | - |
| Culex theileri | 1,413 | 37,512 | - | - |
| Culiseta annulata | 114 | 212 | - | - |
| Culiseta longiareolata | 340 | 851 | - | - |
| Culiseta subochrea | 17 | 691 | - | - |
| Culiseta sp. | 4 | 4 | - | - |
| Ochlerotatus berlandi | 2 | 2 | - | - |
| Ochlerotatus caspius | 3,621 | 83,651 | - | - |
| Ochlerotatus detritus | 486 | 2,998 | - | - |
| Ochlerotatus geniculatus | 4 | 13 | - | - |
| Ochlerotatus pulcritarsis | 4 | 5 | - | - |
| Ochlerotatus sp. | 2 | 3 | - | - |
| Uranotaenia unguiculata | 7 | 8 | - | - |
| Species not reported | 437 | 2 | - | - |
| Total | 12,844 | 218,507 | 8 | 2 |
2.4. Switzerland
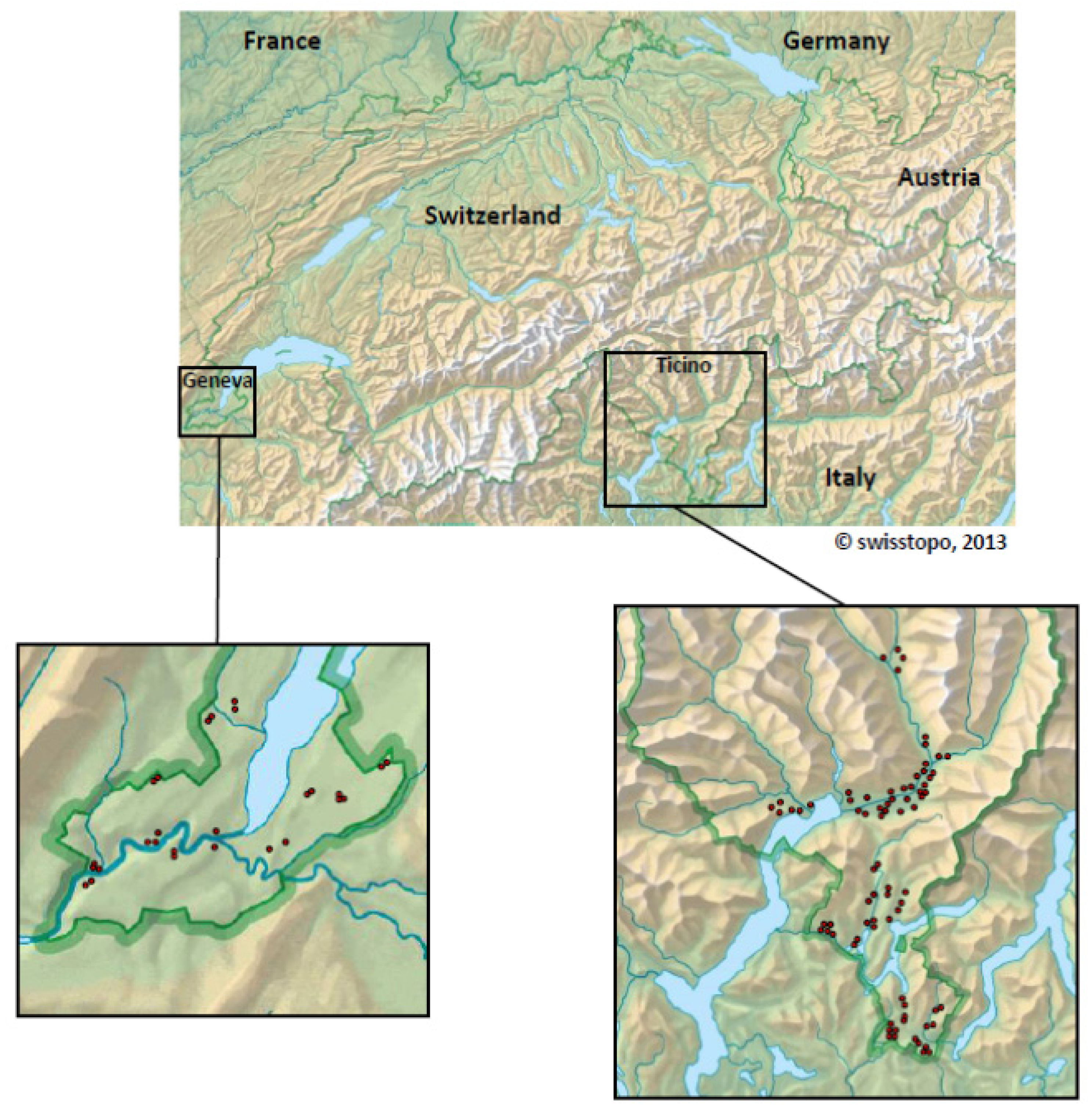
| Mosquito species | Mosquitoes | WNV+ pools | Usutu+ pools | Mosquito-Flaviviruses | |
|---|---|---|---|---|---|
| Ticino | Geneva | ||||
| Aedes albopictus | 3,938 | - | 0 | 0 | 0 |
| Aedes cinereus/geminus | 1,791 | 1 | -- | -- | -- |
| Aedes vexans | 4,597 | - | 0 | 0 | 33 |
| Aedes sp. | 1 | 1 | -- | -- | -- |
| Anopheles claviger | 2 | 31 | -- | -- | -- |
| Anopheles plumbeus | 17 | 2 | -- | -- | -- |
| Anopheles maculipennis | 349 | 28 | -- | -- | -- |
| Coquillettidia buxtoni | 2 | - | -- | -- | -- |
| Coquillettidia richiardii | 53 | 46 | -- | -- | -- |
| Culex hortensis | 9 | - | -- | -- | -- |
| Culex modestus | - | - | -- | -- | |
| Culex pipiens/torrentium | 12,780 | 2,129 | 0 | 41 | 3 |
| Culex sp. | 2,061 | 12 | 0 | 0 | 0 |
| Culiseta annulata | 40 | 14 | -- | -- | -- |
| Culiseta sp. | 1 | 6 | -- | -- | -- |
| Ochlerotatus cantans | 299 | 5 | -- | -- | -- |
| Ochlerotatus caspius | 5 | 1 | -- | -- | -- |
| Ochlerotatus geniculatus | 9 | 1 | -- | -- | -- |
| Ochlerotatus sticticus | 5,654 | 1 | -- | -- | -- |
| Total | 31,608 | 2,278 | 0 | 41 | 36 |
2.5. United Kingdom
2.6. Germany
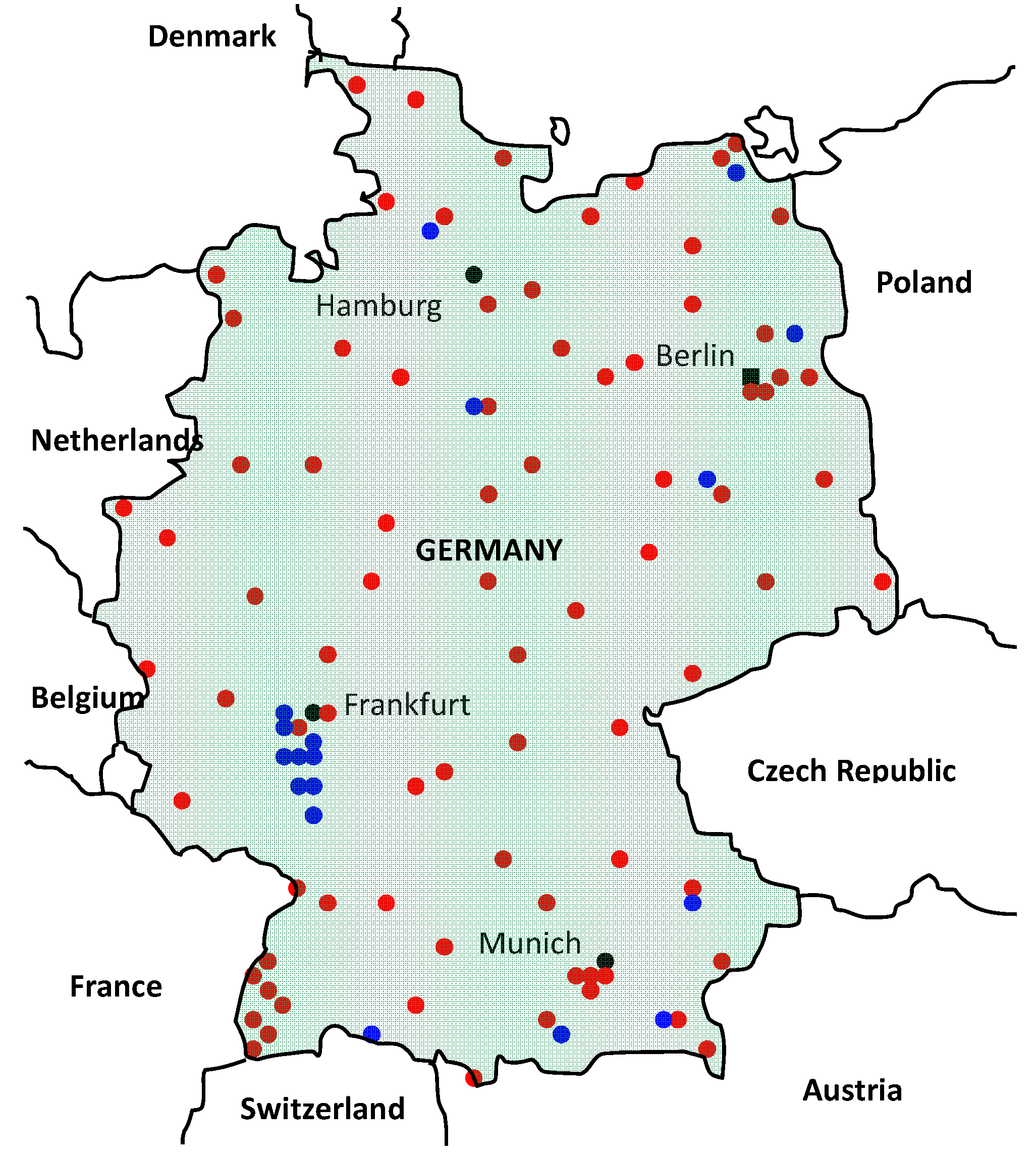
3. Discussion
4. Conclusions
Acknowledgements
Conflicts of Interest
References
- Jupp, P.G. The ecology of West Nile virus in South Africa and the occurence of outbreaks in humans. Ann. N. Y. Acad. Sci. 2001, 951, 143–152. [Google Scholar] [CrossRef]
- Reisen, W.K.; Hayes, C.G.; Azra, K.; Niaz, S.; Mahmood, F.; Parveen, T.; Boreham, P.F. West nile virus in pakistan. II. Entomological studies at Changa Manga national forest, Punjab province. Trans. Roy. Soc. Trop. Med. Hyg. 1982, 76, 437–448. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Lindsay, M.D.; Coelen, R.J.; Broom, A.K.; Hall, R.A.; Smith, D.W. Arboviruses causing human disease in the Australasian zoogeographic region. Arch. Virol. 1994, 136, 447–467. [Google Scholar] [CrossRef]
- Ciccozzi, M.; Peletto, S.; Cella, E.; Giovanetti, M.; Lai, A.; Gabanelli, E.; Acutis, P.L.; Modesto, P.; Rezzam, G.; Platonov, A.E.; et al. Epidemiological history and phylogeography of West Nile virus lineage 2. Infect. Gen. Evol. 2013, 17, 46–50. [Google Scholar]
- Bakonyi, T.; Ivanics, E.; Erdélyi, K.; Ursu, K.; Ferenczi, E.; Weissenböck, H.; Nowotny, N. Lineage 1 and 2 strains of encephalitis West Nile virus, central Europe. Emerg. Infect. Dis. 2006, 12, 618–623. [Google Scholar] [CrossRef]
- Pripilov, A.G.; Kinney, R.M.; Sanokhvalov, E.I.; Savage, H.M.; Al’kjovskii, S.V.; Tsuchiya, K.R.; Gramashevshii, V.L.; Sadykova, G.K.; Shatalov, A.G.; Vyshemirskii, O.I.; et al. Analysis of new variants of West Nile fever. Vopr. Virusol. 2002, 47, 36–41. [Google Scholar]
- Bondre, V.P.; Jadi, R.S.; Mishra, A.C.; Yergollear, P.N.; Anranhalle, V.A. West Nile virus isolates from India: Evidence for a distinct genetic lineage. J. Gen. Virol. 2007, 88, 875–884. [Google Scholar] [CrossRef]
- Vázquez, A.; Sanchez-Seco, M.P.; Ruiz, S.; Molero, F.; Hernandez, L.; Moreno, J.; Magallanes, A.; Tejedor, C.G.; Tenorio, A. Putative new lineage of West Nile virus, Spain. Emerg. Infect. Dis. 2010, 16, 549–552. [Google Scholar] [CrossRef]
- Malkinson, M.; Banet, C. The role of birds in the ecology of West Nile virus in Europe and Africa. Curr. Top. Microbiol. Immunol. 2002, 267, 309–322. [Google Scholar] [CrossRef]
- Kilpatrick, A.M. Globalization, land use, and the invasion of West Nile virus. Science 2011, 334, 323–327. [Google Scholar] [CrossRef]
- McLean, R.G.; Ubico, S.R.; Bourne, D.; Komar, N. West Nile virus in livestock and wildlife. Curr. Top. Microbiol. Immunol. 2002, 267, 271–308. [Google Scholar]
- Reiter, P. West Nile virus in Europe: Understanding the present to gauge the future. Euro Surveill. 2010, 15, 19508. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19538 (accessed on 25 September 2013). [Google Scholar]
- Platonov, A.E. West Nile encephalitis in Russia 1999–2001: Were we ready? Are we ready? Ann. N. Y. Acad. Sci. 2001, 951, 102–116. [Google Scholar] [CrossRef]
- Solomon, T.; Ooi, M.M.; Beasley, D.W.C.; Mallewa, M. West Nile encephalitis. Br. Med. J. 2003, 326, 865–869. [Google Scholar] [CrossRef]
- Gyure, K.A. West Nile virus infections. J. Neuropath. Exp. Neurol. 2009, 68, 1053–1060. [Google Scholar] [CrossRef]
- Castillo-Olivares, J.; Wood, J.N. West Nile infection of horses. Vet. Res. 2004, 35, 467–483. [Google Scholar] [CrossRef]
- Bunning, M.L.; Bowen, R.A.; Cropp, C.B.; Sullivan, K.G.; Davis, B.S.; Komar, N.; Godsey, M.S.; Baker, D.; Hettler, D.L.; Holmes, D.A.; et al. Experimental infection of horses with West Nile virus. Emerg. Infect. Dis. 2002, 8, 380–386. [Google Scholar] [CrossRef]
- Dauphin, G.; Zientara, S. West Nile virus: Recent trends in diagnosis and vaccine development. Vaccine 2007, 25, 5573–5576. [Google Scholar]
- Colpitts, T.M.; Conway, M.J.; Montgomery, R.R.; Fikrig, E. West Nile virus: Biology, transmission and human infection. Clin. Microbiol. Rev. 2012, 25, 635–648. [Google Scholar] [CrossRef]
- Taylor, R.M.; Hurlbut, H.S.; Dressler, H.R.; Spangler, E.W.; Thrasher, D. Isolation of West Nile virus from Culex mosquitoes. J. Egypt. Med. Assoc. 1953, 36, 199–208. [Google Scholar]
- Sardelis, M.R.; Turrell, M.J.; Dohm, D.J.; O’Guinn, M.L. Vector competence of selected North American Culex and Coquillettidia mosquitoes for West Nile virus. Emerg. Infect. Dis. 2001, 7, 1018–1022. [Google Scholar] [CrossRef]
- Balenghien, T.; Vazeille, M.; Grandadam, M.; Schaffner, F.; Zeller, Reiter, P.; Sabatier, P.; Fouque, F.; Bicout, D.J. Vector competence of some French Culex and Aedes mosquitoes for West Nile virus. Vector Borne Zoonotic Dis. 2007, 8, 589–595. [Google Scholar]
- Muñoz, J.; Ruiz, S.; Soriguer, R.; Alcaide, M.; Viana, D.S.; Roiz, D.; Vazquez, A.; Figuerola, J. Feeding patterns of potential West Nile virus vectors in south-west Spain. PLoS One 2012, 7, e39549. [Google Scholar] [CrossRef] [Green Version]
- Hall, R.A.; Broom, A.K.; Smith, D.W.; Mackenzie, J.S. The ecology and epidemiology of Kunjin virus. Curr Top. Microbiol. Immunol. 2003, 267, 253–269. [Google Scholar]
- Jupp, P.G. Laboratory studies on the transmission of West Nile virus by Culex (Culex univittatus) Theobald; Factors influencing transmission rate. J. Med. Entomol. 1974, 11, 455–458. [Google Scholar]
- Zeller, H.G.; Schuffenecker, I. West Nile virus: An overview of its spread in Europe and the Mediterranean Basin in contrast to its spread in the Americas. Eur. J. Clin. Microbiol. Infect. Dis. 2004, 23, 147–156. [Google Scholar] [CrossRef]
- Anderson, J.R.; Rico-Hesse, R. Aedes aegypti vectorial capacity is determined by the infecting genotype of dengue virus. Am. J. Trop. Med. Hyg. 2006, 75, 886–892. [Google Scholar]
- Hubálek, Z.; Halouzka, J. West Nile fever—A reemerging mosquito-borne viral disease in Europe. Emerg. Infect. Dis. 1999, 5, 643–650. [Google Scholar] [CrossRef]
- Lvov, D.; Lvov, D.K.; Kovtunov, A.I.; Butenko, A.M.; Zhukov, A.N. West Nile Fever in Southern Russia—Epidemiological, Clinical, Genetic Peculiarities (1999–2001). In Proceedings of the 12th International Congress of Virology, Paris, France, 27 July–1 August 2002; p. 46.
- Schuffenecker, I.; Peyfritte, C.N.; El Harrak, M.; Murri, S.; Leblond, A.; Zeller, H.G. West Nile virus in Morocco, 2003. Emerg. Infect. Dis. 2005, 11, 306–309. [Google Scholar] [CrossRef]
- Esteves, A.; Almeida, A.P.; Galão, R.P.; Parreira, R.; Rodrigues, J.C.; Sousa, C.A.; Novo, M.T. West Nile virus in Southern Portugal, 2004. Vector Borne Zoonotic Dis. 2005, 5, 410–413. [Google Scholar] [CrossRef]
- Savini, G.; Monaco, F.; Calistri, P.; Lelli, R. Phylogenetic analysis of West Nile virus isolated in Italy in 2008. Eurosurveillance 2008, 13. [Google Scholar] [CrossRef]
- Papa, A.; Bakonyi, T.; Xanthopoulou, K.; Vázquez, A.; Tenorio, A.; Nowotny, N. Genetic characterization of West Nile virus lineage 2, Greece 2010. Emerg. Infect. Dis. 2011, 17, 920–922. [Google Scholar] [CrossRef]
- Calistri, P.; Giovannini, A.; Hubalek, Z.; Ionescu, A.; Monaco, F.; Savini, G.; Lelli, R. Epidemiology of West Nile in Europe and the Mediterranean Basin. Open Virol. J. 2010, 4, 29–37. [Google Scholar]
- Ben Hassine, T.; Hammami, S.; Ghram, A. Detection of circulation of West Nile virus in equine in the north-west of Tunisia. Bull. Soc. Pathol. Exot. 2011, 104, 266–271. [Google Scholar] [CrossRef]
- Gárcia-Bocanegra, I.; Jaén-Téllez, J.A.; Napp, S.; Arenas-Montes, A.; Fernández-Molera, V.; Arenas, A. Monitoring of the West Nile virus epidemic in Spain between 2010 and 2011. Transbound. Emerg. Dis. 2012, 59, 448–455. [Google Scholar] [CrossRef]
- Calzolari, M.; Gaibani, P.; Bellini, R.; Defilippo, F.; Pierro, A.; Alvieri, A.; Maioli, G.; Luppi, A.; Rossini, G.; Balzani, A.; et al. Mosquito, bird and human surveillance of West Nile and Usutu viruses in Emilia-Romagna Region (Italy) in 2010. PLoS One 2012, 7, e38058. [Google Scholar] [CrossRef]
- Medlock, J.M.; Snow, K.R.; Leach, S. Possible ecology and epidemiology of medically important mosquito-borne arboviruses in Great Britain. Epidemiol. Infect. 2007, 135, 466–482. [Google Scholar] [CrossRef]
- Autorino, G.L.; Battisti, A.; Deubel, V.; Ferrari, G.; Forletta, R.; Giovannini, A.; Lelli, R.; Murri, S.; Scicluna, M.T. West Nile virus epidemic in horses, Tuscany region, Italy. Emerg. Infect. Dis. 2002, 8, 1372–1378. [Google Scholar] [CrossRef]
- Monaco, F.; Lelli, R.; Teodori, L.; Pinoni, C.; di Gennaro, A.; Polci, A.; Calistri, P.; Savini, G. Re-emergence of West Nile virus in Italy. Zoonoses Public Health 2010, 57, 476–486. [Google Scholar] [CrossRef]
- Toma, L.; Cipriani, M.; Goredo, M.; Romi, R.; Lelli, R. First report on entomological field activities for the surveillance of West Nile disease in Italy. Vet. Ital. 2008, 44, 483–497. [Google Scholar]
- Severini, F.; Toma, L.; di Luca, M.; Romi, R. Le zanzare italiane: Generalità e identificazione degli adulti (Diptera, Culicidae). Fragm. Entomol. 2009, 41, 213–372. [Google Scholar]
- Calzolari, M.; Bonilauri, P.; Bellini, R.; Albieri, A.; Defilippo, F.; Tamba, M.; Tassinari, M.; Gelati, A.; Cordioli, P.; Angelini, P.; et al. Usutu virus persistence and West Nile Virus inactivity in the Emilia-Romagna region (Italy) in 2011. PLoS One 2013, 8, e63978. [Google Scholar] [CrossRef]
- Savini, G.; Capelli, G.; Monaco, F.; Polci, A.; Russo, F.; Di Gennaro, A.; Marini, V.; Teodori, L.; Montarsi, F.; Pinoni, C.; et al. Evidence of West Nile virus lineage 2 circulation in Northern Italy. Vet. Microbiol. 2012, 158, 267–273. [Google Scholar] [CrossRef]
- Savini, G.; Puggioni, G.; Di Gennaro, A.; Di Francesco, G.; Rocchigiani, A.M.; Polci, A.; Marini, V.; Pinoni, C.; Rolesu, S.; Marruchella, G.; et al. West Nile virus lineage 2 in Sardinian wild birds in 2012: A further threat to public health. Epidemiol. Infect. 2013. [Google Scholar] [CrossRef]
- Papa, A. West Nile virus infection in Greece—An update. Exp. Rev. Antinfect. Ther. 2012, 10, 743–750. [Google Scholar] [CrossRef]
- Papa, A. West Nile virus infections in humans-Focus on Greece. J. Clin. Virol. 2013, 58, 351–353. [Google Scholar]
- Papa, A.; Xanthopoulou, K.; Gewehr, S.; Mourelatos, S. Detection of West Nile virus lineage 2 in mosquitoes during a human outbreak in Greece. Clin. Microbiol. Infect. 2011, 17, 1176–1180. [Google Scholar]
- Valiakos, G.; Touloudi, A.; Iacovakis, C.; Athanasiou, L.; Birtsas, P.; Spyrou, V.; Billinis, C. Molecular detection and phylogenetic analysis of West Nile virus lineage 2 in sedentary wild birds (Eurasian magpie), Greece, 2010. Euro Surveill. 2011, 16. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19862 (accessed on 25 September 2013). [Google Scholar]
- Chaskopoulou, A.; Dovas, C.; Chaintoutis, S.; Bouzalas, I.; Ara, G.; Papanastassopoulou, M. Evidence of enzootic circulation of West Nile virus (Nea Santa-Greece-2010, lineage 2), Greece, May to July 2011. Euro Surveill. 2011, 16. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19933 (accessed on 25 September 2013). [Google Scholar]
- Papa, A.; Papadopoulou, E.; Gavana, E.; Kalaitzopoulou, S.; Mourelatos, S. Detection of West Nile virus lineage 2 in Culex mosquitoes, Greece, 2012. Vector Borne Zoonotic Dis. 2013, 13, 682–684. [Google Scholar]
- Papa, A.; Xanthopoulou, K.; Tsioka, A.; Kalaitzopoulou, S.; Mourelatos, S. West Nile virus in mosquitoes in Greece. Parasitol. Res. 2013, 112, 1551–1555. [Google Scholar] [CrossRef]
- Samanidou-Voyadjoglou, A.; Darsie, R.F., Jr. New country records for mosquito species in Greece. J. Am. Mosq. Control Assoc. 1993, 9, 465–466. [Google Scholar]
- Samanidou-Voyadjoglou, A.; Patsoula, E.; Spanakos, G.; Vakalis, N. Confirmation of Aedes albopictus (Skuse) (Diptera: Culicidae) in Greece. Eur. Mosq. Bull. 2005, 19, 10–12. [Google Scholar]
- Giatropoulos, A.; Emmanouel, N.; Koliopoulos, G.; Michaelakis, A. A study on distribution and seasonal abundance of Aedes albopictus (Diptera: Culicidae) population in Athens, Greece. J. Med. Entomol. 2012, 49, 262–269. [Google Scholar] [CrossRef]
- Giatropoulos, A.; Michaelakis, A.; Koliopoulos, G.; Pontikakos, C. Records of Aedes albopictus and Aedes cretinus (Diptera: Culicidae) in Greece from 2009 to 2011. Hell. Plant Prot. J. 2012, 5, 49–56. [Google Scholar]
- Aranda, C.; Sánchez-Seco, M.P.; Cáceres, F.; Escosa, R.; Gálvez, J.C.; Masià, M.; Marques, E.; Ruiz, S.; Alba, A.; Busquets, N.; et al. Detection and monitoring of mosquito flaviviruses in Spain between 2001 and 2005. Vector Borne Zoonotic Dis. 2009, 9, 171–178. [Google Scholar] [CrossRef]
- Busquets, N.; Alba, A.; Allepuz, A.; Aranda, C.; Nuñez, J.I. Usutu virus sequences in Culex pipiens (Diptera: Culicidae), Spain. Emerg. Infect. Dis. 2008, 14, 861–863. [Google Scholar] [CrossRef]
- Alba, A.; Allepuz, A.; Napp, S.; Soler, M.; Selga, I.; Aranda, C.; Casal, J.; Pages, N.; Hayes, E.B.; Busquets, N. Ecological surveillance for West Nile in Catalonia (Spain), learning from a five-year period of follow-up. Zoonoses Public Health 2013. [Google Scholar] [CrossRef]
- Vazquez, A.; Ruiz, S.; Herrero, L.; Moreno, J.; Molero, F.; Magallanes, A.; Sánchez-Seco, M.P.; Figuerola, J.; Tenorio, A. West Nile and Usutu viruses in mosquitoes in Spain, 2008–2009. Am. J. Trop. Med. Hyg. 2011, 85, 178–181. [Google Scholar] [CrossRef]
- Vazquez, A.; Sánchez-Seco, M.P.; Palacios, G.; Molero, F.; Reyes, N.; Ruiz, S.; Aranda, C.; Marqués, E.; Escosa, R.; Moreno, J.; et al. Novel flaviviruses detected in different species of mosquitoes in Spain. Vector Borne Zoonotic Dis. 2012, 12, 223–229. [Google Scholar] [CrossRef] [Green Version]
- Calzolari, M.; Ze-Ze, L.; Ruzek, D.; Vazquez, A.; Jeffries, C.; Defilippo, F.; Costa Osorio, H.; Kilian, P.; Ruiz, S.; Fooks, A.R.; et al. Detection of mosquito-only flaviviruses in Europe. J. Gen. Virol. 2012, 93, 1215–1225. [Google Scholar] [CrossRef] [Green Version]
- Sotelo, E.; Fernández-Pinero, J.; Llorente, F.; Vázquez, A.; Moreno, A.; Agüero, M.; Cordioli, P.; Tenorio, A.; Jiménez-Clavero, M.A. Phylogenetic relationships of Western Mediterranean West Nile virus strains (1996–2010) using full-length genome sequences: Single or multiple introductions? J. Gen. Virol. 2011, 92, 2512–2522. [Google Scholar] [CrossRef]
- Gäumann, R.; Mühlemann, K.; Strasser, M.; Beuret, C.M. High-throughput procedure for tick surveys of tick-borne encephalitis virus and its application in a national surveillance study in Switzerland. Appl. Environ. Microbiol. 2010, 76, 4241–4249. [Google Scholar] [CrossRef]
- Lambrechts, L.; Paaijmans, K.P.; Fansiri, T.; Carrington, L.B.; Kramer, L.D.; Thomas, M.B.; Scott, T.W. Impact of daily temperature fluctuations on dengue virus transmission by Aedes aegypti. Proc. Natl. Acad. Sci. USA 2011, 108, 7460–7465. [Google Scholar] [CrossRef]
- Paaijmans, K.P.; Blanford, S.; Bell, A.S.; Blanford, J.I.; Read, A.F.; Thomas, M.B. Influence of climate on malaria transmission depends on daily temperature variation. Proc. Natl. Acad. Sci. USA 2010, 107, 15135–15139. [Google Scholar]
- Schaffner, F.; Kaufmann, C.; Hegglin, D; Mathis, A. The invasive mosquito Aedes japonicus in Central Europe. Med. Vet. Entomol. 2009, 23, 448–451. [Google Scholar] [CrossRef]
- Huber, K.; Pluskota, B.; Jöst, A.; Hoffman, K.; Becker, N. Status of the invasive species Aedes japonicus (Diptera: Culicidae) in southwest Germany in 2011. J. Vector Ecol. 2012, 37, 462–465. [Google Scholar] [CrossRef]
- Kampen, H.; Zielke, D.; Werner, D. A new focus of Aedes japonicus (Theobald, 1901) (Diptera, Culicidae) distribution in Western Germany: Rapid spread or a further introduction event? Parasit. Vectors 2012, 5, 284. [Google Scholar] [CrossRef]
- Williges, E.; Farajollahi, A.; Scott, J.J.; McCuiston, L.J.; Crans, W.J.; Gaugler, R. Laboratory colonization of Aedes japonicus. J. Am. Mosq. Control Assoc. 2008, 24, 591–593. [Google Scholar] [CrossRef]
- Schaffner, F.; Medlock, J.; van Bortel, W. Public health significance of invasive mosquitos in Europe. Clin. Microbiol. Infect. 2013, 19, 685–692. [Google Scholar] [CrossRef]
- Flacio, E.; Lüthy, P.; Patocchi, N.; Guidotti, F.; Tonolla, M.; Peduzzi, R. Primo ritrovamento di Aedes albopictus in Svizzera. Boll. Della Soc. Ticinese Sci. Nat. 2004, 92, 141–142. [Google Scholar]
- Wymann, M.N.; Flacio, E.; Radczuweit, S.; Patocchi, N.; Lüthy, P. Asian tiger mosquito (Aedes albopictus)—A threat for Switzerland? Euro Surveill. 2008, 13, 8058. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=8058 (accessed on 25 September 2013). [Google Scholar]
- Buckley, A.; Dawson, A.; Moss, S.R.; Hinsley, S.A.; Bellamy, P.E.; Gould, E.A. Serological evidence of West Nile virus, Usutu virus and Sindbis virus infection of birds in the UK. J. Gen. Virol. 2003, 84, 2807–2817. [Google Scholar] [CrossRef]
- Medlock, J.M.; Vaux, A.G.C. Aedes (Aedes) geminus Peus (Diptera: Culicidae)—An addition to the British mosquito fauna. Dipterists Digest 2010, 17, 1–6. [Google Scholar]
- Medlock, J.M.; Hansford, K.M.; Anderson, M.; Mayho, R.; Snow, K.R. Mosquito nuisance and control in the UK—A questionnaire-based survey of local authorities. Eur. Mosq. Bull. 2012, 30, 15–29. [Google Scholar]
- National Biodiversity Network. Available online: http://www.nbn.org.uk/ (accessed on 29 September 2013).
- Medlock, J.M.; Snow, K.R.; Leach, S. Potential transmission of West Nile virus in the British Isles: An ecological review of candidate mosquito bridge vectors. Med. Vet. Entomol. 2005, 19, 2–21. [Google Scholar] [CrossRef]
- Medlock, J.M.; Vaux, A.G.C. Distribution of West Nile virus vector, Culex modestus, in England. Vet. Record 2012, 15, 278. [Google Scholar] [CrossRef]
- Golding, N.; Nunn, M.A.; Medlock, J.M.; Purse, B.V.; Vaux, A.G.C.; Schafer, S.M. West Nile virus vector Culex modestus established in southern England. Parasites Vectors 2012. [Google Scholar] [CrossRef] [Green Version]
- Medlock, J.M.; Avenell, D.; Barrass, I.; Leach, S. Analysis of the potential for survival and seasonal activity of Aedes albopictus (Diptera: Culicidae) in the United Kingdom. J. Vector Ecol. 2006, 31, 292–304. [Google Scholar] [CrossRef]
- Vaux, A.G.C.; Murphy, G.; Baskerville, N.; Burden, N.; Convery, N.; Crossley, L.; Dettman, L.; Haden, P.; Jarrold, L.; Massey, C.; et al. Monitoring for invasive and endemic mosquitoes at UK ports. Eur. Mosq. Bull. 2011, 29, 133–140. [Google Scholar]
- Murphy, G.; Vaux, A.G.C.; Medlock, J.M. Challenges in undertaking mosquito surveillance at UK seaports and airports to prevent the entry and establishment of invasive vector species. Int. J. Envirol. Health 2012, 23, 181–190. [Google Scholar] [CrossRef]
- Medlock, J.M.; Vaux, A.G.C. Assessing the possible implications of wetland expansion and management on mosquitoes in Britain. Eur. Mosq. Bull. 2011, 29, 38–65. [Google Scholar]
- Medlock, J.M.; Vaux, A.G.C. Colonisation of UK coastal re-alignment sites by mosquitoes: Implications for design, management and public health. J. Vector Ecol. 2013, 38, 53–62. [Google Scholar] [CrossRef]
- Spieckermann, D.; Ackennann, R. Isolierung von Viren der California-Enzephalitis-Gruppe aus Stechmücken m Nordbayern. Zbl. Bakt Hyg. I. Abt. Orig. A 1972, 221, 283–295. [Google Scholar]
- Seidowski, D.; Ziegler, U.; von Rönn, J.A.; Müller, K.; Hüppop, K.; Müller, T.; Freuling, C.; Mühle, R.U.; Nowotny, N.; Ulrich, R.G.; Niedrig, M.; Groschup, M.H. West Nile virus monitoring of migratory and resident birds in Germany. Vector Borne Zoonotic Dis. 2010, 10, 639–647. [Google Scholar] [CrossRef]
- Ziegler, U.; Seidowski, D.; Angenvoort, J.; Eiden, M.; Müller, T.; Nowotny, N.; Groshup, M.H. Monitoring of West Nile virus infection in Germany. Zoonoses Public Health 2012, 59, 95–101. [Google Scholar] [CrossRef]
- Timmermann, U.; Becker, N. Mosquito-borne West Nile virus (WNV) surveillance in the Upper Rhine Valley, Germany. J. Vector Ecol. 2010, 35, 1–4. [Google Scholar] [CrossRef]
- Forschung Laboruntersuchungen von Stechmücken. Available online: http://www.kabsev.de/Seiten/Extern/Forschung%20Laboruntersuchungen.htm (accessed on 29 September 2013).
- Werner, D.; Kronefeld, M.; Schaffner, F.; Kampen, H. Two invasive mosquito species, Aedes albopictus and Aedes japonicus, trapped in south-west Germany, July to August 2011. Euro Surveill. 2012, 17, 20067. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=20067 (accessed on 25 September 2013). [Google Scholar]
- Kronefeld, M.; Dittmann, M.; Zielke, D.; Werner, D.; Kampen, H. Molecular confirmation of the occurrence in Germany of Anopheles daciae (Diptera, Culicidae). Parasite Vectors 2012, 5, 250. [Google Scholar] [CrossRef]
- Jöst, H.; Bialonski, A.; Storch, V.; Günther, S.; Becker, N.; Schmidt-Chanasit, J. Isolation and phylogenetic analysis of Sindbis viruses from mosquitoes in Germany. J. Clin. Microbiol. 2010, 48, 1900–1903. [Google Scholar] [CrossRef]
- Jöst, H.; Bialonski, A.; Schmetz, C.; Günther, S.: Becker, N.; Schmidt-Chanasit, J. Isolation and phylogenetic analysis of Batai virus, Germany. Am. J. Trop. Med. Hyg. 2011, 84, 241–243. [Google Scholar] [CrossRef]
- Jöst, H.; Bialonski, A.; Maus, D.; Sambri, V.; Eiden, M.; Groschup, M.H.; Günther, S.; Becker, N.; Schmidt-Chanasit, J. Isolation of Usutu virus in Germany. Am. J. Trop. Med. Hyg. 2011, 85, 551–553. [Google Scholar] [CrossRef]
- Becker, N.; Jöst, H.; Ziegler, U.; Eiden, M.; Höper, D.; Emmerich, P.; Fichet-Calvet, E.; Ehichioya, D.U.; Czajka, C.; Gabriel, M.; et al. Epizootic emergence of Usutu virus in wild and captive birds in Germany. PLoS One 2012, 7, e32604. [Google Scholar] [CrossRef]
- Chevalier, V.; Lecollinet, S.; Durand, B. West Nile virus in Europe: A comparison of surveillance system designs in a changing epidemiological context. Vector Borne Zoonotic Dis. 2011, 11, 1085–1091. [Google Scholar] [CrossRef]
- Parreira, R.; Severino, P.; Freitas, F.; Piedade, J.; Almeida, A.P.; Esteves, A. Two distinct introductions of the West Nile virus in Portugal disclosed by phylogenetic analysis of genomic sequences. Vector Borne Zoonotic Dis. 2007, 7, 344–352. [Google Scholar]
- Fonseca, D.M.; Keyghobadi, N.; Malcolm, C.A.; Mehmet, C.; Schaffner, F.; Mogi, M.; Fleischer, R.C.; Wilkerson, R.C. Emerging vectors in the Culex pipiens complex. Science 2004, 303, 1535–1538. [Google Scholar] [CrossRef]
- Johnson, N.; Wakeley, P.R.; Mansfield, K.L.; McCracken, F.; Haxton, B.; Phipps, L.P.; Fooks, L.P. Assessment of a novel real-time pan-flavivirus RT-polymerase chain reaction. Vector Borne Zoonotic Dis. 2010, 10, 665–671. [Google Scholar] [CrossRef]
- Huhtamo, E.; Moureau, G.; Cook, S.; Julkunen, O.; Putkuri, N.; Kurkela, S.; Uzcátegui, N.Y.; Harbach, R.E.; Gould, E.; Valpalahti, O.; et al. Novel insect-specific flavivirus isolated from northern Europe. Virology 2012, 433, 471–478. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Engler, O.; Savini, G.; Papa, A.; Figuerola, J.; Groschup, M.H.; Kampen, H.; Medlock, J.; Vaux, A.; Wilson, A.J.; Werner, D.; et al. European Surveillance for West Nile Virus in Mosquito Populations. Int. J. Environ. Res. Public Health 2013, 10, 4869-4895. https://doi.org/10.3390/ijerph10104869
Engler O, Savini G, Papa A, Figuerola J, Groschup MH, Kampen H, Medlock J, Vaux A, Wilson AJ, Werner D, et al. European Surveillance for West Nile Virus in Mosquito Populations. International Journal of Environmental Research and Public Health. 2013; 10(10):4869-4895. https://doi.org/10.3390/ijerph10104869
Chicago/Turabian StyleEngler, Olivier, Giovanni Savini, Anna Papa, Jordi Figuerola, Martin H. Groschup, Helge Kampen, Jolyon Medlock, Alexander Vaux, Anthony J. Wilson, Doreen Werner, and et al. 2013. "European Surveillance for West Nile Virus in Mosquito Populations" International Journal of Environmental Research and Public Health 10, no. 10: 4869-4895. https://doi.org/10.3390/ijerph10104869







