The Sponge-associated Bacterium Bacillus licheniformis SAB1: A Source of Antimicrobial Compounds
Abstract
:1. Introduction
2. Results and Discussion
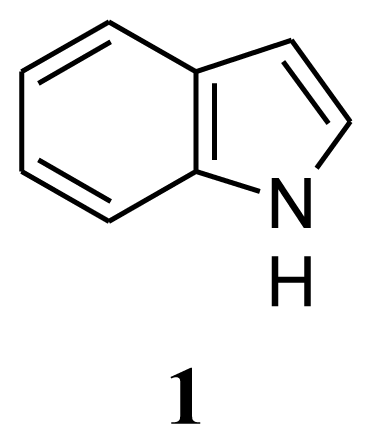
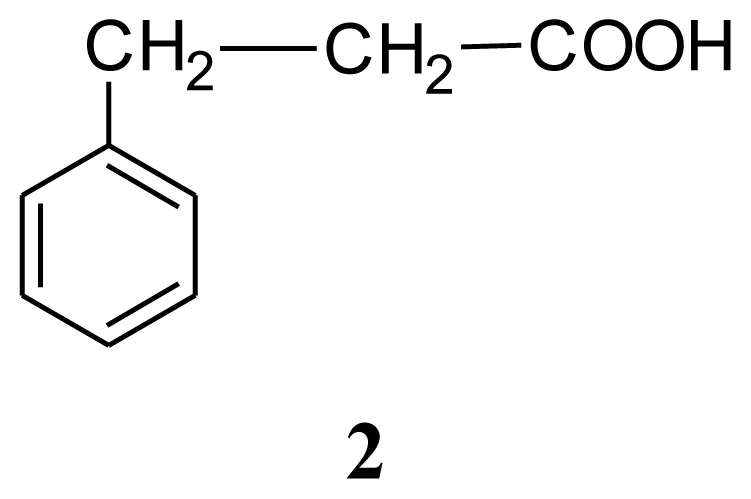
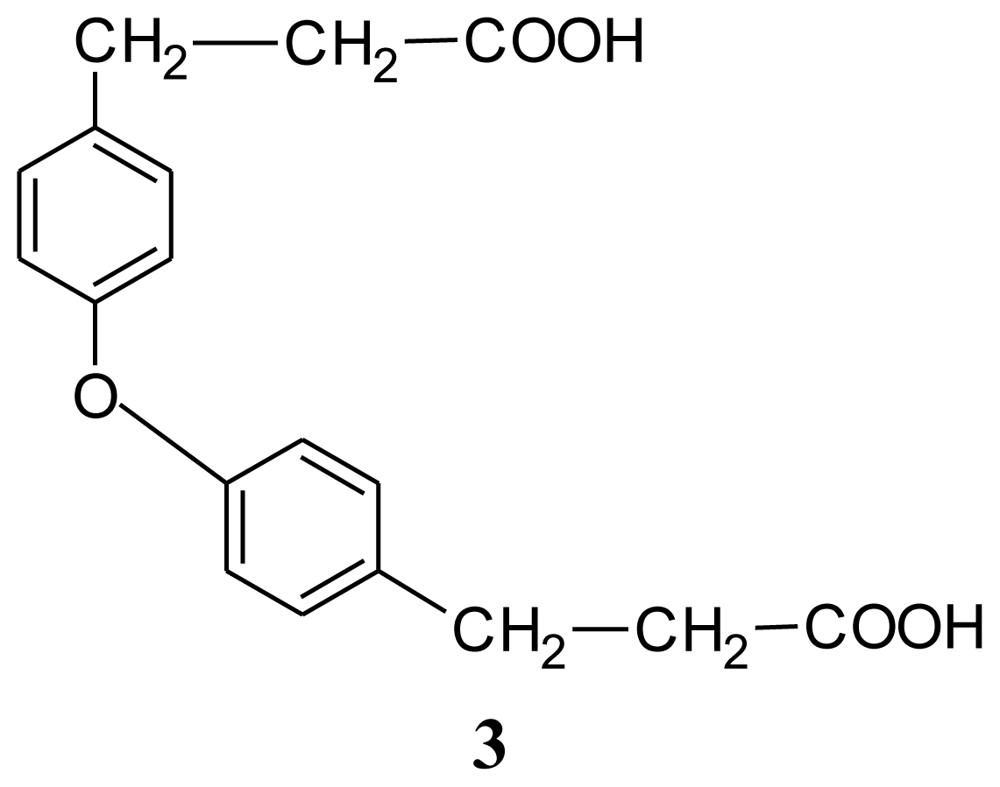
3. Experimental Section
3.1. Isolation of Bacteria Associated with Marine Sponge
3.2. Polymerase Chain Reaction
3.3. Cultivation of Bacterial Isolates for Screening
3.4. Agar Disc Diffusion Assay
3.5. Cultivation of Sponge-Associated Bacterium (SAB1) for Secondary Metabolite Production
3.6. Extraction and Purification of Active Metabolites
3.7. Analytical Methods/Instrumentation
4. Conclusions
Acknowledgements
- Sample Availability: Available from the authors.
References
- Radjasa, OK; Kencana, DS; Sabdono, A; Hutagalung, RA; Lestari, ES. Antibacterial activity of marine bacteria associated with sponge Aaptos sp. against Multi drug resistant (MDR) strains. J Matematika dan Sains 2007, 12, 147–152. [Google Scholar]
- Pawlik, JR; McFall, G; Zea, S. Does the odor from Sponges of the genus Ircinia protect them from fish predators? J Chem Ecol 2002, 28, 1103–1115. [Google Scholar]
- Burgess, JG; Jordan, EM; Bregu, M; Mearns-Spragg, A; Boyd, KG. Microbial antagonism: A neglected avenue of natural products research. J Biotechnol 1999, 70, 27–32. [Google Scholar]
- Slattery, M; Rajbhandari, I; Wesson, K. Competition-mediated antibiotic induction in the marine bacterium Streptomyces tenjimariensis. Microbiol Ecol 2001, 41, 90–96. [Google Scholar]
- Lemos, ML; Toranzo, AE; Barja, LJ. Antibiotic activity of epiphytic bacteria isolated from intertidal seaweeds. Microb Ecol 1986, 11, 149–163. [Google Scholar]
- Jensen, PR; Fenical, W. Strategies for the discovery of secondary metabolites from marine bacteria: ecological perspectives. Annu Rev Microbiol 1994, 48, 559–584. [Google Scholar]
- Boyd, KG; Mearns-Spragg, A; Burgess, JG. Screening of marine bacteria for the production of microbial repellents using a spectrophotometric chemotaxis assay. Mar Biotechnol 1999, 1, 359–363. [Google Scholar]
- Boyd, KG; Adams, DR; Burgess, JG. Antimicrobial and repellent activities of marine bacteria associated with algal surfaces. Biofouling 1999, 14, 227–236. [Google Scholar]
- Lu, Y; Dong, X; Liu, S; Bie, X. Characterization and identification of a novel marine Streptomyces sp. produced antibacterial substance. Mar Biotechnol 2009, 1436–222. [Google Scholar]
- Proksch, P; Edrada, RA; Ebel, R. Drugs from the Seas-current status and microbiological implications. Appl Microbiol Biotechnol 2002, 59, 125–134. [Google Scholar]
- Thiel, V; Imhoff, JF. Phylogenetic identification of bacteria with antimicrobial activities isolated from Mediterranean sponges. Biomol Eng 2003, 20, 421–423. [Google Scholar]
- Radjasa, OK; Martens, T; Grossart, HP; Brinkoff, T; Sabdono, A; Simon, M. Antagonistic activity of a marine bacterium Pseudoalteromonas luteoviolacea TAB4.2 associated with Coral Acropora sp. J Biol Sci 2007, 7, 239–246. [Google Scholar]
- Todar, K. The Good, the Bad, and the Deadly. Sci Mag. 2004, p. 1421. available online: http://www.textbookofbacteriology.net/science_review.html (accessed on 2 March 2010).
- Kennedy, J; Baker, P; Piper, C; Cotter, PD; Walsh, M; Mooij, MJ; Bourke, MB; Rea, MC; O’Connor, PM; Ross, RP; Hill, C; O’Gara, F; Marchesi, JR; Dobson, AD. Isolation and analysis of bacteria with antimicrobial activities from the marine sponge Haliclona simulans collected from Irish waters. Mar Biotechnol 2009, 11, 384–396. [Google Scholar]
- DellaGreca, M; Di Marino, C; Previtera, L; Purcaro, R; Zarrelli, A; Apteniols, A-F. Oxyneolignans from the Leaves of Aptenia cordifolia. Tetrahedron 2005, 61, 11924–11929. [Google Scholar]
- Mao, S; Lee, SJ; Hwangbo, H; Kim, YW; Park, KK; Cha, GS; Park, RD; Kim, KY. Isolation and characterization of antifungal substances from Burkholderia sp. Culture Broth. Curr Microbiol 2006, 53, 358–364. [Google Scholar]
- Narayana, KJP; Prabhakar, P; Vijayalakshmi, M; Venkateswarlu, Y; Krishna, PSJ. Biological activity of phenylpropionic acid isolated from a terrestrialStreptomycetes. Pol J Microbiol 2007, 56, 191–197. [Google Scholar]
- Donia, M; Hamann, MT. Marine natural products and their potential applications as anti-infective agents. Lancet Infect Dis 2003, 3, 338–348. [Google Scholar]
- Isnansetyo, A; Kamei, Y. Pseudoalteromonas phenolica sp. nov., a novel marine bacterium that produces phenolic anti-methicillin-resistant Staphylococcus aureus substances. Int J Syst Evol Microbiol 2003, 53, 583–588. [Google Scholar]
- Anand, TP; Bhat, AW; Shouche, YS; Roy, U; Siddharth, J; Sarma, SP. Antimicrobial activity of marine bacteria associated with sponges from the waters off the coast of south east India. Microbiol Res 2006, 161, 252–262. [Google Scholar]
- Uzair, B; Ahmed, N; Ahmed, V; Kousar, F. A new antibacterial compound produced by indigenous marine bacteria; fermentation, isolation and biological activity. Nat Prod Res 2006, 20, 1326–1331. [Google Scholar]
- Austin, B. Novel pharmaceutical compounds from marine bacteria. J Appl Bacteriol 1989, 67, 461–470. [Google Scholar]
- Prabha, D; D’Souza, L; Kamat, T; Rodrigues, C; Naik, CG. Batch culture fermentation of Penicillium chrysogenum and a report on the isolation, purification, identification and antibiotic activity of citrinin. Indian J Mar Sci 2009, 38, 38–44. [Google Scholar]
| Sr. No. | Isolate no. | Nature of pathogen | Name of pathogen | Disease it causes |
|---|---|---|---|---|
| 1. | B1 | Bacterial pathogens | Escherichia coli | Gastrointestinal infection |
| 2. | B2 | Pseudomonas aeruginosa | Urinary tract infection | |
| 3. | B3 | Staphylococcus aureus | Skin infection | |
| 4. | B4 | Salmonella typhi | Typhoid | |
| 5. | B5 | Shigella flexineri | Gastrointestinal infection | |
| 6. | B6 | Klebsiella sp. | Urinary tract infection | |
| 7. | B7 | Vibrio cholerae | Cholera | |
| 8. | F1 | Fungal pathogens | Aspergillus fumigatus | Skin infection |
| 9. | F2 | Rhodotorula sp. | Skin infection | |
| 10. | F3 | Candida albicans | Candidiasis | |
| 11. | F4 | Cryptococcus neoformans | Skin infection | |
| 12. | F5 | Aspergillus niger | Skin infection | |
| 13. | D1 | Multi-drug resistant bacteria | Streptococcus pyogenes | Skin infection |
| 14. | D2 | Acinetobacter sp. | Urinary tract infection | |
| 15. | D3 | Salmonella typhi | Typhoid | |
| 16. | D4 | Methicillin Resistant Staphylococcus aureus | Skin infection |
| Compound 1: indole | Compound 2: 3-phenylpropionic acid | Compound 3: 4,4′-oxybis(3-phenylpropionic acid) | ||||||
|---|---|---|---|---|---|---|---|---|
| Position | 1HNMR | 13CNMR | Position | 1HNMR | 13CNMR | Position | 1HNMR | 13CNMR |
| 1 | 8. 13 (b,s) | - | 1 | - | 179.45 | 1,1′ | - | 132.46 |
| 2 | 7. 64 (d, 7.5Hz) | 124.13 | 2 | 2.96(t,7.5,7.8Hz) | 35.61 | 2,6,2′,6′ | 7.07(d, 8.4Hz) | 115.43 |
| 3 | 7. 28 (d, 7.8Hz) | 121.89 | 3 | 2.68(t, 7.5,7.8Hz) | 30.54 | 3,5,3′,5′ | 6.75(d, 8.4Hz) | 129.48 |
| 4 | 6. 55 (s) | 102.42 | 1′ | - | 140.13 | 4,4′ | - | 154.11 |
| 5 | 7.09–7.23 (3H, m) | 120.65 | 2′,6′ | 7.19–7.38(5H,m) | 128.54 | 7,7′ | 2.89(t, 7.5,7.8Hz) | 29.88 |
| 6 | 119.74 | 3′,5′ | 128.24 | 8,8′ | 2.64(t, 7.5,7.8Hz) | 34.49 | ||
| 7 | 111.01 | 4′ | 126.38 | |||||
| 8 | 135.69 | |||||||
| 9 | 127.76 | |||||||
| No. | Pathogens used for study | Crude Extract (100μg/disc) | Comp 1 (50μg/disc) | Comp 2 (50μg/disc) | Comp 3 (50μg/disc) | STD Antibiotics (50μg/disc) | Solvent Methanol |
|---|---|---|---|---|---|---|---|
| B1 | Escherichia coli | - | - | - | - | ++++ | - |
| B2 | Pseudomonas aeruginosa | +++ | ++ | ++ | - | +++++ | - |
| B3 | Staphylococcus aureus | ++ | +++ | ++ | - | +++ | - |
| B4 | Salmonella typhi | + | +++ | ++ | ++ | ++ | - |
| B5 | Shigella flexineri | - | - | - | - | +++++ | - |
| B6 | Klebsiella sp. | - | - | - | - | +++++ | - |
| B7 | Vibrio cholerae | + | - | - | ++ | +++++ | - |
| D1 | Streptococcus pyogenes | + | + | ++ | - | ++ | - |
| D2 | Acinetobacter sp. | + | + | ++ | - | - | - |
| D3 | Salmonella typhi | - | - | - | - | - | - |
| D4 | Methicillin Resistant S.aureus | + | ++ | ++ | - | ++ | - |
| F1 | Aspergillus fumigatus | ++ | - | - | +++ | ++++ | - |
| F2 | Rhodotorula sp. | + | ++ | +++ | - | - | - |
| F3 | Candida albicans | + | +++ | ++ | - | - | - |
| F4 | Aspergillus niger | + | + | ++ | - | - | - |
| F5 | Cryptococcus neoformans | - | - | - | - | - | - |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Devi, P.; Wahidullah, S.; Rodrigues, C.; Souza, L.D. The Sponge-associated Bacterium Bacillus licheniformis SAB1: A Source of Antimicrobial Compounds. Mar. Drugs 2010, 8, 1203-1212. https://doi.org/10.3390/md8041203
Devi P, Wahidullah S, Rodrigues C, Souza LD. The Sponge-associated Bacterium Bacillus licheniformis SAB1: A Source of Antimicrobial Compounds. Marine Drugs. 2010; 8(4):1203-1212. https://doi.org/10.3390/md8041203
Chicago/Turabian StyleDevi, Prabha, Solimabi Wahidullah, Cheryl Rodrigues, and Lisette D. Souza. 2010. "The Sponge-associated Bacterium Bacillus licheniformis SAB1: A Source of Antimicrobial Compounds" Marine Drugs 8, no. 4: 1203-1212. https://doi.org/10.3390/md8041203




