FVIIa-sTF and Thrombin Inhibitory Activities of Compounds Isolated from Microcystis aeruginosa K-139
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of Aeruginosin K139, Micropeptin K139, and Microviridin B from M. aeruginosa K139
2.2. Aeruginosin K139
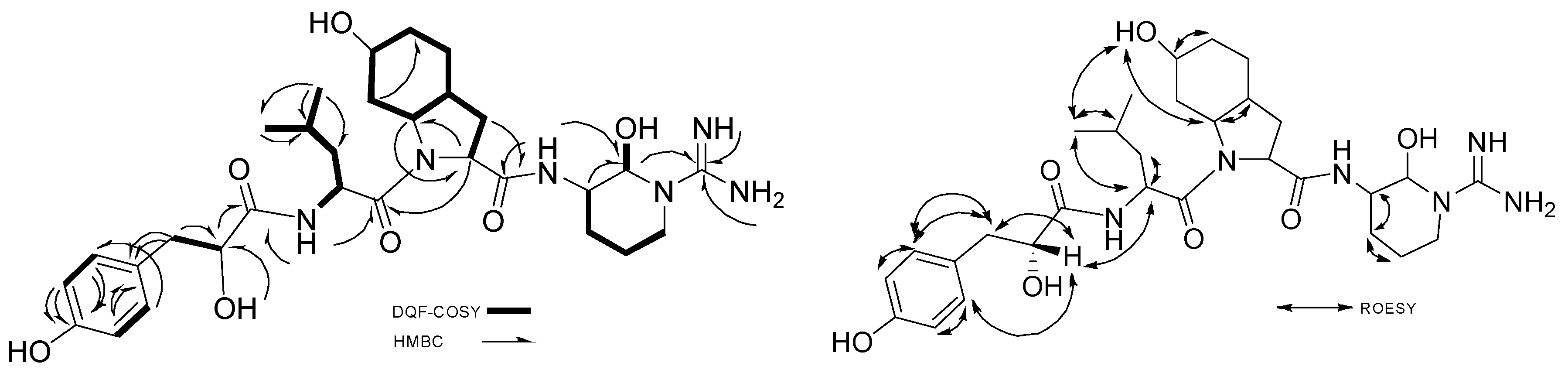
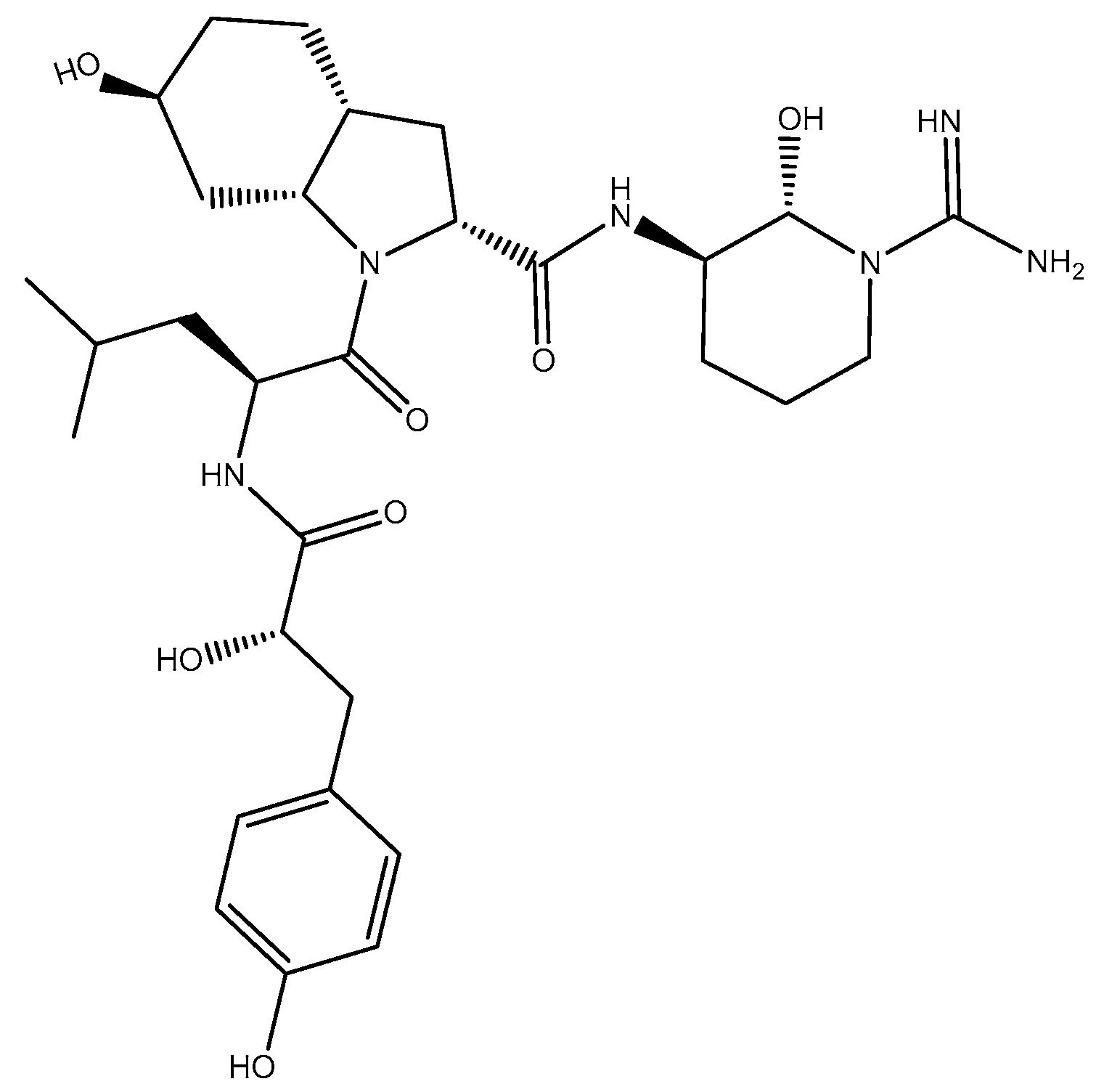
2.2.1. Structure Elucidation of Aeruginosin K139
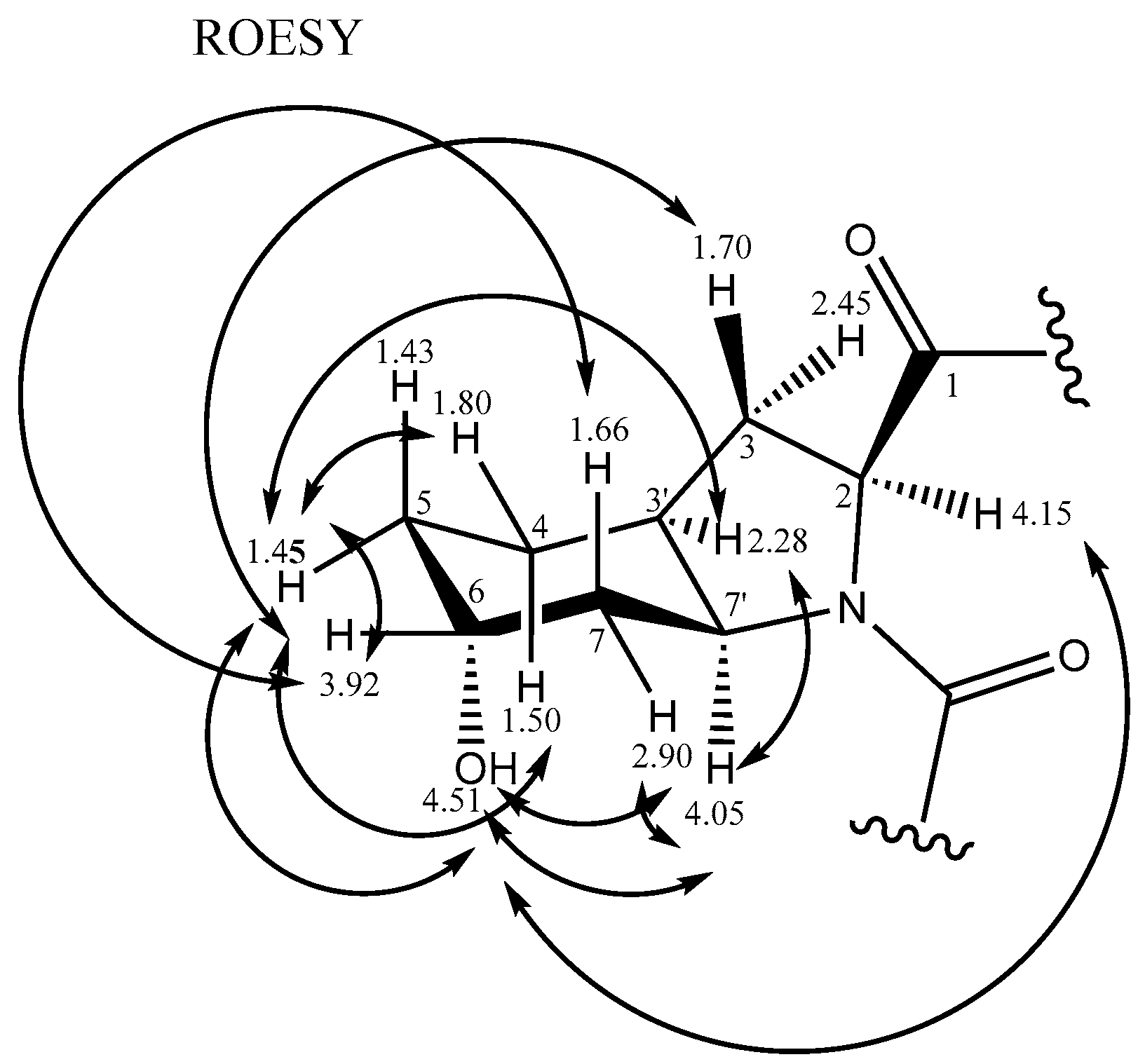
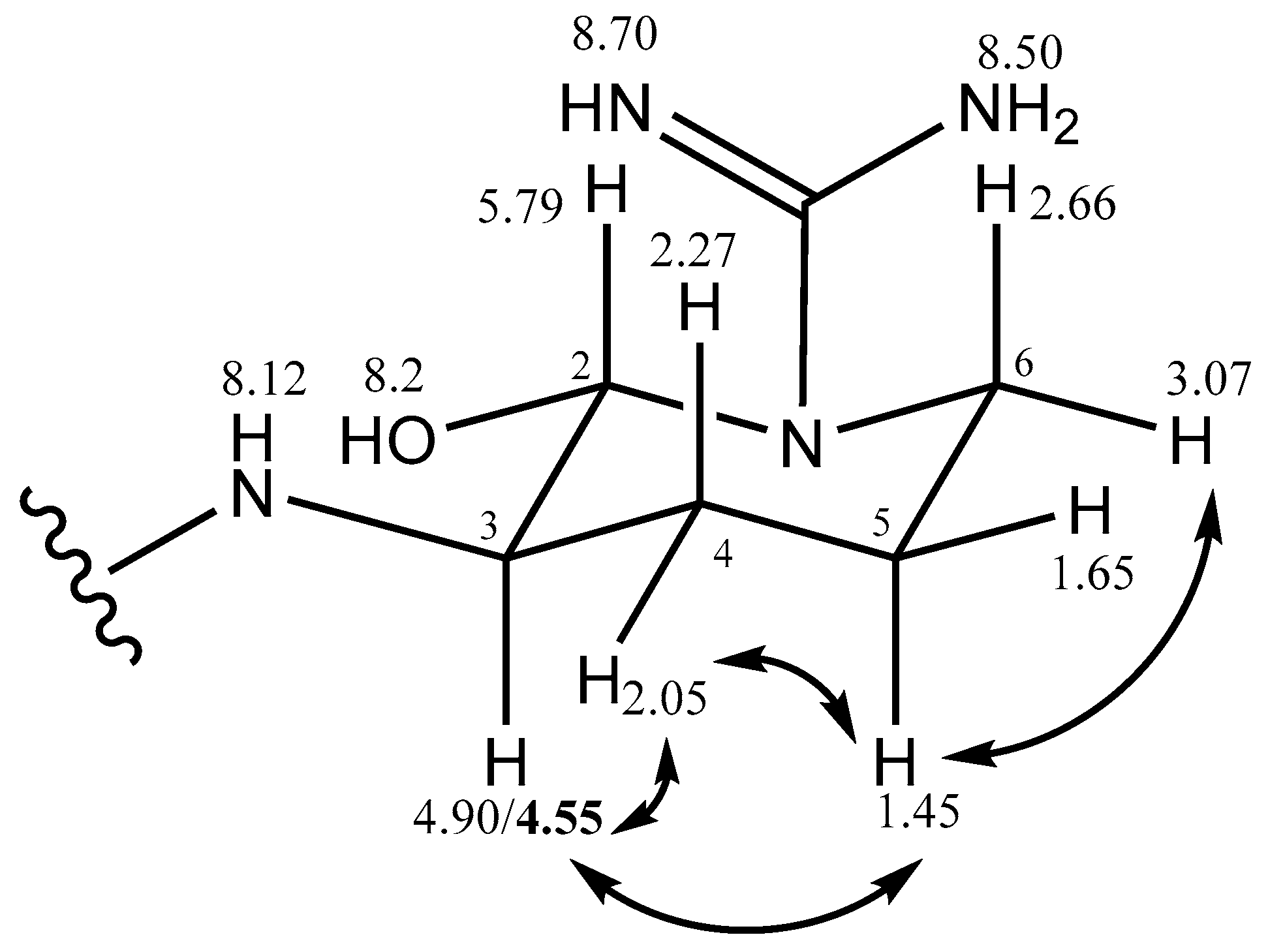
2.2.2. Stereochemistry of Aeruginosin K139
Hpla
Leu
Choi
Argininal
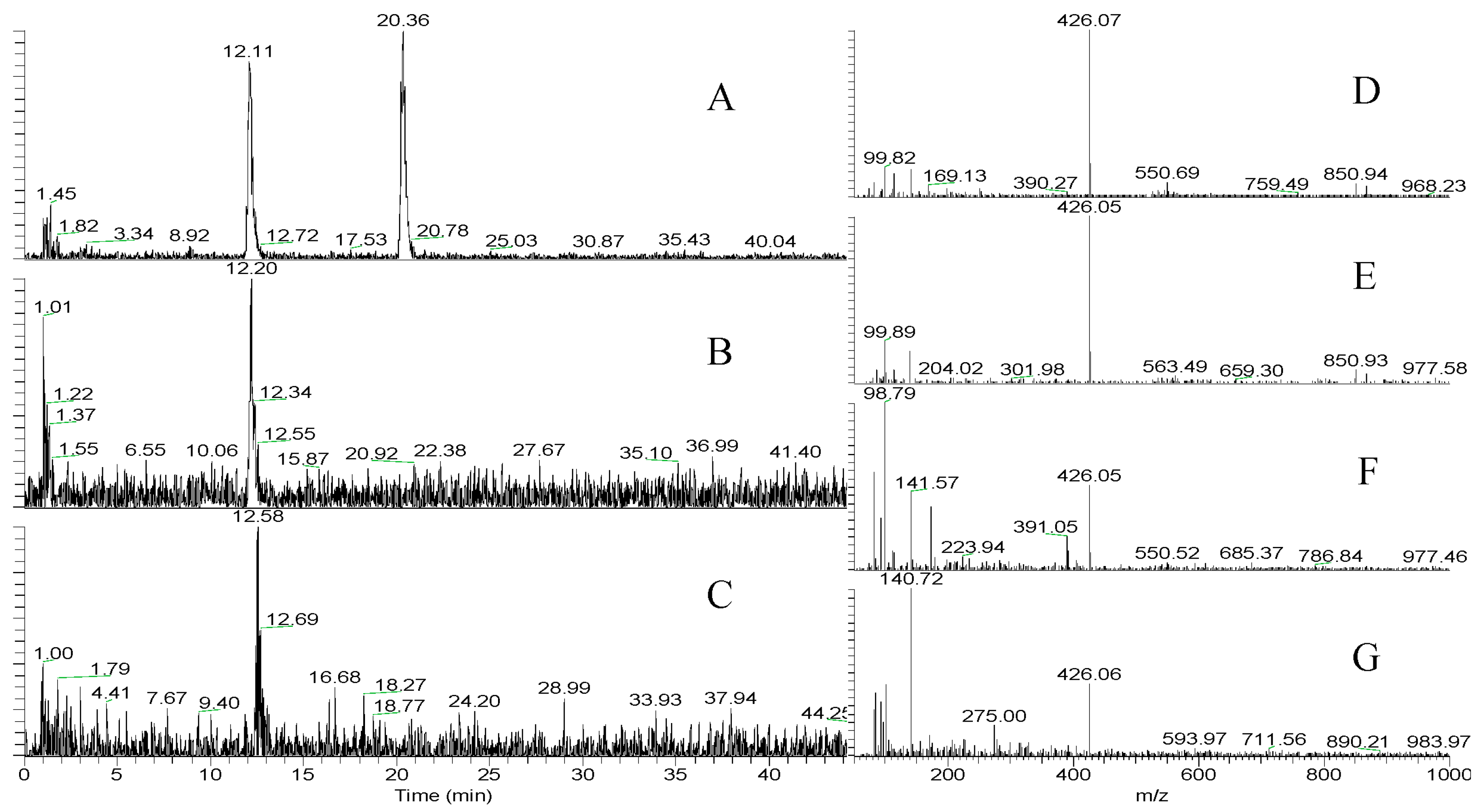
2.2.3. LC-MS and HR-MS of Aeruginosin K139
2.3. FVIIa-sTF and Thrombin Assays
3. Experimental Section
3.1. Laboratory Culture of M. aeruginosa K139
3.2. Isolation of fVIIa-sTF and Thrombin Inhibitors from M. aeruginosa K139
3.3. Thin Layer Chromatography (TLC)
3.4. Open Column Chromatography
3.4.1. Aeruginosin K139
3.4.2. Micropeptin K139 and Microviridin B
3.5. LC-MS and LC-MS/MS
3.6. 1D-NMR and 2D-NMR
3.7. Hydrolysis and Advanced Marfey Analysis
3.8. FVIIa-sTF and Thrombin Assays
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Palareti, G.; Leali, N.; Coccheri, S.; Poggi, M.; Manotti, C.; D’Angelo, A.; Pengo, V.; Erba, N.; Moia, M.; Ciavarella, N.; et al. Bleeding complications of oral anticoagulant treatment: An inception-cohort, prospective collaborative study (ISCOAT). Lancet 1996, 348, 423–428. [Google Scholar] [CrossRef]
- Anas, A.R.J.; Harada, K.-I. Evaluation of Serine Protease Inhibitors as Potent FVIIa-sTF Inhibitors in the Blood Coagulation Cascade. Lett. Drug Des. Discov. 2016, 13, 3–23. [Google Scholar] [CrossRef]
- Nakagura, T.; Tabata, K.; Kira, K.; Hirota, S.; Clark, R.; Matsuura, F.; Hiyoshi, H. Selective tissue factor/factor VIIa Inhibitor, ER-410660, and its prodrug, E5539, have anti-venous and anti-arterial thrombotic effects with a low risk of bleeding. Thromb. Res. 2013, 132, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Eikelboom, J.W.; Weitz, J.I. New anticoagulants. Circulation 2010, 121, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Weitz, J.I.; Bates, S.M. New anticoagulants. J. Thromb. Haemost. 2005, 3, 1843–1853. [Google Scholar] [CrossRef] [PubMed]
- Davie, E.W. A brief historical review of the waterfall/cascade of blood coagulation. J. Biol. Chem. 2003, 278, 50819–50832. [Google Scholar] [CrossRef] [PubMed]
- Davie, E.W.; Fujikawa, K.; Kisiel, W. The coagulation cascade: Initiation, maintenance, and regulation. Biochemistry 1991, 30, 10363–10370. [Google Scholar] [CrossRef] [PubMed]
- Davie, E.W.; Fujikawa, K.; Kurachi, K.; Kisiel, W. The role of serine proteases in the blood coagulation cascade. Adv. Enzymol. Relat. Areas Mol. Biol. 1979, 48, 277–318. [Google Scholar] [PubMed]
- Davie, E.W.; Ratnoff, O.D. Waterfall sequence for intrinsic blood clotting. Science 1964, 145, 1310–1312. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, K.; Naito, S.; Kondo, F.; Watanabe, M.F.; Suzuki, S.; Nakazawa, H.; Suzuki, M.; Shimada, T.; Harada, K.-I. A clean-up method for analysis of trace amounts of microcystins in lake water. Toxicon 1994, 32, 1251–1259. [Google Scholar] [CrossRef]
- Harada, K.-I. Production of secondary metabolites by freshwater cyanobacteria. Chem. Pharm. Bull. 2004, 52, 889–899. [Google Scholar] [CrossRef] [PubMed]
- Anas, A.R.J.; Nakajima, A.; Naruse, C.; Tone, M.; Asukabe, H.; Harada, K.I. Determination of FVIIa-sTF Inhibitors in Toxic Microcystis Cyanobacteria by LC-MS Technique. Mar. Drugs 2015, 14, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.E. Cyclic peptides and depsipeptides from cyanobacteria: A review. J. Ind. Microb. 1996, 16, 134–143. [Google Scholar] [CrossRef]
- Matsuda, H.; Okino, T.; Murakami, M.; Yamaguchi, K. Aeruginosins 102-A and B, new thrombin inhibitors from the cyanobacterium Microcystis viridis (NIES-102). Tetrahedron 1996, 52, 14501–14506. [Google Scholar] [CrossRef]
- Shin, H.J.; Matsuda, H.; Murakami, M.; Yamaguchi, K. Aeruginosins 205A and -B, serine protease inhibitory glycopeptides from the cyanobacterium Oscillatoria agardhii (NIES-205). J. Org. Chem. 1997, 62, 1810–1813. [Google Scholar] [CrossRef]
- Anas, A.R.J.; Kisugi, T.; Umezawa, T.; Matsuda, F.; Campitelli, M.R.; Quinn, R.J.; Okino, T. Thrombin inhibitors from the freshwater cyanobacterium Anabaena compacta. J. Nat. Prod. 2012, 75, 1546–1552. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Okita, Y.; Matsuda, H.; Okino, T.; Yamaguchi, K. Aeruginosin 298-A, a thrombin and trypsin inhibitor from the blue-green alga Microcystis aeruginosa (NIES-298). Tetrahedron Lett. 1994, 35, 3129–3132. [Google Scholar] [CrossRef]
- Ishida, K.; Okita, Y.; Matsuda, H.; Okino, T.; Murakami, M. Aeruginosins, protease inhibitors from the cyanobacterium Microcystis aeruginosa. Tetrahedron 1999, 55, 10971–10988. [Google Scholar] [CrossRef]
- Kodani, S.; Ishida, K.; Murakami, M. Aeruginosin 103-A, a thrombin inhibitor from the cyanobacterium Microcystis viridis. J. Nat. Prod. 1998, 61, 1046–1048. [Google Scholar] [CrossRef] [PubMed]
- Ersmark, K.; Del Valle, J.R.; Hanessian, S. Chemistry and biology of the aeruginosin family of serine protease inhibitors. Angew. Chem. Int. Ed. 2008, 47, 1202–1223. [Google Scholar] [CrossRef] [PubMed]
- Okino, T.; Murakami, M.; Haraguchi, R.; Munekata, H.; Matsuda, H.; Yamaguchi, K. Micropeptins A and B, plasmin, and trypsin inhibitors from the blue-green alga Microcystis aeruginosa. Tetrahedron Lett. 1993, 34, 8131–8134. [Google Scholar] [CrossRef]
- Kisugi, T.; Okino, T. Micropeptins from the freshwater cyanobacterium Microcystis aeruginosa (NIES-100). J. Nat. Prod. 2009, 72, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Ishitsuka, M.O.; Kusumi, T.; Kakisawa, H.; Kaya, K.; Watanabe, M.M. Microviridin. A novel tricyclic depsipeptide from the toxic cyanobacterium Microcystis viridis. J. Am. Chem. Soc. 1990, 112, 8180–8182. [Google Scholar] [CrossRef]
- Nakano, M.; Nakano, Y.; Saito-Taki, T.; Mori, N.; Kojima, M.; Ohtake, A.; Shirai, M. Toxicity of Microcystis aeruginosa K139 Strain. Microbiol. Immunol. 1989, 33, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, T.; Ueda, A.; Nakano, T.; Nishizawa, A.; Miura, T.; Asayama, M.; Fujii, K.; Harada, K.-I.; Shirai, M. Characterization of the locus of genes encoding enzymes producing heptadepsipeptide micropeptin in the unicellular cyanobacterium Microcystis. J. Biochem. 2011, 149, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Nishizawa, A.; Arshad, A.B.; Nishizawa, T.; Asayama, M.; Fujii, K.; Nakano, T.; Harada, K.-I.; Shirai, M. Cloning and characterization of a new hetero-gene cluster of nonribosomal peptide synthetase and polyketide synthase from the cyanobacterium Microcystis aeruginosa K139. J. Gen. Appl. Microbiol. 2007, 53, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Fewer, D.P.; Jokela, J.; Paukku, E.; Österholm, J.; Wahlsten, M.; Permi, P.; Aitio, O.; Rouhiainen, L.; Gomez-Saez, G.V.; Sivonen, K. New structural variants of aeruginosin produced by the toxic bloom forming cyanobacterium Nodularia spumigena. PLoS ONE 2013, 8, e73618. [Google Scholar] [CrossRef] [PubMed]
- Harada, K.I.; Nakano, T.; Fujii, K.; Shirai, M. Comprehensive analysis system using liquid chromatography–mass spectrometry for the biosynthetic study of peptides produced by cyanobacteria. J. Chromatogr. A 2004, 1033, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, M.; Pinto, F.C.R.; Vieira, J.M.S.; Honda, R.Y.; Pimenta, A.M.; Bemquerer, M.P.; Carvalho, L.R.; Kiyota, S. Isolation and structural characterization of microcystin-LR and three minor oligopeptides simultaneously produced by Radiocystis feernandoi (Chroococcales, Cyanobacteriae): A Brazilian toxic cyanobacterium. Toxicon 2006, 47, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Harada, K.I.H. Study on Non-Ribosomal Peptide Synthesis of Peptides by Cyanobacteria. Bachelor’s Thesis, Meijo University, Nagoya, Japan, 2003. [Google Scholar]
- Vegman, M.; Carmeli, S. Three aeruginosins and a microviridin from a bloom assembly of Microcystis spp. collected from a fishpond near Kibbutz Lehavot HaBashan, Israel. Tetrahedron 2014, 70, 6817–6824. [Google Scholar] [CrossRef]
- Fujii, K.; Ikai, Y.; Oka, H.; Suzuki, M.; Harada, K.I. A nonempirical method using LC/MS for determination of the absolute configuration of constituent amino acids in a peptide: Combination of Marfey’s method with mass spectrometry and its practical application. Anal. Chem. 1997, 69, 5146–5151. [Google Scholar] [CrossRef]
- Marfey, P. Determination of d-amino acids. II. Use of a bifunctional reagent, 1,5-difluoro-2,4-dinitrobenzene. Carlsberg Res. Commun. 1984, 49, 591–596. [Google Scholar] [CrossRef]
- GraphPad Prism 7; GraphPad Software, Inc.: La Jolla, CA, USA, 2016.
- Okino, T.; Matsuda, H.; Murakami, M.; Yamaguchi, K. New microviridins, elastase inhibitors from the blue-green alga Microcystis aeruginosa. Tetrahedron 1995, 51, 10679–10686. [Google Scholar] [CrossRef]
- Kadono, S.; Sakamoto, A.; Kikuchi, Y.; Oh-eda, M.; Yabuta, N.; Koga, T.; Hattori, K.; Shiraishi, T.; Haramura, M.; Kodama, H.; et al. Structure of human factor VIIa/tissue factor in complex with a peptide-mimetic inhibitor: High selectivity against thrombin by introducing two charged groups in P2 and P4. Acta Cryst. 2005, 61, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.J.; Murakami, M.; Matsuda, H.; Yamaguchi, K. Microviridins D-F, serine protease inhibitors from the cyanobacterium Oscillatoria agardhii (NIES-204). Tetrahedron 1996, 52, 8159–8168. [Google Scholar] [CrossRef]
- Ohtake, A.; Shirai, M.; Aida, T.; Mori, N.; Harada, K.; Matsuura, K.; Suzuki, M.; Nakano, M. Toxicity of Microcystis species isolated from natural blooms and purification of the toxin. Appl. Environ. Microbiol. 1989, 55, 3202–3207. [Google Scholar] [PubMed]

| Unit | 1H | J (Hz) | HSQC | HMBC | Cosy Double Quantum Filter (DQF-COSY) | Rotational-Frame Nuclear Overhauser Effect SpectroscopY (ROESY) | |
|---|---|---|---|---|---|---|---|
| Hpla | 1 | 174.9 | |||||
| 2 | 4.05 | dd (3.4,7.2) | 72.0 | 1 | 3a,3b | 3b,5,9,2-Leu | |
| 3a | 2.66 | dd (14.0,7.2) | 39.0 | 1,9 | 2 | 3b,5,9 | |
| 3b | 2.86 | dd (14.0,3.4) | 4 | 2 | 2,3a,5,9 | ||
| 4 | 128.0 | 6,8 | |||||
| 5,9 | 6.99 | d (8.3) | 130.4 | 5,7,9 | 6,8 | 2,3a,3b,6,8 | |
| 6,8 | 6.64 | d (8.3) | 114.6 | 7 | 5,9 | 5,9 | |
| 7 | 155.6 | 6,8 | |||||
| 2-OH | 8.70 | s | |||||
| 7-OH | 9.16 | s | |||||
| Leu | 1 | 166.0 | 5, Hpla-2 | ||||
| 2 | 4.66 | t (8.4,8.9) | 59.5 | 3,NH-Leu | 5 | ||
| 3 | 1.36 | m | 42.0 | 2,4 | 2 | ||
| 4 | 1.35 | m | 24.5 | 3,5,5′ | 5 | ||
| 5 | 0.89 | d (6.2) | 21.4 | 4 | 4 | 2,4,Choi 6-OH | |
| 5′ | 0.83 | d (6.2) | 23.3 | 3,4,5 | 4 | ||
| NH | 7.41 | d (8.1) | Hpla-1,1 | 2 | |||
| Choi | 1 | 173.0 | |||||
| 2 | 4.15 | t (2.5,6.3) | 59.5 | Leu-1,7′ | 2,3a,3b | 6-OH | |
| 3a | 1.70 (eq) | solvent overlap | 29.5 | 1 | 2,3′ | 6 | |
| 3b | 2.45 (ax) | solvent overlap | 2,3′ | ||||
| 3′ | 2.28 | m (broad) | 36.0 | 3a,3b,4a,4b,7′ | 5b,7′ | ||
| 4a | 1.50 | m | 20.0 | 3′ | |||
| 4b | 1.80 | m | 3′,5 | 5b | |||
| 5a | 1.43 | m, overlap | 26.0 | 4b,6 | 6-OH | ||
| 5b | 1.45 | 3′,4b,6 | |||||
| 6 | 3.92 | broad | 64.0 | 5,7a | 3a,5b,7b | ||
| 7a | 2.90 (eq) | inc dd | 33.7 | 5 | 6,7′ | 7′ | |
| 7b | 1.66 (ax) | s | 7′ | 6 | |||
| 7′ | 4.05 | dd (3.4,3.6) | 54.0 | 2 | 3′,7a,7b | 3′, 6-OH,7a | |
| 6-OH | 4.51 | broad s | 2, 5a,7′ | ||||
| Argal (cyclic) | 2 | 5.79 | broad | 74.0 | C=N | 6-OH | 4a,4b |
| 3 | 4.90/4.55 | m | 56.5 | 2 | 4a,4b | 4a,5a | |
| 4a | 2.05 | m (broad) | 36.0 | C=N | 3,5a,5b | 2,3,5a | |
| 4b | 2.27 | m (broad) | 3,5a,5b | 2 | |||
| 5a | 1.45 | m, overlap | 26.0 | 4a,4b | 3,4a,6a | ||
| 5b | 1.65 | 4a,4b | 6a | ||||
| 6a | 2.66 | 2 | |||||
| 6b | 3.07 | m | 40.5 | 5a,5b | |||
| NH | 8.12 | broad | 1-Choi, 2 | ||||
| C=NH | 7.35 | broad | C=N | ||||
| C=NH | 8.53 | s | C=N | ||||
| C=N | 158.0 | ||||||
| 2-OH | 8.15 | broad s |
| Compounds | FVIIa-sTF: EC50, μM * | Thrombin: EC50, μM * | Thrombin/FVIIa-sTF EC50 Ratio |
|---|---|---|---|
| APMSF | 19.07 | 2.10 | 0.11 |
| Leupeptin | 13.97 | 18.31 | 1.31 |
| Aeruginosin K139 | ~166 | 0.66 | 0.004 |
| Micropeptin K139 | 10.62 | 26.94 | 2.54 |
| Microviridin B | (-) | 4.58 | NA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anas, A.R.J.; Mori, A.; Tone, M.; Naruse, C.; Nakajima, A.; Asukabe, H.; Takaya, Y.; Imanishi, S.Y.; Nishizawa, T.; Shirai, M.; et al. FVIIa-sTF and Thrombin Inhibitory Activities of Compounds Isolated from Microcystis aeruginosa K-139. Mar. Drugs 2017, 15, 275. https://doi.org/10.3390/md15090275
Anas ARJ, Mori A, Tone M, Naruse C, Nakajima A, Asukabe H, Takaya Y, Imanishi SY, Nishizawa T, Shirai M, et al. FVIIa-sTF and Thrombin Inhibitory Activities of Compounds Isolated from Microcystis aeruginosa K-139. Marine Drugs. 2017; 15(9):275. https://doi.org/10.3390/md15090275
Chicago/Turabian StyleAnas, Andrea Roxanne J., Akane Mori, Mineka Tone, Chiaki Naruse, Anna Nakajima, Hirohiko Asukabe, Yoshiaki Takaya, Susumu Y. Imanishi, Tomoyasu Nishizawa, Makoto Shirai, and et al. 2017. "FVIIa-sTF and Thrombin Inhibitory Activities of Compounds Isolated from Microcystis aeruginosa K-139" Marine Drugs 15, no. 9: 275. https://doi.org/10.3390/md15090275





