Acetylated Triterpene Glycosides and Their Biological Activity from Holothuroidea Reported in the Past Six Decades
Abstract
:1. Introduction
2. Sea Cucumbers Triterpene Glycosides as Chemotaxonomic Markers
3. The Structural Features of Holothurian Triterpene Glycosides
Aglycones
4. Distribution of Saponina
5. Acetylated Triterpene Glycosides
6. Sulfated-Acetylated Compounds
7. Non-Holostane Acetylated Saponins
8. Acetylated Saponins Having an Uncommon Structure
9. Immunomodulatory Properties of Acetylated Saponins
10. Cytotoxicity and Anticancer Activity of Saponins
11. Antifungal Activity
12. Molecular Effect of Saponins on Membranes
13. Chemical Nomenclature
14. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kiew, P.L.; Don, M.M. Jewel of the seabed: Sea cucumbers as nutritional and drug candidates. Int. J. Food Sci. Nutr. 2012, 63, 616–636. [Google Scholar] [CrossRef] [PubMed]
- Janakiram, N.B.; Mohammed, A.; Zhang, Y.; Choi, C.-I.; Woodward, C.; Collin, P.; Steele, V.E.; Rao, C.V. Chemopreventive effects of Frondanol A5, a Cucumaria frondosa extract, against rat colon carcinogenesis and inhibition of human colon cancer cell growth. Cancer Prev. Res. Phila. 2010, 3, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, H.; Yuan, W.; Gong, W.; Tang, H.; Liu, B.; Krohn, K.; Li, L.; Yi, Y.; Zhang, W. Antifungal nortriterpene and triterpene glycosides from the sea cucumber Apostichopus japonicus Selenka. Food Chem. 2012, 132, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.W.; Lovatelli, A.; Vasconcellos, M.; Ye, Y. Managing Sea Cucumber Fisheries with an Ecosystem Approach; FAO Fisheries and Aquaculture Technical Paper No 520; FAO: Rome, Italy, 2010. [Google Scholar]
- Kim, C.G.; Kwak, J.-Y. Anti-cancer effects of triterpene glycosides, frondoside A and cucumarioside A2-2 isolated from sea cucumbers. In Handbook of Anticancer Drugs from Marine Origin; Kim, S.-K., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 673–682. [Google Scholar]
- Zhao, Q.; Xue, Y.; Liu, Z.D.; Li, H.; Wang, J.F.; Li, Z.J.; Wang, Y.M.; Dong, P.; Xue, C.H. Differential effects of sulfated triterpene glycosides, holothurin A1, and 24-dehydroechinoside A, on antimetastasic activity via regulation of the MMP-9 signal pathway. J. Food Sci. 2010, 75, H280–H288. [Google Scholar] [CrossRef] [PubMed]
- Al Marzouqi, N.; Iratni, R.; Nemmar, A.; Arafat, K.; Ahmed Al Sultan, M.; Yasin, J.; Collin, P.; Mester, J.; Adrian, T.E.; Attoub, S. Frondoside A inhibits human breast cancer cell survival, migration, invasion and the growth of breast tumor xenografts. Eur. J. Pharmacol. 2011, 668, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Kundu, N.; Collin, P.D.; Goloubeva, O.; Fulton, A.M. Frondoside A inhibits breast cancer metastasis and antagonizes prostaglandin E receptors EP4 and EP2. Breast Cancer Res. Treat. 2012, 132, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Arafat, K.; Gélaude, A.; Al Sultan, M.A.; Bracke, M.; Collin, P.; Takahashi, T.; Adrian, T.E.; de Wever, O. Frondoside A suppressive effects on lung cancer survival, tumor growth, angiogenesis, invasion, and metastasis. PLoS ONE 2013, 8, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aminin, D.L.; Koy, C.; Dmitrenok, P.S.; Muller-Hilke, B.; Koczan, D.; Arbogast, B.; Silchenko, A.A.; Kalinin, V.I.; Avilov, S.A.; Stonik, V.A.; et al. Immunomodulatory effects of holothurian triterpene glycosides on mammalian splenocytes determined by mass spectrometric proteome analysis. J. Proteom. 2009, 72, 886–906. [Google Scholar] [CrossRef] [PubMed]
- Gorshkova, I.A.; Kalinin, V.I.; Gorshkov, B.A.; Stonik, V.A. Two different modes of inhibition of the rat brain Na+,K+-ATPase by triterpene glycosides, psolusosides A and B from the holothurian Psolus fabricii. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1999, 122, 101–108. [Google Scholar] [CrossRef]
- Jin, J.O.; Shastina, V.V.; Shin, S.W.; Xu, Q.; Park, J.I.; Rasskazov, V.A.; Avilov, S.A.; Fedorov, S.N.; Stonik, V.A.; Kwak, J.Y. Differential effects of triterpene glycosides, frondoside A and cucumarioside A2-2 isolated from sea cucumbers on caspase activation and apoptosis of human leukemia cells. FEBS Lett. 2009, 583, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Roginsky, A.B.; Ding, X.Z.; Woodward, C.; Collin, P.; Newman, R.A.; Bell, R.H., Jr.; Adrian, T.E. Review of the apoptosis pathways in pancreatic cancer and the anti-apoptotic effects of the novel sea cucumber compound, frondoside A. Ann. N. Y. Acad. Sci. 2008, 1138, 181–198. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.H.; Park, E.S.; Shin, S.W.; Na, Y.W.; Han, J.Y.; Jeong, J.S.; Shastina, V.V.; Stonik, V.A.; Park, J.I.; Kwak, J.Y. Stichoposide C induces apoptosis through the generation of ceramide in leukemia and colorectal cancer cells and shows in vivo antitumor activity. Clin. Cancer Res. 2012, 18, 5934–5948. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Smirnov, A.V. Sea cucumbers triterpene glycosides, the recent progress in structural elucidation and chemotaxonomy. Phytochem. Rev. 2005, 4, 221–236. [Google Scholar] [CrossRef]
- Moraes, G.; Norhcote, P.C.; Kalinin, V.I.; Avilov, S.A.; Silchenko, A.S.; Dmitrenok, P.S.; Stonik, V.A.; Levin, V.S. Structure of the major triterpene glycoside from the sea cucumber Stichopus mollis and evidence to reclassify this species into the new genus Australostichopus. Biochem. Syst. Ecol. 2004, 32, 637–650. [Google Scholar] [CrossRef]
- Antonov, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Anastyuk, S.D.; Dmitrenok, P.S.; Kalinin, V.I.; Taboada, S.; Bosh, A.; Avila, C.; Stonik, V.A. Triterpene glycosides from Antarctic sea cucumbers. 2. Structure of Achlioniceosides A1, A2, and A3 from the sea cucumber Achlionice violaecuspidata (=Rhipidothuria racowitzai). J. Nat. Prod. 2009, 72, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Blunt, J.W.; Buckingham, J.; Munro, M.H.G. Taxonomy and marine natural products research. In Handbook of Marine Natural Products; Fattorusso, E., Gerwick, W.H., Taglialatela-Scafati, O., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 3–54. [Google Scholar]
- Bahrami, Y.; Zhang, W.; Franco, C. Discovery of novel saponins from the viscera of the sea cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 2633–2667. [Google Scholar] [CrossRef] [PubMed]
- Chapagain, B.P.; Wiesman, Z. Metabolite profiling of saponins in Balanites aegyptiaca plant tissues using LC (RI)-ESI/MS and MALDI-TOF/MS. Metabolomics 2008, 4, 357–366. [Google Scholar] [CrossRef]
- Kerr, R.G.; Chen, Z. In vivo and in vitro biosynthesis of saponins in sea cucumbers. J. Nat. Prod. 1995, 58, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Hostettmann, K.; Marston, A. Saponins; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Chaieb, I. Saponins as Insecticides: A Review. Tunis. J. Plant Prot. 2010, 5, 39–50. [Google Scholar]
- Kjellin, M.; Johansson, I. (Eds.) Surfactants from Renewable Resources; Wiley: Stockholm, Sweden, 2010.
- Güçlü-Üstünda, Ö.; Mazza, G. Saponins: Properties, applications and processing. Crit. Rev. Food Sci. Nutr. 2007, 47, 231–258. [Google Scholar] [CrossRef] [PubMed]
- Dang, N.H.; Thanh, N.V.; Kiem, P.V.; Huong, L.M.; Minh, C.V.; Kim, Y.H. Two new triterpene glycosides from the Vietnamese sea cucumber Holothuria scabra. Arch. Pharm. Res. 2007, 30, 1387–1391. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Himaya, S.W. Triterpene glycosides from sea cucumbers and their biological activities. Adv. Food Nutr. Res. 2012, 65, 297–319. [Google Scholar] [PubMed]
- Van Dyck, S.; Gerbaux, P.; Flammang, P. Qualitative and quantitative saponin contents in five sea cucumbers from the Indian Ocean. Mar. Drugs 2010, 8, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Chludil, H.D.; Muniain, C.C.; Seldes, A.M.; Maier, M.S. Cytotoxic and antifungal triterpene glycosides from the Patagonian sea cucumber Hemoiedema spectabilis. J. Nat. Prod. 2002, 65, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Zhang, W.; Yi, Y.H.; Liu, B.S.; Pan, M.X.; Wang, X.H. A novel sulfated holostane glycoside from sea cucumber Holothuria leucospilota. Chem. Biodivers. 2010, 7, 1764–1769. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-L.; Li, L.; Yi, Y.-H.; Sun, P. Philinopsides E and F, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Nat. Prod. Res. 2006, 20, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, Y.; Zhang, W.; Chataway, T.; Franco, C. Structural elucidation of novel saponins in the sea cucumber Holothuria lessoni. Mar. Drugs 2014, 12, 4439–4473. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Antonov, A.S.; Silchenko, A.S.; Kalinin, V.I.; Kalinovsky, A.I.; Dmitrenok, P.S.; Stonik, V.A.; Riguera, R.; Jimenez, C. Triterpene glycosides from the far eastern sea cucumber Cucumaria conicospermium. J. Nat. Prod. 2003, 66, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, T.; Togawa, K.; Higuchi, R.; Komori, T.; Sasaki, T. Structures of four new triterpenoid oligoglycosides: DS-penaustrosides A, B, C, and D from the sea cucumber Pentacta australis. J. Nat. Prod. 1992, 55, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A. New triterpene glycoside from the holothurian Neothyonidium magnum. Chem. Nat. Compd. 1990, 26, 42–45. [Google Scholar] [CrossRef]
- Zurita, M.B.; Ahond, A.; Poupat, C.; Potier, P.; Menou, J.L. Invertébrés marins du lagon néo-calédonien, VII. Étude structurale d’un nouveau saponoside sulfaté extrait de l’holothurie, Neothyonidium magnum. J. Nat. Prod. 1986, 49, 809–813. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A. Structure of psolusoside A the major triterpene glycoside from holothurian Psolus fabricii. Chem. Nat. Compd. 1985, 21, 197–202. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovskii, A.I.; Stonik, V.A.; Dmitrenok, P.S.; Elkin, Y.N. Structure of psolusoside-B, triterpenoid glycoside from Psolus holothurians. Khim. Prir. Soedin. 1989, 3, 361–368. [Google Scholar]
- Silchenko, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Morre, J.; Deinzer, M.L.; Woodward, C.; Collin, P.D. Glycosides from the North Atlantic sea cucumber Cucumaria frondosa V-Structures of five new minor trisulfated triterpene oligoglycosides, frondosides A7-1, A7-2, A7-3, A7-4, and isofrondoside C. Can. J. Chem. 2007, 85, 626–636. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjashchenko, P.V.; Dmitrenok, P.S.; Kalinin, V.I.; Stonik, V.A. 3β-O-Glycosylated 16β-acetoxy-9β-H-lanosta-7,24-diene-3β,18,20β-triol, an intermediate metabolite from the sea cucumber Eupentacta fraudatrix and its biosynthetic significance. Biochem. Syst. Ecol. 2012, 44, 53–60. [Google Scholar] [CrossRef]
- Wu, J.; Yi, Y.H.; Tang, H.F.; Wu, H.M.; Zou, Z.R.; Lin, H.W. Nobilisides A–C, three new triterpene glycosides from the sea cucumber Holothuria nobilis. Planta Med. 2006, 72, 932–935. [Google Scholar] [CrossRef] [PubMed]
- Drozdova, O.A.; Avilov, S.S.; Kalinovskii, A.I.; Stonik, V.A. A new acetylated glycoside from the holothurian Cucumaria japonica. Chem. Nat. Compd. 1992, 28, 518–519. [Google Scholar] [CrossRef]
- Bahrami, Y.; Franco, M.M.C. Structure elucidation of new acetylated saponins, Lessoniosides A, B, C, D, and E, and non-acetylated saponins, Lessoniosides F and G, from the viscera of the sea cucumber Holothuria lessoni. Mar. Drugs 2015, 13, 597–617. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, Y. Discovery of Novel Saponins as Potential Future Drugs from Sea Cucumber Viscera. Ph.D. Thesis, Flinders University, Adelaide, Australia, 2015. [Google Scholar]
- Demeyer, M.; De Winter, J.; Caulier, G.; Eeckhaut, I.; Flammang, P.; Gerbaux, P. Molecular diversity and body distribution of saponins in the sea star Asterias rubens by mass spectrometry. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2014, 168, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjashchenko, P.V.; Fedorov, S.N.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I.; Rogacheva, A.V.; Gebruk, A.V. Kolgaosides A and B, two new triterpene glycosides from the Arctic deep water sea cucumber Kolga hyalina (Elasipodida: Elpidiidae). Nat. Prod. Commun. 2014, 9, 1259–1264. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Kalinin, V.I.; Stonik, V.A. Structure and biological action of Cladolosides B1, B2, C, C1, C2 and D, six new triterpene glycosides from the sea cucumber Cladolabes schmeltzii. Nat. Prod. Commun. 2013, 8, 1527–1534. [Google Scholar] [PubMed]
- Kumar, R.; Chaturvedi, A.K.; Shukla, P.K.; Lakshmi, V. Antifungal activity in triterpene glycosides from the sea cucumber Actinopyga lecanora. Bioorg. Med. Chem. Lett. 2007, 17, 4387–4391. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.S.; Yi, Y.H.; Li, L.; Zhang, S.L.; Han, H.; Weng, Y.Y.; Pan, M.X. Arguside A: A new cytotoxic triterpene glycoside from the sea cucumber Bohadschia argus Jaeger. Chem. Biodivers. 2007, 4, 2845–2851. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.S.; Yi, Y.H.; Li, L.; Sun, P.; Han, H.; Sun, G.Q.; Wang, X.H.; Wang, Z.L. Argusides D and E, two new cytotoxic triterpene glycosides from the sea cucumber Bohadschia argus Jaeger. Chem. Biodivers. 2008, 5, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Elbandy, M.; Rho, J.; Afifi, R. Analysis of saponins as bioactive zoochemicals from the marine functional food sea cucumber Bohadschia cousteaui. Eur. Food Res. Technol. 2014, 238, 937–955. [Google Scholar] [CrossRef]
- Yuan, W.H.; Yi, Y.H.; Tang, H.F.; Liu, B.S.; Wang, Z.L.; Sun, G.Q.; Zhang, W.; Li, L.; Sun, P. Antifungal triterpene glycosides from the sea cucumber Bohadschia marmorata. Planta Med. 2009, 75, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.H.; Yi, Y.H.; Li, L.; Liu, B.S.; Zhang, H.W.; Sun, P. Two triterpene glycosides from the sea cucumber Bohadschia marmorata Jaeger. Chin. Chem. Lett. 2008, 19, 457–460. [Google Scholar] [CrossRef]
- Yuan, W.H.; Yi, Y.H.; Tan, R.X.; Wang, Z.L.; Sun, G.Q.; Xue, M.; Zhang, H.W.; Tang, H.F. Antifungal triterpene glycosides from the sea cucumber Holothuria (Microthele) axiloga. Planta Med. 2009, 75, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.; Castro, R.; Riguera, R. Holothurinosides: New antitumour non sulphated triterpenoid glycosides from the sea cucumber Holothuria forskalii. Tetrahedron 1991, 47, 4753–4762. [Google Scholar] [CrossRef]
- Zhang, S.-Y.; Yi, Y.-H.; Tang, H.-F. Bioactive triterpene glycosides from the sea cucumber Holothuria fuscocinerea. J. Nat. Prod. 2006, 69, 1492–1495. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yi, Y.H.; Tang, H.F.; Wu, H.M.; Zhou, Z.R. Hillasides A and B, two new cytotoxic triterpene glycosides from the sea cucumber Holothuria hilla Lesson. J. Asian Nat. Prod. Res. 2007, 9, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Chludil, H.D.; Murray, A.P.; Seldes, A.M.; Maier, M.S. Biologically active triterpene glycosides from sea cucumbers (Holothuroidea, Echinodermata). In Bioactive Natural Products; Atta-ur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 28, pp. 587–615. [Google Scholar]
- Kitagawa, I.; Kobayashi, M.; Son, B.W.; Suzuki, S.; Kyogoku, Y. Marine natural products. XIX: Pervicosides A, B, and C, lanostane-type triterpene-oligoglycoside sulfates from the sea cucumber Holothuria pervicax. Chem. Pharm. Bull. 1989, 37, 1230–1234. [Google Scholar] [CrossRef]
- Stonik, V.A.; Kalinin, V.I.; Avilov, S.A. Toxins from sea cucumbers (holothuroids): Chemical structures, properties, taxonomic distribution, biosynthesis and evolution. J. Nat. Toxins 1999, 8, 235–248. [Google Scholar] [PubMed]
- Elyakov, G.B.; Kuznetsova, T.A.; Stonik, V.A.; Levin, V.S.; Albores, R. Glycosides of marine invertebrates. IV. A comparative study of the glycosides from Cuban sublittoral holothurians. Comp. Biochem. Physiol. B Comp. Biochem. 1975, 52, 413–417. [Google Scholar] [CrossRef]
- Stonik, V.A.; Mal’tsev, I.I.; Kalinovskii, A.I.; Conde, C.; Elyakov, G.B. Glycosides of marine invertebrates. XI. Two new triterpene glycosides from holothurians of the family Stichopadidae. Chem. Nat. Compd. 1982, 18, 177–182. [Google Scholar] [CrossRef]
- Kitagawa, I.; Kobayashi, M.; Imamoto, T.; Yasuzawa, T.; Kyogoku, Y. The structures of six antifungal oligoglycosides, stichlorosides A1, A2, B1, B2, C1 and C2, from the sea cucumber Stichopus chloronotus Brandt. Chem. Pharm. Bull. 1981, 29, 2387–2391. [Google Scholar] [CrossRef]
- Maltsev, I.I.; Stonik, V.A.; Kalinovsky, A.I. Stichoposide-E—A new triterpene glycoside from holoturian stichopodeidae family. Chem. Nat. Compd. 1983, 19, 308–312. [Google Scholar]
- Kobayashi, M.; Hori, M.; Kan, K.; Yasuzawa, T.; Matsui, M.; Suzuki, S.; Kitagawa, I. Marine natural products. XXVII: Distribution of lanostane-type triterpene oligoglycosides in ten kinds of Okinawan Sea cucumbers. Chem. Pharm. Bull. 1991, 39, 2282–2287. [Google Scholar] [CrossRef]
- Stonik, V.A.; Mal’tsev, I.I.; Kalinovskii, A.I.; Elyakov, G.B. Glycosides of marine invertebrates. XII. Structure of a new triterpene oligoglycoside from holothurians of family Stichopodidae. Chem. Nat. Compd. 1982, 18, 182–186. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Anisimov, M.M.; Prokofieva, N.G.; Avilov, S.A.; Afiyatullov, S.S.; Stonik, V.A. Biological activities and biological role of triterpene glycosides from holothuroids (Echinodermata). Echinoderm Stud. 1996, 5, 139–181. [Google Scholar]
- Kelecom, A.; Daloze, D.; Tursch, B. Chemical studies of marine invertebrates—XX: The structures of the genuine aglycones of thelothurins A and B, defensive saponins of the Indo-pacific sea cucumber Thelonota ananas Jaeger (Echinodermata). Tetrahedron 1976, 32, 2313–2319. [Google Scholar] [CrossRef]
- Kelecom, A.; Daloze, D.; Tursch, B. Chemical studies of marine invertebrates—XXI: Six triterpene genins artifacts from thelothurins A and B, toxic saponins of the sea cucumber Thelonota ananas Jaeger (echinodermata). Biosynthesis of the thelothurins. Tetrahedron 1976, 32, 2353–2359. [Google Scholar] [CrossRef]
- Kelecom, A.; Tursch, B.; Vanhaelen, M. Chemical studies of marine invertebrates XIX. Glycosidic chain structure of thelothurins A and B, two new saponins from the Indo-pacific sea cucumber Thelonota ananas Jaeger (Echinodermata). Bull. Soc. Chim. Belg. 1976, 85, 277–292. [Google Scholar] [CrossRef]
- Stonik, V.A.; Mal’tsev, I.I.; Elyakov, G.B. The structure of thelenotosides A and B from the holothurian Thelenota ananas. Chem. Nat. Compd. 1982, 18, 590–593. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Avilov, S.A.; Antonov, A.A.; Kalinin, V.I.; Kalinovsky, A.I.; Smirnov, A.V.; Riguera, R.; Jimenez, C. Triterpene glycosides from the deep-water north-pacific sea cucumber Synallactes nozawai Mitsukuri. J. Nat. Prod. 2002, 65, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I.; Jayasandhya, P.; Rajan, G.C.; Padmakumar, K.P. Structures and biological activities of Typicosides A1, A2, B1, C1 and C2, triterpene glycosides from the sea cucumber Actinocucumis typica. Nat. Prod. Commun. 2013, 8, 301–310. [Google Scholar] [PubMed]
- Rodriguez, J.; Riguera, R. Lefevreiosides: Four new triterpene glycosides from the sea cucumber Cucumaria lefevrei. J. Chem. Res. 1989, 11, 342–343. [Google Scholar]
- Yayli, N.; Findlay, J.A. A triterpenoid saponin from Cucumaria frondosa. Phytochemistry 1999, 50, 135–138. [Google Scholar] [CrossRef]
- Girard, M.; Bélanger, J.; ApSimon, J.W.; Garneau, F.X.; Harvey, C.; Brisson, J.R. Frondoside A. A novel triterpene glycoside from the holothurian Cucumaria frondosa. Can. J. Chem. 1990, 68, 11–18. [Google Scholar] [CrossRef]
- Avilov, S.A.; Kalinin, V.I.; Drozdova, O.A.; Kalinovsky, A.I. Triterpene glycosides from the holothurian Cucumaria frondosa. Chem. Nat. Compd. 1993, 29, 216–218. [Google Scholar] [CrossRef]
- Yayli, N. Minor saponins from the sea cucumber Cucumaria frondosa. Indian J. Chem. Sect. B Org. Chem. Incl. Med. Chem. 2001, 40, 399–404. [Google Scholar]
- Avilov, S.A.; Drozdova, O.A.; Kalinin, V.I.; Kalinovsky, A.I.; Stonik, V.A.; Gudimova, E.N.; Riguera, R.; Jimenez, C. Frondoside C, a new nonholostane triterpene glycoside from the sea cucumber Cucumaria frondosa: Structure and cytotoxicity of its desulfated derivative. Can. J. Chem. 1998, 76, 137–141. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Avilov, S.A.; Antonov, A.S.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Woodward, C.; Collin, P.D. Glycosides from the sea cucumber Cucumaria frondosa. IV. Structure of frondosides A2-4, A2-7, and A2-8, three new minor monosulfated triterpene glycosides. Can. J. Chem. Rev. Can. Chim. 2005, 83, 2120–2126. [Google Scholar] [CrossRef]
- Avilov, S.A.; Kalinin, V.I.; Smirnov, A.V. Use of triterpene glycosides for resolving taxonomic problems in the sea cucumber genus Cucumaria (Holothurioidea, Echinodermata). Biochem. Syst. Ecol. 2004, 32, 715–733. [Google Scholar] [CrossRef]
- Drozdova, O.A.; Avilov, S.A.; Kalinovsky, A.I.; Stonik, V.A. A minor glycoside from the sea cucumber Cucumaria japonica. Khim. Prir. Soedin. 1992, 5, 593. [Google Scholar]
- Drozdova, O.A.; Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A.; Mil’grom, Y.M.; Rashkes, Y.V. New glycosides from the holothurian Cucumaria japonica. Chem. Nat. Compd. 1993, 29, 200–205. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Kalinovsky, A.I.; Dmitrenok, P.S.; Fedorov, S.N.; Stepanov, V.G.; Dong, Z.; Stonik, V.A. Constituents of the sea cucumber Cucumaria okhotensis. Structures of okhotosides B1-B3 and cytotoxic activities of some glycosides from this species. J. Nat. Prod. 2008, 71, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Pislyagin, E.A.; Menchinskaya, E.S.; Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I. Immunomodulatory and anticancer activity of sea cucumber triterpene glycosides. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2014; Volume 41, pp. 75–94. [Google Scholar]
- Aminin, D.L.; Silchenko, A.S.; Avilov, S.A.; Stepanov, V.G.; Kalinin, V.I. Immunomodulatory action of monosulfated triterpene glycosides from the sea cucumber Cucumaria okhotensis: Stimulation of activity of mouse peritoneal macrophages. Nat. Prod. Commun. 2010, 5, 1877–1880. [Google Scholar] [PubMed]
- Silchenko, A.S.; Avilov, S.A.; Kalinin, V.I.; Stonik, V.A.; Kalinovskii, A.I.; Dmitrenok, P.S.; Stepanov, V.G. Monosulfated triterpene glycosides from Cucumaria okhotensis Levin et Stepanov, a new species of sea cucumbers from sea of Okhotsk. Russ. J. Bioorgan. Chem. 2007, 33, 81–90. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Kalinovskii, A.I.; Afiyatullov, S.S. Triterpene glycosides of the holothurian Eupentacta pseudoquinquisemita. Chem. Nat. Compd. 1988, 24, 187–189. [Google Scholar] [CrossRef]
- Zou, Z.; Yi, Y.; Wu, H.; Wu, J.; Liaw, C.-C.; Lee, K.-H. Intercedensides A−C, three new cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod. 2003, 66, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Maier, M.S. Biological activities of sulfated glycosides from echinoderms. In Bioactive Natural Products; Atta-ur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; Volume 35, pp. 311–354. [Google Scholar]
- Zou, Z.; Yi, Y.; Wu, H.; Yao, X.; Du, L.; Jiuhong, W.; Liaw, C.-C.; Lee, K.-H. Intercedensides D−I, cytotoxic triterpene glycosides from the sea cucumber Mensamaria intercedens Lampert. J. Nat. Prod. 2005, 68, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, V.I.; Aminin, D.L.; Avilov, S.A.; Silchenko, A.S.; Stonik, V.A. Triterpene glycosides from sea cucucmbers (Holothuroidea, Echinodermata). Biological activities and functions. In Bioactive Natural Products; Atta-ur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; Volume 35, pp. 135–196. [Google Scholar]
- Han, H.; Xu, Q.Z.; Tang, H.F.; Yi, Y.H.; Gong, W. Cytotoxic holostane-type triterpene glycosides from the sea cucumber Pentacta quadrangularis. Planta Med. 2010, 76, 1900–1904. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-L.; Li, L.; Yi, Y.-H.; Zou, Z.-R.; Sun, P. Philinopgenin A, B, and C, three new triterpenoid aglycones from the sea cucumber Pentacta quadrangularis. Mar. Drugs 2004, 2, 185–191. [Google Scholar] [CrossRef]
- Tong, Y.; Zhang, X.; Tian, F.; Yi, Y.; Xu, Q.; Li, L.; Tong, L.; Lin, L.; Ding, J. Philinopside A, a novel marine-derived compound possessing dual anti-angiogenic and anti-tumor effects. Int. J. Cancer 2005, 114, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.H.; Xu, Q.Z.; Li, L.; Zhang, S.L.; Wu, H.M.; Ding, J.; Tong, Y.G.; Tan, W.F.; Li, M.H.; Tian, F.; et al. Philinopsides A and B, two new sulfated triterpene glycosides from the sea cucumber Pentacta quadrangularis. Helv. Chim. Acta 2006, 89, 54–63. [Google Scholar] [CrossRef]
- Han, H.; Xu, Q.Z.; Yi, Y.H.; Gong, W.; Jiao, B.H. Two new cytotoxic disulfated holostane glycosides from the sea cucumber Pentacta quadrangularis. Chem. Biodivers. 2010, 7, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-Y.; Tang, H.-F.; Yi, Y.-H. Cytotoxic triterpene glycosides from the sea cucumber Pseudocolochirus violaceus. Fitoterapia 2007, 78, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Kalinin, V.I.; Yurchenko, E.A.; Dautov, S.S. Structures of Violaceusosides C, D, E and G, sulfated triterpene glycosides from the sea cucumber Pseudocolochirus violaceus (Cucumariidae, Dendrochirotida). Nat. Prod. Commun. 2014, 9, 391–399. [Google Scholar] [PubMed]
- Zhang, S.-Y.; Yi, Y.-H.; Tang, H.-F.; Li, L.; Sun, P.; Wu, J. Two new bioactive triterpene glycosides from the sea cucumber Pseudocolochirus violaceus. J. Asian Nat. Prod. Res. 2006, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Maier, M.S.; Roccatagliata, A.J.; Kuriss, A.; Chludil, H.; Seldes, A.M.; Pujol, C.A.; Damonte, E.B. Two new cytotoxic and virucidal trisulfated triterpene glycosides from the Antarctic sea cucumber Staurocucumis liouvillei. J. Nat. Prod. 2001, 64, 732–736. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Dmitrenok, P.S.; Kalinin, V.I.; Taboada, S.; Ballesteros, M.; Avila, C. Triterpene glycosides from Antarctic sea cucumbers III. Structures of liouvillosides A4 and A5, two minor disulphated tetraosides containing 3-O-methylquinovose as terminal monosaccharide units from the sea cucumber Staurocucumis liouvillei (Vaney). Nat. Prod. Res. 2011, 25, 1324–1333. [Google Scholar] [CrossRef] [PubMed]
- Antonov, A.S.; Avilov, S.A.; Kalinovsky, A.I.; Anastyuk, S.D.; Dmitrenok, P.S.; Evtushenko, E.V.; Kalinin, V.I.; Smirnov, A.V.; Taboada, S.; Ballesteros, M.; et al. Triterpene glycosides from antarctic sea cucumbers. 1. Structure of liouvillosides A1, A2, A3, B1, and B2 from the sea cucumber Staurocucumis liouvillei: New procedure for separation of highly polar glycoside fractions and taxonomic revision. J. Nat. Prod. 2008, 71, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Kalinin, V.I.; Yurchenko, E.A.; Dolmatov, I.Y. Colochirosides B1, B2, B3 and C, novel sulfated triterpene glycosides from the sea cucumber Colochirus robustus (Cucumariidae, Dendrochirotida). Nat. Prod. Commun. 2015, 10, 1687–1694. [Google Scholar] [PubMed]
- Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A. Two new triterpene glycosides from the holothurian Duasmodactyla kurilensis. Chem. Nat. Compd. 1991, 27, 188–192. [Google Scholar] [CrossRef]
- Avilov, S.A.; Antonov, A.S.; Drozdova, O.A.; Kalinin, V.I.; Kalinovsky, A.I.; Riguera, R.; Lenis, L.A.; Jimenez, C. Triterpene glycosides from the far Eastern sea cucumber Pentamera calcigera II: Disulfated glycosides. J. Nat. Prod. 2000, 63, 1349–1355. [Google Scholar] [CrossRef] [PubMed]
- Bonnard, I.; Rinehart, K.L. Thyonosides A and B, two new saponins isolated from the holothurian Thyone aurea. Tetrahedron 2004, 60, 2987–2992. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Avilov, S.A.; Kalinina, E.Y.; Korolkova, O.G.; Kalinovsky, A.I.; Stonik, V.A.; Riguera, R.; Jimenez, C. Structure of eximisoside A, a novel triterpene glycoside from the Far-Eastern sea cucumber Psolus eximius. J. Nat. Prod. 1997, 60, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Makarieva, T.N.; Stonik, V.A.; Kapustina, I.I.; Boguslavsky, V.M.; Dmitrenoik, A.S.; Kalinin, V.I.; Cordeiro, M.L.; Djerassi, C. Biosynthetic studies of marine lipids. 42. Biosynthesis of steroid and triterpenoid metabolites in the sea cucumber Eupentacta fraudatrix. Steroids 1993, 58, 508–517. [Google Scholar] [CrossRef]
- Afiyatullov, S.S.; Kalinovsky, A.I.; Stonik, V.A. Structure of cucumariosides C1 and C2—Two new triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Chem. Nat. Compd. 1987, 23, 691–696. [Google Scholar] [CrossRef]
- Kalinin, V.I.; Avilov, S.A.; Kalinovskii, A.I.; Stonik, V.A.; Mil’grom, Y.M.; Rashkes, Y.V. Cucumarioside G4—A new triterpenglycoside from the holothurian Eupentacta fraudatrix. Chem. Nat. Compd. 1992, 28, 600–603. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I. Structure of cucumariosides H5, H6, H7 and H8, triterpene glycosides from the sea cucumber Eupentacta fraudatrix and unprecedented aglycone with 16,22-epoxy-group. Nat. Prod. Commun. 2011, 6, 1075–1082. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological activity of Cucumariosides B1 and B2, two new minor non-sulfated unprecedented triosides. Nat. Prod. Commun. 2012, 7, 1157–1162. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological action of cucumariosides I1, I3, I4, three new minor disulfated pentaosides. Nat. Prod. Commun. 2013, 8, 1053–1058. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and biological action of Cucumariosides A1, A3, A4, A5, A6, A12 and A15, seven new minor non-sulfated tetraosides and unprecedented 25-keto, 27-norholostane aglycone. Nat. Prod. Commun. 2012, 7, 517–525. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Martyyas, E.A.; Kalinin, V.I. Triterpene glycosides from the sea cucumber Eupentacta fraudatrix. Structure and cytotoxic action of Cucumariosides A2, A7, A9, A10, A11, A13 and A14, seven new minor non-sulfated tetraosides and an aglycone with an uncommon 18-hydroxy group. Nat. Prod. Commun. 2012, 7, 845–852. [Google Scholar] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Menchinskaya, E.S.; Aminin, D.L.; Kalinin, V.I. Structure of cucumarioside I2 from the sea cucumber Eupentacta fraudatrix (Djakonov et Baranova) and cytotoxic and immunostimulatory activities of this saponin and relative compounds. Nat. Prod. Res. 2013, 27, 1776–1783. [Google Scholar] [CrossRef] [PubMed]
- Popov, R.S.; Avilov, S.A.; Silchenko, A.S.; Kalinovsky, A.I.; Dmitrenok, P.S.; Grebnev, B.B.; Ivanchina, N.V.; Kalinin, V.I. Cucumariosides F1 and F2, two new triterpene glycosides from the sea cucumber Eupentacta fraudatrix and their LC-ESI MS/MS identification in the starfish Patiria pectinifera, a predator of the sea cucumber. Biochem. Syst. Ecol. 2014, 57, 191–197. [Google Scholar] [CrossRef]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Savchenko, A.M.; Kalinin, V.I. Triterpene glycosides from the sea cucumber cladolabes schmeltzii. II. Structure and biological action of cladolosides A1–A6. Nat. Prod. Commun. 2014, 9, 1421–1428. [Google Scholar]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Dolmatov, I.Y.; Kalinin, V.I. Structures and biological activities of cladolosides C3, E1, E2, F1, F2, G, H1 and H2, eight triterpene glycosides from the sea cucumber Cladolabes schmeltzii with one known and four new carbohydrate chains. Carbohydr. Res. 2015, 414, 22–31. [Google Scholar] [CrossRef]
- Encarnación, D.R.; Murillo, J.I.; Nielsen, J.; Christophersen, C. Neothyoside B, a triterpenoid diglycoside from the Pacific sea cucumber Neothyone gibbosa. Acta Chem. Scand. 1996, 50, 848–849. [Google Scholar] [CrossRef]
- Encarnacion, R.; Carrasco, G.; Espinoza, M.; Anthoni, U.; Nielsen, P.H.; Christophersen, C. Neothyoside A, proposed structure of a triterpenoid tetraglycoside from the Pacific sea-cucumber Neothyone gibbosa. J. Nat. Prod. 1989, 52, 248–251. [Google Scholar] [CrossRef]
- Honey-Escandón, M.; Arreguín-Espinosa, R.; Solís-Marín, F.A.; Samyn, Y. Biological and taxonomic perspective of triterpenoid glycosides of sea cucumbers of the family Holothuriidae (Echinodermata, Holothuroidea). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2015, 180, 16–39. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Agafonova, I.G.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Stonik, V.A.; Collin, P.D.; Woodward, C. Immunomodulatory properties of frondoside A, a major triterpene glycoside from the North Atlantic commercially harvested sea cucumber Cucumaria frondosa. J. Med. Food 2008, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Aminin, D.L.; Agafonova, I.G.; Berdyshev, E.V.; Isachenko, E.G.; Avilov, S.A.; Stonik, V.A. Immunomodulatory properties of cucumariosides from the edible far-eastern holothurian Cucumaria japonica. J. Med. Food 2001, 4, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I. Bioactive marine natural products. Yakugaku Zasshi 1988, 108, 398–416. [Google Scholar] [PubMed]
- Podolak, I.; Galanty, A.; Sobolewska, D. Saponins as cytotoxic agents: A review. Phytochem. Rev. 2010, 9, 425–474. [Google Scholar] [CrossRef] [PubMed]
- Mujoo, K.; Haridas, V.; Hoffmann, J.J.; Wächter, G.A.; Hutter, L.K.; Lu, Y.; Blake, M.E.; Jayatilake, G.S.; Bailey, D.; Mills, G.B.; et al. Triterpenoid saponins from Acacia victoriae (Bentham) decrease tumor cell proliferation and induce Apoptosis. Cancer Res. 2001, 61, 5486–5490. [Google Scholar] [PubMed]
- Kuroda, M.; Mimaki, Y.; Hasegawa, F.; Yokosuka, A.; Sashida, Y.; Sakagami, H. Steroidal glycosides from the bulbs of Camassia leichtlinii and their cytotoxic activities. Chem. Pharm. Bull. 2001, 49, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Mimaki, Y.; Yokosuka, A.; Kuroda, M.; Sashida, Y. Cytotoxic activities and structure-cytotoxic relationships of steroidal saponins. Biol. Pharm. Bull. 2001, 24, 1286–1289. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Tishchenko, L.Y.; Stonik, V.A. Structure of cucumarioside A2–2A triterpene glycoside from the holothurian Cucumaria japonica. Chem. Nat. Compd. 1984, 20, 759–760. [Google Scholar] [CrossRef]
- Park, J.-I.; Bae, H.-R.; Kim, C.G.; Stonik, V.A.; Kwak, J.-Y. Relationships between chemical structures and functions of triterpene glycosides isolated from sea cucumbers. Front. Chem. 2014, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kim, Y.H.; Kim, Y.; Lee, S.J. Frondoside A has an anti-invasive effect by inhibiting TPA-induced MMP-9 activation via NF-κB and AP-1 signaling in human breast cancer cells. Int. J. Oncol. 2012, 41, 933–940. [Google Scholar] [PubMed]
- Miyamoto, T.; Togawa, K.; Higuchi, R.; Komori, T.; Sasaki, T. Constituents of holothuroidea, II. Six newly identified biologically active triterpenoid glycoside sulfates from the sea cucumber Cucumaria echinata. Liebigs Ann. Chem. 1990, 1990, 453–460. [Google Scholar] [CrossRef]
- Taha, T.A.; Mullen, T.D.; Obeid, L.M. A house divided: Ceramide, sphingosine, and sphingosine-1-phosphate in programmed cell death. Biochim. Biophys. Acta 2006, 1758, 2027–2036. [Google Scholar] [CrossRef] [PubMed]
- Yun, S.H.; Park, E.S.; Shin, S.W.; Ju, M.H.; Han, J.Y.; Jeong, J.S.; Kim, S.H.; Stonik, V.A.; Kwak, J.Y.; Park, J.I. By activating Fas/ceramide synthase 6/p38 kinase in lipid rafts, Stichoposide D inhibits growth of leukemia xenografts. Oncotarget 2015, 6, 27596–27612. [Google Scholar] [CrossRef] [PubMed]
- Careaga, V.P.; Maier, M.S. Cytotoxic triterpene glycosides from sea cucumbers. In Handbook of Anticancer Drugs from Marine Origin; Kim, S.-K., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 515–528. [Google Scholar]
- Wang, J.; Han, H.; Chen, X.; Yi, Y.; Sun, H. Cytotoxic and apoptosis-inducing activity of triterpene glycosides from Holothuria scabra and Cucumaria frondosa against HepG2 Cells. Mar. Drugs 2014, 12, 4274–4290. [Google Scholar] [CrossRef] [PubMed]
- Silchenko, A.S.; Kalinovsky, A.I.; Avilov, S.A.; Andryjaschenko, P.V.; Dmitrenok, P.S.; Yurchenko, E.A.; Kalinin, V.I. Structures and cytotoxic properties of cucumariosides H2, H3 and H4 from the sea cucumber Eupentacta fraudatrix. Nat. Prod. Res. 2012, 26, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Ricci-Vitiani, L.; Pallini, R.; Biffoni, M.; Todaro, M.; Invernici, G.; Cenci, T.; Maira, G.; Parati, E.A.; Stassi, G.; Larocca, L.M.; et al. Tumour vascularization via endothelial differentiation of glioblastoma stem-like cells. Nature 2010, 468, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.R.; Tang, H.F.; Lin, H.W.; Cheng, G.; Wang, S.W.; Zhang, X. Saponins: The potential chemotherapeutic agents in pursuing new anti-glioblastoma drugs. Mini Rev. Med. Chem. 2013, 13, 1709–1724. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Ye, X.; Huang, H.; Peng, R.; Su, Z.; Lian, X.Y.; Zhang, Z. Bioactive sulfated saponins from sea cucumber Holothuria moebii. Planta Med. 2015, 81, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I.; Kobayashi, M.; Inamoto, T.; Yasuzawa, T.; Kyogoku, Y.; Kido, M. Stichlorogenol and dehydrostichlorogenol, genuin aglycones of stichlorosides A1, A2, B1, B2, C1, and C2, from the sea cucumber Stichopus chloronotus (Brandt). Chem. Pharm. Bull. 1981, 29, 1189–1192. [Google Scholar] [CrossRef]
- Wang, Z.; Gong, W.; Sun, G.; Tang, H.; Liu, B.; Li, L.; Yi, Y.; Zhang, W. New holostan-type triterpene glycosides from the sea cucumber Apostichopus japonicus. Nat. Prod. Commun. 2012, 7, 1431–1434. [Google Scholar] [PubMed]
- Wang, X.-H.; Zou, Z.-R.; Yi, Y.-H.; Han, H.; Li, L.; Pan, M.-X. Variegatusides: New non-sulphated triterpene glycosides from the sea cucumber Stichopus variegates Semper. Mar. Drugs 2014, 12, 2004–2018. [Google Scholar] [CrossRef] [PubMed]
- Avilov, S.A.; Silchenko, A.S.; Antonov, A.S.; Kalinin, V.I.; Kalinovsky, A.I.; Smirnov, A.V.; Dmitrenok, P.S.; Evtushenko, E.V.; Fedorov, S.N.; Savina, A.S.; et al. Synaptosides A and A1, triterpene glycosides from the sea cucumber Synapta maculata containing 3-O-methylglucuronic acid and their cytotoxic activity against tumor cells. J. Nat. Prod. 2008, 71, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, I.; Kobayashi, M.; Inamoto, T.; Fuchida, M.; Kyogoku, Y. Marine natural products. XIV. Structures of echinosides A and B, antifungal lanostane-oligosides from the sea cucumber Actinopyga echinites (Jaeger). Chem. Pharm. Bull. 1985, 33, 5214–5224. [Google Scholar] [CrossRef] [PubMed]
- Pislyagin, E.A.; Gladkikh, R.V.; Kapustina, I.I.; Kim, N.Y.; Shevchenko, V.P.; Nagaev, I.Y.; Avilov, S.A.; Aminin, D.L. Interaction of holothurian triterpene glycoside with biomembranes of mouse immune cells. Int. Immunopharmacol. 2012, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, M.M. Triterpene glycosides and the structural-functional properties of membranes. Nauchnye Doki. Vyss. Shkoly Biol. Nauki 1987, 10, 49–63. [Google Scholar] [PubMed]
- Augustin, J.M.; Kuzina, V.; Andersen, S.B.; Bak, S. Molecular activities, biosynthesis and evolution of triterpenoid saponins. Phytochemistry 2011, 72, 435–457. [Google Scholar] [CrossRef] [PubMed]
- Gorshkova, I.A.; Kalinovsky, A.I.; Ilyin, S.G.; Gorshkov, B.A.; Stonik, V.A. Physicochemical characteristics of interaction of toxic triterpene glycosides from holothurians with rat brain Na+-K+-ATPase. Toxicon 1989, 27, 937–945. [Google Scholar] [CrossRef]
- Popov, A.M.; Rovin Iu, G.; Anisimov, M.M.; Likhatskaia, G.N.; Strigina, L.I. Effect of triterpene glycosides on the stability of bilayer lipid membranes, containing different sterols. Biofizika 1982, 27, 827–831. [Google Scholar] [PubMed]
- Aminin, D.L.; Chaykina, E.L.; Agafonova, I.G.; Avilov, S.A.; Kalinin, V.I.; Stonik, V.A. Antitumor activity of the immunomodulatory lead Cumaside. Int. Immunopharmacol. 2010, 10, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Popov, A.M. Comparative study of effects of various sterols and triterpenoids on permeability of model lipid membranes. J. Evol. Biochem. Physiol. 2003, 39, 314–320. [Google Scholar] [CrossRef]
- Menchinskaya, E.S.; Aminin, D.L.; Avilov, S.A.; Silchenko, A.S.; Andryjashchenko, P.V.; Kalinin, V.I.; Stonik, V.A. Inhibition of tumor cells multidrug resistance by cucumarioside A2-2, frondoside A and their complexes with cholesterol. Nat. Prod. Commun. 2013, 8, 1377–1380. [Google Scholar] [PubMed]
- Wink, M.; Ashour, M.L.; El-Readi, M.Z. Secondary metabolites from plants inhibiting ABC transporters and reversing resistance of cancer cells and microbes to cytotoxic and antimicrobial agents. Front. Microbiol. 2012, 3, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Mal’tsev, I.I.; Stekhova, S.I.; Shentsova, E.B.; Anisimov, M.M.; Stonik, V.A. Antimicrobial activity of glycosides from holothurians of the family Stichopodidae. Pharm. Chem. J. 1985, 19, 44–46. [Google Scholar] [CrossRef]
- Yudt, M.R.; Cidlowski, J.A. The glucocorticoid receptor: Coding a diversity of proteins and responses through a single gene. Mol. Endocrinol. 2002, 16, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-F.; Fan, X.-J.; Li, X.; Peng, L.-L.; Wang, G.-H.; Ke, K.-F.; Jiang, Z.-L. Ginsenoside Rg1 protects neurons from hypoxic–ischemic injury possibly by inhibiting Ca2+ influx through NMDA receptors and L-type voltage-dependent Ca2+ channels. Eur. J. Pharmacol. 2008, 586, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.; Yeh, C.-H.; Yeh, M.; Cheng, J.-T. Increase of adipogenesis by ginsenoside (Rh2) in 3T3-L1 cell via an activation of glucocorticoid receptor. Horm. Metab. Res. 2009, 41, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Gorshkova, I.A.; Gorshkov, B.A.; Stonik, V.A. Inhibition of rat brain Na+-K+-ATPase by triterpene glycosides from holothurians. Toxicon 1989, 27, 927–936. [Google Scholar] [CrossRef]
- Zhang, J.J.; Dai, J.B.; Chen, L.L.; Ding, P.Y.; Wu, J. Screening of bioactive constituents from sea cucumber Holothuria nobilis using conidia of Pyricularia oryzae. Zhong Yao Cai 2008, 31, 1001–1003. [Google Scholar] [PubMed]
- Wu, J.; Yi, Y.; Zou, Z. Two new triterpene glycosides from sea cucumber Holothuria nobilis. Chin. Tradit. Herb. Drugs 2006, 37, 497. [Google Scholar]
- Caulier, G.; Van Dyck, S.; Gerbaux, P.; Eeckhaut, I.; Flammang, P. Review of Saponin Diversity in Sea Cucumbers Belonging to the Family Holothuriidae; SPC Beche-de-mer Information Bulletin #31; SPC: Noumea Cedex, New Caledonia, 2011; pp. 48–54. [Google Scholar]
- Zhang, J.J. Extraction, isolation, and structure elucidation of two new triterpene glycosides from sea cucumber Holothuria nobilis. Chin. Tradit. Herb. Drugs 2011, 42, 1467–1472. [Google Scholar]




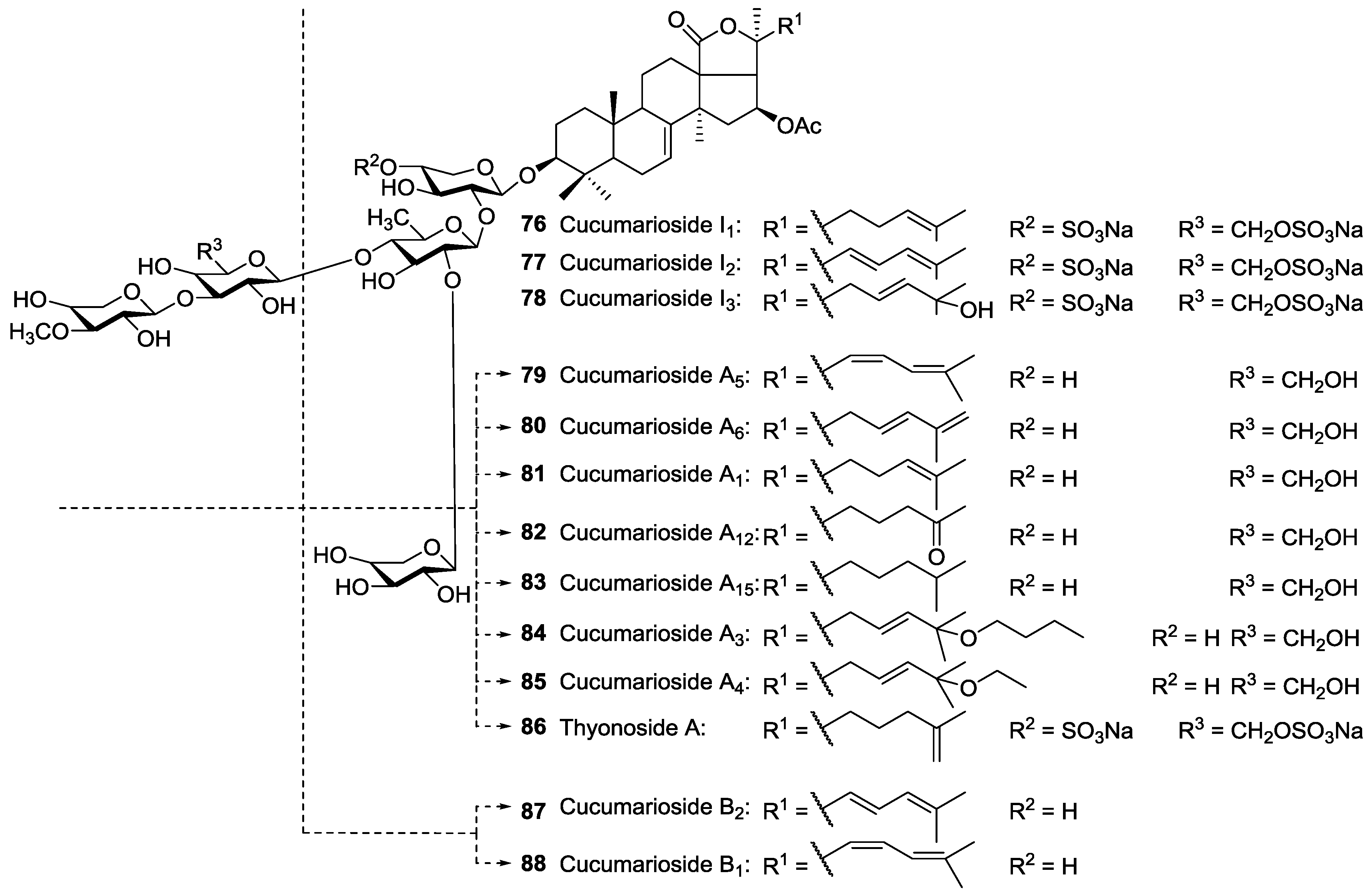
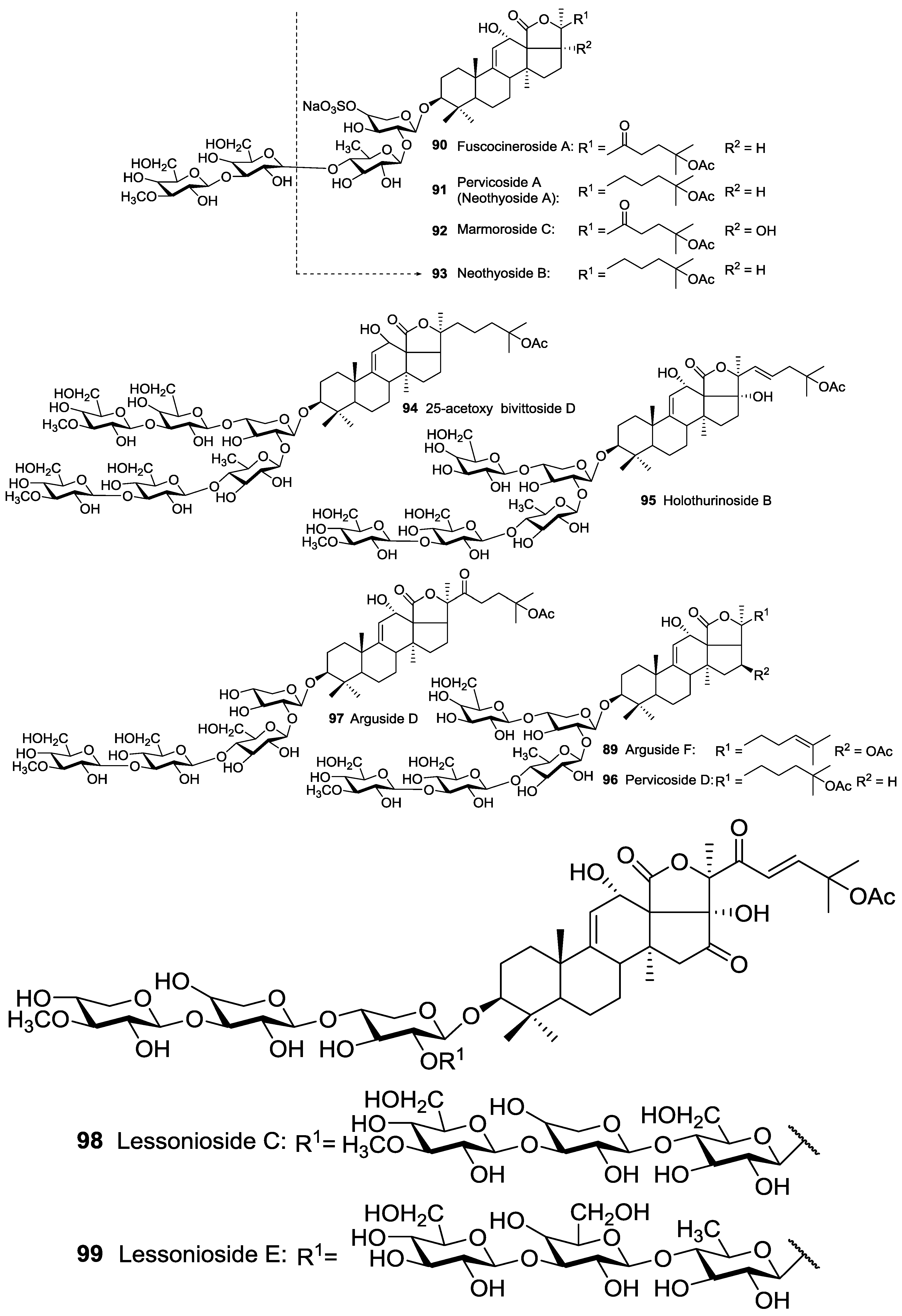

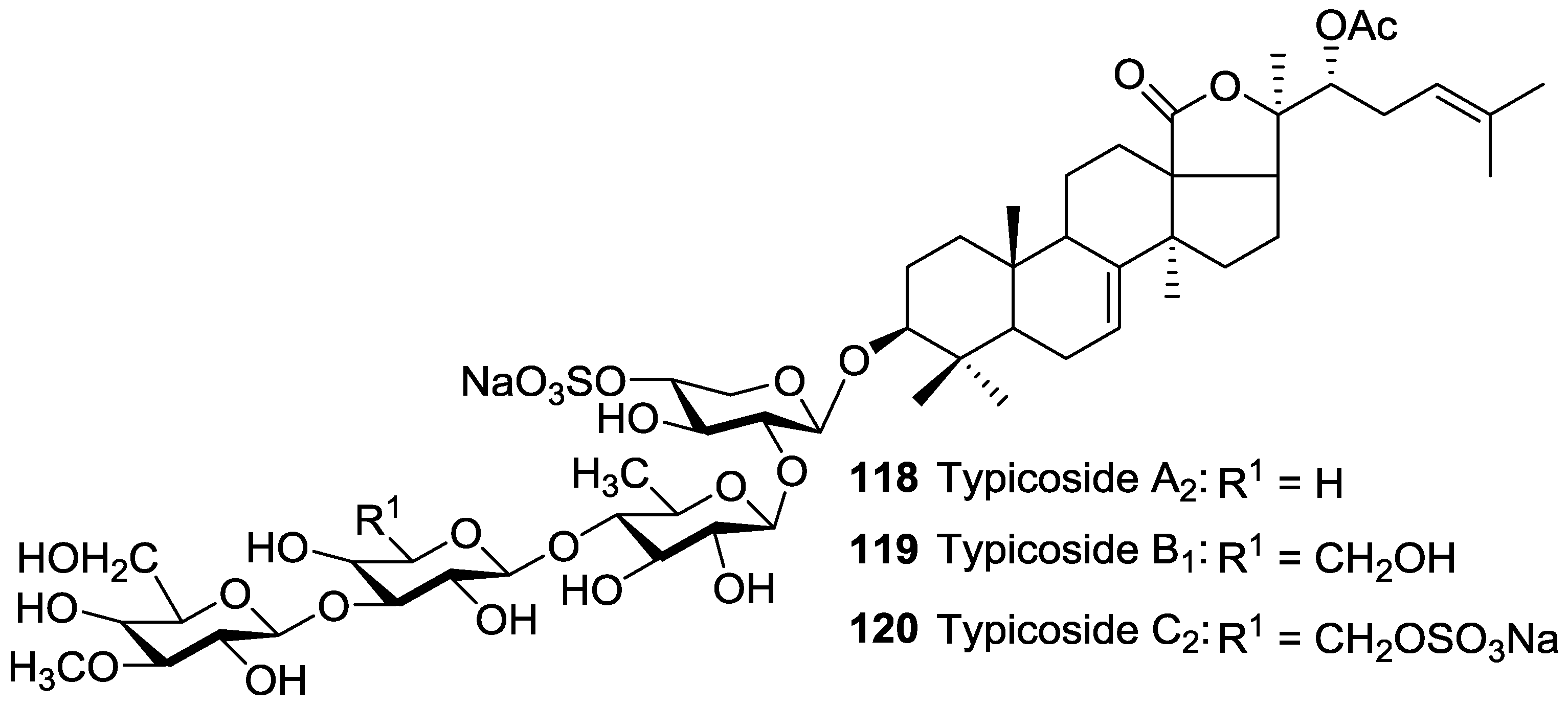
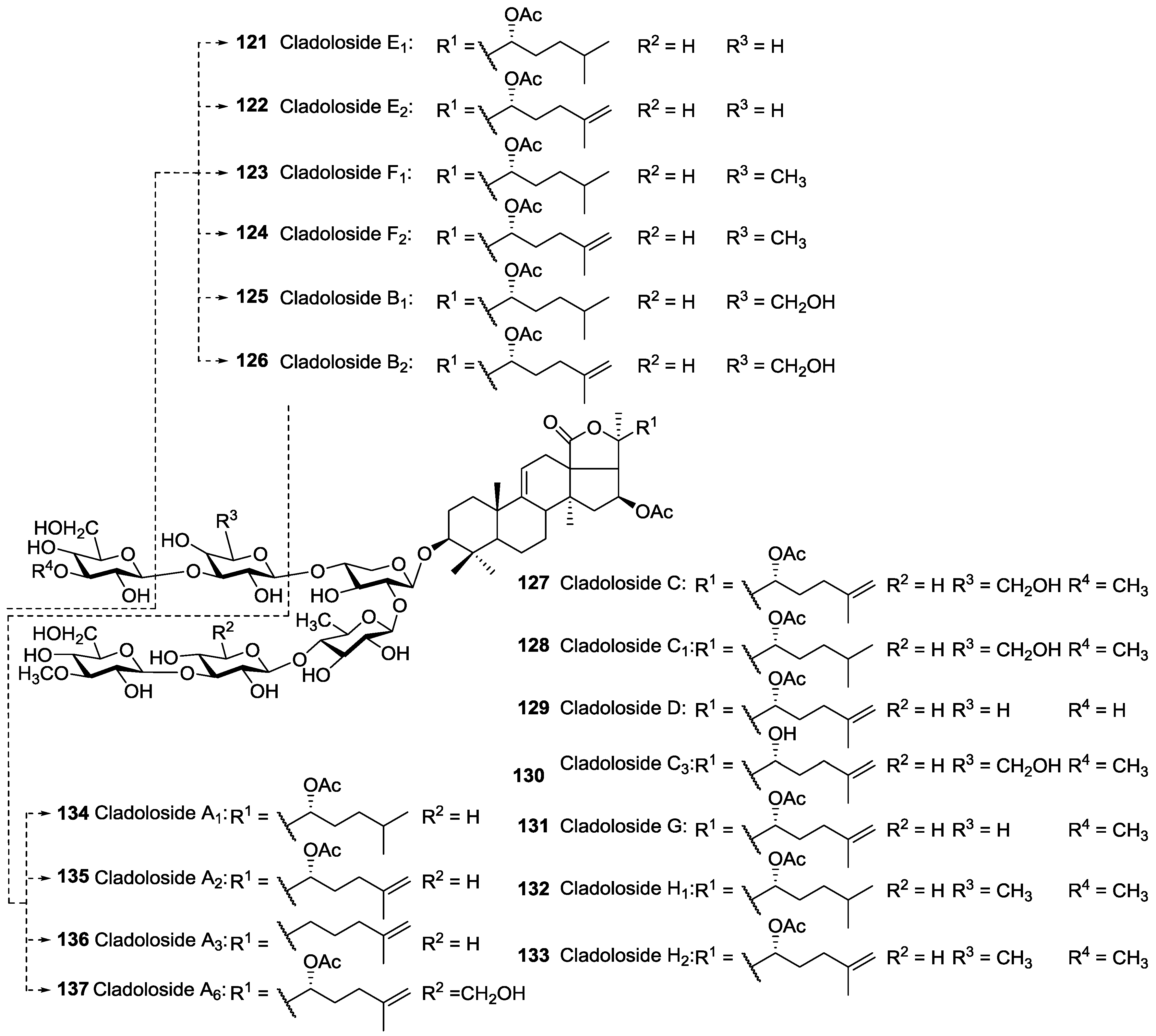

| Order | Family | Taxon | Glycosides (Holothurins) N = Non-holostane | Type of Genins H = Holostane N = Non-holostane | Place (Site) of Collection | References |
|---|---|---|---|---|---|---|
| Aspidochirotida | Holothuridae | Actinopyga lecanora | A novel triterpene glycoside 68 | H | Indian Ocean | [48] |
| Aspidochirotida | Holothuridae | Bohadschia argus | Argusides A 69 and D 97 | H | Sanya Bay, South China Sea | [49,50] |
| Aspidochirotida | Holothuridae | Bohadschia cousteaui | Cousteside D 75 | H | Gulf of Aqaba, Red Sea | [51] |
| Aspidochirotida | Holothuridae | Bohadschia marmorata | 25-acetoxy bivittoside D 94, Marmoroside C 92, Fuscocineroside A 90 | H | Hainan Island, South China Sea | [52,53] |
| Aspidochirotida | Holothuridae | Holothuria (Microthele) axiloga | Arguside F 89, Pervicoside D 96 | H | Hainan Island, South China Sea | [54] |
| Aspidochirotida | Holothuridae | Holothuria forskalii | Holothurinoside B 95 | H | Villagarcia de Arosa, Spain | [55] |
| Aspidochirotida | Holothuridae | Holothuria fuscocinerea | Fuscocineroside A 90 | H | South China Sea | [56] |
| Aspidochirotida | Holothuridae | Holothuria hilla | Hillaside B 70 | H | Dongshan Island, South China Sea | [57] |
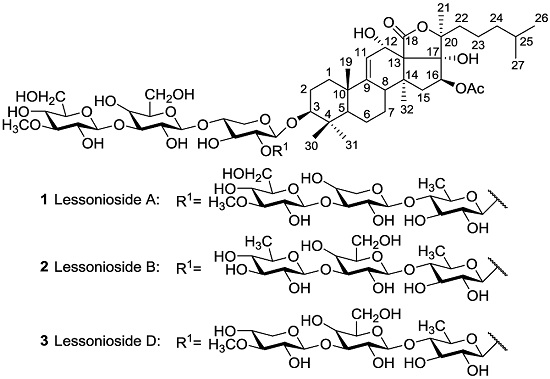
| Aspidochirotida | Holothuridae | Holothuria lessoni | Lessoniosides A 1, B 2, C 98, D 3, and E 99 | H | near Lizard Island, Australia | [43,44] |
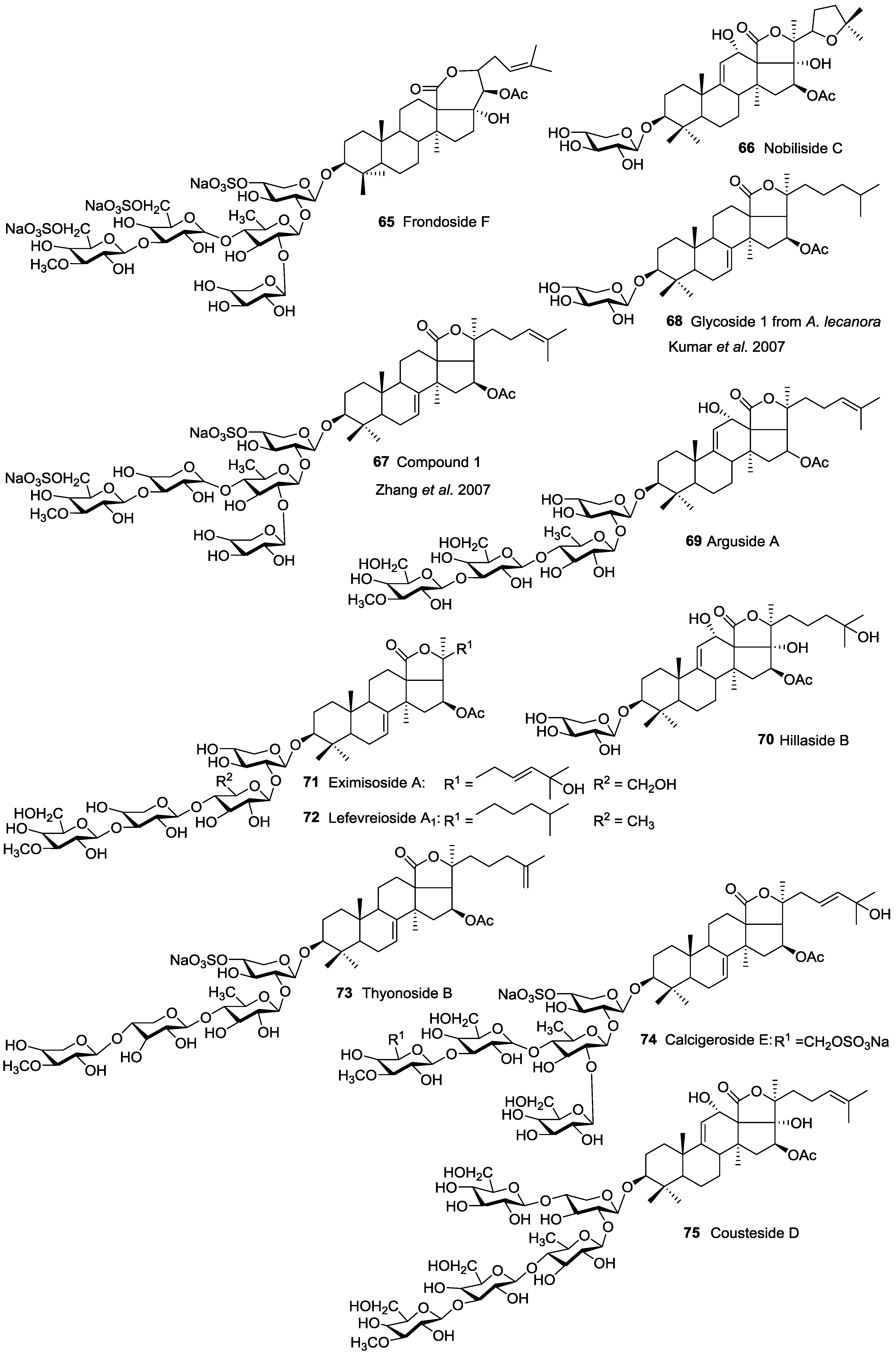
| Aspidochirotida | Holothuridae | Holothuria nobilis | Nobiliside C 66 | H | Dongshan Island, South China Sea | [41] |
| Aspidochirotida | Holothuriidae | Holothuria pervicax | Pervicoside A (Neothyoside A) 91 | H | Kushimoto, Japan | [58,59,60] |
| Aspidochirotida | Stichopodidae | Astichopus multifidus | Stichoposide B 101, Astichoposide C 103 | H | Islas de Pinos, Cuba | [61,62] |
| Aspidochirotida | Stichopodidae | Genera Stichopus | 25(26)-dihydro Stichoposide C (Stichloroside C2) 105, 25(26)-dihydro Stichoposide D (Stichloroside B2) 106, 25(26)-dihydro Stichoposide E (Stichloroside A2) 108, 25(26)-dihydro Stichoposide A 102 | H | - | [60] |
| Aspidochirotida | Stichopodidae | Stichopus chloronotus | Stichoposides A 100, B 101, C (Stichloroside C1) 103, D (Stichloroside B1) 104, E (Stichloroside A1) 107, Stichlorosides A1 107, A2 108, C1 103, C2 105, B1 104 and B2 106, Stichloroside C1 (Stichoposide C) 103, Stichloroside B1 (Stichoposide D) 104, Stichloroside A1 (Stichoposide E) 107 | H | The Great Barrier Reef, Australia; Koetivi Island, Seychelles; Okinawa, Japan | [16,58,62,63,64] |
| Aspidochirotida | Stichopodidae | Stichopus hermanni | Stichoposides C 103, D 104, E 107 and their dehydro derivatives, Stichlorosides A1 107, A2 108, B1 104, B2 106, C1 103 and C2 105 | H | Okinawa, Japan | [65] |
| Aspidochirotida | Stichopodidae | Stichopus mollis (Australostichopus) | Stichoposides A 100, B 101, C 103, D 104 | H | Wellington Harbor, New Zealand | [16] |
| Aspidochirotida | Stichopodidae | Stichopus variegatus | Stichoposides C 103 and D 104, and their 25(26)-dehydro derivatives, Stichlorosides C2 105, B2 106, A2 108 | H | The Great Barrier Reef, Australia | [63,66] |
| Aspidochirotida | Stichopodidae | Thelenota ananas | Stichoposide A 100 and its 25(26)-dehydro derivative 102, Thelenotosides A 109 and B 110, 25(26)-dihydro Thelenotoside A 111, 25(26)-dihydro Thelenotoside B 112, Stichoposide C 103 and its 25(26)-dehydro analog 105, Stichoposides D 104, E 107 and their dehydro derivatives 106, 108, Stichlorosides A1 107, A2 108, B1 104, B2 106, C1 103 and C2 105 | H | Albatross Rocks, Seychelles; Okinawa, Japan | [16,58,65,67,68,69,70,71] |
| Aspidochirotida | Stichopodidae | Thelenota anax | Stichoposides C 103, D 104, E 107, Stichlorosides A1 107, B1 104 and C1 103 | H | Okinawa, Japan | [65] |
| Aspidochirotida | Synallactidae | Synallactes nozawai | Synallactosides A1 115, A2 116, B1 117, B2 113 and C 114 | H | The southern part of the Sea of Japan | [72] |
| Dendrochirotida | Cucumariidae | Actinocucumis typica | Typicosides A1 42, A2 118, B1 119, C2 120, Intercedenside A 53 | H | Vizhinjam coast, Arabian Sea | [73] |
| Dendrochirotida | Cucumariidae | Cucumaria (Aslia) lefevrei | Lefevreiosides A1 72, A2 11 and 49, B 12, C 13 | H | Galicia, Spain | [58,74] |
| Dendrochirotida | Cucumariidae | Cucumaria frondosa | Frondosides A 43, A1 49, NA2-7 138, NA2-8 142, NC 145, D 48, F 65 | H and N | Gulf of St. Lawrence Canada; Kolsky shore, Barents Sea, Iles de Mai, Quebec, Canada | [58,75,76,77,78,79,80] |
| Dendrochirotida | Cucumariidae | Cucumaria japonica | Cucumariosides A0-1 47, A0-2 46, A1-2 146 | H | Gulf of Posiet, Sea of Japan | [42,81,82,83] |
| Dendrochirotida | Cucumariidae | Cucumaria okhotensis | Frondosides A 43 and A1 49, Okhotosides A1-1 51, A2-1 44, B1 50, B2 52, B3 62, Cucumariosides A0-1 47, A2-5 45 | H | Okhotsk Sea near Paramushir Island, | [84,85,86,87] |
| Dendrochirotida | Cucumariidae | Eupentacta pseudoquinquiesemita | Cucumariosides C2 25, H 21 | H | Ushishir, Kurile Islands | [88] |
| Dendrochirotida | Cucumariidae | Mensamaria intercedens | Intercedensides A 53, B 54, C 55, D 56, E 57, F 58, G 59, H 60, I 61 | H | South Chinese Sea | [89,90,91,92] |
| Dendrochirotida | Cucumariidae | Pentacta quadrangularis | Pentactasides I 37, II 38 and III 36, Pentactasides B 63 and C 64, Philinopsides A 39 and B 40 | H | Zhanjiang, near Guangdong, South China Sea | [92,93,94,95,96,97] |
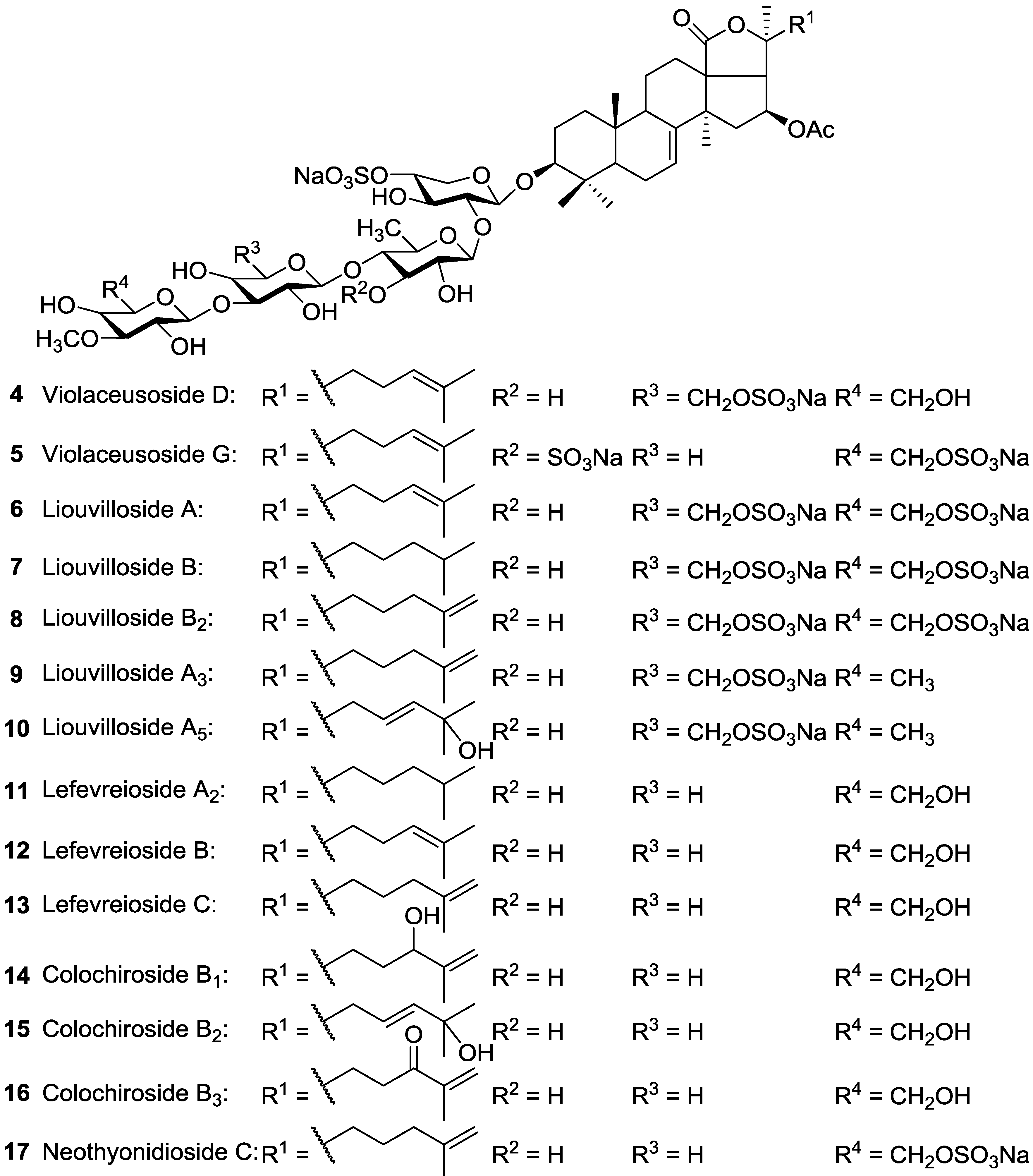
| Dendrochirotida | Cucumariidae | Pseudocolochirus violaceus (=Cucumaria tricolor) | Violaceusides A (Philinopside A) 39, B (Philinopside F) 41, Violaceusosides D 4, G 5, Intercedenside B 54, a new glycoside 67, Liouvilloside A 6, Lefevreioside C 13 | H | Sanya Bay, Nha Trang Gulf, Vietnam, South China Sea | [98,99,100] |
| Dendrochirotida | Cucumariidae | Staurocucumis liouvillei | Liouvillosides A 6, B 7, B2 8, A3 9, A5 10 | H | Island of Bouvet and South Georgia (Antarctic) | [101,102,103] |
| Dendrochirotida | Cucumariidae | Colochirus robustus | Colochirosides B1 14, B2 15, B3 16, Lefevreioside C 13, Violaceusides A 39 and B 41 | H | Nha Trang Gulf, Vietnam | [104] |
| Dendrochirotida | Cucumariidae | Cucumaria conicospermium | Cucumarioside A2-5 45 | H and N | North Western shore, Sea of Japan | [33] |
| Dendrochirotida | Phyllophoridae | Duasmodactyla Kurilensis | Kurilosides A 139 and C 140 | N | Kurile Island, Sea of Okhotsh | [105] |
| Dendrochirotida | Phyllophoridae | Neothyonidium magnum | Neothyonidioside C 17 | H | Shores of south Vietnam | [35,60] |
| Dendrochirotida | Phyllophoridae | Pentamera calcigera | Calcigeroside E 74 | H | Peter-the-Great Gulf, Sea of Japan | [106] |
| Dendrochirotida | Phyllophoridae | Thyone aurea | Thyonosides A 86, B 73 | H | Namibia | [107] |
| Dendrochirotida | Psolidae | Psolus eximius | Eximisoside A 71 | H | Okhotsk Sea near Paramushir Island, Kurile Islands | [108] |
| Dendrochirotida | Psolidae | Psolus fabricii | Psolusoside B 141 | N | Onekotan, Kurile Island | [38] |
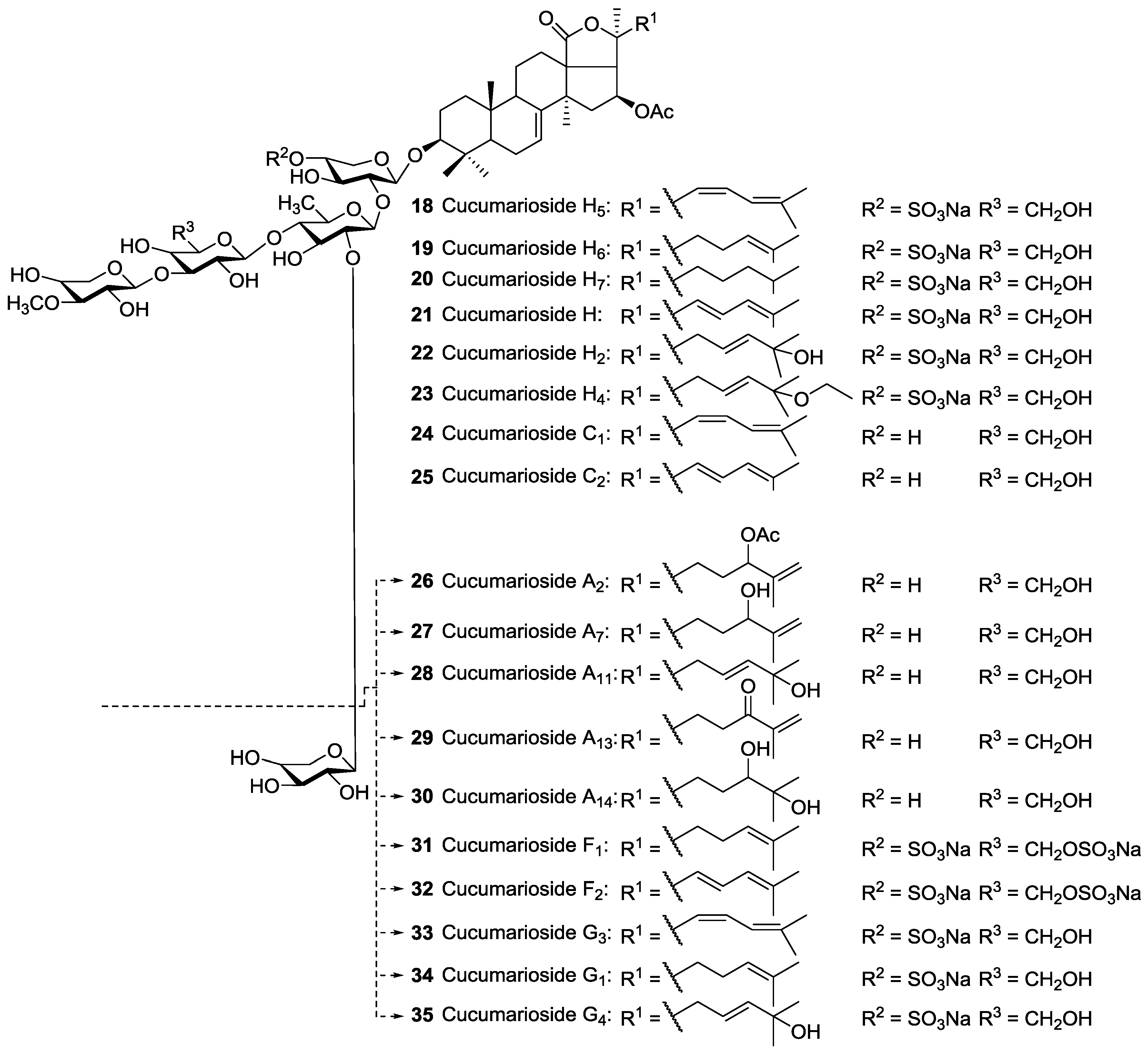
| Dendrochirotida | Sclerodactilidae | Eupentacta fraudatrix (=Cucumaria fraudatrix, =Cucumaria obunca) | Cucumariosides A2 26, A5 79, A6 80, A7 27, NA8 143, NA9 144, A11 28, A13 29, A14 30, B1 88, B2 87, C1 24, C2 25, F1 31, F2 32, G1 34, G3 33, G4 35, H 21, H5 18, H6 19, H7 20, I1 76, I2 77, I3 78 | H and N | Troitsa Bay, Sea of Japan | [40,58,60,109,110,111,112,113,114,115,116,117,118] |
| Dendrochirotida | Sclerodactylidae | Cladolabes schmeltzii | Cladolosides A1 134, A2 135, A3 136, A6 137, B1 125, B2 126, C 127, C1 128, C3 130, D 129, E1 121, E2 122, F1 123, F2 124, G 131, H1 132, H2 133 | H | Nha Trang Gulf, Vietnam | [47,119,120] |
| Dendrochirotida | Sclerodactylidae | Neothyone gibbosa | Neothyosides A 91 and B 93 | H | Esperitu Santo Island, Mexico | [121,122] |
| Elasipodida | Elpidiidae | Kolga hyalina | Holothurinoside B 95 | H | Central Arctic Ocean, Amundsen Basin | [46] |
| Species | Compounds | Mode of Action | References |
|---|---|---|---|
| Psolus fabricii | Psolusosides A and B 141 | Inhibit membrane transporter Na+-K+-ATPase | [11] |
| Cucumaria okhotensis | Frondoside А 43 | Membrane transport P-gp, MDR1 | [155] |
| Stichopus variegatus | Stichoposides C 103 and D 104 | Inhibit membrane transporter Na+-K+-ATPase | [161] |
| Stichopus chloronotus | Stichoposide E 107 | Inhibit membrane transporter Na+-K+-ATPase | [161] |
| Astichopus multifidus | Astichoposide C 103 | Inhibit membrane transporter Na+-K+-ATPase | [161] |
| Thelenota ananas | Thelenotosides A 109 and B 110 | Inhibit membrane transporter Na+-K+-ATPase | [161] |
| Cucumaria fraudatrix | Cucumarioside G1 34 | Inhibit membrane transporter Na+-K+-ATPase | [161] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahrami, Y.; Franco, C.M.M. Acetylated Triterpene Glycosides and Their Biological Activity from Holothuroidea Reported in the Past Six Decades. Mar. Drugs 2016, 14, 147. https://doi.org/10.3390/md14080147
Bahrami Y, Franco CMM. Acetylated Triterpene Glycosides and Their Biological Activity from Holothuroidea Reported in the Past Six Decades. Marine Drugs. 2016; 14(8):147. https://doi.org/10.3390/md14080147
Chicago/Turabian StyleBahrami, Yadollah, and Christopher M. M. Franco. 2016. "Acetylated Triterpene Glycosides and Their Biological Activity from Holothuroidea Reported in the Past Six Decades" Marine Drugs 14, no. 8: 147. https://doi.org/10.3390/md14080147