APO-9′-Fucoxanthinone Extracted from Undariopsis peteseniana Protects Oxidative Stress-Mediated Apoptosis in Cigarette Smoke-Exposed Human Airway Epithelial Cells
Abstract
:1. Introduction
2. Results
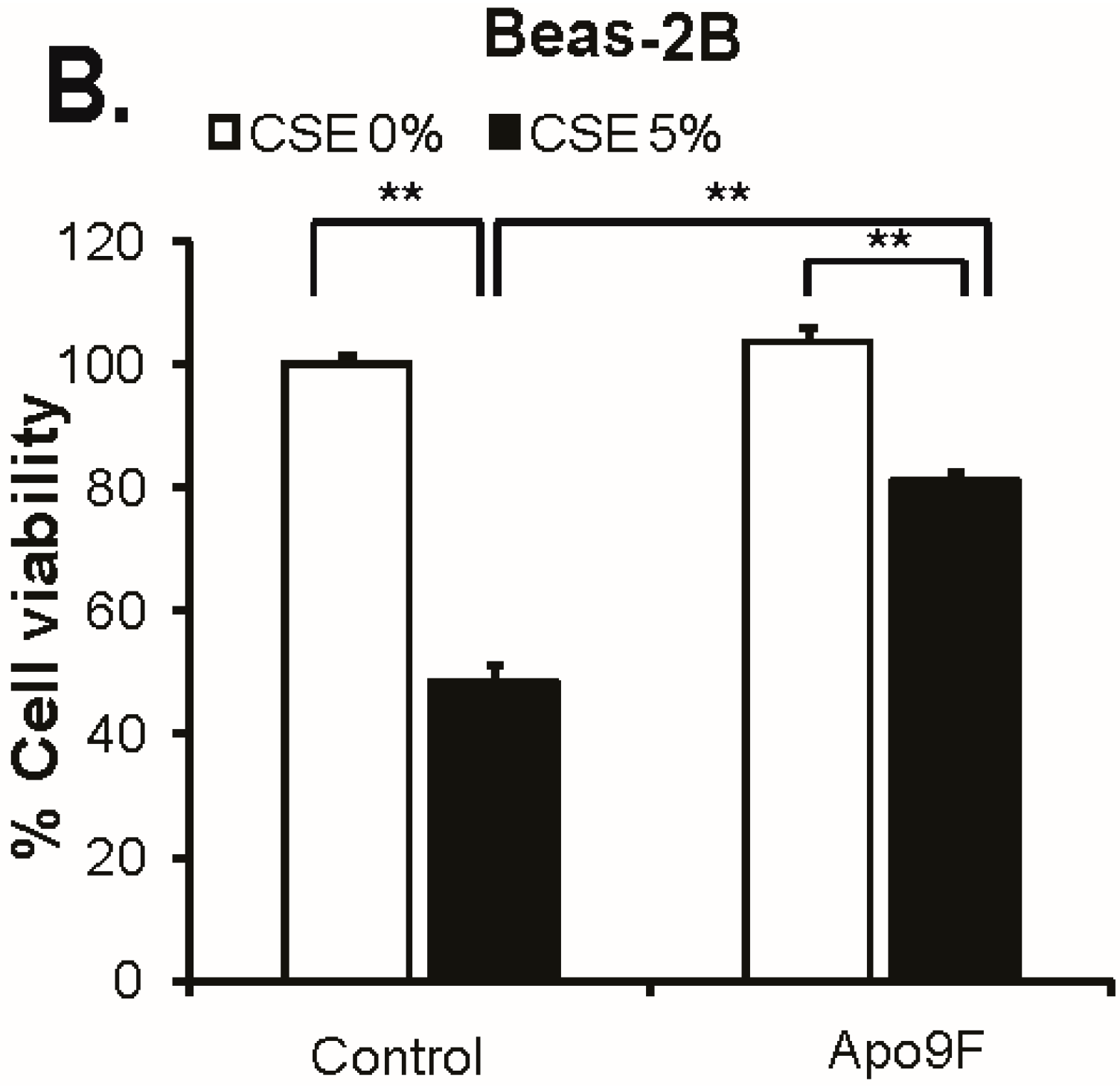
2.1. Apo9F Protects against Cigarette Smoke-Induced Cytotoxicity in Immortalized Human Bronchial Epithelial Cells
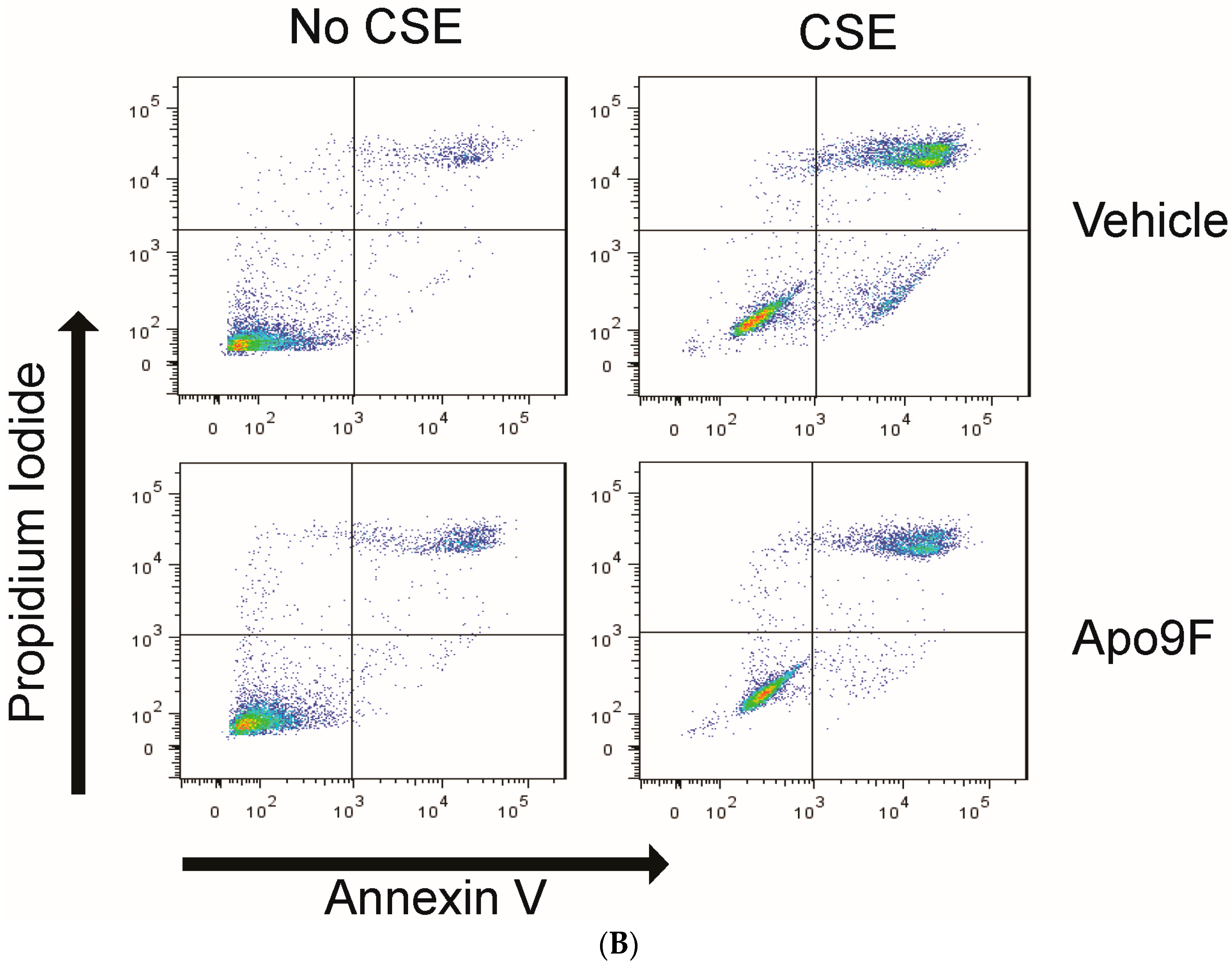
2.2. Apo9F Suppresses Cigarette Smoke-Induced Apoptotic Cell Death in HBEC2 Cells
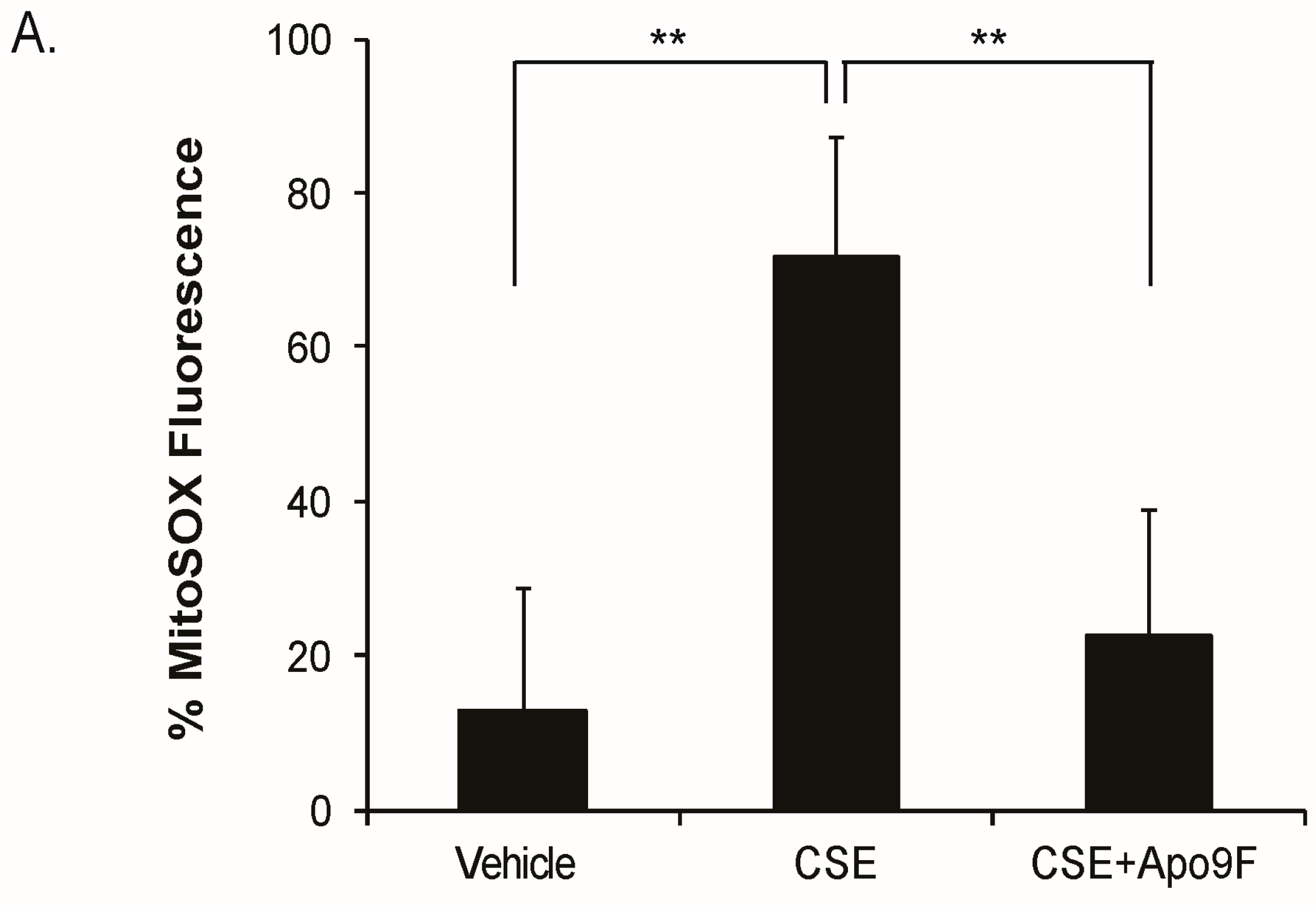
2.3. Apo9F Decreases Mitochondria-Derived ROS Production in Cigarette Smoke-Exposed HBEC2 Cells
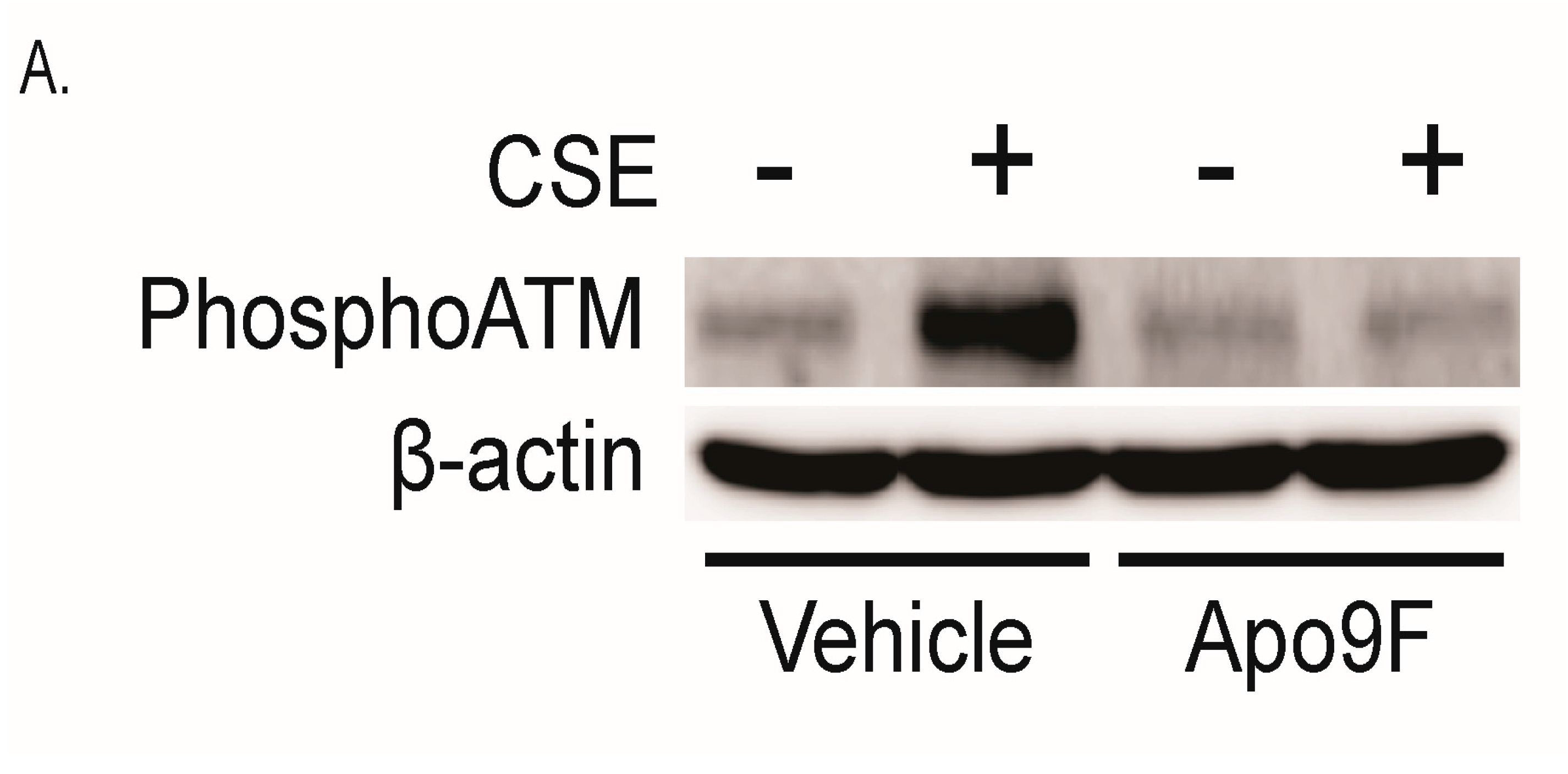
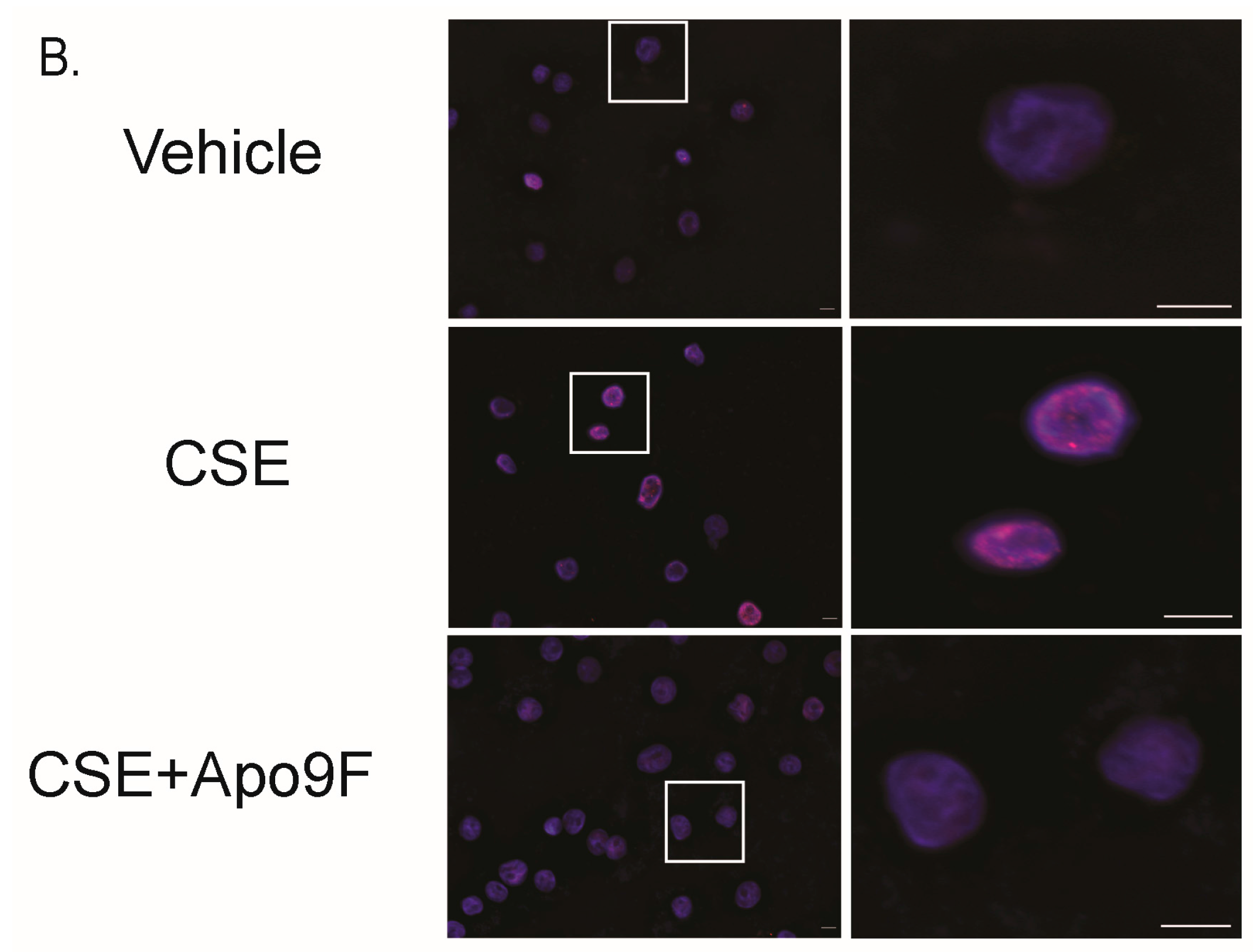
2.4. Apo9F Attenuates Cigarette Smoke-Induced DNA Damage in HBEC2 Cells
3. Discussion
4. Materials and Methods
4.1. Chemicals, Reagents, and Antibodies
4.2. Natural Marine Compounds Isolated from the Brown Algae
4.3. Cigarette Smoke Extract Preparation
4.4. Cell Culture and Cell Viability
4.5. Flow Cytometric Analysis of Apoptotic Cells
4.6. Immunoblot Analysis
4.7. Immunocytofluorescence (ICF)
4.8. Mitochondrial Reactive Oxygen Species
4.9. DNA Damage Comet Assay
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| COPD | chronic obstructive pulmonary disease |
| CS | cigarette smoke |
| CSE | cigarette smoke extract |
| Apo9F | apo-9′-fucoxanthinone |
| RONS | reactive oxygen/nitrogen species |
| HBEC | human bronchial epithelial cells |
| DDR | DNA damage response |
| PIKKs | phosphoinositide 3-kinase related protein kinases |
| ATM | ataxia teleangiectasia mutated |
| DSB | DNA double-strand break |
| DK | dieckol |
| PFFA | phlorofucofuroeckol |
| PGB | phloroglucinol 6,6-bieckol |
| 2,7PGB | 2,7-phyrogalyol-6,6-bieckol |
| OPA | octaphlorethol |
| DPHC | diphlorethohydroxycarmalol |
| SA | saringosterol acetate |
| LC-ESI-MS | liquid chromatography-electrospray ionization-mass spectrometry |
| NMR | nuclear magnetic resonance |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide |
| DSMO | dimethyl sulfoxide |
| PI | propidium iodide |
| FITC | fluorescein isothiocyanate |
| FACS | fluorescence-activated cell sorting |
| ICF | immunocytofluorescence |
| DAPI | 4′,6-diamidino-2-phenylindole |
| MDA | malonic dialdehyde |
References
- Lee, J.C.; Hou, M.F.; Huang, H.W.; Chang, F.R.; Yeh, C.C.; Tang, J.Y.; Chang, H.W. Marine algal natural products with anti-oxidative, anti-inflammatory, and anti-cancer properties. Cancer Cell. Int. 2013, 13, 55. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Chung, H.Y.; Kim, J.Y.; Son, B.W.; Jung, H.A.; Choi, J.S. Inhibitory phlorotannins from the edible brown alga Ecklonia stolonifera on total reactive oxygen species (ROS) generation. Arch. Pharm. Res. 2004, 27, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.C.; Hwang, H.J.; Kang, K.J.; Lee, B.H. An antioxidative and antiinflammatory agent for potential treatment of osteoarthritis from Ecklonia cava. Arch. Pharm. Res. 2006, 29, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.I.; Shin, H.C.; Kim, S.H.; Park, W.Y.; Lee, K.T.; Choi, J.H. 6,6′-Bieckol, isolated from marine alga Ecklonia cava, suppressed LPS-induced nitric oxide and PGE(2) production and inflammatory cytokine expression in macrophages: The inhibition of NFkappaB. Int. Immunopharmacol. 2012, 12, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Nagayama, K.; Iwamura, Y.; Shibata, T.; Hirayama, I.; Nakamura, T. Bactericidal activity of phlorotannins from the brown alga Ecklonia kurome. J. Antimicrob. Chemother. 2002, 50, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Artan, M.; Li, Y.; Karadeniz, F.; Lee, S.H.; Kim, M.M.; Kim, S.K. Anti-HIV-1 activity of phloroglucinol derivative, 6,6′-bieckol, from Ecklonia cava. Bioorg. Med. Chem. 2008, 16, 7921–7926. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lee, S.H.; Le, Q.T.; Kim, M.M.; Kim, S.K. Anti-allergic effects of phlorotannins on histamine release via binding inhibition between IgE and Fc epsilonRI. J. Agric. Food Chem. 2008, 56, 12073–12080. [Google Scholar] [CrossRef] [PubMed]
- Mercado, N.; Ito, K.; Barnes, P.J. Accelerated ageing of the lung in COPD: New concepts. Thorax 2015, 70, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Church, D.F.; Pryor, W.A. Free-radical chemistry of cigarette smoke and its toxicological implications. Environ. Health Perspect. 1985, 64, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Perfetti, T.A.; Garg, R.; Hansch, C. IARC carcinogens reported in cigarette mainstream smoke and their calculated log P values. Food Chem. Toxicol. 2003, 41, 807–817. [Google Scholar] [CrossRef]
- Nyunoya, T.; Monick, M.M.; Klingelhutz, A.; Yarovinsky, T.O.; Cagley, J.R.; Hunninghake, G.W. Cigarette smoke induces cellular senescence. Am. J. Respir. Cell. Mol. Biol. 2006, 35, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Volonte, D.; Kahkonen, B.; Shapiro, S.; Di, Y.; Galbiati, F. Caveolin-1 expression is required for the development of pulmonary emphysema through activation of the ATM-p53-p21 pathway. J. Biol. Chem. 2009, 284, 5462–5466. [Google Scholar] [CrossRef] [PubMed]
- Firsanov, D.V.; Solovjeva, L.V.; Svetlova, M.P. H2AX phosphorylation at the sites of DNA double-strand breaks in cultivated mammalian cells and tissues. Clin. Epigenet. 2011, 2, 283–297. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.P.; Bartek, J. The DNA-damage response in human biology and disease. Nature 2009, 461, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Nyunoya, T.; Mebratu, Y.; Contreras, A.; Delgado, M.; Chand, H.S.; Tesfaigzi, Y. Molecular processes that drive cigarette smoke-induced epithelial cell fate of the lung. Am. J. Respir. Cell. Mol. Biol. 2014, 50, 471–482. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Liu, X.; Kobayashi, T.; Conner, H.; Kohyama, T.; Wen, F.Q.; Fang, Q.; Abe, S.; Bitterman, P.; Rennard, S.I. Reversible cigarette smoke extract-induced DNA damage in human lung fibroblasts. Am. J. Respir. Cell. Mol. Biol. 2004, 31, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Nyunoya, T.; Monick, M.M.; Klingelhutz, A.L.; Glaser, H.; Cagley, J.R.; Brown, C.O.; Matsumoto, E.; Aykin-Burns, N.; Spitz, D.R.; Oshima, J.; et al. Cigarette smoke induces cellular senescence via Werner’s syndrome protein down-regulation. Am. J. Respir. Crit. Care Med. 2009, 179, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Balansky, R.; Izzotti, A.; Scatolini, L.; D’Agostini, F.; de Flora, S. Induction by carcinogens and chemoprevention by N-acetylcysteine of adducts to mitochondrial DNA in rat organs. Cancer Res. 1996, 56, 1642–1647. [Google Scholar] [PubMed]
- Ito, S.; Araya, J.; Kurita, Y.; Kobayashi, K.; Takasaka, N.; Yoshida, M.; Hara, H.; Minagawa, S.; Wakui, H.; Fujii, S.; et al. PARK2-mediated mitophagy is involved in regulation of HBEC senescence in COPD pathogenesis. Autophagy 2015, 11, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qian, Z.J.; Ryu, B.; Lee, S.H.; Kim, M.M.; Kim, S.K. Chemical components and its antioxidant properties in vitro: An edible marine brown alga, Ecklonia cava. Bioorg. Med. Chem. 2009, 17, 1963–1973. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Ko, J.Y.; Oh, J.Y.; Kim, C.Y.; Lee, H.J.; Kim, J.; Jeon, Y.J. Preparative isolation and purification of phlorotannins from Ecklonia cava using centrifugal partition chromatography by one-step. Food Chem. 2014, 158, 433–437. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kang, S.M.; Ko, S.C.; Lee, D.H.; Jeon, Y.J. Octaphlorethol A, a novel phenolic compound isolated from a brown alga, Ishige foliacea, increases glucose transporter 4-mediated glucose uptake in skeletal muscle cells. Biochem. Biophys. Res. Commun. 2012, 420, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.J.; Kim, J.P.; Jung, W.K.; Lee, N.H.; Kang, H.S.; Jun, E.M.; Park, S.H.; Kang, S.M.; Lee, Y.J.; Park, P.J.; et al. Identification of chemical structure and free radical scavenging activity of diphlorethohydroxycarmalol isolated from a brown alga, Ishige okamurae. J. Microbiol. Biotechnol. 2008, 18, 676–681. [Google Scholar] [PubMed]
- Doi, Y.; Ishibashi, M.; Yamaguchi, N.; Kobayashi, J. Isolation of Apo-9′-Fucoxanthinone from the Cultured Marine Dinoflagellate Amphidinium sp. J. Nat. Prod. 1995, 58, 1097–1099. [Google Scholar] [CrossRef]
- Kurata, K.; Taniguchi, K.; Shiraishi, K.; Suzuki, M. A C26 Sterol From The Brown Alga Eisenia Bicyclis. Phytochemistory 1990, 29, 3678–3680. [Google Scholar] [CrossRef]
- Bruse, S.; Moreau, M.; Bromberg, Y.; Jang, J.H.; Wang, N.; Ha, H.; Picchi, M.; Lin, Y.; Langley, R.J.; Qualls, C.; et al. Whole exome sequencing identifies novel candidate genes that modify chronic obstructive pulmonary disease susceptibility. Hum. Genom. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Van der Toorn, M.; Rezayat, D.; Kauffman, H.F.; Bakker, S.J.; Gans, R.O.; Koeter, G.H.; Choi, A.M.; van Oosterhout, A.J.; Slebos, D.J. Lipid-soluble components in cigarette smoke induce mitochondrial production of reactive oxygen species in lung epithelial cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2009, 297, L109–L114. [Google Scholar] [CrossRef] [PubMed]
- Mahalik, N.P.; Kim, K. Aquaculture monitoring and control systems for seaweed and fish farming. World J. Agric. Res. 2014, 2, 176–182. [Google Scholar] [CrossRef]
- Ahn, G.; Lee, W.; Kim, K.N.; Lee, J.H.; Heo, S.J.; Kang, N.; Lee, S.H.; Ahn, C.B.; Jeon, Y.J. A sulfated polysaccharide of Ecklonia cava inhibits the growth of colon cancer cells by inducing apoptosis. EXCLI J. 2015, 14, 294–306. [Google Scholar] [PubMed]
- Kang, M.C.; Cha, S.H.; Wijesinghe, W.A.; Kang, S.M.; Lee, S.H.; Kim, E.A.; Song, C.B.; Jeon, Y.J. Protective effect of marine algae phlorotannins against AAPH-induced oxidative stress in zebrafish embryo. Food Chem. 2013, 138, 950–955. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.H.; Yang, Y.I.; Lee, K.T.; Choi, J.H. Dieckol, isolated from the edible brown algae Ecklonia cava, induces apoptosis of ovarian cancer cells and inhibits tumor xenograft growth. J. Cancer Res. Clin. Oncol. 2015, 141, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Jeon, Y.J. Anti-diabetic effects of brown algae derived phlorotannins, marine polyphenols through diverse mechanisms. Fitoterapia 2013, 86, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Safe, L.M.; Wong, C.J.; Chandler, R.F. Sterols of marine algae. J. Pharm. Sci. 1974, 63, 464–466. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, Y.S.; Jung, S.H.; Kang, S.S.; Shin, K.H. Anti-oxidant activities of fucosterol from the marine algae Pelvetia siliquosa. Arch. Pharm. Res. 2003, 26, 719–722. [Google Scholar] [CrossRef] [PubMed]
- Heo, S.J.; Yoon, W.J.; Kim, K.N.; Ahn, G.N.; Kang, S.M.; Kang, D.H.; Affan, A.; Oh, C.; Jung, W.K.; Jeon, Y.J. Evaluation of anti-inflammatory effect of fucoxanthin isolated from brown algae in lipopolysaccharide-stimulated RAW 264.7 macrophages. Food Chem. Toxicol. 2010, 48, 2045–2051. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Jin, S.E.; Ahn, B.R.; Lee, C.M.; Choi, J.S. Anti-inflammatory activity of edible brown alga Eisenia bicyclis and its constituents fucosterol and phlorotannins in LPS-stimulated RAW264.7 macrophages. Food Chem. Toxicol. 2013, 59, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Shin, K.H.; Kim, B.K.; Lee, S. Anti-diabetic activities of fucosterol from Pelvetia siliquosa. Arch. Pharm. Res. 2004, 27, 1120–1122. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.; Sasikala, S.L.; Aldous, V.; Huxley, J. Isolation and identification of MDR-Mycobacterium tuberculosis and screening of partially characterized antimycobacterial compounds from chosen marine micro algae. Asian Pac. J. Trop. Med. 2010, 3, 655–661. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Ji, W.; Du, J.R.; Yu, D.K.; He, Y.; Yu, C.X.; Li, D.S.; Zhao, C.Y.; Qiao, K.Y. Preventive effects of low molecular mass potassium alginate extracted from brown algae on DOCA salt-induced hypertension in rats. Biomed. Pharm. 2010, 64, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.J.; Ham, Y.M.; Lee, W.J.; Lee, N.H.; Hyun, C.G. Anti-inflammatory effects of apo-9′-fucoxanthinone from the brown alga, Sargassum muticum. Daru 2013, 21, 62. [Google Scholar] [CrossRef] [PubMed]
- Chae, D.; Manzoor, Z.; Kim, S.C.; Kim, S.; Oh, T.H.; Yoo, E.S.; Kang, H.K.; Hyun, J.W.; Lee, N.H.; Ko, M.H.; et al. Apo-9′-fucoxanthinone, isolated from Sargassum muticum, inhibits CpG-induced inflammatory response by attenuating the mitogen-activated protein kinase pathway. Mar. Drugs 2013, 11, 3272–3287. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.H.; Kim, H.P.; Sciurba, F.C.; Lee, S.J.; Feghali-Bostwick, C.; Stolz, D.B.; Dhir, R.; Landreneau, R.J.; Schuchert, M.J.; Yousem, S.A.; et al. Egr-1 regulates autophagy in cigarette smoke-induced chronic obstructive pulmonary disease. PLoS ONE 2008, 3, e3316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, W.; Yuan, C.; Zhang, J.; Li, L.; Yu, L.; Wiegman, C.H.; Barnes, P.J.; Adcock, I.M.; Huang, M.; Yao, X. Klotho expression is reduced in COPD airway epithelial cells: Effects on inflammation and oxidant injury. Clin. Sci. 2015, 129, 1011–1023. [Google Scholar] [CrossRef] [PubMed]
- Petrache, I.; Natarajan, V.; Zhen, L.; Medler, T.R.; Richter, A.T.; Cho, C.; Hubbard, W.C.; Berdyshev, E.V.; Tuder, R.M. Ceramide upregulation causes pulmonary cell apoptosis and emphysema-like disease in mice. Nat. Med. 2005, 11, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Ryter, S.W.; Choi, A.M. Functional significance of apoptosis in chronic obstructive pulmonary disease. COPD 2007, 4, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.H.; Lam, H.C.; Jin, Y.; Kim, H.P.; Cao, J.; Lee, S.J.; Ifedigbo, E.; Parameswaran, H.; Ryter, S.W.; Choi, A.M. Autophagy protein microtubule-associated protein 1 light chain-3B (LC3B) activates extrinsic apoptosis during cigarette smoke-induced emphysema. Proc. Natl. Acad. Sci. USA 2010, 107, 18880–18885. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.I.; Yeh, C.C.; Lee, J.C.; Yi, S.C.; Huang, H.W.; Tseng, C.N.; Chang, H.W. Aqueous extracts of the edible Gracilaria tenuistipitata are protective against H2O2-induced DNA damage, growth inhibition, and cell cycle arrest. Molecules 2012, 17, 7241–7254. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.S.; Liu, L.J.; OuYang, X.K.; Qu, Y.L.; Chen, Y.; Ding, G.F. Protective effect of polysaccharides from Sargassum horneri against oxidative stress in RAW264.7 cells. Int. J. Biol. Macromol. 2014, 68, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Cao, J.; Gao, J.; Zheng, L.; Goodwin, A.; An, C.H.; Patel, A.; Lee, J.S.; Duncan, S.R.; Kaminski, N.; et al. Retinoic acid-related orphan receptor-alpha is induced in the setting of DNA damage and promotes pulmonary emphysema. Am. J. Respir. Crit. Care Med. 2012, 186, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Bruse, S.; Liu, Y.; Duffy, V.; Zhang, C.; Oyamada, N.; Randell, S.; Matsumoto, A.; Thompson, D.C.; Lin, Y.; et al. Aldehyde dehydrogenase 3A1 protects airway epithelial cells from cigarette smoke-induced DNA damage and cytotoxicity. Free Radic. Biol. Med. 2014, 68, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Bruse, S.; Huneidi, S.; Schrader, R.M.; Monick, M.M.; Lin, Y.; Carter, A.B.; Klingelhutz, A.J.; Nyunoya, T. Acrolein-Exposed Normal Human Lung Fibroblasts: Cellular Senescence, Enhanced Telomere Erosion, and Degradation of Werner’s Syndrome Protein. Environ. Health Perspect. 2014, 122, 955–962. [Google Scholar] [CrossRef] [PubMed]
- Contreras, A.U.; Mebratu, Y.; Delgado, M.; Montano, G.; Hu, C.A.; Ryter, S.W.; Choi, A.M.; Lin, Y.; Xiang, J.; Chand, H.; et al. Deacetylation of p53 induces autophagy by suppressing Bmf expression. J. Cell. Biol. 2013, 201, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, C.; Kampka, J.M.; Huppertz, S.; Weber, H.; Schlosser, A.; Muller, A.M.; Becker, M. Inhibition of KDM6 activity during murine ESC differentiation induces DNA damage. J. Cell. Sci. 2016, 129, 788–803. [Google Scholar] [CrossRef] [PubMed]
- Olive, P.L.; Banath, J.P. The comet assay: A method to measure DNA damage in individual cells. Nat. Protoc. 2006, 1, 23–29. [Google Scholar] [CrossRef] [PubMed]
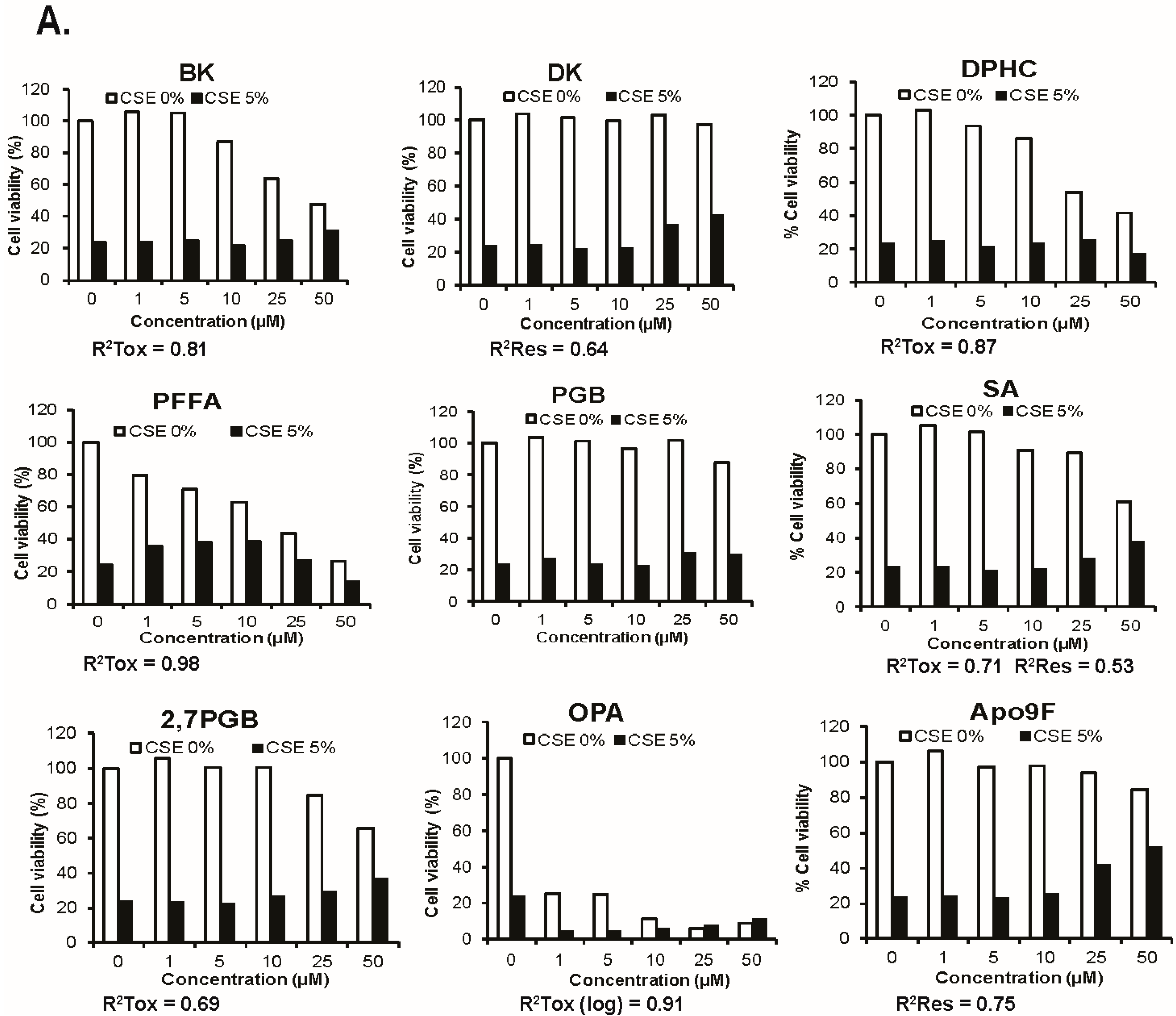



| Species | Structure Name | Abbreviation | Molecular Weight | References for the Extraction Methods |
|---|---|---|---|---|
| Ecklonia cava | 6,6-bieckol | BK | 742 | |
| Dieckol | DK | 742 | ||
| Phlorofucofuroeckol A | PFFA | 602 | [20,21] | |
| Phloroglucinol 6,6-bieckol | PGB | 972 | ||
| 2,7-phyrogalyol-6,6-biekol | 2,7PGB | 972 | ||
| Ishige foliacea | Octaphlorethol A | OPA | 993 | [22] |
| Ishige okamura | Diphlorethohydroxycarmalol | DPHC | 512 | [23] |
| Undariopsis peteseniana | Apo-9′-fucoxanthinone | Apo9F | 266 | [24] |
| Hizikia fusiformis | Saringosterol acetate | SA | 470 | [25] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, J.-H.; Lee, J.-H.; Chand, H.S.; Lee, J.-S.; Lin, Y.; Weathington, N.; Mallampalli, R.; Jeon, Y.-J.; Nyunoya, T. APO-9′-Fucoxanthinone Extracted from Undariopsis peteseniana Protects Oxidative Stress-Mediated Apoptosis in Cigarette Smoke-Exposed Human Airway Epithelial Cells. Mar. Drugs 2016, 14, 140. https://doi.org/10.3390/md14070140
Jang J-H, Lee J-H, Chand HS, Lee J-S, Lin Y, Weathington N, Mallampalli R, Jeon Y-J, Nyunoya T. APO-9′-Fucoxanthinone Extracted from Undariopsis peteseniana Protects Oxidative Stress-Mediated Apoptosis in Cigarette Smoke-Exposed Human Airway Epithelial Cells. Marine Drugs. 2016; 14(7):140. https://doi.org/10.3390/md14070140
Chicago/Turabian StyleJang, Jun-Ho, Ji-Hyeok Lee, Hitendra S. Chand, Jong-Soo Lee, Yong Lin, Nathaniel Weathington, Rama Mallampalli, You-Jin Jeon, and Toru Nyunoya. 2016. "APO-9′-Fucoxanthinone Extracted from Undariopsis peteseniana Protects Oxidative Stress-Mediated Apoptosis in Cigarette Smoke-Exposed Human Airway Epithelial Cells" Marine Drugs 14, no. 7: 140. https://doi.org/10.3390/md14070140







