Fucoxanthin, a Marine Carotenoid, Reverses Scopolamine-Induced Cognitive Impairments in Mice and Inhibits Acetylcholinesterase in Vitro
Abstract
:1. Introduction
2. Results
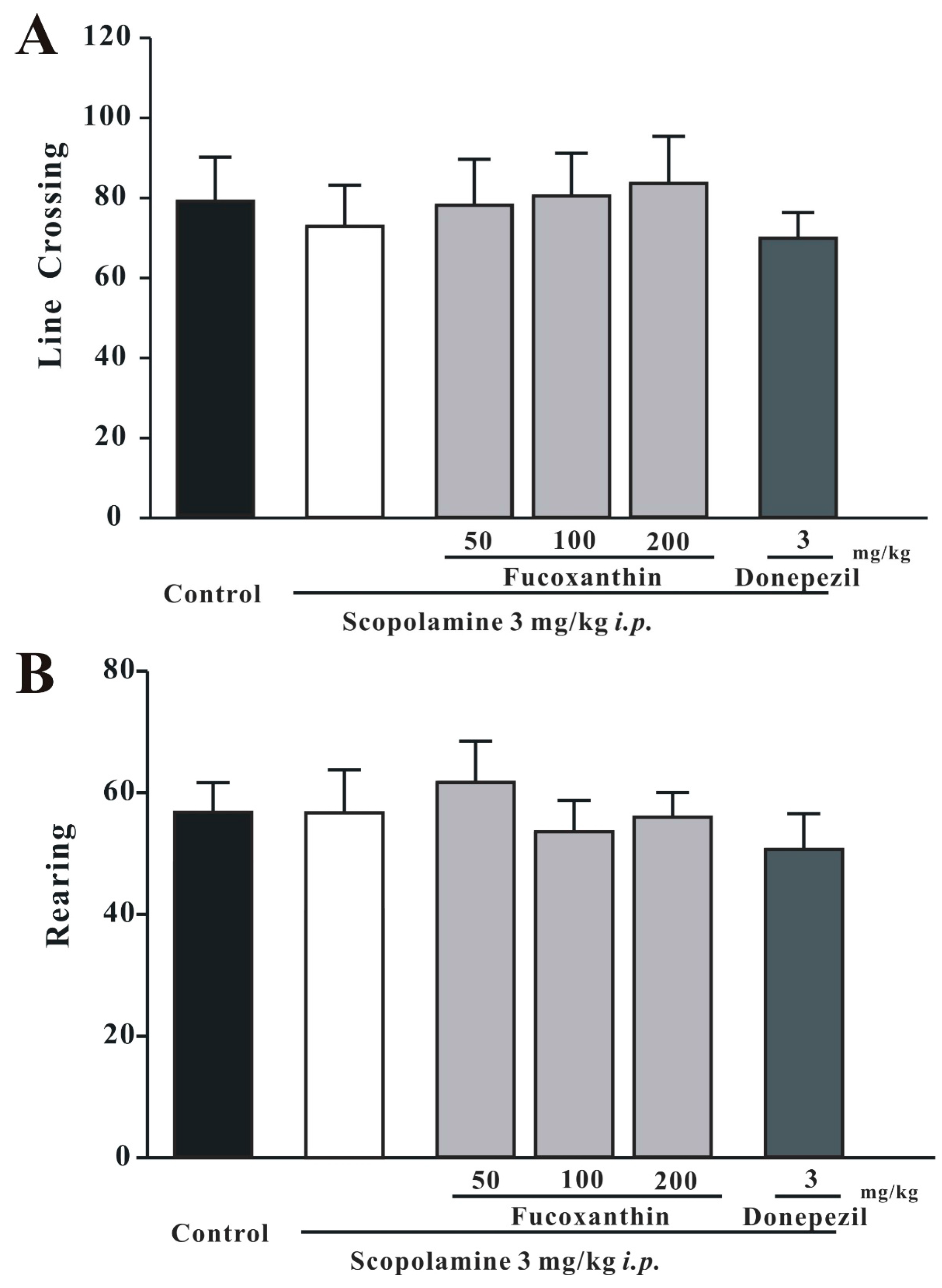
2.1. Fucoxanthin Does Not Affect Locomotor Activity in the 5-min Open-Field Test
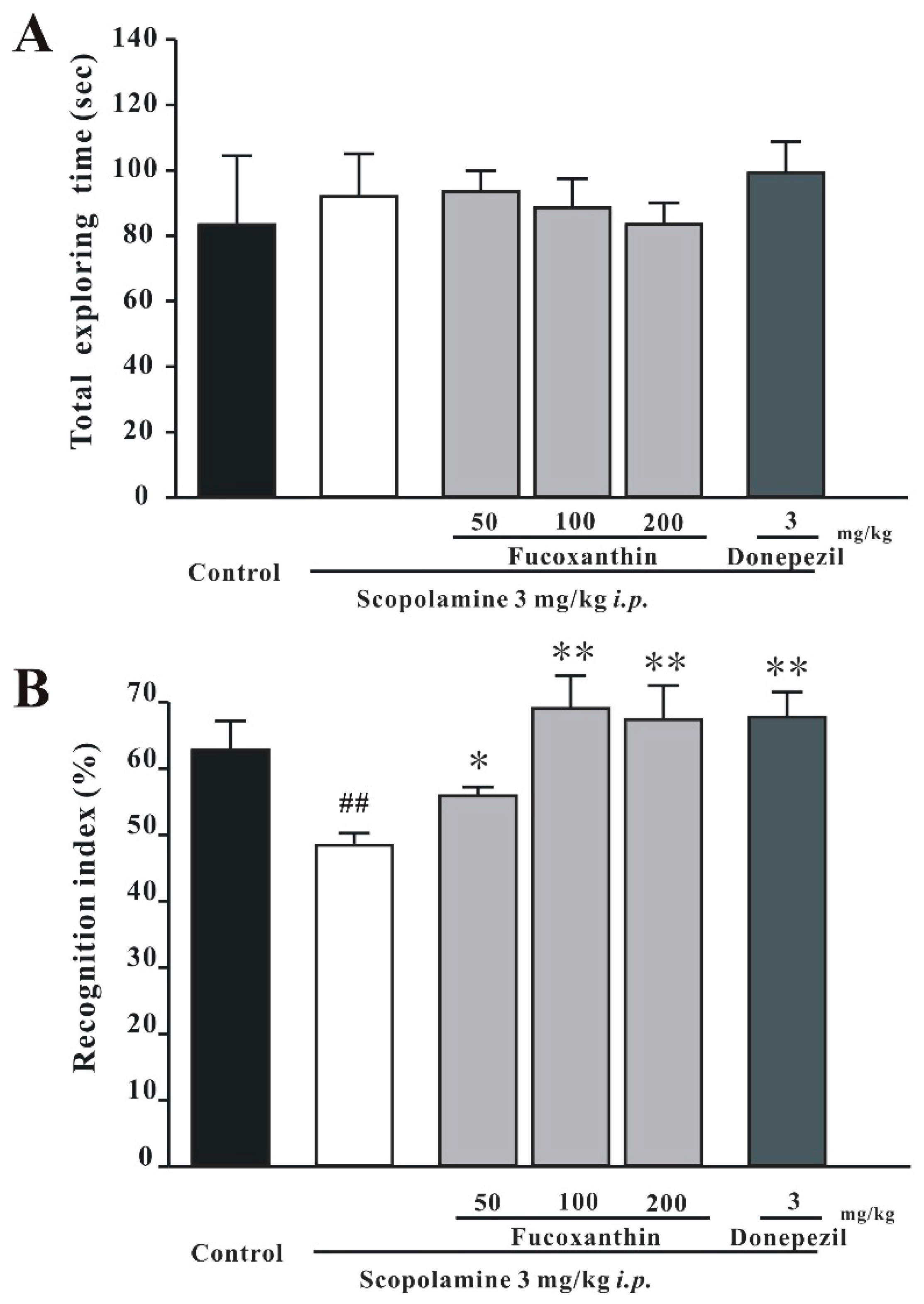
2.2. Fucoxanthin Reverses Scopolamine-Induced Recognition Impairment in the NOR Test
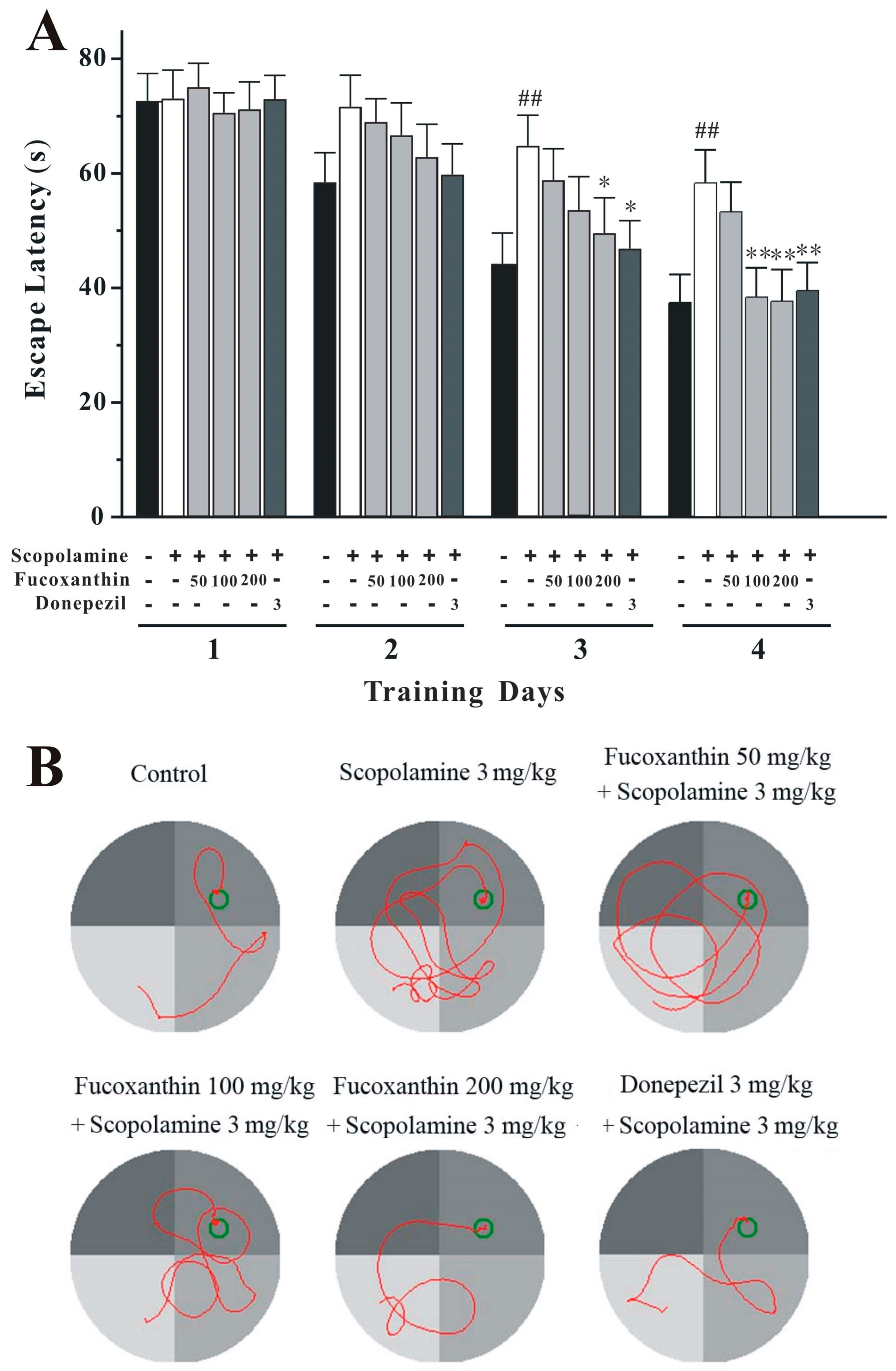
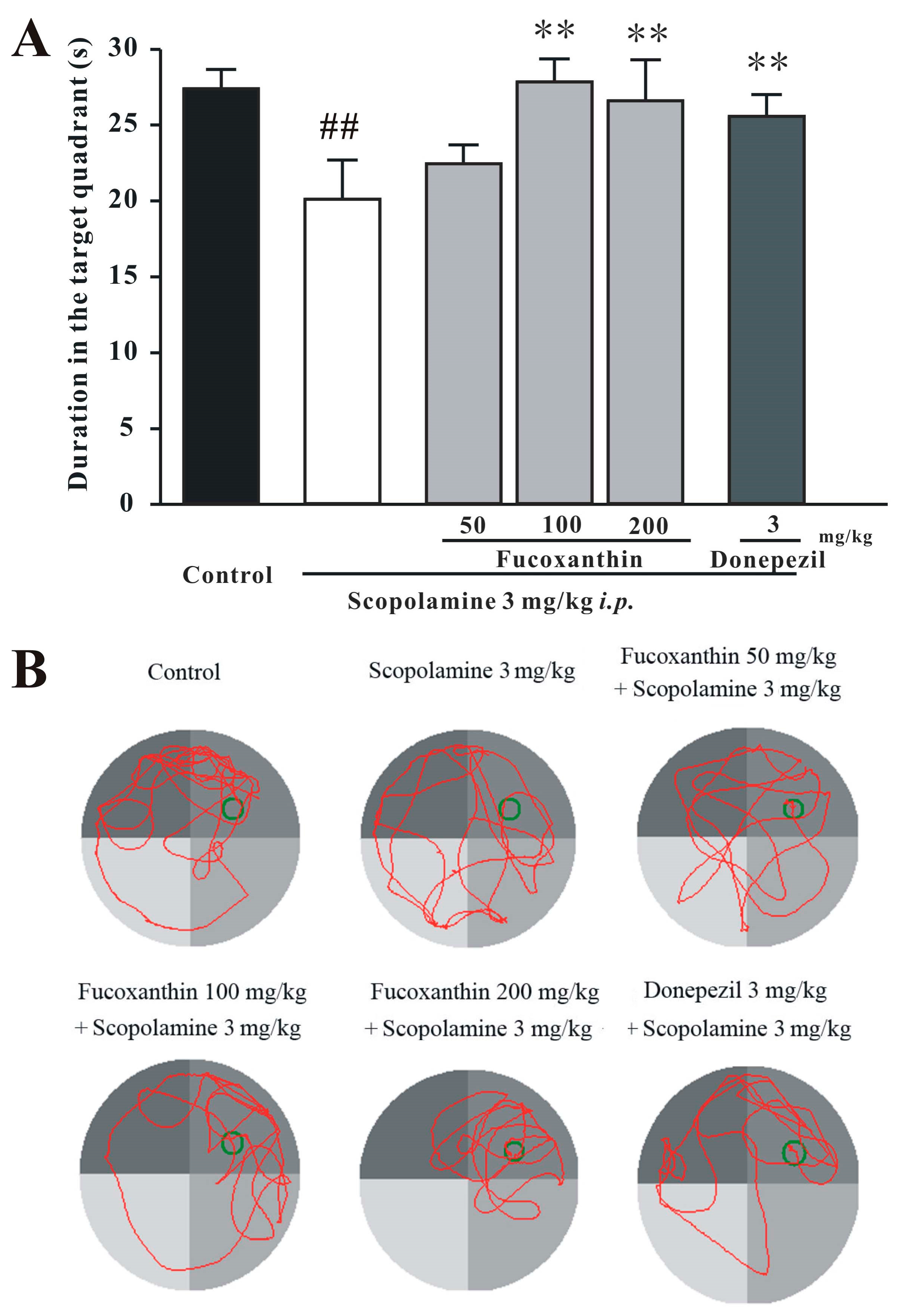
2.3. Fucoxanthin Reverses Scopolamine-Induced Spatial Learning and Memory Impairments in the Morris Water Maze Test
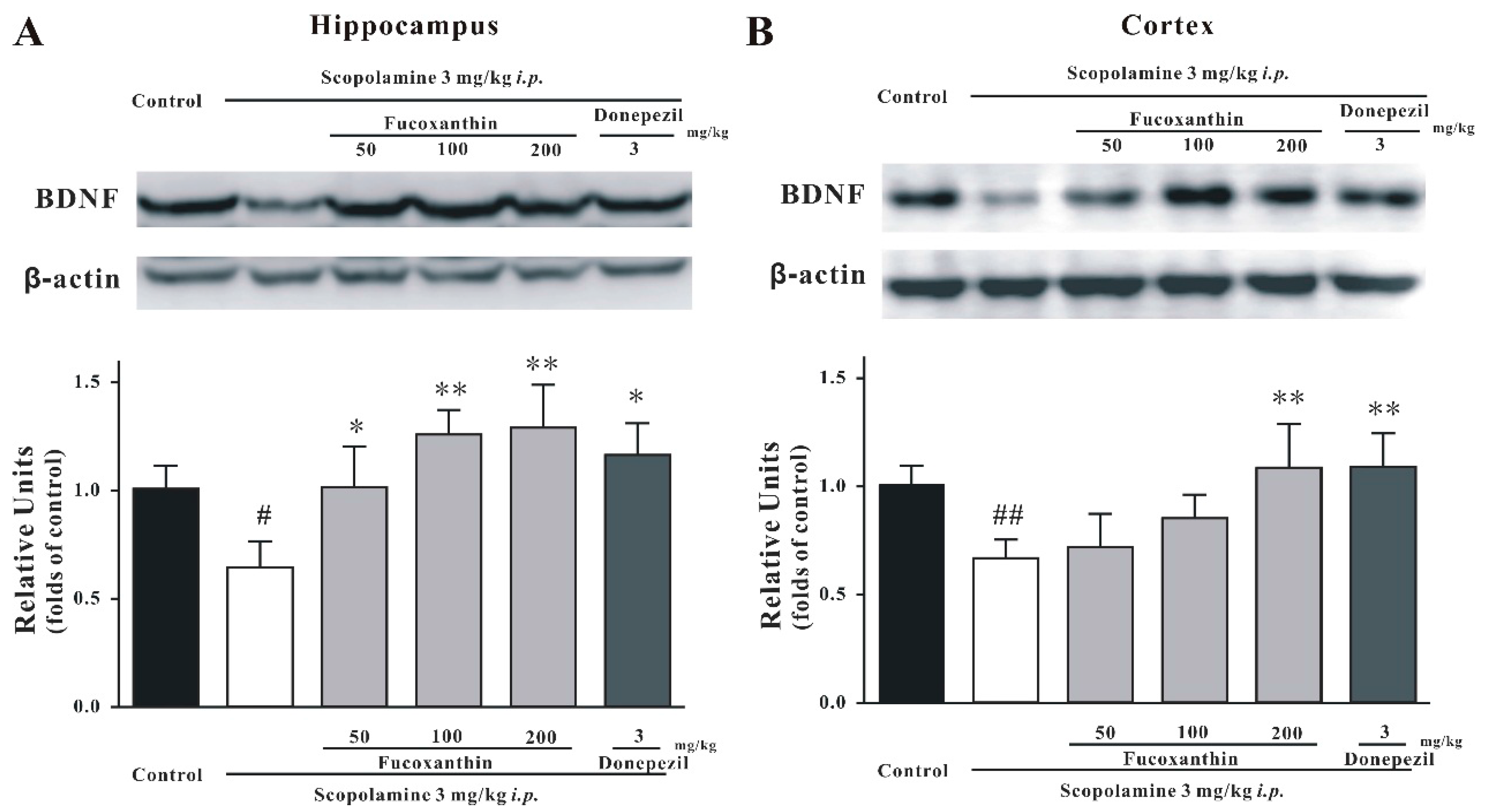
2.4. Fucoxanthin Increases the Expression of BDNF in the Hippocampus and Cortex of Mice
2.5. Fucoxanthin Increases ChAT Activity and Decreases AChE Activity in the Hippocampus and Cortex of Mice
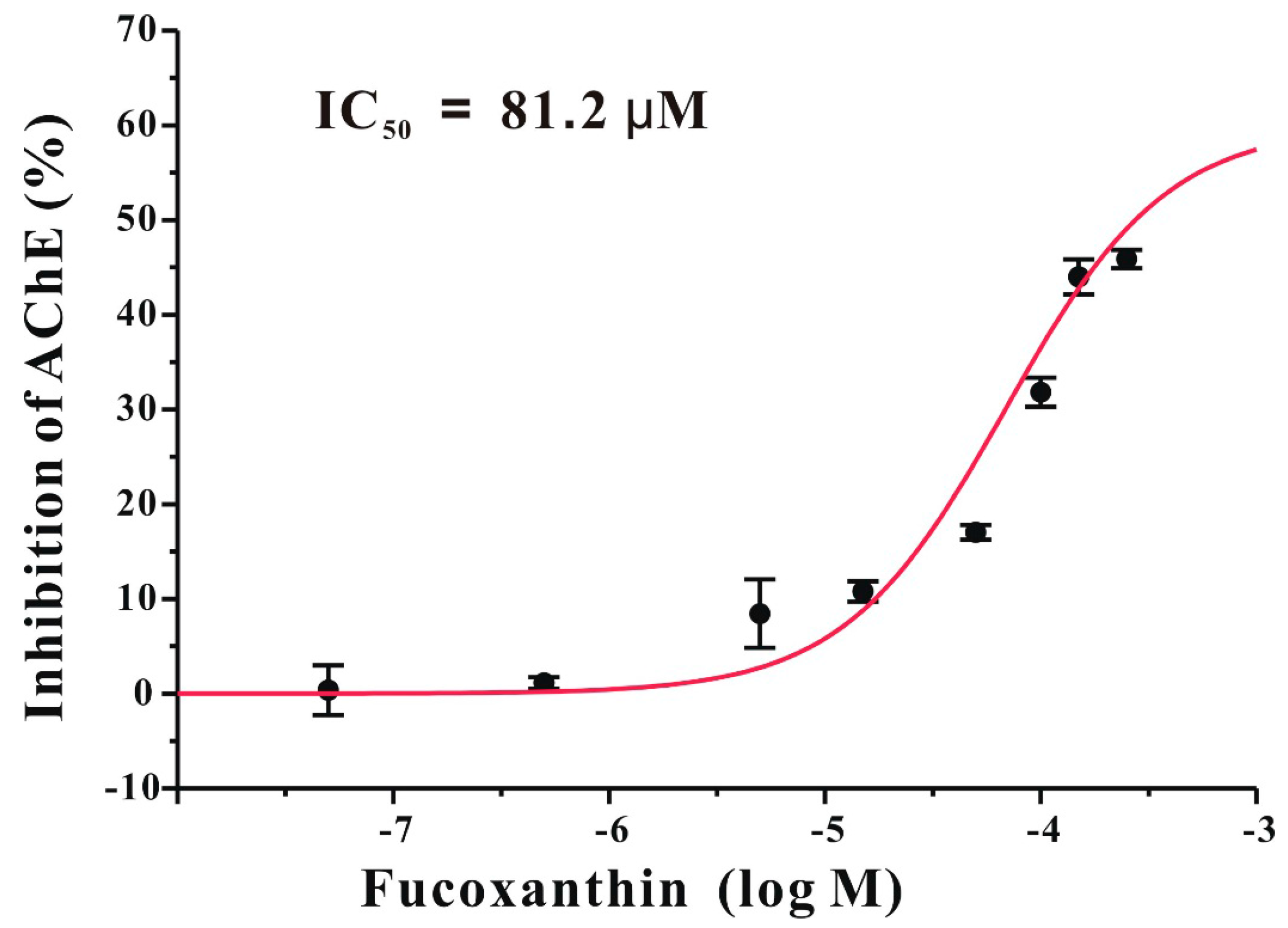
2.6. Fucoxanthin Directly Inhibits AChE in a Non-Competitive Manner in Vitro
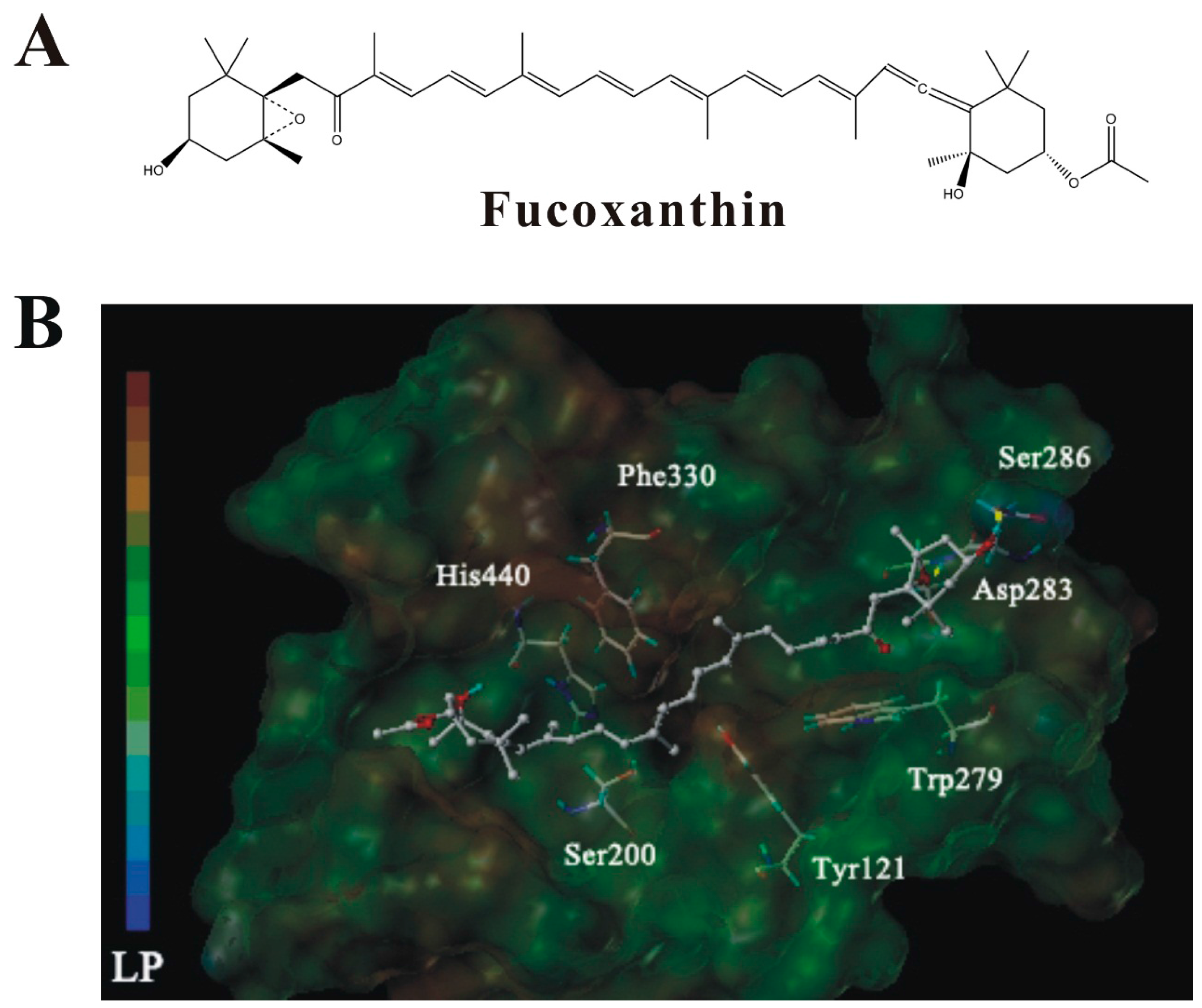
2.7. Molecular Docking Analysis of the Interaction between Fucoxanthin and AChE
3. Discussion
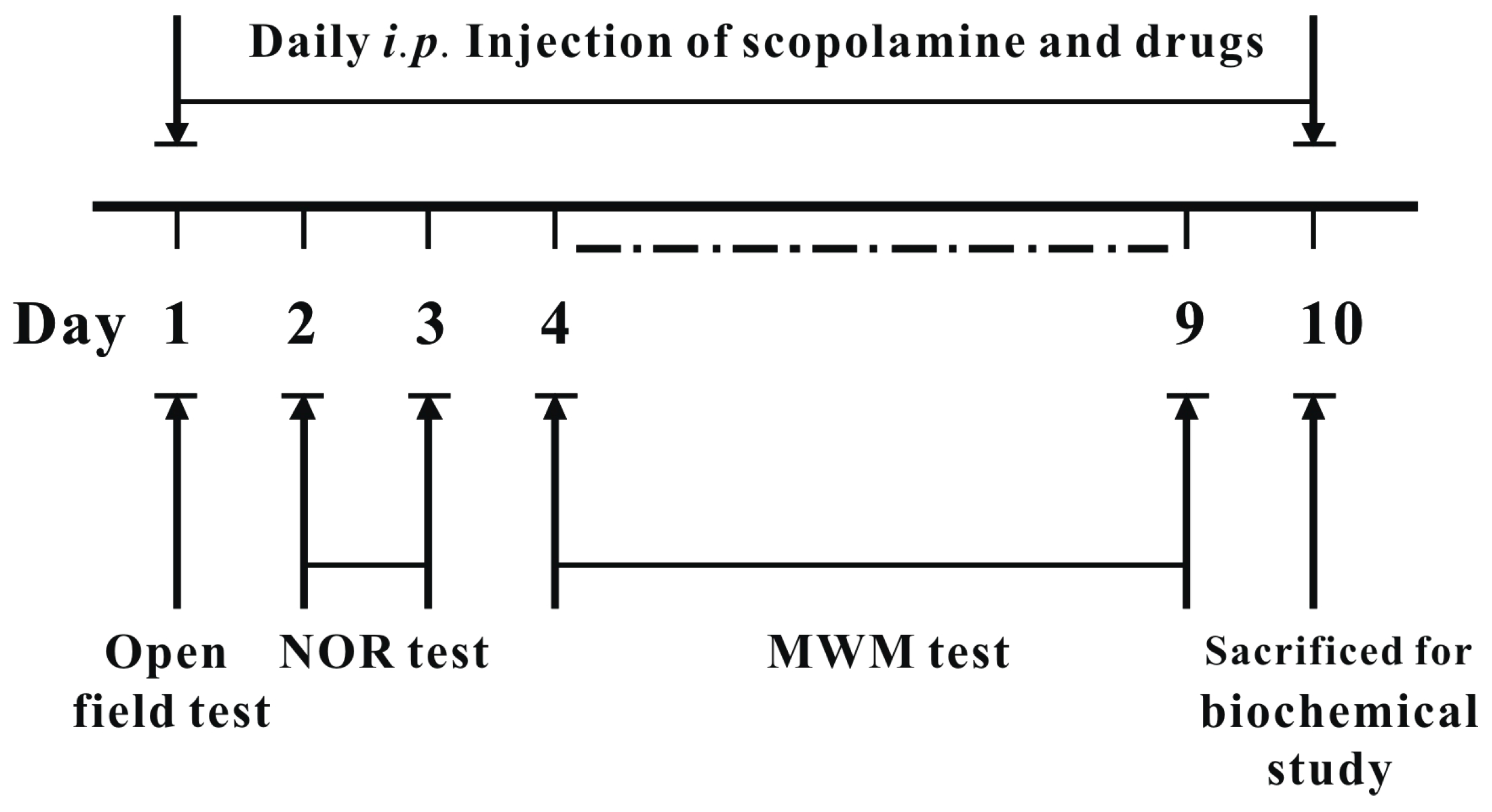
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Drug Treatment for Animal Study
4.3. Open-Field Test
4.4. Novel Object Recognition Test
4.5. Morris Water Maze Task
4.6. Western Blot Analysis
4.7. Measurement of Choline ChAT Activity ex Vivo
4.8. Measurement of AChE Activity ex Vivo
4.9. Measurement of AChE Activity in Vitro
4.10. Molecular Docking
4.11. Data Analysis and Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kumar, A.; Singh, A.; Ekavali. A review on Alzheimer’s disease pathophysiology and its management: An update. Pharmacol. Rep. 2015, 67, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Anand, R.; Gill, K.D.; Mahdi, A.A. Therapeutics of Alzheimer’s disease: Past, present and future. Neuropharmacology 2014, 76, 27–50. [Google Scholar] [CrossRef] [PubMed]
- Zemek, F.; Drtinova, L.; Nepovimova, E.; Sepsova, V.; Korabecny, J.; Klimes, J.; Kuca, K. Outcomes of Alzheimer’s disease therapy with acetylcholinesterase inhibitors and memantine. Expert Opin. Drug Saf. 2014, 13, 759–774. [Google Scholar] [PubMed]
- Min, A.Y.; Doo, C.N.; Son, E.J.; Sung, N.Y.; Lee, K.J.; Sok, D.E.; Kim, M.R. N-palmitoyl serotonin alleviates scopolamine-induced memory impairment via regulation of cholinergic and antioxidant systems, and expression of BDNF and p-CREB in mice. Chem. Biol. Interact. 2015, 242, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Konar, A.; Kumar, A.; Srivas, S.; Thakur, M.K. Hippocampal chromatin-modifying enzymes are pivotal for scopolamine-induced synaptic plasticity gene expression changes and memory impairment. J. Neurochem. 2015, 134, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Araujo, J.A.; Studzinski, C.M.; Milgram, N.W. Further evidence for the cholinergic hypothesis of aging and dementia from the canine model of aging. Prog. Neuropsychopharmacol. Biol. Psychiatry 2005, 29, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Beatty, W.W.; Butters, N.; Janowsky, D.S. Patterns of memory failure after scopolamine treatment: Implications for cholinergic hypotheses of dementia. Behav. Neural Biol. 1986, 45, 196–211. [Google Scholar] [CrossRef]
- Kopelman, M.D.; Corn, T.H. Cholinergic “blockade” as a model for cholinergic depletion. A comparison of the memory deficits with those of Alzheimer-type dementia and the alcoholic Korsakoff syndrome. Brain 1988, 111, 1079–1110. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.H.; Lee, H.K.; Kim, J.A.; Hong, S.I.; Kim, H.C.; Jo, T.H.; Park, Y.I.; Lee, C.K.; Kim, Y.B.; Lee, S.Y.; et al. Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase and anti-oxidative activities in mice. Eur. J. Pharmacol. 2010, 649, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Ghumatkar, P.J.; Patil, S.P.; Jain, P.D.; Tambe, R.M.; Sathaye, S. Nootropic, neuroprotective and neurotrophic effects of phloretin in scopolamine induced amnesia in mice. Pharmacol. Biochem. Behav. 2015, 135, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Luo, X.; Liu, X.; Liu, D.; Wang, X.; Guo, Z.; Zhu, L.; Tian, Q.; Yang, X.; Wang, J.Z. Intraperitoneal Administration of a Novel TAT-BDNF Peptide Ameliorates Cognitive Impairments via Modulating Multiple Pathways in Two Alzheimer’s Rodent Models. Sci. Rep. 2015, 5, 15032. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, K. Function of marine carotenoids. Forum Nutr. 2009, 61, 136–146. [Google Scholar] [PubMed]
- D’Orazio, N.; Gammone, M.A.; Gemello, E.; De Girolamo, M.; Cusenza, S.; Riccioni, G. Marine bioactives: Pharmacological properties and potential applications against inflammatory diseases. Mar. Drugs 2012, 10, 812–833. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Chuda, Y.; Suzuki, M.; Nagata, T. Fucoxanthin as the major antioxidant in Hijikia fusiformis, a common edible seaweed. Biosci. Biotechnol. Biochem. 1999, 63, 605–607. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Hashimoto, T.; Kanazawa, K. Growth inhibition of human hepatic carcinoma HepG2 cells by fucoxanthin is associated with down-regulation of cyclin D. Biochim. Biophys. Acta 2008, 1780, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Woo, M.N.; Jeon, S.M.; Shin, Y.C.; Lee, M.K.; Kang, M.A.; Choi, M.S. Anti-obese property of fucoxanthin is partly mediated by altering lipid-regulating enzymes and uncoupling proteins of visceral adipose tissue in mice. Mol. Nutr. Food Res. 2009, 53, 1603–1611. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Miyashita, K. Dietary combination of fucoxanthin and fish oil attenuates the weight gain of white adipose tissue and decreases blood glucose in obese/diabetic KK-Ay mice. J. Agric. Food Chem. 2007, 55, 7701–7706. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; D’Orazio, N. Anti-obesity activity of the marine carotenoid fucoxanthin. Mar. Drugs 2015, 13, 2196–2214. [Google Scholar] [CrossRef] [PubMed]
- Ha, A.W.; Na, S.J.; Kim, W.K. Antioxidant effects of fucoxanthin rich powder in rats fed with high fat diet. Nutr. Res. Pract. 2013, 7, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Pangestuti, R.; Vo, T.S.; Ngo, D.H.; Kim, S.K. Fucoxanthin ameliorates inflammation and oxidative reponses in microglia. J. Agric. Food Chem. 2013, 61, 3876–3883. [Google Scholar] [CrossRef] [PubMed]
- Beppu, F.; Niwano, Y.; Sato, E.; Kohno, M.; Tsukui, T.; Hosokawa, M.; Miyashita, K. In vitro and in vivo evaluation of mutagenicity of fucoxanthin (FX) and its metabolite fucoxanthinol (FXOH). J. Toxicol. Sci. 2009, 34, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Beppu, F.; Niwano, Y.; Tsukui, T.; Hosokawa, M.; Miyashita, K. Single and repeated oral dose toxicity study of fucoxanthin (FX), a marine carotenoid, in mice. J. Toxicol. Sci. 2009, 34, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, C.R.; Yang, H.; Liu, J.; Zhang, T.; Jiao, S.S.; Wang, Y.J.; Xu, Z.Q. proBDNF Attenuates Hippocampal Neurogenesis and Induces Learning and Memory Deficits in Aged Mice. Neurotox. Res. 2015, 29, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Shim, I.; Lee, H.; Hahm, D.H. Rehmannia glutinosa ameliorates scopolamine-induced learning and memory impairment in rats. J. Microbiol. Biotechnol. 2011, 21, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Siow, N.L.; Xie, H.Q.; Choi, R.C.; Tsim, K.W. ATP induces the post-synaptic gene expression in neuron-neuron synapses: Transcriptional regulation of AChE catalytic subunit. Chem. Biol. Interact. 2005, 157–158, 423–426. [Google Scholar] [CrossRef]
- Anand, P.; Singh, B. A review on cholinesterase inhibitors for Alzheimer’s disease. Arch. Pharm. Res. 2013, 36, 375–399. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Zhang, X.J. Acetylcholinesterase and apoptosis. A novel perspective for an old enzyme. FEBS J. 2008, 275, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Tsim, K.; Soreq, H. Acetylcholinesterase: Old questions and new developments. Front Mol. Neurosci. 2012, 5, 101. [Google Scholar] [PubMed]
- Ota, T.; Shinotoh, H.; Fukushi, K.; Kikuchi, T.; Sato, K.; Tanaka, N.; Shimada, H.; Hirano, S.; Miyoshi, M.; Arai, H.; et al. Estimation of plasma IC50 of donepezil for cerebral acetylcholinesterase inhibition in patients with Alzheimer disease using positron emission tomography. Clin. Neuropharmacol. 2010, 33, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.D.; Yaffe, K. Dementia (including Alzheimer’s disease) can be prevented: Statement supported by international experts. J. Alzheimers Dis. 2014, 38, 699–6703. [Google Scholar] [PubMed]
- Chen, X.; Hu, J.; Jiang, L.; Xu, S.; Zheng, B.; Wang, C.; Zhang, J.; Wei, X.; Chang, L.; Wang, Q. Brilliant Blue G improves cognition in an animal model of Alzheimer’s disease and inhibits amyloid-beta-induced loss of filopodia and dendrite spines in hippocampal neurons. Neuroscience 2014, 279, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Ketcha Wanda, G.J.; Djiogue, S.; Zemo Gamo, F.; Guemnang Ngitedem, S.; Njamen, D. Anxiolytic and sedative activities of aqueous leaf extract of Dichrocephala integrifolia (Asteraceae) in mice. J. Ethnopharmacol. 2015, 176, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wang, C.; Xue, Z.; Li, C.; Zhang, J.; Zhao, X.; Liu, A.; Wang, Q.; Zhou, W. PI3K/AKT/mTOR signaling-mediated neuropeptide VGF in the hippocampus of mice is involved in the rapid onset antidepressant-like effects of GLYX-13. Int. J. Neuropsychopharmacol. 2015, 18, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, J.; Zhao, X.; Chen, Z.; Wang, G.; Liu, A.; Wang, Q.; Zhou, W.; Xu, Y.; Wang, C. Phosphodiesterase-5 inhibitor sildenafil prevents neuroinflammation, lowers beta-amyloid levels and improves cognitive performance in APP/PS1 transgenic mice. Behav. Brain Res. 2013, 250, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Cui, W.; Yang, Y.; Xu, S.; Zhou, W.; Fu, H.; Hu, S.; Mak, S.; Hu, J.; Wang, Q.; et al. Protection against beta-amyloid-induced synaptic and memory impairments via altering beta-amyloid assembly by bis(heptyl)-cognitin. Sci. Rep. 2015, 5, 10256. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Zhang, Z.; Li, W.; Mak, S.; Hu, S.; Zhang, H.; Yuan, S.; Rong, J.; Choi, T.C.; Lee, S.M.; Han, Y. Unexpected neuronal protection of SU5416 against 1-Methyl-4-phenylpyridinium ion-induced toxicity via inhibiting neuronal nitric oxide synthase. PLoS ONE 2012, 7, e46253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mak, S.; Luk, W.W.; Cui, W.; Hu, S.; Tsim, K.W.; Han, Y. Synergistic inhibition on acetylcholinesterase by the combination of berberine and palmatine originally isolated from Chinese medicinal herbs. J. Mol. Neurosci. 2014, 53, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Kryger, G.; Silman, I.; Sussman, J.L. Structure of acetylcholinesterase complexed with E2020 (Aricept): Implications for the design of new anti-Alzheimer drugs. Structure 1999, 7, 297–307. [Google Scholar] [CrossRef]
- Jain, A.N. Surflex: Fully automatic flexible molecular docking using a molecular similarity-based search engine. J. Med. Chem. 2003, 46, 499–511. [Google Scholar] [CrossRef] [PubMed]

| Brain Region | AChE Activity | |||||
|---|---|---|---|---|---|---|
| Control | Scopolamine 3 mg/kg i.p. | |||||
| Vehicle | Fucoxanthin (mg/kg) | Donepezil (mg/kg) | ||||
| 50 | 100 | 200 | 3 | |||
| Hippocampus | 60.3 ± 2.7 | 80.2 ± 2.3 ## | 76.2 ± 4.0 | 68.1 ± 3.2 ** | 67.6 ± 4.3 ** | 62.4 ± 2.6 ** |
| Cortex | 52.1 ± 2.8 | 71.2 ± 1.3 ## | 60.5 ± 3.0** | 62.4 ± 2.9 ** | 57.4 ± 3.2 ** | 61.6 ± 1.4 ** |
| Brain Region | ChAT Activity | |||||
|---|---|---|---|---|---|---|
| Control | Scopolamine 3 mg/kg i.p. | |||||
| Vehicle | Fucoxanthin (mg/kg) | Donepezil (mg/kg) | ||||
| 50 | 100 | 200 | 3 | |||
| Hippocampus | 32.8 ± 1.1 | 27.6 ± 0.8 # | 30.0 ± 2.0 | 32.9 ± 1.6 * | 30.5 ± 1.8 * | 31.2 ± 1.8 * |
| Cortex | 30.0 ± 1.3 | 25.2±1.0 # | 27.7 ± 1.9 | 34.0 ± 2.4 * | 30.7 ± 1.5 * | 33.7 ± 2.8 * |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, J.; Huang, L.; Yu, J.; Xiang, S.; Wang, J.; Zhang, J.; Yan, X.; Cui, W.; He, S.; Wang, Q. Fucoxanthin, a Marine Carotenoid, Reverses Scopolamine-Induced Cognitive Impairments in Mice and Inhibits Acetylcholinesterase in Vitro. Mar. Drugs 2016, 14, 67. https://doi.org/10.3390/md14040067
Lin J, Huang L, Yu J, Xiang S, Wang J, Zhang J, Yan X, Cui W, He S, Wang Q. Fucoxanthin, a Marine Carotenoid, Reverses Scopolamine-Induced Cognitive Impairments in Mice and Inhibits Acetylcholinesterase in Vitro. Marine Drugs. 2016; 14(4):67. https://doi.org/10.3390/md14040067
Chicago/Turabian StyleLin, Jiajia, Ling Huang, Jie Yu, Siying Xiang, Jialing Wang, Jinrong Zhang, Xiaojun Yan, Wei Cui, Shan He, and Qinwen Wang. 2016. "Fucoxanthin, a Marine Carotenoid, Reverses Scopolamine-Induced Cognitive Impairments in Mice and Inhibits Acetylcholinesterase in Vitro" Marine Drugs 14, no. 4: 67. https://doi.org/10.3390/md14040067







