3.1. Chemistry
The sterols were purchased from the Sinopharm Chemical Reagent Co., Ltd., Shanghai, China. All chemicals and solvents were analytical grade and solvents were purified by general methods before being used. Melting points were determined on an X4 apparatus and were uncorrected. Infrared spectra were measured with a Nicolet FT-360 Spectrophotometer (Thermo Fisher Scientific, New York, NY, USA). The 1H and 13C NMR spectra were recorded in CDCl3 on a Bruker AV-600 spectrometer (Bruker Corporation, Billerica, MA, USA) at working frequencies 600 and 150 MHz, and a Bruker AV-300 spectrometer at working frequencies 300 and 75 MHz, respectively. Chemical shifts are expressed in parts per million (δ) values and coupling constants (J) in Hertz. LREIMS were recorded on a Thermo-DSQ instrument (Thermo Fisher Scientific, New York, NY, USA), while HREIMS were measured on a Agilent 6210 TOFMS instrument (Agilent Technologies, Palo Alto, CA, USA). The cell proliferation assay was undertaken by a MTT method using 96-well plates on MLLTISKAN MK3 analysis spectrometer (Thermo Scientific, Shanghai, China). Annexin V assay was performed using FACS Calibur flow cytometry (Becton Dickinson, Biosciences, Franklin Lakes, NJ, USA).
Compounds
3,
16 were prepared according to the procedures in the literature [
7].
3.1.1. 3-Acetoxy-7-aza-B-homocholest-4-ene-6-one (4)
The solution of thionyl chloride (1.5 mL) in 5 mL dry THF was added to a solution of the oxime 3 (300 mg) in dry THF (30 mL). The solution was stirred under anhydrous condition for 1 h at 0 °C. The reaction was terminated and water was added to the solution. The solution was neutralized with ammonia and the product was extracted with CH2Cl2 (20 × 3 mL). The combined extract was washed with water, 5% NaHCO3, and saturated brine, dried over anhydrous Na2SO4 and evaporated under reduced pressure to give a crude product which was chromatographed on silica gel (elution: Dichloromethane/methanol = 20/1) to give white solid 216 mg. Yield: 72%, mp 157–159 °C; IR (KBr) ν: 3195, 2938, 1740, 1659, 1470, 1377, 1230, 1136, 1230, 1058 cm−1; 1H NMR (CDCl3, 300MHz) δ: 0.70 (3H, s, 18-CH3), 0.86 (3H, d, J = 6.6, 26- or 27-CH3), 0.88 (3H, d, J = 6.6, 26- or 27-CH3), 0.91 (3H, d, J = 6.6, 21-CH3), 1.21 (3H, s, 19-CH3), 2.14 (3H, s, -OCOCH3), 3.16-3.21 (2H, m, C7a-H), 5.53 (1H, dd, J = 12.3, 4.5, C3-αH), 5.91 (1H, s, C4-H), 6.72 (1H, s, 6-NH); 13C NMR (CDCl3, 75MHz) δ: 169.9 (6-C), 169.8 (COCH3), 156.7 (5-C), 114.7 (4-C), 71.2 (3-C), 56.0 (17-C), 55.5 (14-C), 53.3 (9-C), 45.5 (7a-C), 42.9 (13-C), 42.6 (24-C), 39.6 (12-C), 39.5 (8-C), 36.6 (10-C), 36.1 (22-C), 35.7 (20-C), 34.6 (1-C), 28.1 (16-C), 28.0 (25-C), 24.1 (15-C), 23.8 (2-C), 22.8 (26 or 27-C), 22.5 (26 or 27-C), 21.8 (23-C), 21.4 (11-C), 21.3 (CH3CO), 20.8 (19-C), 18.6 (21-C), 12.0 (18-C); LRESI-MS(m/z): 458.3 [M + H]+ (Supplementary Information: Figure S1).
3.1.2. 3-Hydroxy-7-aza-B-homocholest-4-ene-6-one (5)
20 mL of K2CO3 solution (13%) was added to a solution of compound 4 (1520 mg, 3.32 mmol) in CH3OH (200 mL). The reaction mixture was heated under reflux condition for 1 h. After completion of the reaction as indicated by TLC, the solvent was removed under reduced pressure. 200 mL of CH2Cl2 was added to dissolve solid and the resulting solution was washed with cold water and saturated brine. After drying over anhydrous sodium sulfate, the solvent was removed under reduced pressure, and the resulting crude product was purified by chromatography on silica gel using methanol/dichloromethane (30:1) as elution to give 988 mg of 5 as white solid. Yield: 65%, mp 233–234 °C; IR (KBr) ν: 3366, 2941, 2872, 1654, 1593, 1470, 1380, 1319, 1123, 898 cm−1; 1H NMR (CDCl3, 600MHz) δ: 0.71 (3H, s, 18-CH3), 0.88 (3H, d, J = 6.6, 26- or 27-CH3), 0.89 (3H, d, J = 6.6, 26- or 27-CH3), 0.92 (3H, d, J = 6.6, 21-CH3), 1.15 (3H, s, 19-CH3), 2.16-2.20 (1H, m, C2-H), 2.23 (1H, br s, -OH), 3.16-3.21 (1H, m, C7a-αH), 3.28-3.23 (1H, m, C7a-βH), 4.43 (1H, t, J = 5.4, C3-αH), 6.21 (1H, s, C4-H), 6.38 (1H, s, 7-NH); 13C NMR (CDCl3, 75MHz) δ: 170.7 (6-C), 162.1 (5-C), 114.3 (4-C), 69.6 (3-C), 56.0 (17-C), 55.6 (14-C), 53.1 (9-C), 45.0 (7a-C), 42.7 (13-C), 42.6 (12-C), 39.6 (24-C), 39.5 (8-C), 36.7 (10-C), 36.1 (22-C), 35.7 (20-C), 34.7 (1-C), 28.2 (2-C), 28.1 (16-C), 28.0 (25-C), 24.2 (15-C), 23.8 (23-C), 22.8 (26 or 27-C), 22.6 (26 or 27-C), 21.8 (11-C), 21.7 (19-C), 18.6 (21-C), 12.0 (18-C); HRESI-MS(m/z): 416.3515 [M + H]+ (calcd for C27H46NO2, 416.3529) (Supplementary Information: Figure S2).
3.1.3. 7-Aza-B-homocholest-4-ene-3,6-dione (6)
Jones reagent was added dropwise to the solution of 5 (1 mmol) in 20 mL of acetone in 10 min at room temperature until the reaction solution didn’t fade. The mixture was stirred at room temperature for 1 h, and then neutralized with 10% K2CO3 solution. The suspension was poured over a silica gel column and eluted with ethyl acetate. The solvent was removed under reduced pressure. The residue was purified by chromatography on silica gel using methanol/dichloromethane (30:1) as elution to give 270 mg of 6 as white solid. Yield: 65%, mp 209–210 °C. IR (KBr) ν: 3509, 2933, 2868, 1695, 1658, 1605, 1462, 1376, 1249, 1074 cm−1; 1H NMR (CDCl3, 600MHz) δ: 0.72 (3H, s, 18-CH3), 0.88 (3H, d, J = 6.6, 26-CH3 or 27-CH3), 0.89 (3H, d, J = 6.6, 26 or 27-CH3), 0.93 (3H, d, J = 6.6, 21-CH3), 1.06 (3H, s, 19-CH3), 2.11-2.03 (2H, m, C2-H and C1-H), 2.62 (1H, dd, J = 15, 4.5, C2-H), 3.25-3.21 (1H, m, C7a-αH), 3.35-3.30 (1H, m, C7a-βH), 6.00 (1H, s, C4-H), 6.43 (1H, s, -NH); 13C NMR (75MHz, CDCl3) δ: 205.3 (3-C), 168.7 (6-C), 156.8 (5-C), 121.5 (4-C), 56.4 (17-C), 55.9 (14-C), 51.2 (9-C), 47.1(13-C), 45.9(7a-C), 42.7 (12-C), 40.4 (24-C), 39.5 (8-C), 39.3 (10-C), 36.9(22-C), 36.0 (20-C), 35.7 (1-C), 35.3 (2-C), 28.0 (16-C), 28.0 (25-C), 23.9 (15-C), 23.8 (23-C), 22.8 (26 or 27-C), 22.5 (26-C or 27-C), 21.9 (11-C), 20.5 (19-C), 18.6 (21-C), 12.0 (18-C); HRESI-MS (m/z): 414.3362 [M + H]+ (calcd for C27H44NO2, 414.3372) (Supplementary Information: Figure S3).
3.1.4. 3-Hydroximino-7-aza-B-homocholest-4-ene-6-one (7)
CH3COONa.3H2O (120 mg, 0.33 mmol) and NH2OH.HCl (23 mg, 0.33 mmol) were added to the solution of 120 mg (0.27 mmol) 6 in 20 mL 95% ethanol. After the solution was heated to 60 °C, the mixture was stirred at the temperature for 1 h. Then the reaction was terminated and the majority of solvent was evaporated under reduced pressure. Water was added into the reaction mixture, and the product was extracted with ethyl acetate (20 × 3 mL). The combined extracts were washed with saturated brine, dried, and evaporated under reduced pressure. The residue was subjected to chromatography using petroleum ether/ethyl acetate (5:1) as the eluent to give 50 mg of 7 as white solid. Yield: 42%, mp 285–286 °C. IR (KBr) ν: 3317, 2941, 2859, 2348, 1645, 1600, 1449, 1367, 1257, 963, 930 cm−1; 1H NMR (d6-DMSO, 600MHz) δ: 0.70 (3H, s, 18-CH3), 0.88 (3H, d, J = 6.6, 26- or 27-CH3), 0.89 (3H, d, J = 6.6, 26- or 27-CH3), 0.92 (3H, d, J = 6.6, 21-CH3), 1.08 (3H, s, 19-CH3), 2.07-1.99 (2H, m, ), 3.21-3.16 (1H, m, C7a-αH), 3.38-3.33 (1H, m, C7a-βH), 3.45 (1H, dd, J = 15.3, 4.8, C2-βH), 6.11 (1H, d, J = 1.8, C4-H), 6.33 (1H, s, -NH), 9.50 (H, s, =N-OH); 13C NMR (d6-DMSO, 75MHz) δ: 168.0 (6-C), 158.6 (3-C), 153.3 (5-C), 121.3 (4-C), 56.1 (17-C), 55.9 (14-C), 51.2 (9-C), 44.8 (7a-C), 42.6 (13-C), 36.0 (10-C), 35.5 (22-C), 33.9 (20-C), 30.4 (1-C), 28.2 (2-C), 27.8 (16-C), 27.8 (25-C), 23.6 (15-C), 23.1 (23-C), 22.9 (26 or 27-C), 21.8 (11-C), 20.7 (19-C), 18.9 (21-C), 12.2 (18-C); HRESI-MS (m/z): 429.3463 [M + H]+ (calcd for C27H45N2O2, 429.3481) (Supplementary Information: Figure S4).
Compounds in
Scheme 2 were prepared similarly according to the procedure of
Scheme 1.
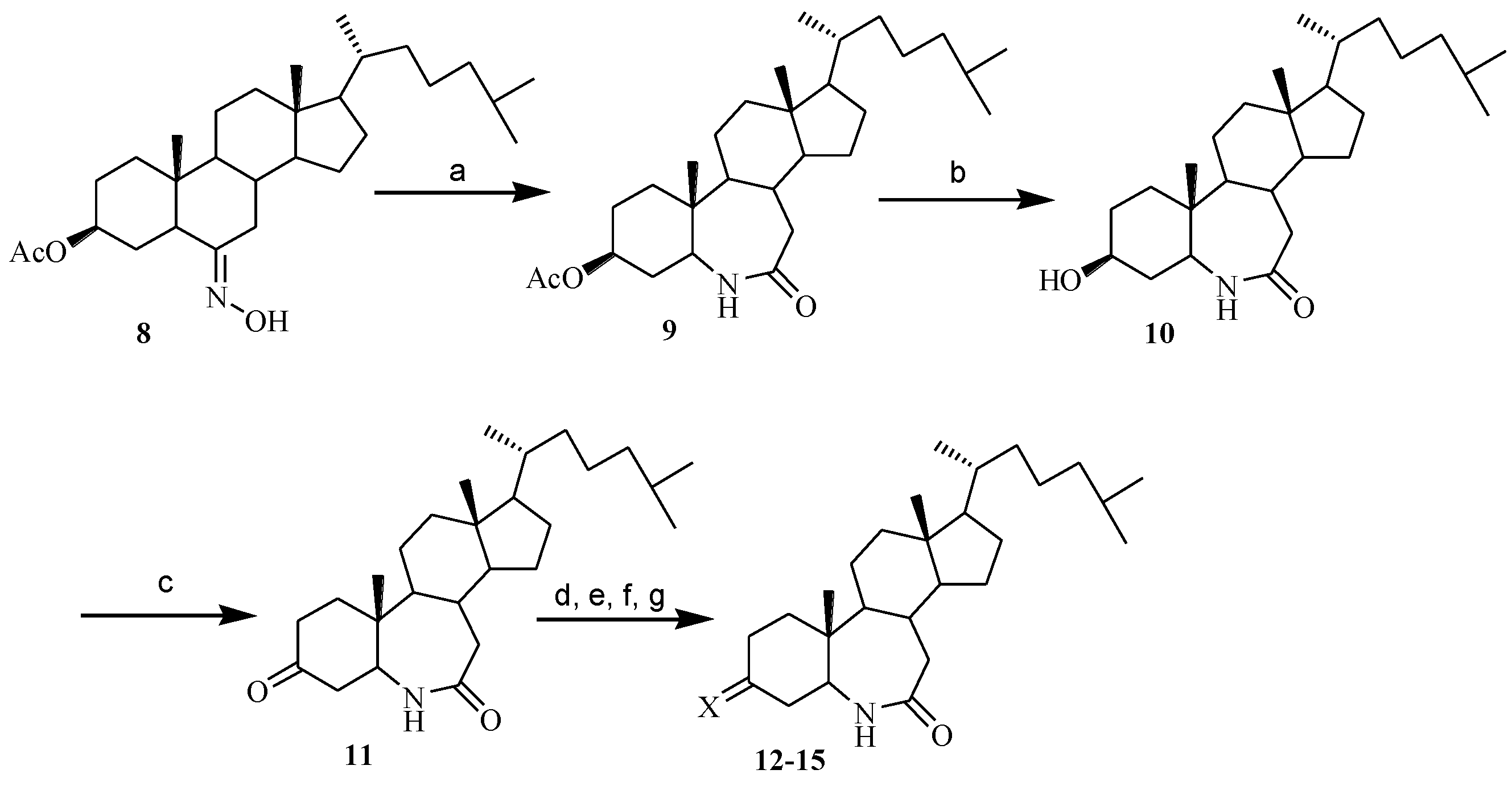
3.1.5. The Synthesis of 3-Acetoxy-6-aza-B-homocholest-7-one (9)
White solid, yield 93%, mp 222–223 °C; IR(KBr) ν: 3346, 2974, 2945, 2864, 1719, 1662, 1654, 1474, 1364, 1249 cm−1; 1H NMR(CDCl3, 300MHz) δ: 0.68 (3H, s, 18-CH3), 0.86 (3H, s, 19-CH3), 0.87 (6H, d, J = 6.3, 26- and 27-CH3), 0.90 (3H, d, J = 6.3, 21-CH3), 2.05 (3H, s, CH3CO-), 2.36-2.22 (2H, m, C7a-H), 3.47-3.40 (1H, m, C5-H), 4.70-4.60 (1H, m, C3-αH), 5.54 (1H, d, J = 5.1, N-H); 13C NMR (CDCl3, 75MHz) δ: 176.2 (-CONH), 170.5 (COCH3), 70.9 (3-C), 58.7 (14-C), 56.7 (17-C), 56.4 (5-C), 55.6 (9-C), 42.5 (7a-C), 40.2 (13-C), 39.9 (12-C), 39.5 (24-C), 38.7 (20-C), 35.9 (10-C), 35.7 (22-C), 35.3 (1-C), 34.6 (4-C), 34.5 (8-C), 28.0 (16-C), 27.6 (25-C), 27.0 (2-C), 25.6 (23-C), 23.8 (15-C), 23.0 (11-C), 22.8 (27-C), 22.6 (C-26), 21.3 (CH3CO-), 18.6 (21-C), 12.4 (19-C), 11.8 (18-C); HRESI-MS (m/z): 460.3784 [M + H]+ (calcd for C29H50NO3, 460.3791) (Supplementary Information: Figure S5).
3.1.6. The Synthesis of 3-Hydroxy-6-aza-B-homocholest-7-one (10)
White solid, yield: 76%. mp 234–235 °C. IR (KBr) ν: 3321, 2953, 2864, 1650, 1446, 1384, 1053 cm−1; 1H NMR (CDCl3, 600MHz) δ: 0.67 (3H, s, 18-CH3), 0.84 (3H, s, 19-CH3), 0.85 (3H, d, J = 6.6, 26 or 27-CH3), 0.86 (3H, d, J = 6.6, 26 or 27-CH3), 0.88 (3H, d, J = 6.6, 21-CH3), 2.24 (1H, t, J = 13.8, C7a-H), 2.33 (1H, d, J = 13.8, C7a-H), 3.38-3.34 (1H, m, C5-H), 3.61-3.57 (1H, m, C3-H), 5.30 (1H, s, N-H); 13C NMR (CDCl3, 75MHz) δ: 176.3 (7-C), 68.8 (3-C), 58.8 (14-C), 56.9 (17-C), 56.5 (5-C), 55.7 (9-C), 42.5 (7a-C), 40.3 (13-C), 39.9 (12-C), 39.5 (24-C), 38.6 (4-C), 36.0 (10-C), 35.7 (22-C), 35.6 (20-C), 34.6 (1-C), 30.9 (8-C), 28.0 (2-C), 28.0 (25-C), 27.6 (16-C), 25.6 (23-C), 23.8 (15-C), 23.0 (11-C), 22.8 (27-C), 22.6 (26-C), 18.6 (21-C), 12.5 (19-C), 11.8 (18-C); LRESI-MS (m/z): 418.3 [M + H]+(Supplementary Information: Figure S6).
3.1.7. The Synthesis of 6-Aza-B-homocholest-3,7-dione (11)
White solid, yield: 80%. mp 238–240 °C. IR (KBr) ν: 3346, 2966, 2864, 1723, 1658, 1458 cm−1; 1H NMR (CDCl3, 300MHz) δ: 0.72 (3H, s, 18-CH3), 0.88 (6H, d, J = 6.6, 26 and 27-CH3), 0.91 (3H, d, J = 6.6, 21-CH3), 1.07 (3H, s, 19-CH3), 2.52-2.22 (6H, m, C2-, C4- and C7a-H), 3.78-3.66 (1H, m, C5-H), 6.10 (1H, s, N-H); 13C NMR (CDCl3, 75MHz) δ: 207.8 (3-C), 176.4 (7-C), 58.5 (14-C), 58.0 (17-C), 56.5 (9-C), 55.7 (5-C), 43.7 (13-C ), 42.5 (7a-C), 40.2 (4-C), 39.9 (12-C), 39.5 (24-C), 39.0 (10-C), 36.8 (2-C), 36.0 (20-C), 35.9 (22-C), 35.7 (1-C), 34.7 (8-C), 28.0 (16-C), 27.6 (25-C), 25.6 (23-C), 23.8 (15-C), 23.6 (11-C), 22.8 (27-C), 22.5 (26-C), 18.6 (21-C), 12.2 (19-C), 11.8 (18-C); HRESI-MS (m/z): 416.3523 [M + H]+ (calcd for C27H46NO2, 416.3529) (Supplementary Information: Figure S7).
3.1.8. The Synthesis of 3-Hydroximino-6-aza-B-homocholest-7-one (12)
White solid, yield: 85%, mp 248–249 °C. IR (KBr) ν: 3235, 2945, 1650, 1458, 1433, 1380, 1119, 972 cm−1; 1H NMR (CDCl3, 300MHz) δ: 0.70 (3H, s, 18-CH3), 0.84 (3H, s, 19-CH3), 0.85 (3H, d, J = 6.6, 26 or 27-CH3), 0.86 (3H, d, J = 6.6, 26 or 27-CH3), 0.94 (3H, d, J = 6.6, 21-CH3), 3.58-3.44 (1H, m, C5-H), 6.19 (1H, s, N-H); 13C NMR (CDCl3, 75MHz) δ: 176.9 (7-C), 157.5 (3-C), 58.4 (14-C), 57.4 (17-C), 56.5 (5-C), 55.7 (9-C), 42.5 (7a-C), 40.1 (13-C), 39.9 (10-C), 39.5 (12-C), 37.0 (24-C), 36.0 (22-C), 35.7 (20-C), 34.6 (8-C), 29.6 (4-C), 28.0 (16-C), 27.6 (25-C), 27.0 (2-C), 25.6 (1-C), 23.8 (23-C), 23.2 (15-C), 22.8 (26-C), 22.6 (27-C), 22.1 (11-C), 18.6 (21-C), 12.0 (19-C), 11.8 (18-C); HRESI-MS (m/z): 431.3630 [M + H]+ (calcd for C27H47N2O2, 431.3638).
3.1.9. The Synthesis of 3-O-Methyloximino-6-aza-B-homocholest-7-one (13)
Compounds 13 and 14 were prepared similarly according to the procedure of 11, but CH3ONH2·HCl and PhCH2ONH2·HCl were used as reagents instead of NH2OH·HCl.
Compound 13 was a mixture of (E) and (Z)-13 (ratio: E:Z = 0.9:1.1). Yield: 68%, mp 204–205 °C. IR (KBr) ν: 3425, 3222, 1954, 2869, 1673, 1048, 1016 cm−1; 1H NMR (CDCl3, 300MHz) δ: 0.66 (3H, s, 18-CH3), 0.83 (3H, d, J = 6.6, 26-CH3 or 27-CH3), 0.84 (3H, d, J = 6.6, 26-CH3 or 27-CH3), 0.86 (3H, d, J = 6.6, 21-CH3), 0.91 (3H, s, 19-CH3), 3.50-3.38 (1H, m, C5-H), 3.01 (Z: 0.55H, br d, J = 14.7, C4-H), 3.31 (E: 0.45H, ddd, J = 14.4, 5.4, 1.5, C2-βH), 3.786 (Z: 3H, s, -OCH3), 3.789 (E: 3H, s, -OCH3), 6.17 (Z: 0.55H, br s, N-H), 6.23 (0.45H, br s, N-H); 13C NMR (CDCl3, 75MHz) δ: 176.6 (7-C), 157.0 (3-C, E-), 156.3 (3-C, Z-), 61.2 (-OCH3), 58.6 (5-C, Z-), 58.4 (14-C), 57.4 (5-C, E-), 56.4 (17-C), 55.6 (9-C), 42.4 (7a-C), 40.2 (13-C), 40.1 (10-C), 39.9 (12-C), 39.4 (24-C), 37.2 (22-C), 36.2 (20-C), 35.9 (4-C, Z-), 35.7(4-C, E-), 34.7 (8-C, Z-), 34.3 (8-C, E-), 28.0 (16-C), 27.7 (2-C, E-), 27.6 (25-C), 27.1 (2-C, Z-), 25.6 (1-C), 23.7 (23-C), 23.1 (15-C), 22.8 (27-C), 22.6 (26-C), 20.8 (11-C), 18.6 (21-C, E-), 12.1 (19-C, Z-), 12.0 (19-C), 11.8 (18-C); HRESI-MS (m/z): 445.3788 [M + H]+ (calcd for C28H49N2O2, 445.3794) (Supplementary Information: Figure S8).
3.1.10. The Synthesis of 3-O-Benzyloximino-6-aza-B-homocholest-7-one (14)
Compound 14 was a mixture of (E) and (Z)-14 (ratio: E:Z = 0.9:1.1). Yield: 63%, mp 195–196 °C. IR(KBr) ν : 3424, 3224, 3088, 3031, 2950, 2868, 1672, 1452, 1366, 1351, 1048, 1015, 734, 697 cm−1; 1H NMR (CDCl3, 300MHz) δ: 0.68 (3H, s, 18-CH3), 0.858 (3H, d, J = 6.6, 26-CH3 or 27-CH3), 0.863 (3H, d, J = 6.6, 26-CH3 or 27-CH3), 0.89 (3H, d, J = 6.6, 21-CH3), 0.93 (3H, s, 19-CH3), 3.13 (0.5H, br d, J = 15.0, C4-H, E-), 3.42-3.36 (0.5H, m, C2-H, Z-), 3.52-3.42 (1H, m, C5-H), 5.05 (2H, s, -OCH2Ph), 5.90 (1H, s, N-H), 7.36-7.30 (5H, m, -C6H5); 13C NMR (CDCl3, 75MHz): 176.4 (7-C), 157.6 (E: 3-C), 156.9 (Z: 3-C), [Z: 137.9, 128.4, 128.4, 128.1, 128.1, 127.8, (-C6H5)], [E: 137.7, 128.4, 128.4, 128.0, 128.0, 127.7, (-C6H5)], 75.5 (-OCH2Ph), 58.5 (Z: 5-C), 58.3 (14-C), 57.4 (E: 5-C), 56.4 (17-C), 55.7 (9-C), 42.5 (7a-C), 42.4 (13-C), 39.9 (10-C), 39.5 (12-C), 37.2 (24-C), 36.2 (22-C), 35.9 (20-C), 35.7 (8-C), 34.7 (Z: 4-C), 34.4 (E: 4-C), 28.0 (16-C), 28.0 (25-C), 27.6 (E: 2-C), 27.1 (Z: 2-C), 25.6 (1-C), 23.8 (22-C), 23.1 (15-C), 22.8 (26-C), 22.6 (27-C), 21.1 (11-C), 18.6 (21-C), 12.1 (E: 19-C), 12.0 (Z: 19-C), 11.8 (18-C); HRESI-MS (m/z): 521.4102 [M + H]+ (calcd for C34H53N2O2, 521.4107) (Supplementary Information: Figure S9).
3.1.11. The Synthesis of 6-Aza-7-oxo-B-homocholest-3-thiosemicarbazone (15)
A mixture of compound 11 (100 mg, 0.24 mmol), thiosemicarbazide (24 mg, 0.26 mmol), and a few drops of glacial acetic acid (0.5 mL) in 95% ethanol (20 mL) was stirred at 60–70 °C for 10 h. After completion of the reaction, the majority of solvent was evaporated and some water was added to this solution. The mixture was extracted with CH2Cl2 and the extract was washed with saturated brine, dried with anhydrous sodium sulfate and evaporated under reduce pressure. The resulting residue was chromatographed on a column of silica gel with a mixture of DCM-methanol (20:1) to give 80 mg of compound 15. Yield: 70%, mp 274–276 °C. Compound 15 was a mixture of (E)- and (Z)-15 (ratio: E:Z = 1:1). IR (KBr) ν: 3424, 3237, 2951, 2668, 1658, 1595, 1467, 1386, 1284, 1134, 1078 cm−1; 1H NMR (DMSO, 600 MHz) δ: 0.64 (3H, s, 18-CH3), 0.814 (3H, d, J = 6.6, 26 or 27-CH3), 0.818 (3H, d, J = 6.6, 26 or 27-CH3), 0.84 (1.5H, s, 19-CH3), 0.85 (1.5H, s, 19-CH3), 0.92 (3H, d, J = 6.0, 21-CH3), 2.69 (0.5H, d, J = 12.6, C2-H, E-), 2.81 (1H, br s, C4-H, Z-), 2.97 (0.5H, d, J = 10.8, C4-H, Z-), 3.51 (0.5H, J = 4.8, C5-H, E-), 3.57 (0.5H, J = 4.8, C5-H, Z-), 7.13 (1H, br s, -NH2), 7.29 (0.5H, br s, CON-H-), 7.36 (1H, br s, -NH2), 7.43 (0.5H, br s, CON-H-), 9.57 (0.5H, br s, -NH-, E-), 10.20 (0.5H, br s, -NH-, Z-); 13C NMR (DMSO, 150 MHz) δ: 178.5 (C=S), 177.4 (7-C, Z-), 177.1 (7-C, E-), 156.3 (3-C, Z-), 155.1 (3-C, E-), 59.0 (5-C, Z-), 58.3 (5-C, E-), 58.2 (14-C), 56.5 (17-C), 55.6 (9-C), 42.6 (7a-C), 40.3 (10-C, Z-), 40.2 (10-C, E-), 39.9 (13-C), 39.6 (12-C), 39.4 (24-C), 37.7 (4-C, Z-), 37.2 (4-C, E-), 36.2 (2-C, E-), 36.0 (22-C), 35.8 (20-C), 34.7 (8-C), 30.6 (2-C, Z-), 28.1 (16-C), 27.7 (1-C), 25.8 (25-C), 23.9 (23-C), 23.4 (15-C), 23.3 (11-C), 22.7 (27-C), 22.9 (26-C), 18.7 (21-C), 12.6 (19-C, Z-), 12.4 (19-C, E-), 12.0 (18-C); HRESI-MS (m/z): 489.3620 [M + H]+ (calcd for C28H49N4OS, 489.3627) (Supplementary Information: Figure S10).
Compounds in
Scheme 3 were prepared similarly according to the procedure of
Scheme 1.
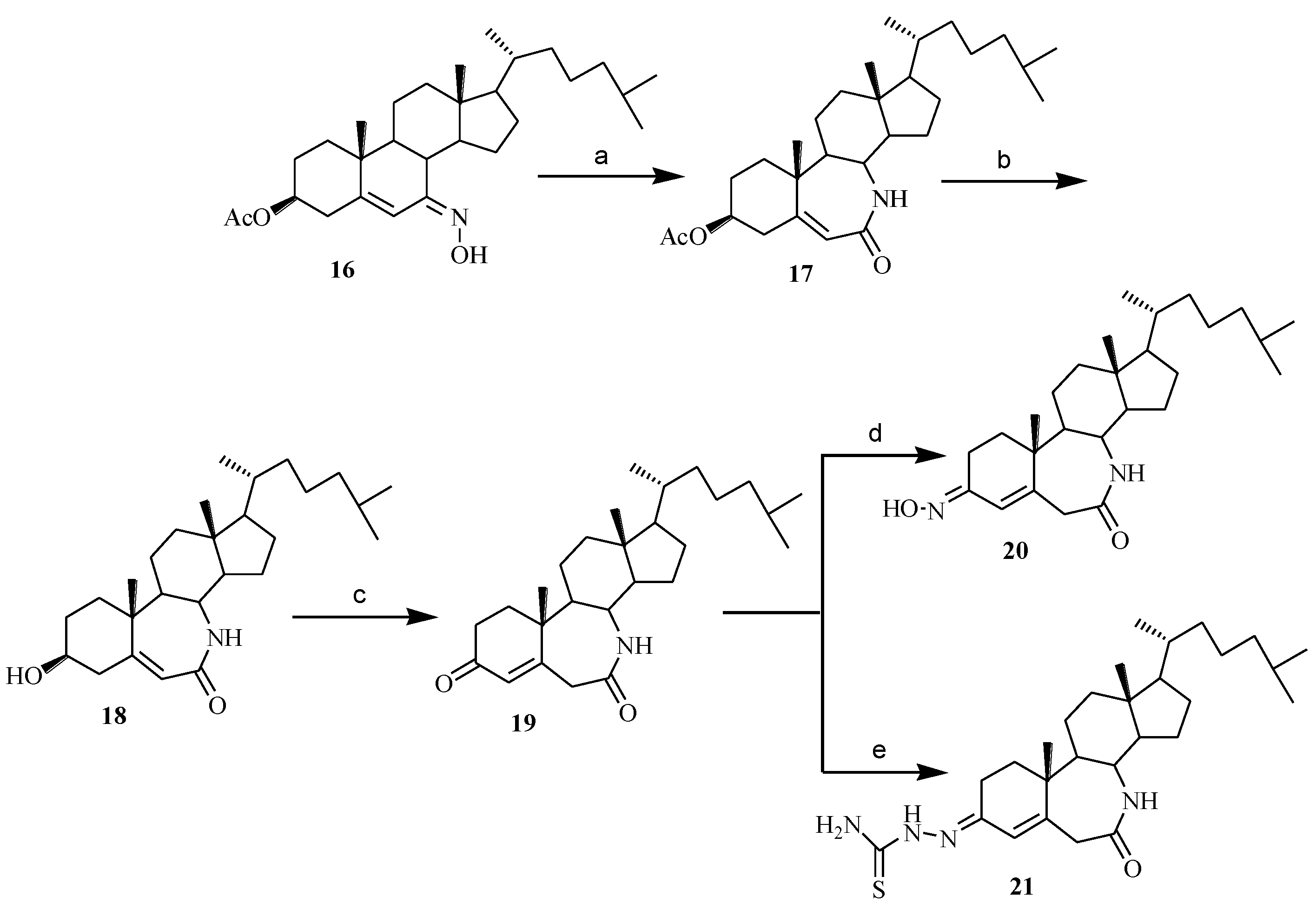
3.1.12. 3-Acetoxy-7a-aza-B-homocholest-5-ene-7-one (17)
Yellow solid, yield: 72%, mp192–194 °C. IR (KBr) ν: 3424, 2942, 2868, 1740, 1659, 1617, 1442 cm−1; 1H NMR (300 MHz, CDCl3) δ: 0.69 (3H, s, 18-CH3), 0.869 (3H, d, J = 6.6, 26 or 27-CH3), 0.874 (3H, s, J = 6.6, 26 or 27-CH3), 0.92 (3H, d, J = 6.6, 21-CH3), 1.27 (3H, s, 19-CH3), 2.05 (3H, s, 2′-CH3), 2.46 (1H, dd, J = 13.2, 3.6, C4-βH), 2.61 (1H, t, J = 11.7, C4-αH), 3.29 (1H, t, J = 8.4, C8-H), 4.76-4.65 (1H, m, C3-αH), 5.66 (1H, br s, -NH-), 5.83 (1H, s, C6-H); 13C NMR (75 MHz, CDCl3) δ: 170.3 (1′-C), 167.4 (7-C), 156.1 (5-C), 122.5 (6-C), 72.5 (3-C), 55.7 (14-C), 55.4 (17-C), 51.7 (8-C), 48.9 (9-C), 43.8 (24-C), 41.8 (12-C), 41.6 (13-C), 39.4 (15-C), 38.6 (10-C), 35.9 (4-C), 35.5 (1-C), 35.4 (22-C), 28.0 (20-C), 27.8 (25-C), 27.0 (2-C), 25.1 (16-C), 23.8 (23-C), 23.2 (11-C), 22.8 (26-C), 22.5 (27-C), 21.3 (2′-C), 19.3 (21-C), 18.6 (19-C), 11.4 (18-C); HRESI-MS (m/z): 458.3623 [M + H]+ (calcd for C29H48NO3, 458.3634) (Supplementary Information: Figure S11).
3.1.13. 3-Hydroxy-7a-aza-B-homocholest-5-ene-7-one (18)
Yellow solid, yield: 74.5%, mp 207–209 °C; IR (KBr) ν: 3383, 2946, 2860, 1663, 1601, 1438 cm−1; 1H NMR (300 MHz, CDCl3) δ: 0.65 (3H, s, 18-CH3), 0.837 (3H, d, J = 6.6, 26 or 27-CH3), 0.842 (3H, s, J = 6.6, 26 or 27-CH3), 0.89 (3H, d, J = 6.6, 21-CH3), 1.24 (3H, s, 19-CH3), 2.40 (1H, dd, J = 13.2, 3.3, C4-βH), 2.53 (1H, t, J = 11.7, 3.6, C4-αH), 3.25 (1H, t, J = 8.1, C8-H), 3.65-3.54 (1H, m, C3-αH), 5.77 (1H, s, C6-H), 6.01 (1H, br s, -NH-); 13C NMR (75 MHz, CDCl3) δ: 168.2 (7-C), 158.6 (5-C), 121.0 (6-C), 70.6 (3-C), 55.6 (14-C), 55.1 (17-C), 51.6 (8-C), 49.1 (9-C), 46.3 (13-C), 44.2 (4-C), 41.7 (24-C), 39.4 (12-C), 38.5 (15-C), 36.0 (1-C), 35.9 (22-C), 35.5 (20-C), 30.5 (10-C), 28.0 (2-C), 27.8 (25-C), 25.1 (16-C), 23.8 (23-C), 23.1 (11-C), 22.8 (26-C), 22.5 (27-C), 19.4 (21-C), 18.6 (19-C), 11.3 (18-C); HRESI-MS (m/z): 416.3515 [M + H]+ (calcd for C27H46NO2, 416.3529) (Supplementary Information: Figure S12).
3.1.14. 7a-Aza-B-homocholest-4-ene-3,7-dione (19)
Faint yellow solid, yield: 43.0%, mp 203–205 °C; IR (KBr) ν: 3367, 2958, 2860, 1675, 1462 cm−1; 1H NMR (300 MHz, CDCl3) δ: 0.72 (3H, s, 18-CH3), 0.845 (3H, d, J = 6.6, 26 or 27-CH3), 0.850 (3H, s, J = 6.6, 26 or 27-CH3), 0.90 (3H, d, J = 6.6, 21-CH3), 1.27 (3H, s, 19-CH3), 2.37-2.28 (2H, m, C2-H), 2.94 (1H, d, J = 12.6, C6-βH), 3.55 (1H, d, J = 12.6, C6-αH), 3.52-3.46 (1H, m, C8-H), 5.82 (1H, br s, -CONH-), 5.93 (1H, s, C4-H); 13C NMR (75 MHz, CDCl3) δ: 198.4 (3-C), 172.5 (7-C), 162.8 (5-C), 126.9 (4-C), 55.8 (17-C), 54.0 (14-C), 52.4 (8-C), 49.2 (9-C), 42.7 (6-C), 41.6 (13-C), 41.3 (24-C), 39.4 (12-C), 38.2 (10-C), 35.9 (15-C), 35.5 (1-C), 34.1 (22-C), 33.4 (20-C), 28.0 (2-C), 27.9 (25-C), 27.7 (16-C), 24.8 (23-C), 23.8 (11-C), 22.8 (26-C), 22.6 (27-C), 21.2 (21-C), 18.6 (19-C), 11.7 (18-C); HRESI-MS (m/z): 414.3362 [M + H]+ (calcd for C28H47N4OS, 414.3372) (Supplementary Information: Figure S13).
3.1.15. 3-Hydroximino-7a-aza-B-homocholest-4-ene-7-one (20)
Faint yellow solid, yield: 77.3%, mp 204–206 °C. IR (KBr) ν: 3268, 2949, 2868, 1658, 1657, 1462, 1366 cm−1; 1H NMR (CDCl3, 300MHz) δ: 0.73 (3H, s, 18-CH3), 0.88 (6H, d, J = 6.6, 26- and 27-CH3), 0.93 (3H, d, J = 6.6, 21-CH3), 1.22 (3H, s, 19-CH3), 2.33-2.22 (2H, m, C2-H), 2.79 (1H, br d, J = 17.4, C2-H), 2.94 (1H, d, J = 13.5, C6-βH), 3.47 (1H, d, J = 13.5, C6-αH), 3.52-3.45 (1H, m, C8-H), 5.77 (1H, br s, -CONH-), 6.12 (1H, s, C4-H), 6.84 (1H, s, -OH); 13C NMR (CDCl3, 75MHz) δ: 174.3 (7-C), 156.1 (3-C), 147.5 (5-C), 121.8 (4-C), 55.9 (14-C), 54.4 (17-C), 52.5 (8-C), 50.0 (13-C), 49.1 (9-C), 42.8 (6-C), 41.0 (24-C), 40.6 (10-C), 39.4 (12-C), 38.5 (15-C), 35.9 (22-C), 35.5 (20-C), 33.0 (1-C), 29.7 (25-C), 28.0 (2-C), 27.7 (16-C), 24.9 (23-C), 23.8 (11-C), 22.8 (26-C), 22.5 (27-C), 21.0 (19-C), 18.6 (21-C), 11.7 (18-C); HRESI-MS (m/z): 429.3463 [M + H]+ (calcd for C27H45N2O2, 429.3481).
3.1.16. 7-Oxo-7a-aza-B-homocholest-4-ene-3-thiosemicarbazone (21)
Faint yellow solid, yield: 31%, mp 230-231 °C; IR(KBr) ν: 3436, 2954, 2856, 1659, 1573, 1409 cm−1; 1 H NMR (CDCl3, 300MHz) δ: 0.75 (3H, s, 18-CH3), 0.88 (3H, d, J = 6.6, 26 or 27-CH3), 0.89 (3H, d, J = 6.6, 26 or 27-CH3), 0.94 (3H, d, J = 6.3, 21-CH3), 1.26 (3H, s, 19-CH3), 2.97 (1H, d, J = 13.2, C6-βH), 3.51 (1H, d, J = 13.2, C6-αH), 3.56-3.48 (1H, m, C8-H), 5.28 (1H, br s, -CONH-), 6.10 (1H, s, C4-H), 6.37 (1H, br s, -NH2), 7.25 (1H, br s, -NH2), 8.64 (1H, s, -NH-CS); 13C NMR (CDCl3, 75MHz) δ: 178.7 (C=S), 173.6 (7-C), 150.5 (5-C), 148.9 (3-C), 124.2 (4-C), 55.9 (14-C), 54.3 (17-C), 52.5 (8-C), 49.2 (9-C), 42.8 (6-C), 41.1 (13-C), 40.5 (24-C), 39.4 (12-C), 38.4 (15-C), 35.9 (22-C), 35.5 (20-C), 32.8 (10-C), 29.7 (1-C), 28.0 (25-C), 24.9 (16-C), 23.8 (23-C), 22.8 (26-C), 22.5 (27-C), 22.4 (2-C), 21.0 (11-C), 20.0 (21-C), 18.6 (19-C), 11.7 (18-C); HRESI-MS (m/z): 487.3491 [M + H]+ (calcd for C28H47N4OS, 487.3471) (Supplementary Information: Figure S14).