New Spirotetronate Antibiotics, Lobophorins H and I, from a South China Sea-Derived Streptomyces sp. 12A35
Abstract
:1. Introduction
2. Results and Discussion
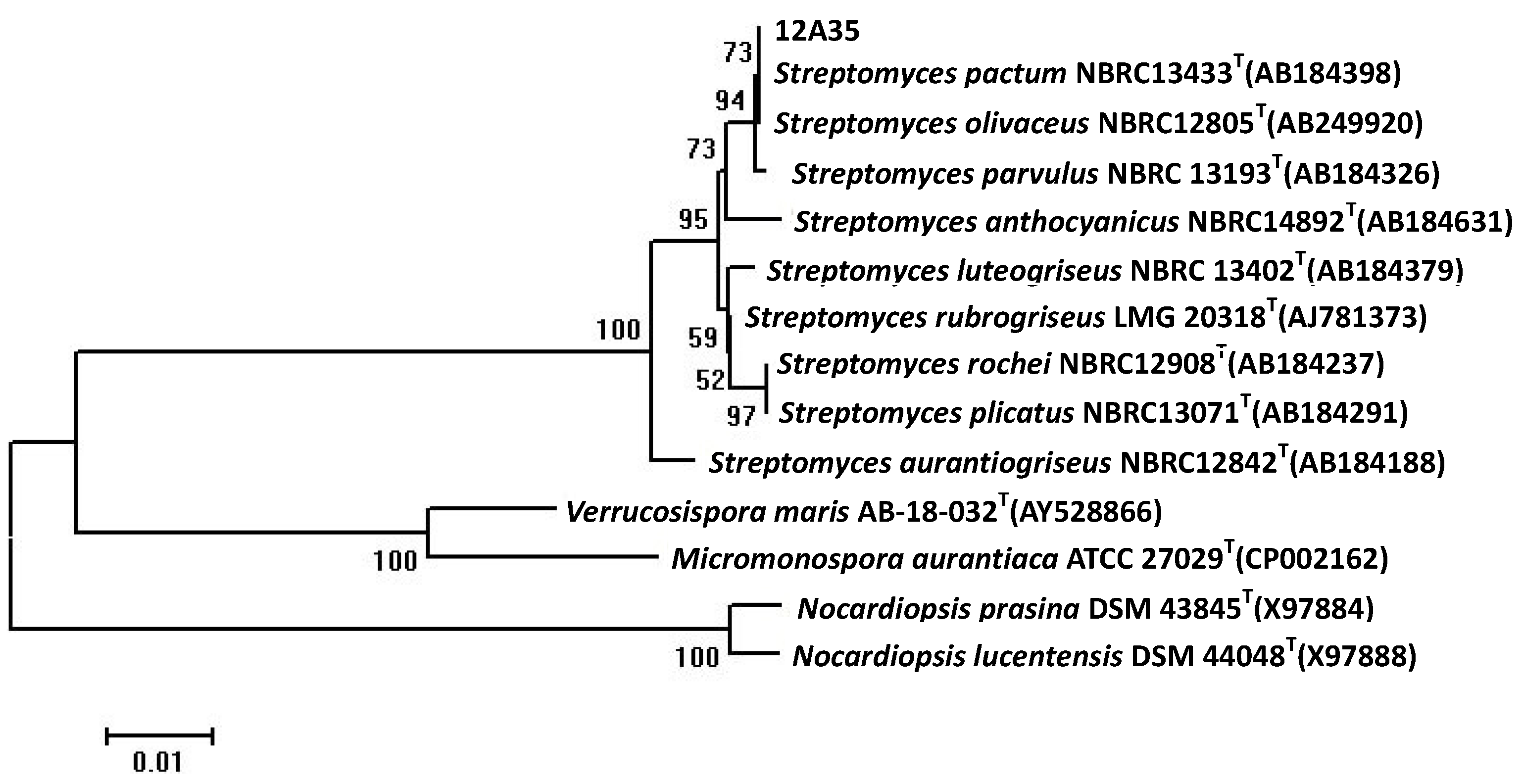
2.1. Taxonomy of the Strain 12A35
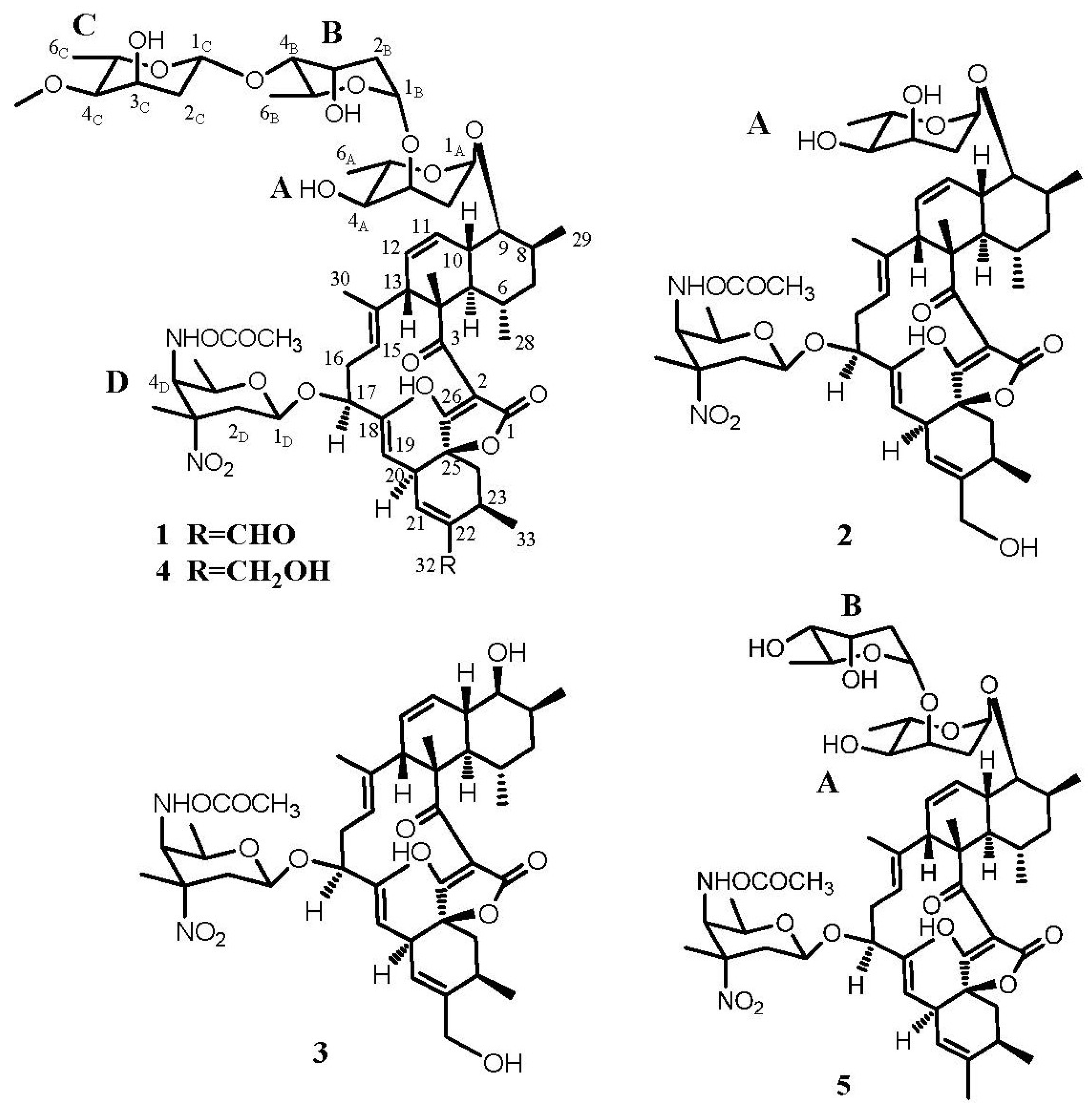
2.2. Structure Determination
| Position | 1 | 2 | 3 | 4 | Lobophorin F [6] | ||
|---|---|---|---|---|---|---|---|
| δC | δH ( J in Hz) | δC | δH ( J in Hz) | δC | δC | δC | |
| 1 | 166.71 | 167.24 | 167.12 | 167.23 | 167.3 | ||
| 2 | 101.68 | 101.85 | 101.89 | 101.76 | 101. 9 | ||
| 3 | 206.47 | 206.07 | 206.43 | 206.33 | 206.3 | ||
| 4 | 50.93 | 50.90 | 51.02 | 50.92 | 51.0 | ||
| 5 | 43.11 | 2.00 (m) | 43.20 | 1.99 (m) | 42.79 | 43.10 | 43.4 |
| 6 | 31.28 | 1.61 (m) | 31.29 | 1.61 (m) | 31.19 | 31.30 | 31.2 |
| 7 | 41.66 | 1.58 (m), 1.50 (m) | 41.73 | 1.59 (m), 1.51 (m) | 41.72 | 41.69 | 41.9 |
| 8 | 34.37 | 2.22 (m) | 34.98 | 1.89 (m) | 34.71 | 34.39 | 34.5 |
| 9 | 84.08 | 3.44 (m) | 85.57 | 3.55 (dd, 10.9, 5.4) | 76.08 | 84.16 | 86.7 |
| 10 | 38.39 | 2.08 (m) | 38.37 | 2.15 (m) | 39.22 | 38.42 | 38.2 |
| 11 | 125.97 | 5.73 (d, 10.6) | 124.98 | 5.71 (d, 10.9) | 125.51 | 125.84 | 125.9 |
| 12 | 126.29 | 5.38 (m) | 127.18 | 5.38 (d, 9.7) | 126.51 | 126.45 | 126.6 |
| 13 | 53.24 | 3.46 (m) | 52.98 | 3.48 (m) | 53.17 | 53.15 | 53.1 |
| 14 | 135.92 | 135.45 | 135.83 | 135.73 | 135.7 | ||
| 15 | 123.22 | 5.17 (d, 9.2) | 123.59 | 5.16 (d, 9.3) | 123.32 | 123.34 | 123.5 |
| 16 | 30.02 | 2.34 (m), 1.71 (m) | 30.94 | 2.34 (m), 2.25 (m) | 31.08 | 30.03 | 31.2 |
| 17 | 78.69 | 4.22 (m) | 78.33 | 4.19 (m) | 78.47 | 78.36 | 78.9 |
| 18 | 139.46 | 137.09 | 137.03 | 137.08 | 136.7 | ||
| 19 | 116.72 | 5.28 (d, 10.5) | 119.17 | 5.11 (d, 10.9) | 119.23 | 119.19 | 119.9 |
| 20 | 41.24 | 3.80 (d, 11.1) | 40.17 | 3.85 (d, 10.9) | 40.18 | 40.17 | 40.4 |
| 21 | 148.12 | 6.56 (s) | 121.52 | 5.49 (s) | 121.43 | 121.52 | 120.6 |
| 22 | 144.59 | 141.24 | 141.35 | 141.28 | 137.8 | ||
| 23 | 25.36 | 3.01 (m) | 27.87 | 2.66 (m) | 27.93 | 27.95 | 31.9 |
| 24 | 34.80 | 2.36 (m), 1.89 (m) | 35.31 | 2.37 (m), 1.82 (m) | 35.33 | 35.35 | 35.5 |
| 25 | 82.94 | 83.32 | 83.24 | 83.30 | 83.3 | ||
| 26 | 200.97 | 201.37 | 201.49 | 201.63 | 201.7 | ||
| 27 (4-CH3) | 15.13 | 1.62 (s, 3H) | 14.98 | 1.59 (s, 3H) | 15.03 | 15.05 | 15.1 |
| 28 (6-CH3) | 22.23 | 0.64 (d, 4.9) | 22.15 | 0.63 (d, 5.8) | 22.28 | 22.23 | 22.2 |
| 29 (8-CH3) | 14.11 | 1.09 (d, 6.4) | 14.39 | 1.09 (d, 7.0) | 12.99 | 14.12 | 14.6 |
| 30 (14-CH3) | 13.73 | 1.35 (s, 3H) | 13.70 | 1.31 (s, 3H) | 13.70 | 13.70 | 13.8 |
| 31 (18-CH3) | 15.18 | 1.45 (s, 3H) | 15.03 | 1.45 (s, 3H) | 15.11 | 15.09 | 15.1 |
| 32 (22-C) | 193.32 | 9.51 (s) | 64.85 | 4.20 (s, 2H) | 64.93 | 64.89 | 21.8 |
| 33 (23-CH3) | 19.97 | 1.36 (d, 6.4, 3H) | 20.14 | 1.30 (m, 3H) | 20.18 | 20.18 | 20.2 |
| A1 | 98.00 | 4.78 (d, 4.7) | 99.58 | 4.90 (d, 2.7) | 97.98 | 99.1 | |
| A2 | 31.02 | 2.30 (m), 2.35 (m) | 34.05 | 2.25 (m, 2H) | 30.95 | 33.4 | |
| A3 | 66.61 | 4.00 (m) | 67.26 | 3.98 (m) | 66.67 | 74.1 | |
| A4 | 71.80 | 3.26 (dd, 8.7, 3.5) | 72.60 | 3.19 (d, 8.9) | 71.81 | 72.4 | |
| A5 | 64.94 | 4.00 (m) | 65.23 | 3.82 (m) | 64.95 | 65.0 | |
| A6 | 17.72 | 1.26 (d, 6.0, 3H) | 17.69 | 1.30 (m, 3H) | 17.71 | 17.7 | |
| B1 | 90.97 | 5.13 (m) | 90.99 | 96.5 | |||
| B2 | 34.08 | 2.12 (m), 1.91 (m) | 34.07 | 35.3 | |||
| B3 | 65.48 | 4.22 (m) | 65.53 | 66.9 | |||
| B4 | 82.10 | 3.23 (dd, 7.7, 2.3) | 82.1 | 72.5 | |||
| B5 | 62.12 | 3.98 (m) | 62.13 | 65.6 | |||
| B6 | 17.89 | 1.20 (d, 6.0, 3H) | 17.87 | 17.7 | |||
| C1 | 98.28 | 4.91 (dd, 9.3, 2.0) | 98.30 | ||||
| C2 | 36.65 | 2.16 (m), 1.67 (m) | 36.64 | ||||
| C3 | 63.96 | 4.25 (d, 2.6) | 63.96 | ||||
| C4 | 82.10 | 2.84 (dd, 9.2, 2.6) | 82.05 | ||||
| C5 | 68.37 | 3.76 (q, 5.8) | 68.37 | ||||
| C6 | 18.25 | 1.25 (d, 6.0, 3H) | 18.25 | ||||
| C4-OCH3 | 57.37 | 3.41 (s, 3H) | 57.37 | ||||
| D1 | 97.39 | 4.46 (dd, 9.2, 1.5) | 96.98 | 4.42 (dd, 9.8, 1.9) | 97.03 | 96.96 | 97.4 |
| D2 | 35.72 | 2.77 (m), 1.63 (m) | 35.65 | 2.75 (m), 1.59 (m) | 35.71 | 35.71 | 35.8 |
| D3 | 91.09 | 90.98 | 91.01 | 91.12 | 90.9 | ||
| D4 | 53.72 | 4.39 (d, 10.4) | 53.66 | 4.35 (d, 10.1) | 53.66 | 53.67 | 53.9 |
| D5 | 69.12 | 3.48 (m) | 69.07 | 3.47 (d, 7.4) | 69.07 | 69.05 | 68.9 |
| D6 | 16.97 | 1.16 (d, 6.0, 3H) | 16.94 | 1.16 (d, 6.4, 3H) | 16.97 | 16.97 | 17.0 |
| D3-CH3 | 25.22 | 1.59 (s, 3H) | 25.25 | 1.56 (s, 3H) | 25.28 | 25.27 | 25.3 |
| D4 C=O | 157.34 | 157.44 | 157.35 | 157.38 | 157.4 | ||
| D4-OCH3 | 52.74 | 3.72 (s, 3H) | 52.72 | 3.72 (s, 3H) | 52.69 | 52.73 | 52.7 |

2.3. Antimicrobial Activity of Compounds 1–5
| Compounds | S. aureus ATCC29213 | B. subtilis CMCC63501 | E. coil ATCC25922 | C. albicans ATCC10231 | F. moniliforme S16 |
|---|---|---|---|---|---|
| 1 | 50 | 1.57 | >100 | >200 | >200 |
| 2 | >100 | 50 | >100 | >200 | >200 |
| 3 | 100 | 50 | >100 | >200 | >200 |
| 4 | 100 | 3.13 | >100 | >200 | >200 |
| 5 | 6.25 | 6.25 | >100 | >200 | >200 |
| ampicillin | 3.13 | 1.57 | 25 | NA | NA |
| nystatin | NA | NA | NA | 6.25 | 25 |
3. Experimental Section
3.1. General Experimental Procedures
3.2. Microorganisms and Fermentation Conditions
3.3. Bioactivity-Guided Isolation and Purification
3.4. Antimicrobial Activity of Compounds 1–5
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef]
- Kwon, H.C.; Kauffman, C.A.; Jensen, P.R.; Fenical, W. Marinisporolides, polyene-polyol macrolides from a marine actinomycete of the new genus Marinispora. J. Org. Chem. 2008, 74, 675–684. [Google Scholar]
- Zhang, W.; Liu, Z.; Li, S.; Yang, T.; Zhang, Q.; Ma, L.; Tian, X.; Zhang, H.; Huang, C.; Zhang, S. Spiroindimicins A–D: New bisindole alkaloids from a deep-sea-derived actinomycete. Org. Lett. 2012, 14, 3364–3367. [Google Scholar] [CrossRef]
- Mallams, A.K.; Puar, M.S.; Rossman, R.R.; McPhail, A.T.; Macfarlane, R.D.; Stephens, R.L. Kijanimicin. Part 3. Structure and absolute stereochemistry of kijanimicin. J. Chem. Soc. Perkin Trans. 1983, 1, 1497–1534. [Google Scholar]
- Jiang, Z.-D.; Jensen, P.R.; Fenical, W. Lobophorins A and B, new antiinflammatory macrolides produced by a tropical marine bacterium. Bioorg. Med. Chem. Lett. 1999, 9, 2003–2006. [Google Scholar] [CrossRef]
- Niu, S.; Li, S.; Chen, Y.; Tian, X.; Zhang, H.; Zhang, G.; Zhang, W.; Yang, X.; Zhang, S.; Ju, J.; et al. Lobophorins E and F, new spirotetronate antibiotics from a South China Sea-derived Streptomyces sp. SCSIO 01127. J. Antibiot. 2011, 64, 711–716. [Google Scholar] [CrossRef]
- Mallams, A.K.; Puar, M.S.; Rossman, R.R.; McPhail, A.T.; Macfarlane, R.D. Kijanimicin. 2. Structure and absolute stereochemistry of kijanimicin. J. Am. Chem. Soc. 1981, 103, 3940–3943. [Google Scholar] [CrossRef]
- Tomita, F.; Tamaoki, T.; Shirahata, K.; Kasai, M.; Morimoto, M.; Ohkubo, S.; Mineura, K.; Ishii, S. Novel antitumor antibiotics, tetrocarcins. J. Antibiot. 1980, 33, 668–670. [Google Scholar] [CrossRef]
- Kusumi, T.; Ichikawa, A.; Kakisawa, H.; Tsunakawa, M.; Konishi, M.; Oki, T. The structures of quartromicins A1, A2, and A3: Novel macrocyclic antiviral antibiotics possessing four tetronic acid moieties. J. Am. Chem. Soc. 1991, 113, 8947–8948. [Google Scholar] [CrossRef]
- Jia, X.Y.; Tian, Z.H.; Shao, L.; Qu, X.D.; Zhao, Q.F.; Tang, J.; Tang, G.L.; Liu, W. Genetic characterization of the chlorothricin gene cluster as a model for spirotetronate antibiotic biosynthesis. Chem. Biol. 2006, 13, 575–585. [Google Scholar] [CrossRef]
- Waitz, J.A.; Horan, A.; Kalyanpur, M.; Lee, B.K.; Loebenberg, D.; Marquez, J.A.; Miller, G.; Patel, M.G. Kijanimicin (Sch 25663), a novel antibiotic produced by Actinomadura kijaniata SCC 1256. Fermentation, isolation, characterization and biological properties. J. Antibiot. 1981, 34, 1101–1106. [Google Scholar] [CrossRef]
- Tamaoki, T.; Kasai, M.; Shirahata, K.; Tomita, F. Tetrocarcins E1, E2, F and F-1, new antibiotics. Fermentation, isolation and characterization. J. Antibiot. 1982, 35, 979–984. [Google Scholar] [CrossRef]
- Momose, I.; Hirosawa, S.; Nakamura, H.; Naganawa, H.; Iinuma, H.; Ikeda, D.; Takeuchi, T. Decatromicins A and B, new antibiotics produced by Actinomadura sp. MK73-NF4. II. Structure determination. J. Antibiot. 1999, 32, 787–796. [Google Scholar]
- Horan, A.C.; Shearer, M.C.; Hedge, V.; Beyazova, M.L.; Brodsky, B.C.; King, A.; Berrie, R.; Cardaci, K.; Nimeck, M. A family of novel macrocyclic lactones, the saccharocarcins produced by Saccharothrix aerocolonigenes subsp. antibiotica. I. Taxonomy, fermentation, isolation and biological properties. J. Antibiot. 1997, 50, 126–134. [Google Scholar] [CrossRef]
- Kobinata, K.; Uramoto, M.; Mizuno, T.; Isono, K. A new antibiotic, antlermicin A. J. Antibiot. 1980, 33, 244–246. [Google Scholar] [CrossRef]
- Park, H.R.; Furihata, K.; Hayakawa, Y.; Shin-ya, K. Versipelostatin, a novel GRP78/Bip molecular chaperone down-regulator of microbial origin. Tetrahedron Lett. 2002, 43, 6941–6945. [Google Scholar] [CrossRef]
- Furumai, T.; Takagi, K.; Igarashi, Y.; Saito, N.; Oki, T. Arisostatins A and B, new members of tetrocarcin class of antibiotics from Micromonospora sp. TP-A0316-I. Taxonomy, fermentation, isolation and biological properties. J. Antibiot. 2000, 53, 227–232. [Google Scholar] [CrossRef]
- Nakai, R.; Kakita, S.; Asai, A.; Chiba, S.; Akinaga, S.; Mizukami, T.; Yamashita, Y. Chrolactomycin, a novel antitumor antibiotic produced by Streptomyces sp. J. Antibiot. 2001, 54, 836–839. [Google Scholar] [CrossRef]
- Wei, R.B.; Xi, T.; Li, J.; Wang, P.; Li, F.C.; Lin, Y.C.; Qin, S. Lobophorin C and D, new kijanimicin derivatives from a marine sponge-associated actinomycetal srain AZS17. Mar. Drugs 2011, 9, 359–368. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J.; Guo, H.; Hou, W.; Yang, N.; Ren, B.; Liu, M.; Dai, H.; Liu, X.; Song, F.; et al. Three antimycobacterial metabolites identified from a marine-derived Streptomyces sp. MS100061. Appl. Microbiol. Biotechnol. 2013, 97, 3885–3892. [Google Scholar]
- Griffith, B.R.; Langenhan, J.M.; Thorson, J.S. “Sweetening” natural products via glycorandomization. Curr. Opin. Biotechnol. 2005, 16, 622–630. [Google Scholar] [CrossRef]
- Engelhardt, K.; Degnes, K.F.; Kemmler, M.; Bredholt, H.; Fjaervik, E.; Klinkenberg, G.; Sletta, H.; Ellingsen, T.E.; Zotchev, S.B. Production of a new thiopeptide antibiotic, TP-1161, by a marine Nocardiopsis species. Appl. Environ. Microbiol. 2010, 76, 4969–4976. [Google Scholar] [CrossRef] [Green Version]
Supplementary Files
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pan, H.-Q.; Zhang, S.-Y.; Wang, N.; Li, Z.-L.; Hua, H.-M.; Hu, J.-C.; Wang, S.-J. New Spirotetronate Antibiotics, Lobophorins H and I, from a South China Sea-Derived Streptomyces sp. 12A35. Mar. Drugs 2013, 11, 3891-3901. https://doi.org/10.3390/md11103891
Pan H-Q, Zhang S-Y, Wang N, Li Z-L, Hua H-M, Hu J-C, Wang S-J. New Spirotetronate Antibiotics, Lobophorins H and I, from a South China Sea-Derived Streptomyces sp. 12A35. Marine Drugs. 2013; 11(10):3891-3901. https://doi.org/10.3390/md11103891
Chicago/Turabian StylePan, Hua-Qi, Song-Ya Zhang, Nan Wang, Zhan-Lin Li, Hui-Ming Hua, Jiang-Chun Hu, and Shu-Jin Wang. 2013. "New Spirotetronate Antibiotics, Lobophorins H and I, from a South China Sea-Derived Streptomyces sp. 12A35" Marine Drugs 11, no. 10: 3891-3901. https://doi.org/10.3390/md11103891




