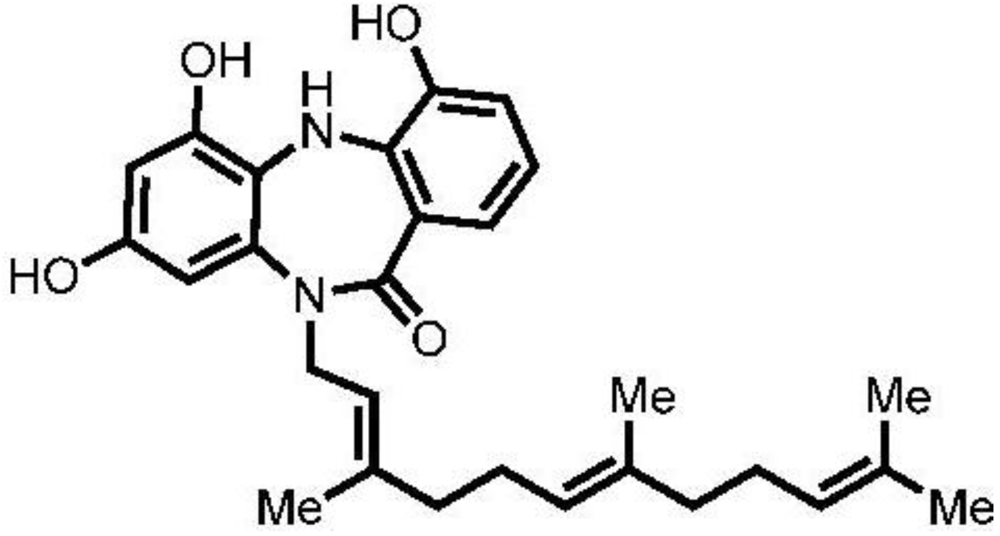
Antioxidant and Anti-Protease Activities of Diazepinomicin from the Sponge-Associated Micromonospora Strain RV115
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cytotoxicity of Diazepinomicin in Human Kidney Cells (HK-2)
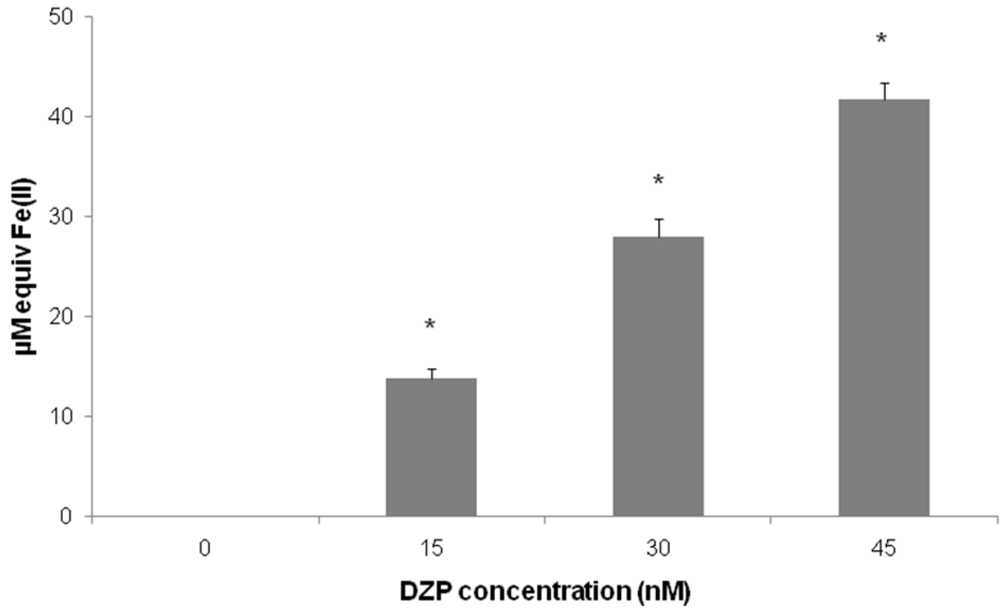
2.2. Antioxidant Potential of Diazepinomicin
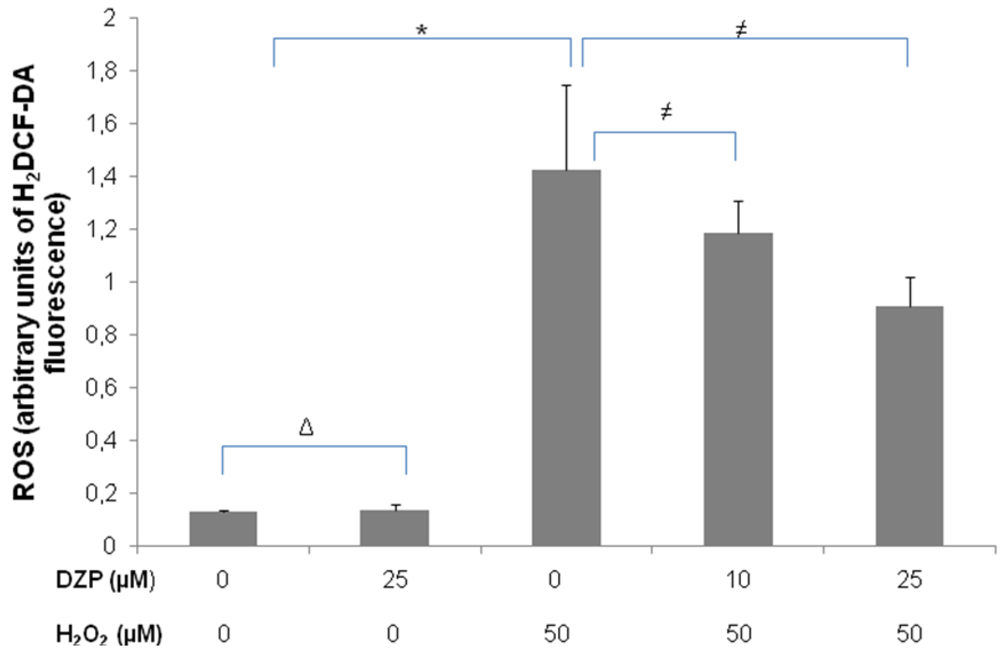
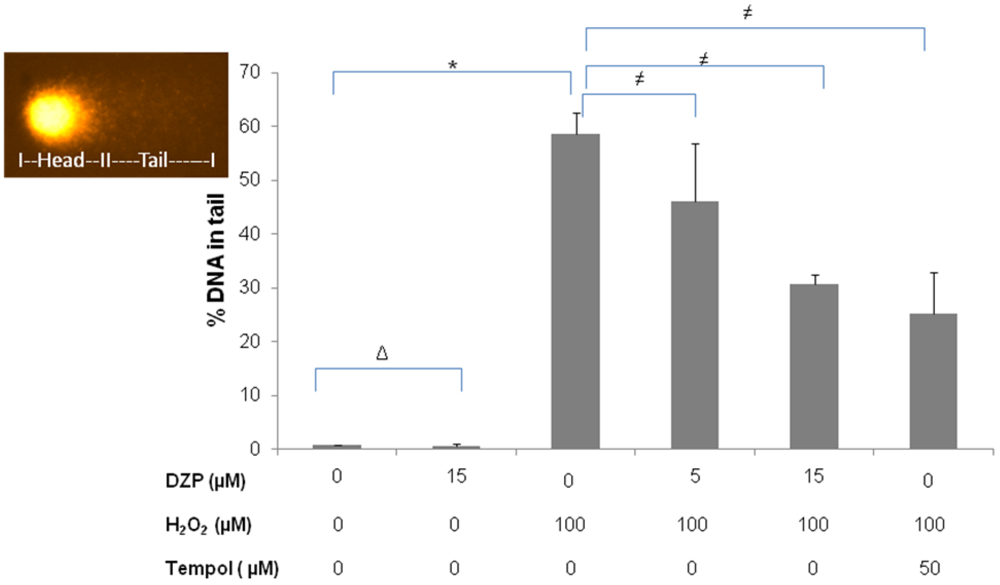
2.3. Protective Effect of Diazepinomicin against Oxidative Stress-Induced Cell Death in HK-2 Cells
| Test substance | Cell death (% dead cells) |
|---|---|
| Control | 4 ± 1.0 |
| 25 µM DZP | 4.5 ± 0.0 (Δ vs. control ) |
| 50 µM tempol | 9.7 ± 4.3 |
| 100 µM H2O2 | 21 ± 5.8 (* vs. control) |
| 100 µM H2O2 + 2 µM DZP | 6.5 ± 4.4 (≠ vs. H2O2) |
| 100 µM H2O2 + 5 µM DZP | 7.5 ± 1.8 (≠ vs. H2O2) |
| 100 µM H2O2 + 10 µM DZP | 5.3 ± 1.0 (≠ vs. H2O2) |
| 100 µM H2O2 + 25 µM DZP | 6.3 ± 2.5 (≠ vs. H2O2) |
| 100 µM H2O2 + 50 µM tempol | 14.7 ± 5.8 |
2.4. Antitrypanosomal and Anti-Protease Activities
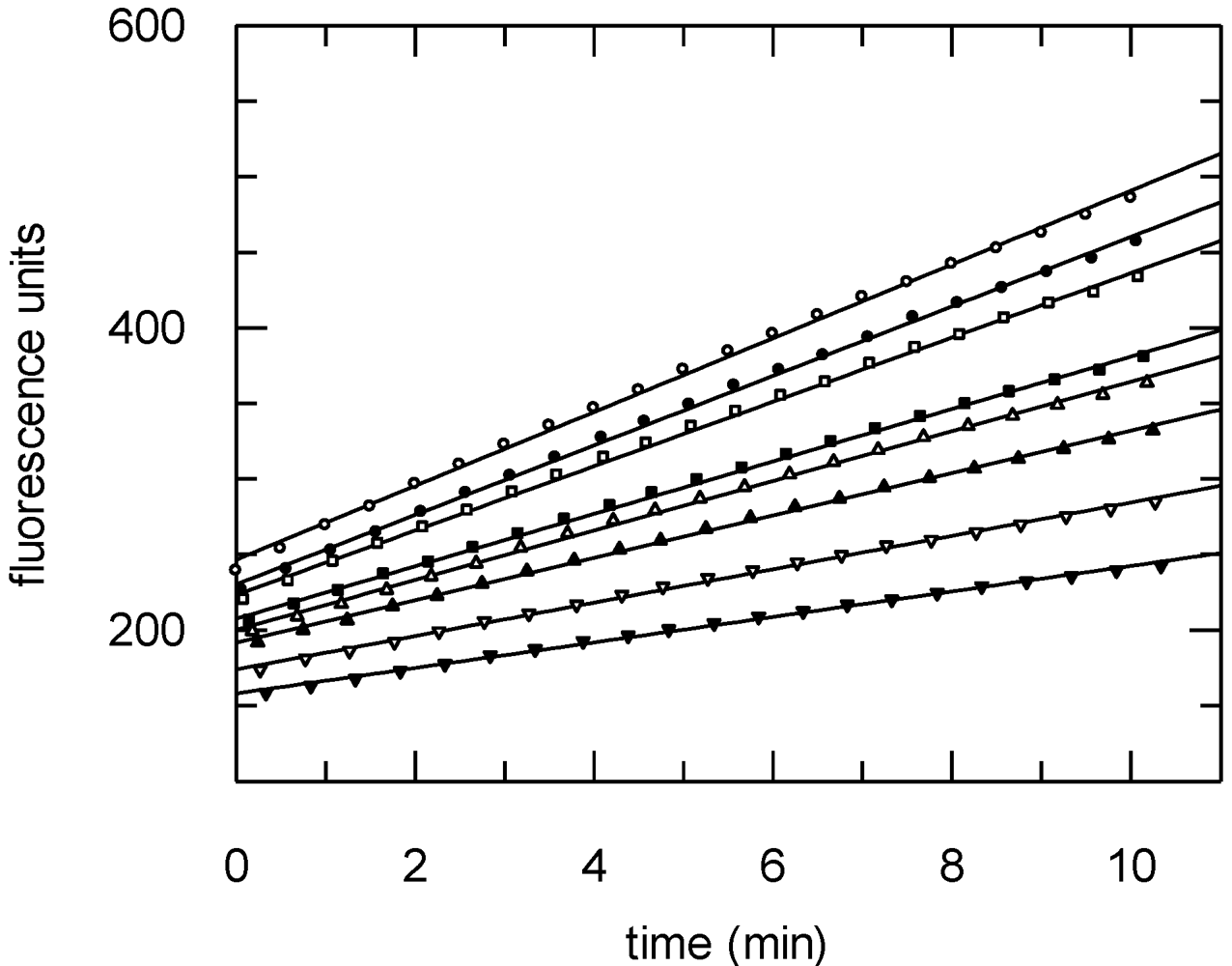
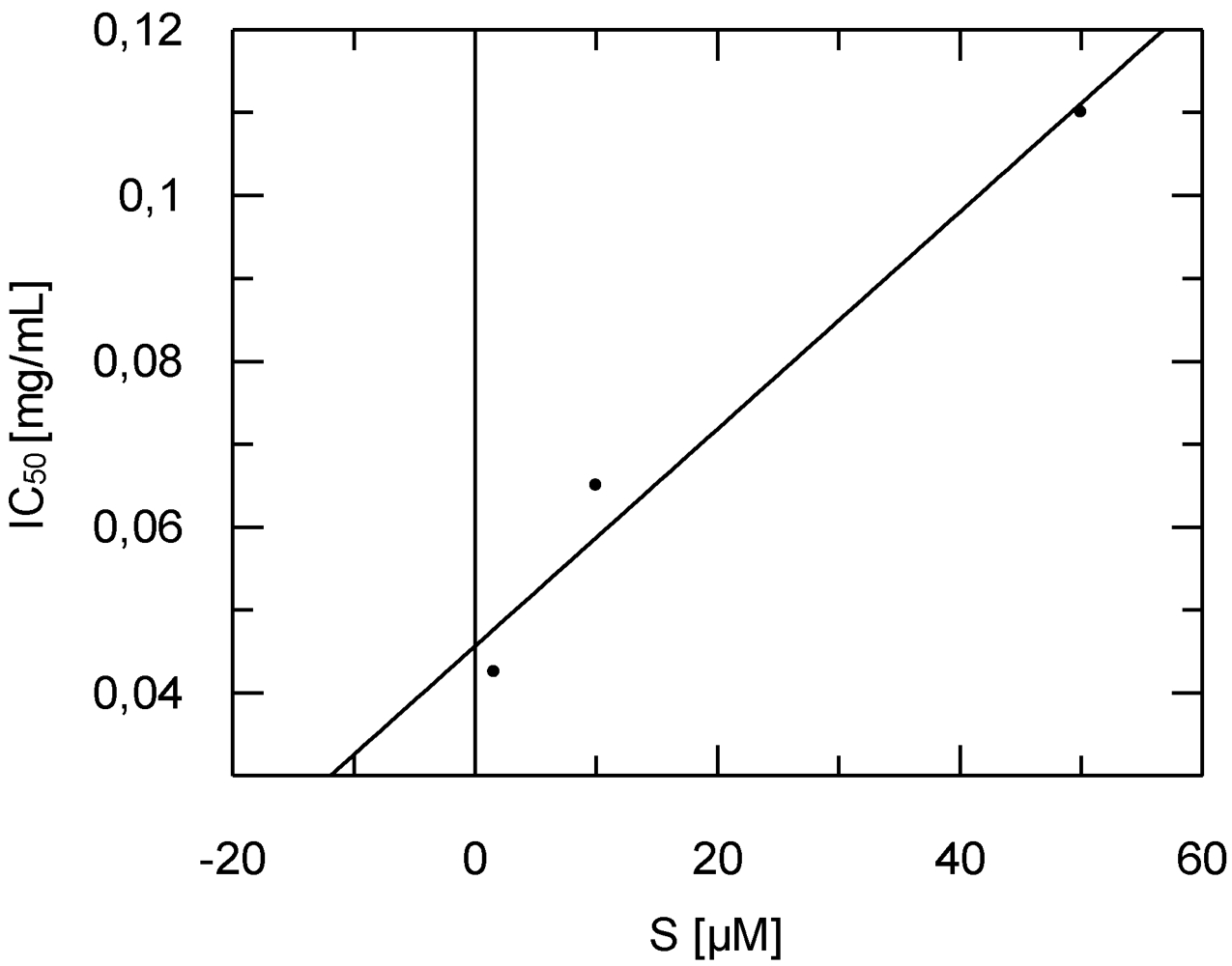
3. Experimental Section
3.1. Cultivation and Purification
3.2. Ferric Reducing Antioxidant Power (FRAP) Assay
3.3. Cell Culture Maintenance
3.4. Flow Cytometric Quantification of Antioxidant Activity
3.5. Vitality Test
3.6. Comet Assay
3.7. Statistics for Cell Based Assays
3.8. Antitrypanosomal Activity
3.9. Protease Assays
4. Conclusions
Acknowledgments
References
- Zazopoulos, E.; Huang, K.; Staffa, A.; Liu, W.; Bachmann, B.O.; Nonaka, K.; Ahlert, J.; Thorson, J.S.; Shen, B.; Farnet, C.M. A genomics-guided approach for discovering and expressing cryptic metabolic pathways. Nat. Biotechnol. 2003, 21, 187–190. [Google Scholar] [CrossRef]
- Charan, R.D.; Schlingmann, G.; Janso, J.; Bernan, V.; Feng, X.; Carter, G.T. Diazepinomicin, a new antimicrobial alkaloid from a marine Micromonospora sp. J. Nat. Prod. 2004, 67, 1431–1433. [Google Scholar] [CrossRef]
- McAlpine, J.B.; Banskota, A.H.; Charan, R.D.; Schlingmann, G.; Zazopoulos, E.; Piraee, M.; Janso, J.; Bernan, V.S.; Aouidate, M.; Farnet, C.M.; et al. Biosynthesis of diazepinomicin/ECO-4601, a Micromonospora secondary metabolite with a novel ring system. J. Nat. Prod. 2008, 71, 1585–1590. [Google Scholar] [CrossRef]
- Ratnayake, A.S.; Janso, J.E.; Feng, X.; Schlingmann, G.; Goljer, I.; Carter, G.T. Evaluating indole-related derivatives as precursors in the directed biosynthesis of diazepinomicin analogues. J. Nat. Prod. 2009, 72, 496–499. [Google Scholar] [CrossRef]
- Campas, C. Diazepinomicin. Drug Fut. 2009, 34, 349–351. [Google Scholar] [CrossRef]
- Wong, K.K. Recent developments in anti-cancer agents targeting the Ras/Raf/ MEK/ERK pathway. Recent Pat. Anticancer Drug Discov. 2009, 4, 28–35. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J. Free Radicals in Biology and Medicine; Clarendon Press: New York, NY, USA, 1999; pp. 20–37. [Google Scholar]
- Sohal, R.S.; Mockett, R.J.; Orr, W.C. Mechanisms of aging: an appraisal of the oxidative stress hypothesis. Free Radic. Biol. Med. 2002, 33, 575–586. [Google Scholar] [CrossRef]
- Chowienczyk, P.J.; Brett, S.E.; Gopaul, N.K.; Meeking, D.; Marchetti, M.; Russell-Jones, D.L.; Anggard, E.E.; Ritter, J.M. Oral treatment with an antioxidant (raxofelast) reduces oxidative stress and improves endothelial function in men with type II diabetes. Diabetologia 2000, 43, 974–977. [Google Scholar]
- Parthasarathy, S.; Santanam, N.; Ramachandran, S.; Meilhac, O. Potential role of oxidized lipids and lipoproteins in antioxidant defense. Free Radic. Res. 2000, 33, 197–215. [Google Scholar] [CrossRef]
- DeNicola, G.M.; Karreth, F.A.; Humpton, T.J.; Gopinathan, A.; Wei, C.; Frese, K.; Mangal, D.; Yu, K.H.; Yeo, C.J.; Calhoun, E.S.; et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 2011, 475, 106–109. [Google Scholar]
- Young, I.S.; Woodside, J.V. Antioxidants in health and disease. J. Clin. Pathol. 2001, 54, 176–186. [Google Scholar]
- Zhang, C.Y.; Wu, W.H.; Wang, J.; Lan, M.B. Antioxidant properties of polysaccharide from the brown seaweed Sargassum graminifolium (Turn.), and its effects on calcium oxalate crystallization. Mar. Drugs 2012, 10, 119–130. [Google Scholar] [CrossRef]
- Song, L.; Li, T.; Yu, R.; Yan, C.; Ren, S.; Zhao, Y. Antioxidant activities of hydrolysates of Arca subcrenata prepared with three proteases. Mar. Drugs 2008, 6, 607–619. [Google Scholar] [CrossRef]
- Sunassee, S.N.; Davies-Coleman, M.T. Cytotoxic and antioxidant marine prenylated quinones and hydroquinones. Nat. Prod. Rep. 2012, 29, 513–535. [Google Scholar] [CrossRef]
- Zhang, C.; Kim, S.K. Matrix metalloproteinase inhibitors (MMPIs) from marine natural products: The current situation and future prospects. Mar. Drugs 2009, 7, 71–84. [Google Scholar] [CrossRef]
- Hsieh, C.C.; Hernandez-Ledesma, B.; Jeong, H.J.; Park, J.H.; de Lumen, B.O. Complementary roles in cancer prevention: Protease inhibitor makes the cancer preventive peptide lunasin bioavailable. PLoS One 2010, 5, e8890. [Google Scholar]
- Cai, H.; Kuang, R.; Gu, J.; Wang, Y. Proteases in malaria parasites—a phylogenomic perspective. Curr. Genomics 2011, 12, 417–427. [Google Scholar] [CrossRef]
- McKerrow, J.H.; Rosenthal, P.J.; Swenerton, R.; Doyle, P. Development of protease inhibitors for protozoan infections. Curr. Opin. Infect. Dis. 2008, 21, 668–672. [Google Scholar] [CrossRef]
- Abdelmohsen, U.R.; Pimentel-Elardo, S.M.; Hanora, A.; Radwan, M.; Abou-El-Ela, S.H.; Ahmed, S.; Hentschel, U. Isolation, phylogenetic analysis and anti-infective activity screening of marine sponge-associated actinomycetes. Mar. Drugs 2010, 8, 399–412. [Google Scholar] [CrossRef]
- Verzola, D.; Bertolotto, M.B.; Villaggio, B.; Ottonello, L.; Dallegri, F.; Salvatore, F.; Berruti, V.; Gandolfo, M.T.; Garibotto, G.; Deferrari, G. Oxidative stress mediates apoptotic changes induced by hyperglycemia in human tubular kidney cells. J. Am. Soc. Nephrol. 2004, 15 (Suppl. 1), S85–S87. [Google Scholar]
- Djamali, A. Oxidative stress as a common pathway to chronic tubulointerstitial injury in kidney allografts. Am. J. Physiol. Renal Physiol. 2007, 293, F445–F455. [Google Scholar] [CrossRef]
- Moyer, R.A.; Hummer, K.E.; Finn, C.E.; Frei, B.; Wrolstad, R.E. Anthocyanins, phenolics, and antioxidant capacity in diverse small fruits: Vaccinium, rubus, and ribes. J. Agric. Food Chem. 2002, 50, 519–525. [Google Scholar] [CrossRef]
- Takamatsu, S.; Hodges, T.W.; Rajbhandari, I.; Gerwick, W.H.; Hamann, M.T.; Nagle, D.G. Marine natural products as novel antioxidant prototypes. J. Nat. Prod. 2003, 66, 605–608. [Google Scholar] [CrossRef]
- Pimentel-Elardo, S.M.; Buback, V.; Gulder, T.A.; Bugni, T.S.; Reppart, J.; Bringmann, G.; Ireland, C.M.; Schirmeister, T.; Hentschel, U. New tetromycin derivatives with anti-trypanosomal and protease inhibitory activities. Mar. Drugs 2011, 9, 1682–1697. [Google Scholar] [CrossRef]
- Tabares, P.; Degel, B.; Schaschke, N.; Hentschel, U.; Schirmeister, T. Identification of the protease inhibitor miraziridine A in the Red sea sponge Theonella swinhoei. Pharmacogn. Res. 2012, 4, 63–66. [Google Scholar]
- Leto, G.; Sepporta, M.V.; Crescimanno, M.; Flandina, C.; Tumminello, F.M. Cathepsin L in metastatic bone disease: therapeutic implications. Biol. Chem. 2010, 391, 655–664. [Google Scholar]
- Yan, J.A.; Xiao, H.; Ji, H.X.; Shen, W.H.; Zhou, Z.S.; Song, B.; Chen, Z.W.; Li, W.B. Cathepsin L is associated with proliferation and clinical outcome of urothelial carcinoma of the bladder. J. Int. Med. Res. 2010, 38, 1913–1922. [Google Scholar]
- Colella, R.; Lu, G.; Glazewski, L.; Korant, B.; Matlapudi, A.; England, M.R.; Craft, C.; Frantz, C.N.; Mason, R.W. Induction of cell death in neuroblastoma by inhibition of cathepsins B and L. Cancer Lett. 2010, 294, 195–203. [Google Scholar] [CrossRef]
- Joyce, J.A.; Baruch, A.; Chehade, K.; Meyer-Morse, N.; Giraudo, E.; Tsai, F.Y.; Greenbaum, D.C.; Hager, J.H.; Bogyo, M.; Hanahan, D. Cathepsin cysteine proteases are effectors of invasive growth and angiogenesis during multistage tumorigenesis. Cancer Cell 2004, 5, 443–453. [Google Scholar]
- Ludewig, S.; Kossner, M.; Schiller, M.; Baumann, K.; Schirmeister, T. Enzyme kinetics and hit validation in fluorimetric protease assays. Curr. Top. Med. Chem. 2010, 10, 368–382. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–27. [Google Scholar]
- Schmitt, E.; Lehmann, L.; Metzler, M.; Stopper, H. Hormonal and genotoxic activity of resveratrol. Toxicol. Lett. 2002, 136, 133–142. [Google Scholar] [CrossRef]
- Huber, W.; Koella, J.C. A comparison of three methods of estimating EC50 in studies of drug resistance of malaria parasites. Acta Trop. 1993, 55, 257–261. [Google Scholar] [CrossRef]
- Baltz, T.; Baltz, D.; Giroud, C.; Crockett, J. Cultivation in a semi-defined medium of animal infective forms of Trypanosoma brucei, T. equiperdum, T. evansi, T. rhodesiense and T. gambiense. EMBO J. 1985, 4, 1273–1277. [Google Scholar]
- Breuning, A.; Degel, B.; Schulz, F.; Buchold, C.; Stempka, M.; Machon, U.; Heppner, S.; Gelhaus, C.; Leippe, M.; Leyh, M.; et al. Michael acceptor based antiplasmodial and antitrypanosomal cysteine protease inhibitors with unusual amino acids. J. Med. Chem. 2010, 53, 1951–1963. [Google Scholar]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abdelmohsen, U.R.; Szesny, M.; Othman, E.M.; Schirmeister, T.; Grond, S.; Stopper, H.; Hentschel, U. Antioxidant and Anti-Protease Activities of Diazepinomicin from the Sponge-Associated Micromonospora Strain RV115. Mar. Drugs 2012, 10, 2208-2221. https://doi.org/10.3390/md10102208
Abdelmohsen UR, Szesny M, Othman EM, Schirmeister T, Grond S, Stopper H, Hentschel U. Antioxidant and Anti-Protease Activities of Diazepinomicin from the Sponge-Associated Micromonospora Strain RV115. Marine Drugs. 2012; 10(10):2208-2221. https://doi.org/10.3390/md10102208
Chicago/Turabian StyleAbdelmohsen, Usama Ramadan, Matthias Szesny, Eman Maher Othman, Tanja Schirmeister, Stephanie Grond, Helga Stopper, and Ute Hentschel. 2012. "Antioxidant and Anti-Protease Activities of Diazepinomicin from the Sponge-Associated Micromonospora Strain RV115" Marine Drugs 10, no. 10: 2208-2221. https://doi.org/10.3390/md10102208






