The Impact of Lipoproteins on Wound Healing: Topical HDL Therapy Corrects Delayed Wound Healing in Apolipoprotein E Deficient Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. LDL Receptor Gene Transfer
2.3. HDL Purification
2.4. Determination of Plasma Cholesterol and of non-HDL and HDL Cholesterol Levels
2.5. Full Thickness Excisional Wound Model
2.6. Macroscopic Analysis of the Wounds
2.7. Statistical Analysis
3. Results
3.1. Severe Hypercholesterolemia in C57BL/6 LDLr−/− Mice Enhances Wound Healing
| Total cholesterol | Non-HDL cholesterol | HDL cholesterol | |
|---|---|---|---|
| C57BL/6 SC diet | 71.0 ± 5.1 | 11.7 ± 1.1 | 59.2 ± 4.9 |
| C57BL/6 CC diet | 78.1 ± 4.1 | 13.9 ± 1.4 | 64.2 ± 3.6 |
| C57BL/6 LDLr−/− SC diet | 189 ± 10 *** | 146 ± 10 *** | 42.9 ± 2.9 * |
| C57BL/6 LDLr−/− CC diet | 489 ± 14 *** | 419 ± 13 *** | 69.8 ± 3.8 |
| C57BL/6 LDLr−/− CC diet AdLDLr | 72.3 ± 3.5 | 27.4 ± 2.7 *** | 45 ± 3.4 * |
| C57BL/6 apo E−/− SC diet | 434 ± 16 *** | 405 ± 5 *** | 29.0 ± 1.4 *** |
 ) and C57BL/6 LDLr−/− SC diet mice (▲) and C57BL/6 LDLr−/− CC diet mice (Δ), respectively. The effect of AdLDLr gene transfer (
) and C57BL/6 LDLr−/− SC diet mice (▲) and C57BL/6 LDLr−/− CC diet mice (Δ), respectively. The effect of AdLDLr gene transfer (  ) on re-epithelialization in C57BL/6 LDLr−/− CC diet mice is illustrated in Panel C. Panel D compares wound coverage by newly formed epithelium in C57BL/6 SC diet mice (
) on re-epithelialization in C57BL/6 LDLr−/− CC diet mice is illustrated in Panel C. Panel D compares wound coverage by newly formed epithelium in C57BL/6 SC diet mice (  ) and C57BL/6 CC diet mice (
) and C57BL/6 CC diet mice (  ). All data represent means ± SEM.
). All data represent means ± SEM.
 ) and C57BL/6 LDLr−/− SC diet mice (▲) and C57BL/6 LDLr−/− CC diet mice (Δ), respectively. The effect of AdLDLr gene transfer (
) and C57BL/6 LDLr−/− SC diet mice (▲) and C57BL/6 LDLr−/− CC diet mice (Δ), respectively. The effect of AdLDLr gene transfer (  ) on re-epithelialization in C57BL/6 LDLr−/− CC diet mice is illustrated in Panel C. Panel D compares wound coverage by newly formed epithelium in C57BL/6 SC diet mice (
) on re-epithelialization in C57BL/6 LDLr−/− CC diet mice is illustrated in Panel C. Panel D compares wound coverage by newly formed epithelium in C57BL/6 SC diet mice (  ) and C57BL/6 CC diet mice (
) and C57BL/6 CC diet mice (  ). All data represent means ± SEM.
). All data represent means ± SEM.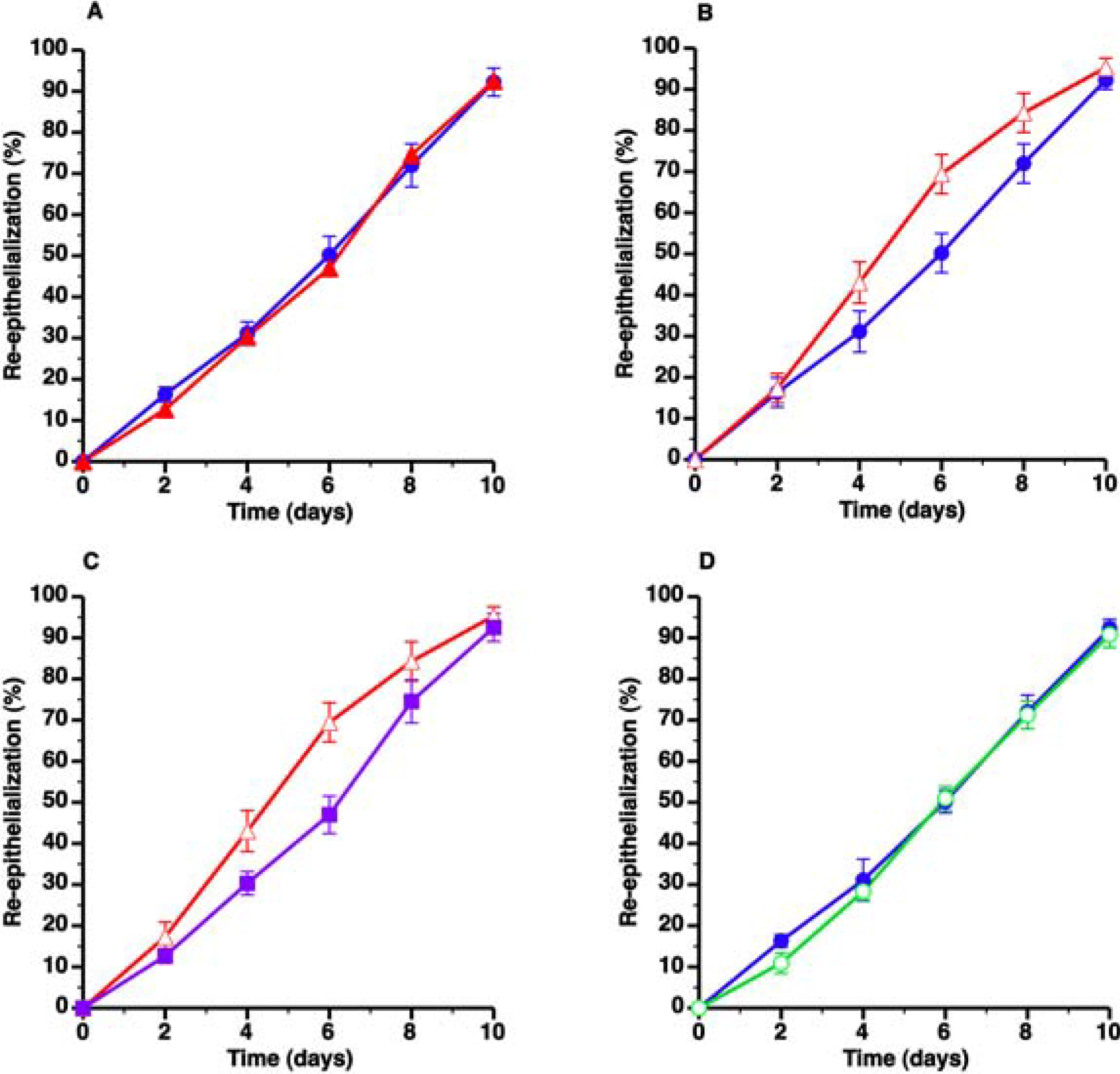
| Number of mice | Area under the curve | |
|---|---|---|
| C57BL/6 SC diet | 10 | 432 ± 26 |
| C57BL/6 CC diet | 12 | 414 ± 18 |
| C57BL/6 LDLr−/− SC diet | 10 | 424 ± 25 |
| C57BL/6 LDLr−/− CC diet | 13 | 523 ± 31 * |
| C57BL/6 LDLr−/− CC diet AdLDLr | 14 | 432 ± 27 |
| C57BL/6 apo E−/− SC diet | 11 | 334 ± 11 *** |
3.2. C57BL/6 Apo E−/− Mice Are Characterized by Delayed Wound Healing
 ) and C57BL/6 apo E−/− SC diet mice (
) and C57BL/6 apo E−/− SC diet mice (  ). Panels A and B illustrate the time course of wound coverage by granulation tissue and by newly formed epithelium, respectively, both expressed as percentage of the original wound surface. All data represent means ± SEM.
). Panels A and B illustrate the time course of wound coverage by granulation tissue and by newly formed epithelium, respectively, both expressed as percentage of the original wound surface. All data represent means ± SEM.
 ) and C57BL/6 apo E−/− SC diet mice (
) and C57BL/6 apo E−/− SC diet mice (  ). Panels A and B illustrate the time course of wound coverage by granulation tissue and by newly formed epithelium, respectively, both expressed as percentage of the original wound surface. All data represent means ± SEM.
). Panels A and B illustrate the time course of wound coverage by granulation tissue and by newly formed epithelium, respectively, both expressed as percentage of the original wound surface. All data represent means ± SEM.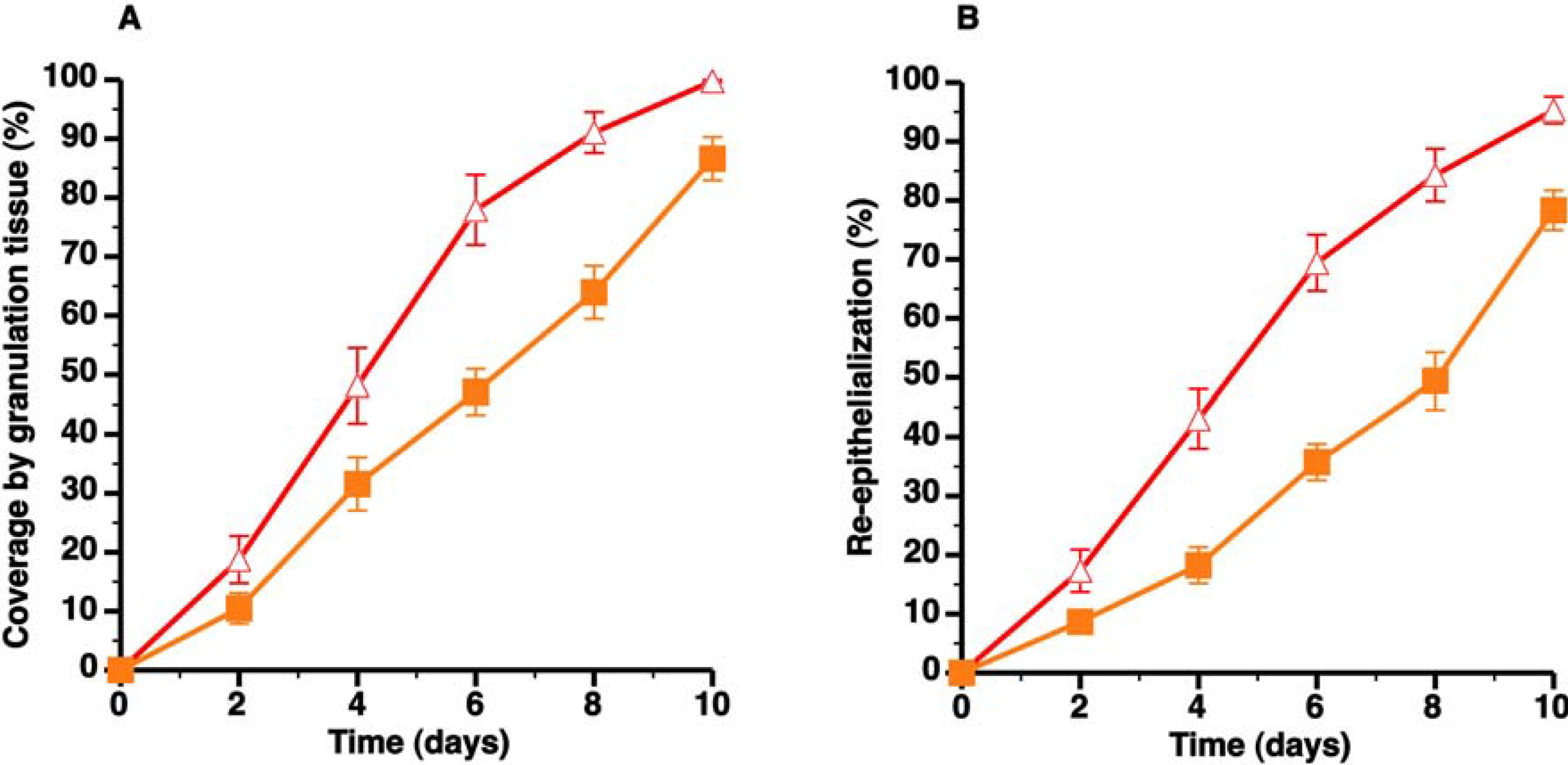
3.3. Topical HDL Therapy Enhances Wound Healing in C57BL/6 Apo E−/− Mice
| Number of mice | Area under the curve | |
|---|---|---|
| C57BL/6 apo E−/− | 11 | 334 ± 11 |
| C57BL/6 apo E−/− control gel | 10 | 284 ± 31 |
| C57BL/6 apo E−/− HDL gel | 10 | 420 ± 28 **§§ |
 ) compared to C57BL/6 mice (
) compared to C57BL/6 mice (  ) whereas panel B shows that topical HDL therapy (
) whereas panel B shows that topical HDL therapy (  ) corrects delayed wound healing. Panel C represents a comparison of wound coverage by newly formed epithelium in C57BL/6 apo E−/− control mice (
) corrects delayed wound healing. Panel C represents a comparison of wound coverage by newly formed epithelium in C57BL/6 apo E−/− control mice (  ) and C57BL/6 apo E−/− mice treated with control pluronic gel (
) and C57BL/6 apo E−/− mice treated with control pluronic gel (  ). The effect of control Pluronic gel (
). The effect of control Pluronic gel (  ) and HDL Pluronic gel (
) and HDL Pluronic gel (  ) is directly compared in panel D. All data represent means ± SEM.
) is directly compared in panel D. All data represent means ± SEM.
 ) compared to C57BL/6 mice (
) compared to C57BL/6 mice (  ) whereas panel B shows that topical HDL therapy (
) whereas panel B shows that topical HDL therapy (  ) corrects delayed wound healing. Panel C represents a comparison of wound coverage by newly formed epithelium in C57BL/6 apo E−/− control mice (
) corrects delayed wound healing. Panel C represents a comparison of wound coverage by newly formed epithelium in C57BL/6 apo E−/− control mice (  ) and C57BL/6 apo E−/− mice treated with control pluronic gel (
) and C57BL/6 apo E−/− mice treated with control pluronic gel (  ). The effect of control Pluronic gel (
). The effect of control Pluronic gel (  ) and HDL Pluronic gel (
) and HDL Pluronic gel (  ) is directly compared in panel D. All data represent means ± SEM.
) is directly compared in panel D. All data represent means ± SEM.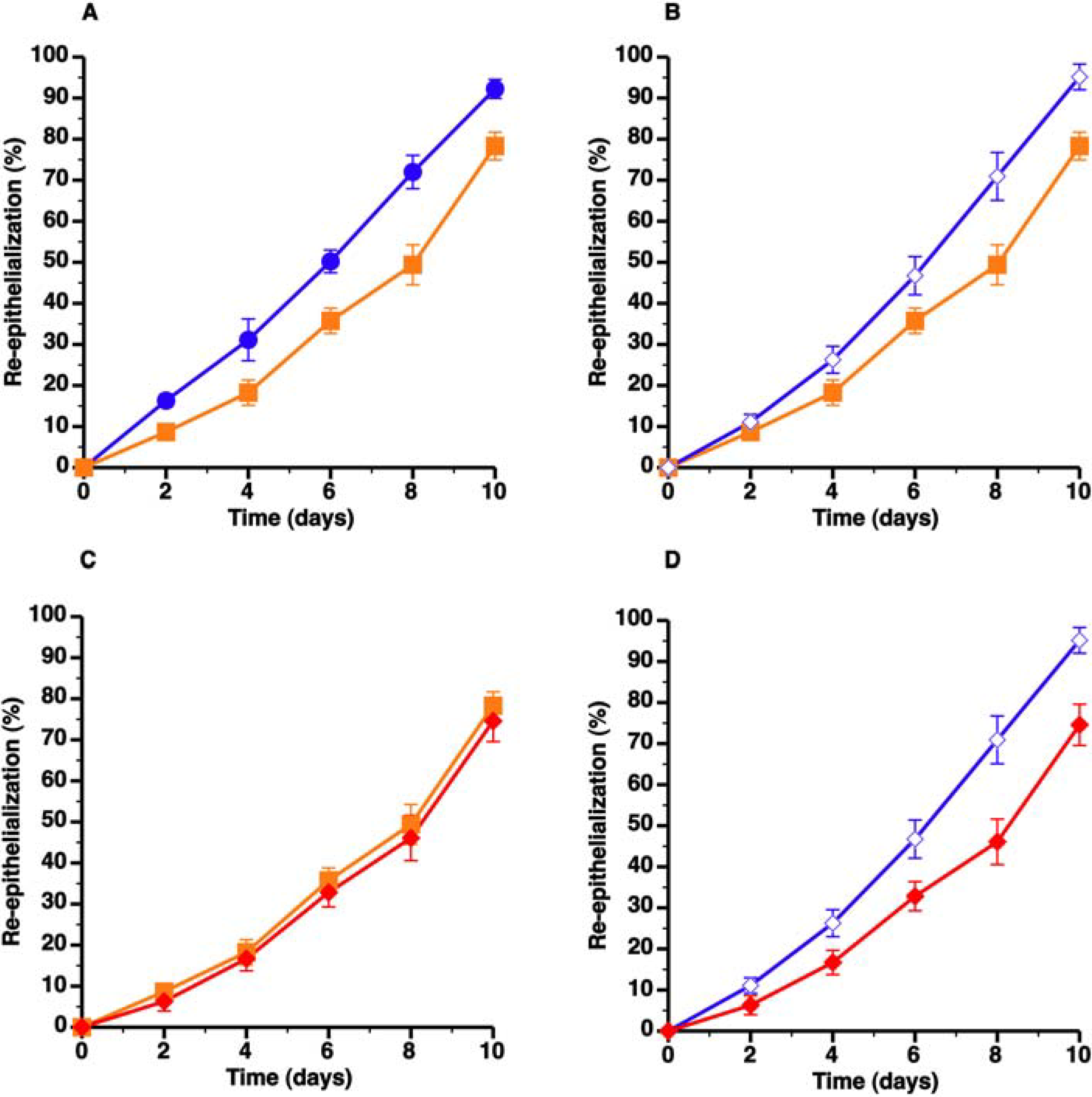
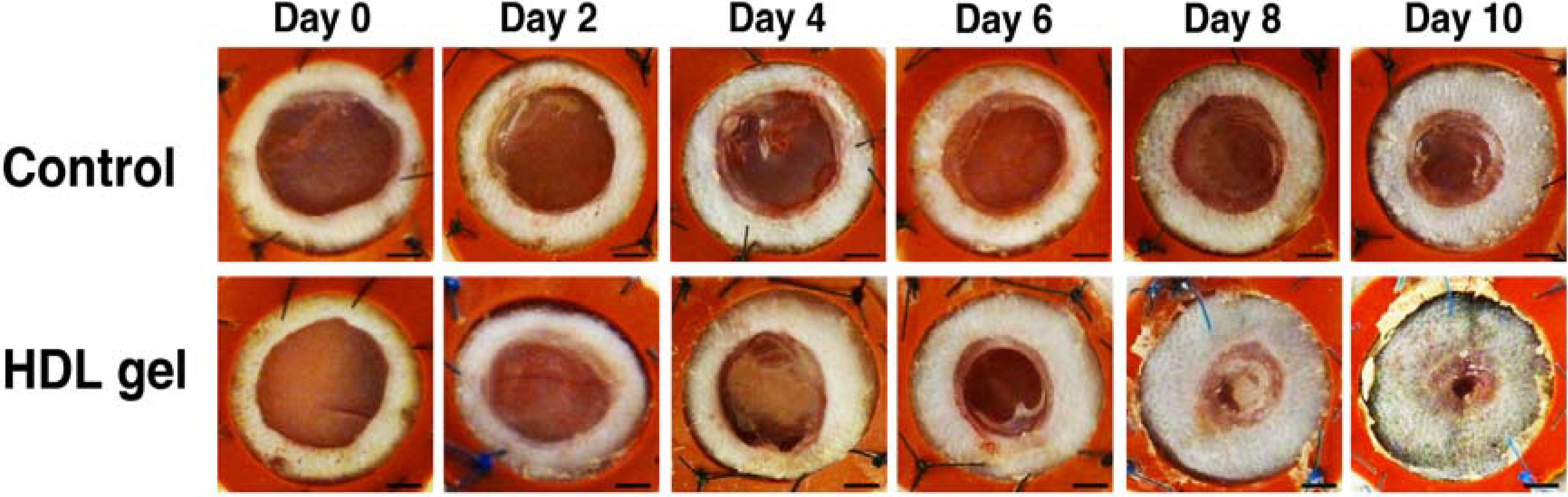
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Di Angelantonio, E.; Sarwar, N.; Perry, P.; Kaptoge, S.; Ray, K.K.; Thompson, A.; Wood, A.M.; Lewington, S.; Sattar, N.; Packard, C.J.; et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 2009, 302, 1993–2000. [Google Scholar] [CrossRef]
- Owen, D.M.; Magenau, A.; Williamson, D.; Gaus, K. The lipid raft hypothesis revisited—New insights on raft composition and function from super-resolution fluorescence microscopy. Bioessays 2012, 34, 739–747. [Google Scholar] [CrossRef]
- Velagaleti, R.S.; Massaro, J.; Vasan, R.S.; Robins, S.J.; Kannel, W.B.; Levy, D. Relations of lipid concentrations to heart failure incidence: The Framingham Heart Study. Circulation 2009, 120, 2345–2351. [Google Scholar] [CrossRef]
- Van Craeyveld, E.; Jacobs, F.; Gordts, S.C.; De Geest, B. Low-density lipoprotein receptor gene transfer in hypercholesterolemic mice improves cardiac function after myocardial infarction. Gene Ther. 2012, 19, 860–871. [Google Scholar] [CrossRef]
- Vance, J.E. Dysregulation of cholesterol balance in the brain: Contribution to neurodegenerative diseases. Dis. Model. Mech. 2012, 5, 746–755. [Google Scholar] [CrossRef]
- Cruz, P.M.; Mo, H.; McConathy, W.J.; Sabnis, N.; Lacko, A.G. The role of cholesterol metabolism and cholesterol transport in carcinogenesis: A review of scientific findings, relevant to future cancer therapeutics. Front. Pharmacol. 2013, 4. [Google Scholar] [CrossRef]
- Duffy, D.; Rader, D.J. Update on strategies to increase HDL quantity and function. Nat. Rev. Cardiol. 2009, 6, 455–463. [Google Scholar] [CrossRef]
- Van Craeyveld, E.; Gordts, S.; Jacobs, F.; De Geest, B. Gene therapy to improve high-density lipoprotein metabolism and function. Curr. Pharm. Des. 2010, 16, 1531–1544. [Google Scholar] [CrossRef]
- Feng, Y.; Jacobs, F.; van Craeyveld, E.; Brunaud, C.; Snoeys, J.; Tjwa, M.; Van Linthout, S.; De Geest, B. Human ApoA-I transfer attenuates transplant arteriosclerosis via enhanced incorporation of bone marrow-derived endothelial progenitor cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 278–283. [Google Scholar]
- Feng, Y.; van Craeyveld, E.; Jacobs, F.; Lievens, J.; Snoeys, J.; De Geest, B. Wild-type apo A-I and apo A-I(Milano) gene transfer reduce native and transplant arteriosclerosis to a similar extent. J. Mol. Med. 2009, 87, 287–297. [Google Scholar] [CrossRef]
- Feng, Y.; van Eck, M.; van Craeyveld, E.; Jacobs, F.; Carlier, V.; van Linthout, S.; Erdel, M.; Tjwa, M.; De Geest, B. Critical role of scavenger receptor-BI-expressing bone marrow-derived endothelial progenitor cells in the attenuation of allograft vasculopathy after human apo A-I transfer. Blood 2009, 113, 755–764. [Google Scholar] [CrossRef]
- Diegelmann, R.F.; Evans, M.C. Wound healing: An overview of acute, fibrotic and delayed healing. Front. Biosci. 2004, 9, 283–289. [Google Scholar] [CrossRef]
- Feng, Y.; Gordts, S.C.; Chen, F.; Hu, Y.; van Craeyveld, E.; Jacobs, F.; Carlier, V.; Zhang, Z.; Xu, Q.; Ni, Y.; et al. Topical HDL administration reduces vein graft atherosclerosis in apo E deficient mice. Atherosclerosis 2011, 214, 271–278. [Google Scholar] [CrossRef]
- Sloop, C.H.; Dory, L.; Roheim, P.S. Interstitial fluid lipoproteins. J. Lipid Res. 1987, 28, 225–237. [Google Scholar]
- Parini, P.; Johansson, L.; Broijersen, A.; Angelin, B.; Rudling, M. Lipoprotein profiles in plasma and interstitial fluid analyzed with an automated gel-filtration system. Eur. J. Clin. Investig. 2006, 36, 98–104. [Google Scholar] [CrossRef]
- Jacobs, F.; Snoeys, J.; Feng, Y.; van Craeyveld, E.; Lievens, J.; Armentano, D.; Cheng, S.H.; De Geest, B. Direct comparison of hepatocyte-specific expression cassettes following adenoviral and nonviral hydrodynamic gene transfer. Gene Ther. 2008, 15, 594–603. [Google Scholar] [CrossRef]
- Van Craeyveld, E.; Gordts, S.C.; Nefyodova, E.; Jacobs, F.; De Geest, B. Regression and stabilization of advanced murine atherosclerotic lesions: A comparison of LDL lowering and HDL raising gene transfer strategies. J. Mol. Med. 2011, 89, 555–567. [Google Scholar] [CrossRef]
- Van Linthout, S.; Lusky, M.; Collen, D.; De Geest, B. Persistent hepatic expression of human apo A–I after transfer with a helper-virus independent adenoviral vector. Gene Ther. 2002, 9, 1520–1528. [Google Scholar] [CrossRef]
- Jacobs, F.; van Craeyveld, E.; Feng, Y.; Snoeys, J.; De Geest, B. Adenoviral low density lipoprotein receptor attenuates progression of atherosclerosis and decreases tissue cholesterol levels in a murine model of familial hypercholesterolemia. Atherosclerosis 2008, 201, 289–297. [Google Scholar] [CrossRef]
- Greenhalgh, D.G. Wound healing and diabetes mellitus. Clin. Plast. Surg. 2003, 30, 37–45. [Google Scholar] [CrossRef]
- Galiano, R.D.; Michaels, J., 5th; Dobryansky, M.; Levine, J.P.; Gurtner, G.C. Quantitative and reproducible murine model of excisional wound healing. Wound Repair Regen. 2004, 12, 485–492. [Google Scholar] [CrossRef]
- Pirro, M.; Bagaglia, F.; Paoletti, L.; Razzi, R.; Mannarino, M.R. Hypercholesterolemia-associated endothelial progenitor cell dysfunction. Ther. Adv. Cardiovasc. Dis. 2008, 2, 329–339. [Google Scholar] [CrossRef]
- Gordts, S.C.; van Craeyveld, E.; Muthuramu, I.; Singh, N.; Jacobs, F.; De Geest, B. Lipid Lowering and HDL Raising Gene Transfer Increase Endothelial Progenitor Cells, Enhance Myocardial Vascularity, and Improve Diastolic Function. PLoS One 2012, 7, e46849. [Google Scholar]
- Suh, W.; Kim, K.L.; Kim, J.M.; Shin, I.S.; Lee, Y.S.; Lee, J.Y.; Jang, H.S.; Lee, J.S.; Byun, J.; Choi, J.H.; et al. Transplantation of endothelial progenitor cells accelerates dermal wound healing with increased recruitment of monocytes/macrophages and neovascularization. Stem Cells 2005, 23, 1571–1578. [Google Scholar] [CrossRef]
- Dobreva, I.; Waeber, G.; James, R.W.; Widmann, C. Interleukin-8 secretion by fibroblasts induced by low density lipoproteins is p38 MAPK-dependent and leads to cell spreading and wound closure. J. Biol. Chem. 2006, 281, 199–205. [Google Scholar] [CrossRef]
- Dobreva, I.; Zschornig, O.; Waeber, G.; James, R.W.; Widmann, C. Cholesterol is the major component of native lipoproteins activating the p38 mitogen-activated protein kinases. Biol. Chem. 2005, 386, 909–918. [Google Scholar]
- Bulat, N.; Waeber, G.; Widmann, C. LDLs stimulate p38 MAPKs and wound healing through SR-BI independently of Ras and PI3 kinase. J. Lipid Res. 2009, 50, 81–89. [Google Scholar] [CrossRef]
- Gu, X.; Kozarsky, K.; Krieger, M. Scavenger receptor class B, type I-mediated [3H]cholesterol efflux to high and low density lipoproteins is dependent on lipoprotein binding to the receptor. J. Biol. Chem. 2000, 275, 29993–30001. [Google Scholar] [CrossRef]
- Webb, N.R.; de Beer, M.C.; de Beer, F.C.; van der Westhuyzen, D.R. ApoB-containing lipoproteins in apoE-deficient mice are not metabolized by the class B scavenger receptor BI. J. Lipid Res. 2004, 45, 272–280. [Google Scholar]
- Feng, Y.; Lievens, J.; Jacobs, F.; Hoekstra, M.; van Craeyveld, E.; Gordts, S.C.; Snoeys, J.; De Geest, B. Hepatocyte-specific ABCA1 transfer increases HDL cholesterol but impairs HDL function and accelerates atherosclerosis. Cardiovasc. Res. 2010, 88, 376–385. [Google Scholar] [CrossRef]
- Zhang, H.; Wu, L.M.; Wu, J. Cross-talk between apolipoprotein E and cytokines. Mediat. Inflamm. 2011, 2011. [Google Scholar] [CrossRef]
- Baitsch, D.; Bock, H.H.; Engel, T.; Telgmann, R.; Muller-Tidow, C.; Varga, G.; Bot, M.; Herz, J.; Robenek, H.; von Eckardstein, A.; et al. Apolipoprotein E induces antiinflammatory phenotype in macrophages. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1160–1168. [Google Scholar] [CrossRef]
- Grainger, D.J.; Reckless, J.; McKilligin, E. Apolipoprotein E modulates clearance of apoptotic bodies in vitro and in vivo, resulting in a systemic proinflammatory state in apolipoprotein E-deficient mice. J. Immunol. 2004, 173, 6366–6375. [Google Scholar]
- Tada, T.; Ito, J.; Asai, M.; Yokoyama, S. Fibroblast growth factor 1 is produced prior to apolipoprotein E in the astrocytes after cryo-injury of mouse brain. Neurochem. Int. 2004, 45, 23–30. [Google Scholar] [CrossRef]
- Harr, S.D.; Uint, L.; Hollister, R.; Hyman, B.T.; Mendez, A.J. Brain expression of apolipoproteins E, J, and A-I in Alzheimer’s diseas. J. Neurochem. 1996, 66, 2429–2435. [Google Scholar]
- Grehan, S.; Allan, C.; Tse, E.; Walker, D.; Taylor, J.M. Expression of the apolipoprotein E gene in the skin is controlled by a unique downstream enhancer. J. Investig. Dermatol. 2001, 116, 77–84. [Google Scholar]
- Hu, Y.; Zou, Y.; Dietrich, H.; Wick, G.; Xu, Q. Inhibition of neointima hyperplasia of mouse vein grafts by locally applied suramin. Circulation 1999, 100, 861–868. [Google Scholar] [CrossRef]
- De Nardo, D.; Labzin, L.I.; Kono, H.; Seki, R.; Schmidt, S.V.; Beyer, M.; Xu, D.; Zimmer, S.; Lahrmann, C.; Schildberg, F.A.; et al. High-density lipoprotein mediates anti-inflammatory reprogramming of macrophages via the transcriptional regulator ATF3. Nat. Immunol. 2014, 15, 152–160. [Google Scholar] [CrossRef]
- Van Linthout, S.; Spillmann, F.; Graiani, G.; Miteva, K.; Peng, J.; van Craeyveld, E.; Meloni, M.; Tolle, M.; Escher, F.; Subasiguller, A.; et al. Down-regulation of endothelial TLR4 signalling after apo A-I gene transfer contributes to improved survival in an experimental model of lipopolysaccharide-induced inflammation. J. Mol. Med. 2011, 89, 151–160. [Google Scholar] [CrossRef]
- Tsuruoka, H.; Khovidhunkit, W.; Brown, B.E.; Fluhr, J.W.; Elias, P.M.; Feingold, K.R. Scavenger receptor class B type I is expressed in cultured keratinocytes and epidermis. Regulation in response to changes in cholesterol homeostasis and barrier requirements. J. Biol. Chem. 2002, 277, 2916–2922. [Google Scholar]
- Rono, B.; Engelholm, L.H.; Lund, L.R.; Hald, A. Gender affects skin wound healing in plasminogen deficient mice. PLoS One 2013, 8, e59942. [Google Scholar]
- Dos Santos, J.S.; Monte-Alto-Costa, A. Female, but not male, mice show delayed cutaneous wound healing following aspirin administration. Clin. Exp. Pharmacol. Physiol. 2013, 40, 90–96. [Google Scholar] [CrossRef]
- Gilliver, S.C.; Ruckshanthi, J.P.; Hardman, M.J.; Nakayama, T.; Ashcroft, G.S. Sex dimorphism in wound healing: The roles of sex steroids and macrophage migration inhibitory factor. Endocrinology 2008, 149, 5747–5757. [Google Scholar] [CrossRef]
- Romana-Souza, B.; Assis de Brito, T.L.; Pereira, G.R.; Monte-Alto-Costa, A. Gonadal hormones differently modulate cutaneous wound healing of chronically stressed mice. Brain Behav. Immun. 2014, 36, 101–110. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gordts, S.C.; Muthuramu, I.; Amin, R.; Jacobs, F.; De Geest, B. The Impact of Lipoproteins on Wound Healing: Topical HDL Therapy Corrects Delayed Wound Healing in Apolipoprotein E Deficient Mice. Pharmaceuticals 2014, 7, 419-432. https://doi.org/10.3390/ph7040419
Gordts SC, Muthuramu I, Amin R, Jacobs F, De Geest B. The Impact of Lipoproteins on Wound Healing: Topical HDL Therapy Corrects Delayed Wound Healing in Apolipoprotein E Deficient Mice. Pharmaceuticals. 2014; 7(4):419-432. https://doi.org/10.3390/ph7040419
Chicago/Turabian StyleGordts, Stephanie C., Ilayaraja Muthuramu, Ruhul Amin, Frank Jacobs, and Bart De Geest. 2014. "The Impact of Lipoproteins on Wound Healing: Topical HDL Therapy Corrects Delayed Wound Healing in Apolipoprotein E Deficient Mice" Pharmaceuticals 7, no. 4: 419-432. https://doi.org/10.3390/ph7040419




