Sensing Characteristics of Flame-Spray-Made Pt/ZnO Thick Films as H2 Gas Sensor
Abstract
:1. Introduction
2. Experimental
2.1. Particles Synthesis and Characterization
2.2. Sensing Films Preparation and Characterization of the Gas Sensing Properties
3. Results and Discussion
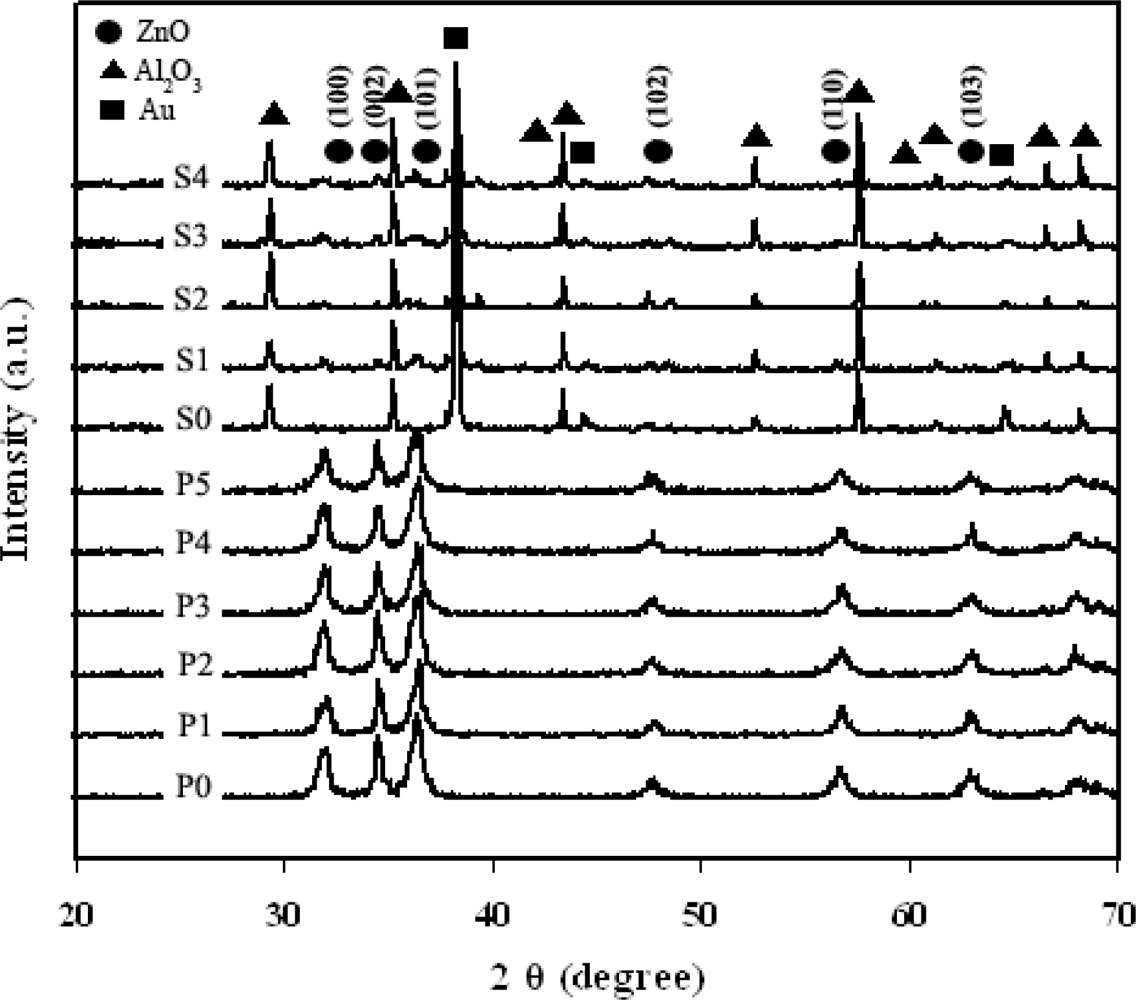
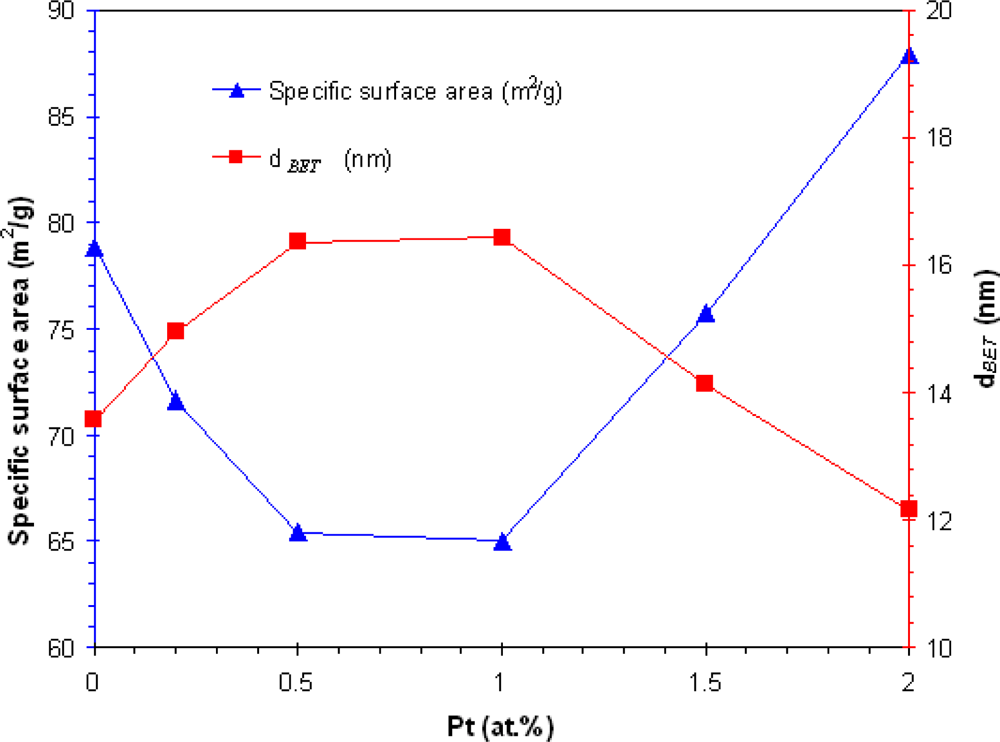
3.1. Particles Properties
3.2. SEM Sensing Layer
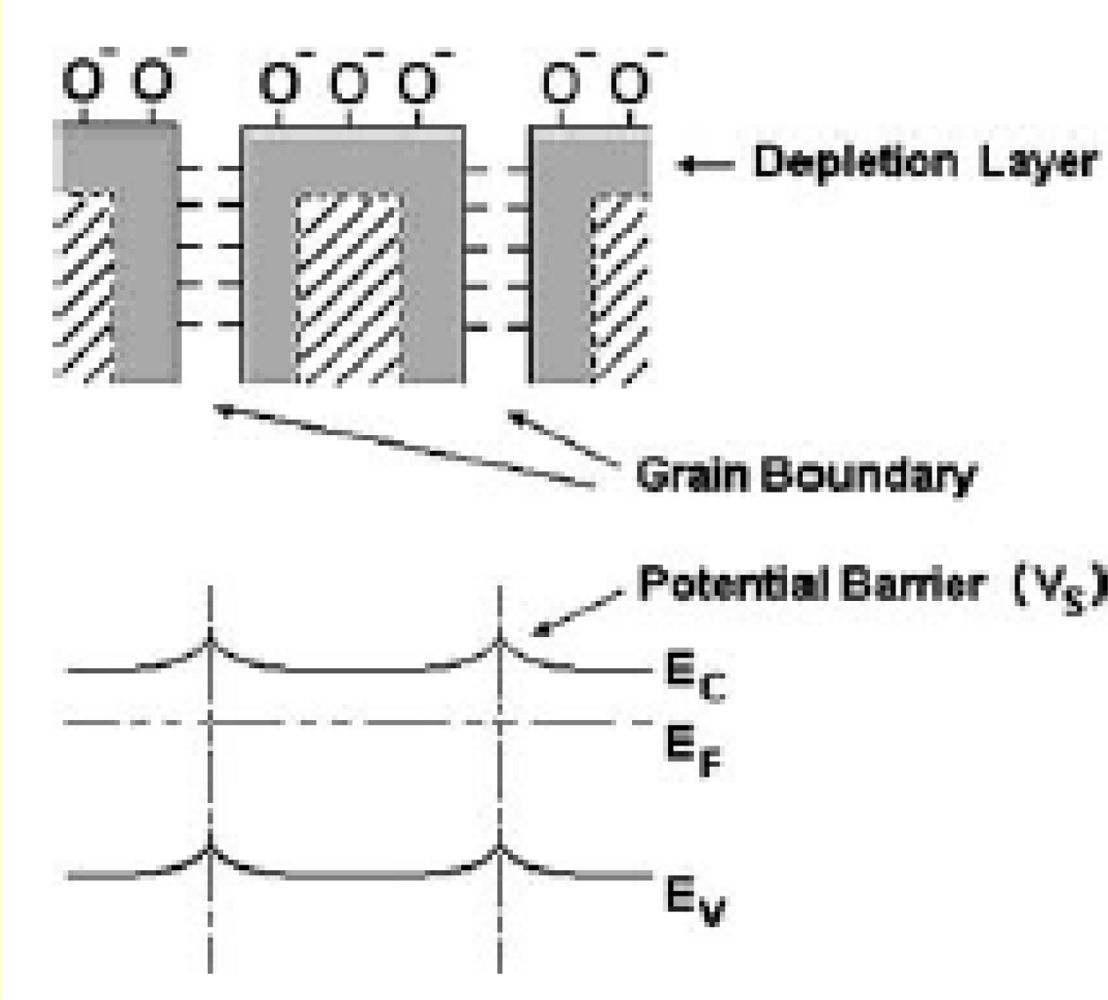
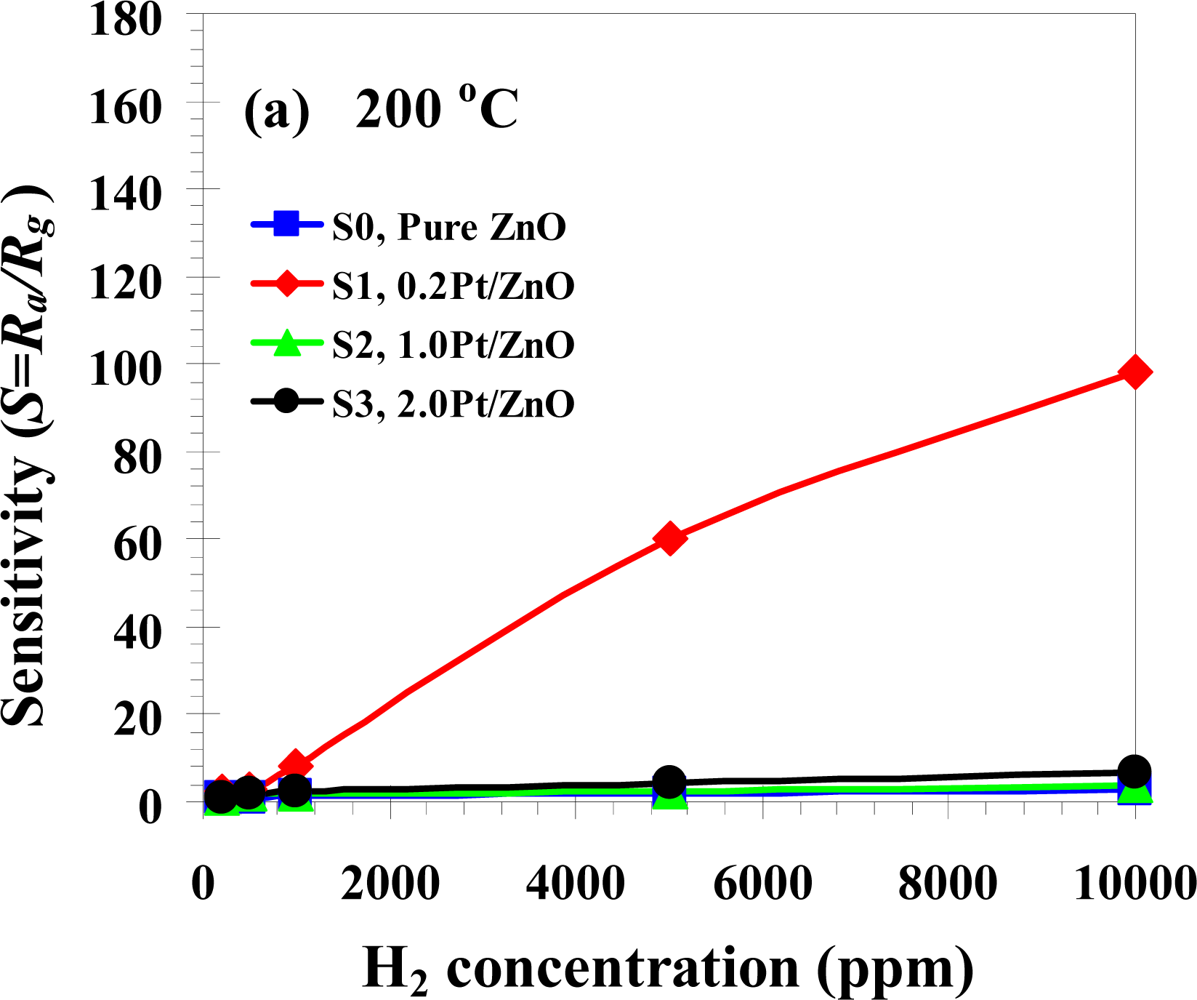
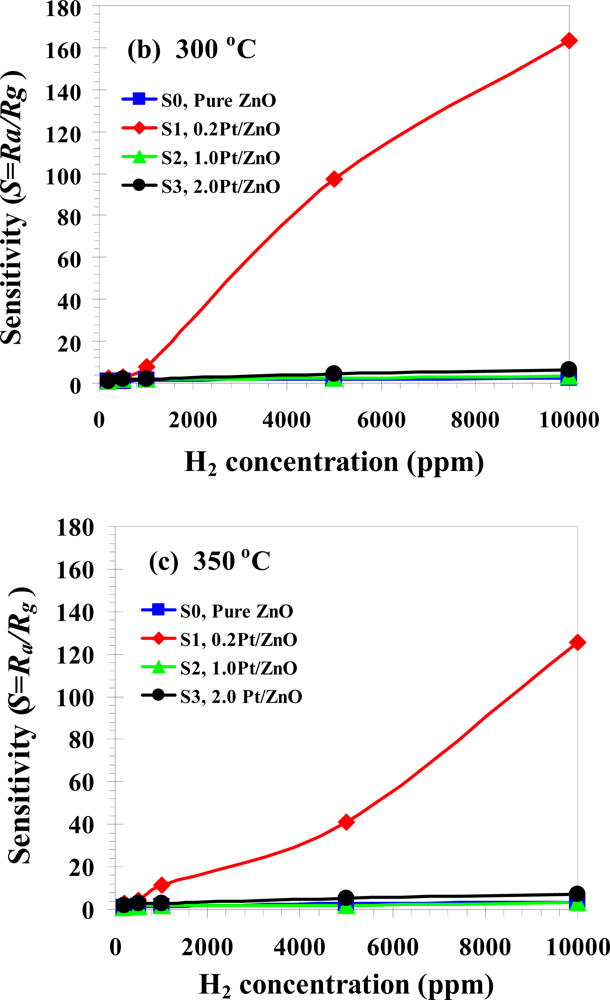
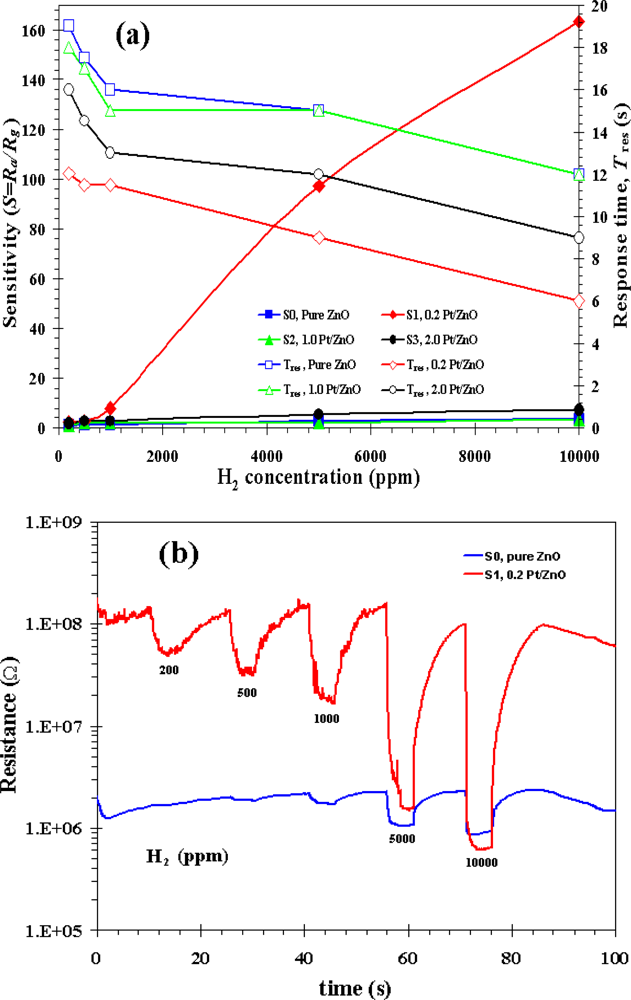
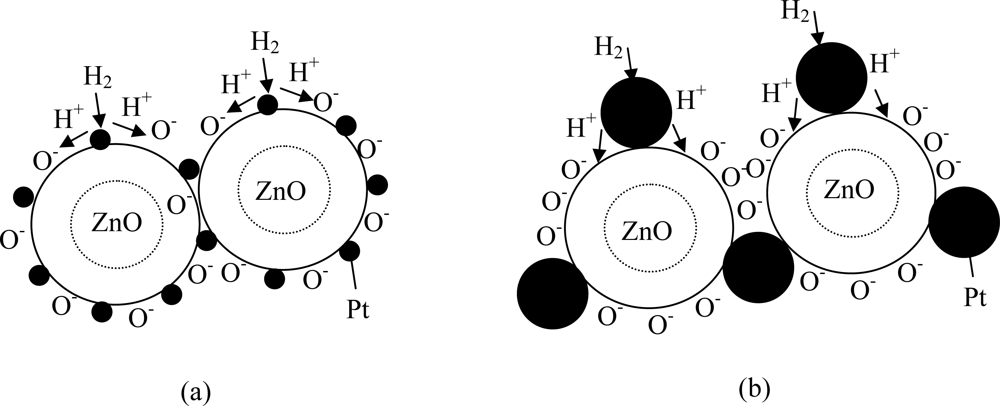
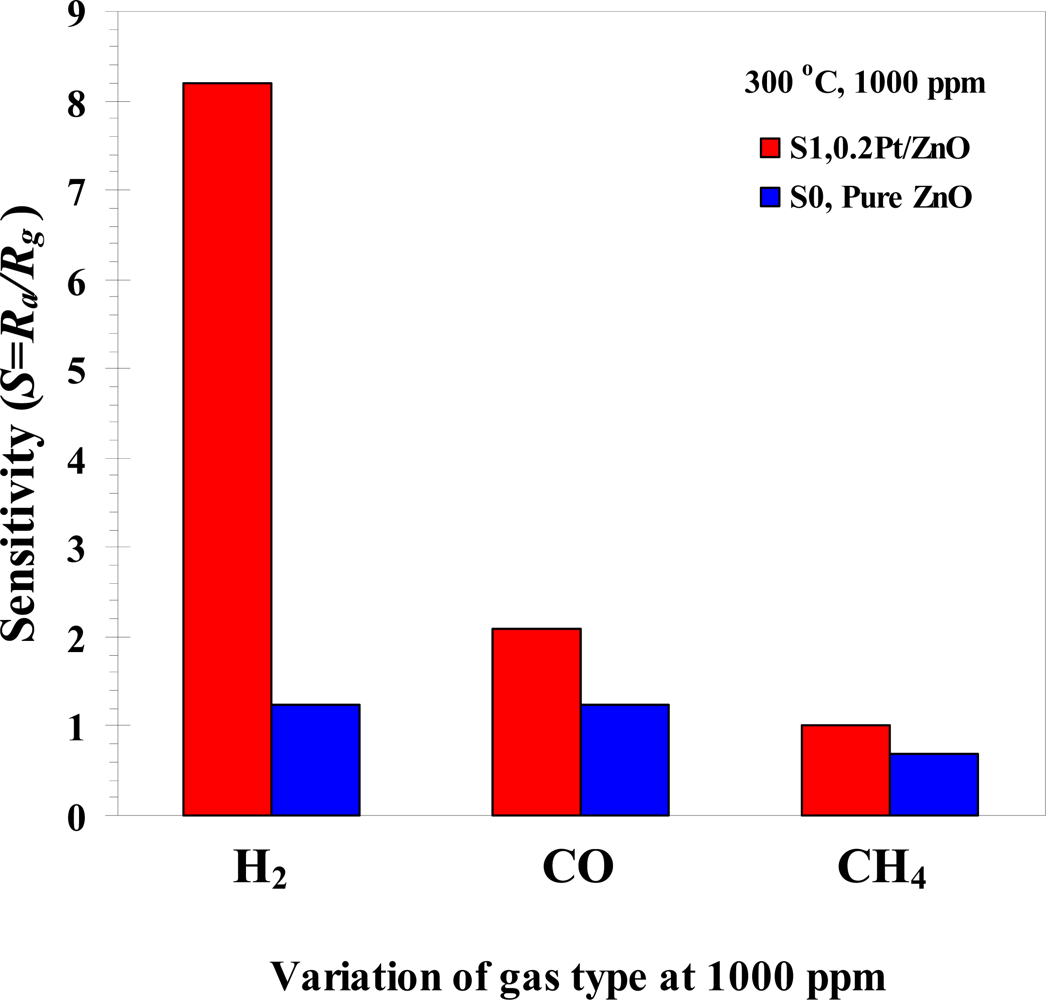
3.3. Gas Sensing Properties
4. Conclusions
Acknowledgments
References and Notes
- Choopun, S.; Hongsith, N.; Mangkorntong, P.; Mangkorntong, N. Zinc oxide nanobelts by RF sputtering for ethanol sensor. Physica E 2007, 39, 53–56. [Google Scholar]
- Wang, Y.; Ouyang, G.; Wang, L.L.; Tang, L.M.; Tang, D.S.; Chang, Q.S. Size- and composition-induced band-gap change of nanostructured compound of II–VI semiconductors. Chem. Phys. Lett 2008, 463, 383–386. [Google Scholar]
- Liewhiran, C.; Seraphin, S.; Phanichphant, S. Synthesis of nano-sized ZnO powders by thermal decomposition of zinc acetate using Broussonetia papyrifera (L.) Vent pulp as a dispersant. Curr. Appl. Phys 2006, 6, 499–502. [Google Scholar]
- Liewhiran, C.; Phanichphant, S. Influence of thickness on ethanol sensing characteristics of doctor-bladed thick film from flame-made ZnO nanoparticles. Sensors 2007, 7, 185–201. [Google Scholar]
- Liewhiran, C.; Phanichphant, S. Improvement of flame-made nanoparticulate thick film morphology for ethanol sensing. Sensors 2007, 7, 650–675. [Google Scholar]
- Purica, M.; Budianu, E.; Rusu, E.; Danila, M.; Gavrila, R. Optical and structural investigation of ZnO thin films prepared by chemical vapor deposition (CVD). Thin Solid Films 2002, 403–404, 485–488. [Google Scholar]
- Xu, J.; Shun, Y.; Pan, Q.; Qin, J. Sensing characteristics of double layer film of ZnO. Sensor. Actuat. B-Chem 2000, 66, 161–163. [Google Scholar]
- Rout, C.S.; Raju, A.R.; Govindaraj, A.; Rao, C.N.R. Hydrogen sensors based on ZnO nanoparticles. Solid State Commun 2006, 138, 136–138. [Google Scholar]
- Rout, C.S.; Krishna, S.H.; Vivekchand, S.R.C.; Govindaraj, A.; Rao, C.N.R. Hydrogen and ethanol sensors based on ZnO nanorods, nanowires and nanotubes. Chem. Phys. Lett 2006, 418, 586–590. [Google Scholar]
- Wang, H.T.; Kang, B.S.; Ren, F.; Tien, L.C.; Sadik, P.W.; Norton, D.P.; Pearton, S.J.; Lin, J. Detection of hydrogen at room temperature with catalyst-coated multiple ZnO nanorods. Appl. Phys. A 2005, 81, 1117–1119. [Google Scholar]
- Tien, L.C.; Sadik, P.W.; Norton, D.P.; Voss, L.F.; Pearton, S.J.; Wang, H.T.; Kang, B.S.; Ren, F.; Jun, J.; Lin, J. Hydrogen sensing at room temperature with Pt-coated ZnO thin films and Nanorods. Appl. Phys. Lett 2005, 87, 222106:1–222106:3. [Google Scholar]
- Kim, S.; Kang, B.S.; Ren, F.; Ip, K.; Heo, Y.W.; Norton, D.P.; Peartona, S.J. Sensitivity of Pt/ZnO Schottky diode characteristics to hydrogen. Appl. Phys. Lett 2004, 84, 1698–1700. [Google Scholar]
- Liu, Y.; Dong, J.; Hesketh, P.J.; Liu, M. Synthesis and gas sensing properties of ZnO single crystal flakes. J. Mater. Chem 2005, 15, 2316–2320. [Google Scholar]
- Xu, J.; Chen, Y.; Li, Y.; Shen, J. Gas sensing properties of ZnO nanorods prepared by hydrothermal method. J. Mater. Sci 2005, 40, 2919–2921. [Google Scholar]
- Feng, P.; Wan, Q.; Wang, T.H. Contact-controlled sensing properties of flowerlike ZnO nanostructure. Appl. Phys. Lett 2005, 87, 21311:1–21311:3. [Google Scholar]
- Xu, H.; Liu, X.; Cui, D.; Li, M.; Jiang, M. A novel method for improving the performance of ZnO gas sensors. Sens. Actuat. B-Chem 2006, 114, 301–307. [Google Scholar]
- Raju, A.R.; Rao, C.N.R. Gas-sensing characteristics of ZnO and copperimpregnated ZnO. Sens. Actuat. B-Chem 1991, 3, 305–310. [Google Scholar]
- Baruwati, B.; Kumar, D.K.; Manorama, S.V. Hydrothermal synthesis of highly crystalline ZnO nanoparticles: A competitive sensor for LPG and EtOH. Sens. Actuat. B-Chem 2006, 119, 676–682. [Google Scholar]
- Rao, B.B. Zinc oxide ceramic semi-conductor gas sensor for ethanol vapour. Mater. Chem. Phys 2000, 64, 62–65. [Google Scholar]
- Cheng, X.L.; Zhao, H.; Huo, L.H.; Gao, S.; Zhao, J.G. ZnO nanoparticulate thin film: preparation, characterization and gas-sensing property. Sens. Actuat. B-Chem 2004, 102, 248–252. [Google Scholar]
- Zhou, X.; Jiang, T.; Zhang, J.; Wang, X.; Zhu, Z. Humidity sensor based on quartz tuning fork coated with sol-gel-derived nanocrystalline zinc oxide thin film. Sens. Actuat. B-Chem 2006, 123, 299–305. [Google Scholar]
- Ge, C.; Xie, C.; Cai, S. Preparation and gas-sensing properties of Ce-doped ZnO thin film sensors by dip-coating. Mater. Sci. Eng. B 2007, 137, 53–58. [Google Scholar]
- Xiangfeng, C.; Dongli, J.; Djurišic, A.B.; Leung, Y.H. Gas-sensing properties of thick film based on ZnO nano-tetrapods. Chem. Phys. Lett 2005, 401, 426–429. [Google Scholar]
- Arshak, K.; Gaidan, I. Development of a novel gas sensor based on oxide thick films. Mater. Sci. Eng. B 2005, 118, 44–49. [Google Scholar]
- Baruwati, B.; Kumar, D.K.; Manorama, S.V. Hydrothermal synthesis of highly crystalline ZnO nanoparticles: A competitive sensor for LPG and EtOH. Sens. Actuat. B-Chem 2006, 119, 676–682. [Google Scholar]
- Dayan, N.J.; Karekar, R.N.; Aiyer, R.C.; Sainkar, S.R. Effect of film thickness and curing temperature on the sensitivity of ZnO:Sb thick-film hydrogen sensor. J. Mater. Sci.–Mater. Electron 1997, 8, 277–279. [Google Scholar]
- Zheng, L.; Xu, T.; Li, G.; Yin, Q. Influence of thickness on oxygen-sensing properties of TiO2 thin films on Al2O3. Jpn. J. Appl. Phys. Part 1 2002, 41, 4655–4658. [Google Scholar]
- Lee, Y.L.; Sheu, C.Y.; Hsiao, R.H. Gas sensing characteristics of copper phthalocyanine films; effects of film thickness and sensing temperature. Sens. Actuat. B-Chem 2004, 99, 281–287. [Google Scholar]
- Babari, F.H.; Orvatinia, M. Analysis of thickness dependence of the sensitivity in thin film resistive gas sensors. Sens. Actuat. B-Chem 2003, 89, 256–261. [Google Scholar]
- Geistlinger, H. Electron theory of thin-film gas sensors. Sens. Actuat. B-Chem 1993, 17, 47–60. [Google Scholar]
- López, M.A.R.; Peiteado, M.; Fernández, J.F.; Caballero, A.C.; Holc, J.; Drnovsek, S.; Kuscer, D.; Macek, S.; Kosec, M. Thick film ZnO based varistors prepared by screen printing. J. Eur. Ceram. Soc 2006, 26, 2985–2989. [Google Scholar]
- Chang, J.F.; Kuo, H.H.; Leu, I.C.; Hon, M.H. The effects of thickness and operation temperature on ZnO:Al thin film CO gas sensor. Sens. Actuat. B-Chem 2002, 84, 258–264. [Google Scholar]
- Christoulakis, S.; Suchea, M.; Koudoumas, E.; Katharakis, M.; Katsarakis, N.; Kiriakidis, G. Thickness influence on surface morphology and ozone sensing properties of nanostructured ZnO transparent thin films grown by PLD. Appl. Surf. Sci 2006, 252, 5351–5354. [Google Scholar]
- Sakai, G.; Baik, N.S.; Miura, N.; Yamazoe, N. Gas sensing properties of tin oxide thin films fabricated from hydrothermally treated nanoparticles dependence of CO and H2 response on film thickness. Sens. Actuat. B-Chem 2001, 77, 116–121. [Google Scholar]
- Maedler, L.; Kammler, H.K.; Mueller, R.; Pratsinis, S.E. Controlled synthesis of nanostructured particles by flame spray pyrolysis. J. Aerosol. Sci 2002, 33, 369–389. [Google Scholar]
- Liewhiran, C.; Phanichphant, S. Effects of palladium loading on the response of a thick film flame-made ZnO gas sensor for detection of ethanol vapor. Sensors 2007, 7, 1159–1184. [Google Scholar]
- Murray, J.H.; Lutz, M.; Sotiris, E.P. Nanorods of ZnO made by flame spray pyrolysis. Chem. Mater 2006, 1, 572–578. [Google Scholar]
- Liewhiran, C.; Phanichphant, S. Doctor-bladed thick films of flame-made Pd/ZnO nanoparticles for ethanol sensing. Curr. Appl. Phys 2008, 8, 336–339. [Google Scholar]
- Kisi, E.H.; Elcombe, M.M. u Parameters for the wurtzite structure of ZnS and ZnO using powderneutron diffraction. Acta Crystallogr. Sect. C: Cryst. Struct. Commun 1989, 45, 1867–1870. [Google Scholar]
- Liu, R.S.; Shi, W.C.; Cheng, Y.C.; Huang, C.Y. Crystal structures and peculiar magnetic properties of alpha- and gamma-(Al2O3) powders. Mod. Phys. Lett. B 1997, 11, 1169–1174. [Google Scholar]
- Swanson, H.E.; Tatge, E. Standard X-ray diffraction powder patterns. Z. Angew. Phys 1956, 8, 202–205. [Google Scholar]
- Wan, Q.; Li, Q.H.; Chen, Y.J.; He, X.L.; Li, J.P.; Lin, C.L.; Wang, T.H. Fabrication and ethanol sensing characteristics of ZnO nanowire gas sensors. Appl. Phys. Lett 2004, 84, 3654–3656. [Google Scholar]
- Wegner, K.; Stark, W.J.; Pratsinis, S.E. Flame-nozzle synthesis of nanoparticles with closely controlled size, morphology and crystallinity. Mater. Lett 2002, 55, 318–321. [Google Scholar]
- Morrison, S.R. Selectivity in semiconductor gas sensors. Sens. Actuat. B-Chem 1987, 12, 425–440. [Google Scholar]




| Authors | Method | Doping level | Range (H2) | Sensitivity |
|---|---|---|---|---|
| Xu et al. [6] | Chemical precipitation | 0.5 wt% Pt | 0.2% in air | 2 at 330 °C |
| 0.5 wt% Ru | 0.2% in air | 6 at 230 °C and 2 at 400 °C | ||
| 0.5 wt% Rh | 0.2% in air | 115 at 300 °C | ||
| 0.5 wt% Ag | 0.2% in air | 9 at 400 °C | ||
| Rout et al. [8] | ZnO nanowires and ZnO nanotubes by electrochemical deposition | 0 at.% Pt | 1,000 ppm in air | 43 at 150 °C |
| 1,000 ppm in air | ||||
| 1 at.% Pt | 1,000 ppm in air | 825 at 150 °C | ||
| 1,000 ppm in air | ||||
| 0 at.% Pt | 18 at 150 °C | |||
| 1 at.% Pt | 740 at 150 °C | |||
| Wang et al. [9] | ZnO nanorods deposited by Molecular Beam Epitaxy (MBE) | Pt-coated | 500 ppm in N2 | Relative response of up to 8% H2 at room temp. |
| Tien et al. [10] | ZnO nanorods / thin films deposited by Molecular Beam Epitaxy (MBE) | Pt-coated | 500 ppm in N2 | Response of Pt-coated nanorods was 3 times of thin films as prepared ZnO |
| Phanichphant et al. [this work] | Flame spray pyrolysis | 0 at.% | 1% (10,000 ppm in air) | 0.2 at.%Pt showed the highest sensitivity of 164 at 350 °C |
| 0.2 at.% | ||||
| 1 at.% | ||||
| 2 at.% | 0.2 at.%Pt showed the highest sensitivity of 8.2 at 350 °C | |||
| 0.1% (1,000 ppm in air) | ||||
© 2009 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tamaekong, N.; Liewhiran, C.; Wisitsoraat, A.; Phanichphant, S. Sensing Characteristics of Flame-Spray-Made Pt/ZnO Thick Films as H2 Gas Sensor. Sensors 2009, 9, 6652-6669. https://doi.org/10.3390/s90906652
Tamaekong N, Liewhiran C, Wisitsoraat A, Phanichphant S. Sensing Characteristics of Flame-Spray-Made Pt/ZnO Thick Films as H2 Gas Sensor. Sensors. 2009; 9(9):6652-6669. https://doi.org/10.3390/s90906652
Chicago/Turabian StyleTamaekong, Nittaya, Chaikarn Liewhiran, Anurat Wisitsoraat, and Sukon Phanichphant. 2009. "Sensing Characteristics of Flame-Spray-Made Pt/ZnO Thick Films as H2 Gas Sensor" Sensors 9, no. 9: 6652-6669. https://doi.org/10.3390/s90906652




