Nanomaterials - Acetylcholinesterase Enzyme Matrices for Organophosphorus Pesticides Electrochemical Sensors: A Review
Abstract
:1. Introduction
2. Nanomaterials Based AChE Sensors
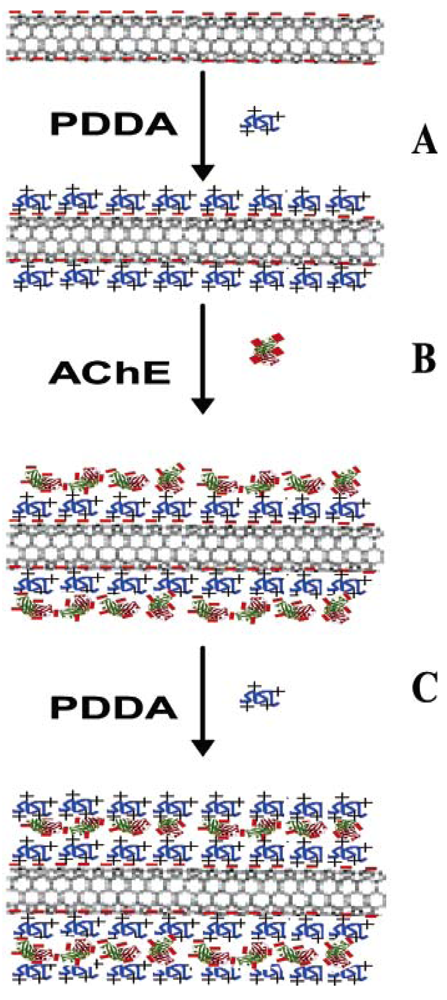
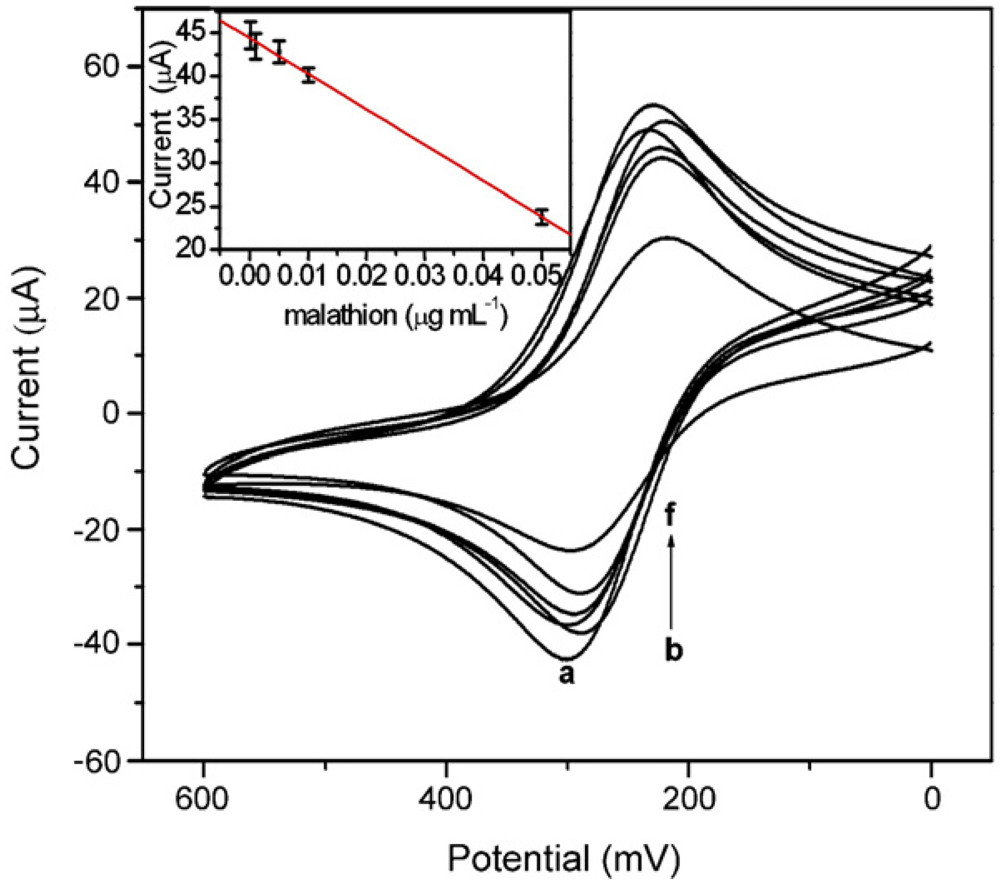
2.1. Carbon Nanotubes based AChE Sensors
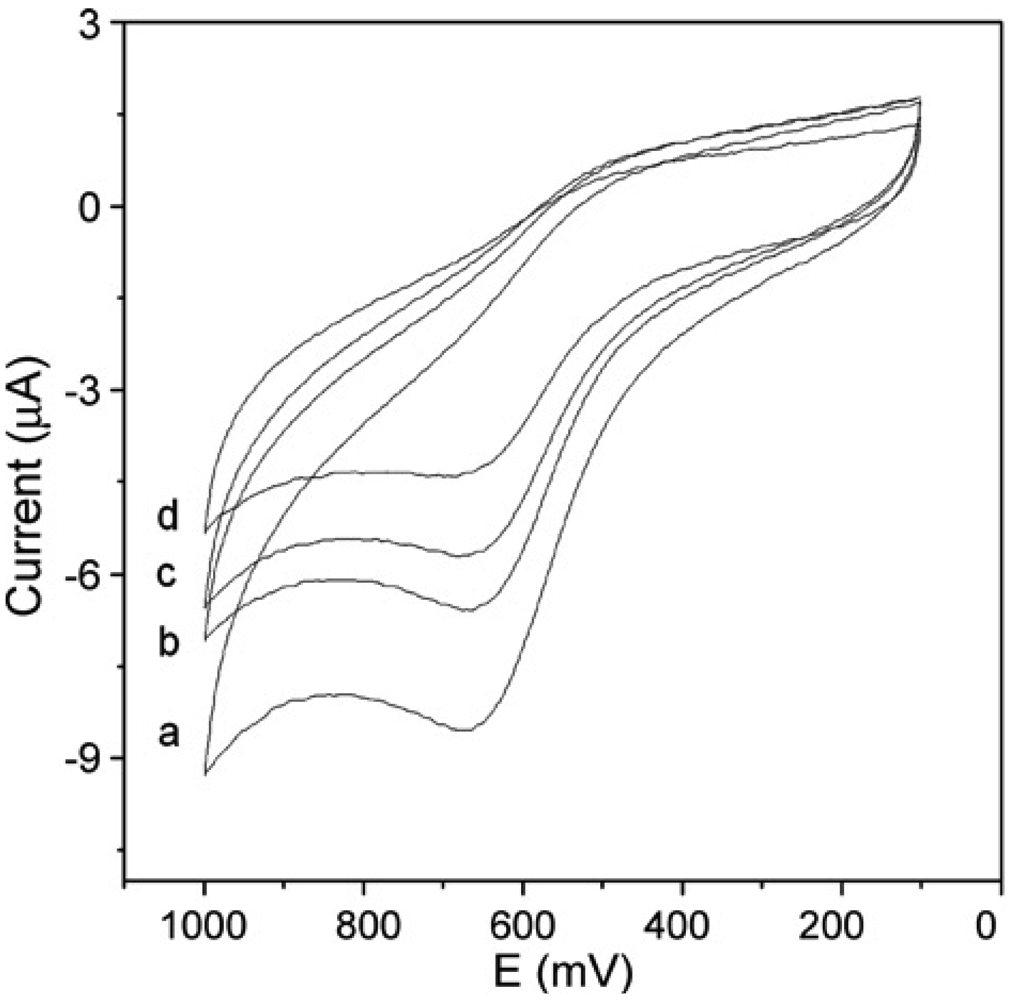
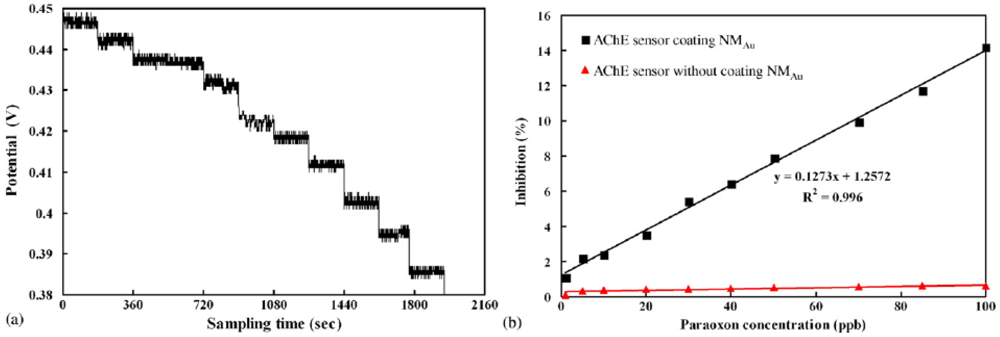
2.2. Gold Nanoparticles Based AChE Sensors
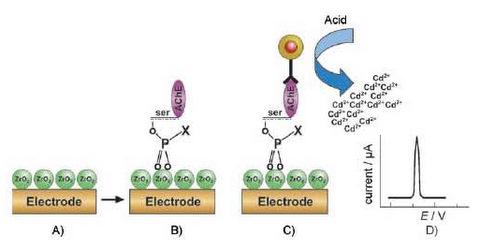
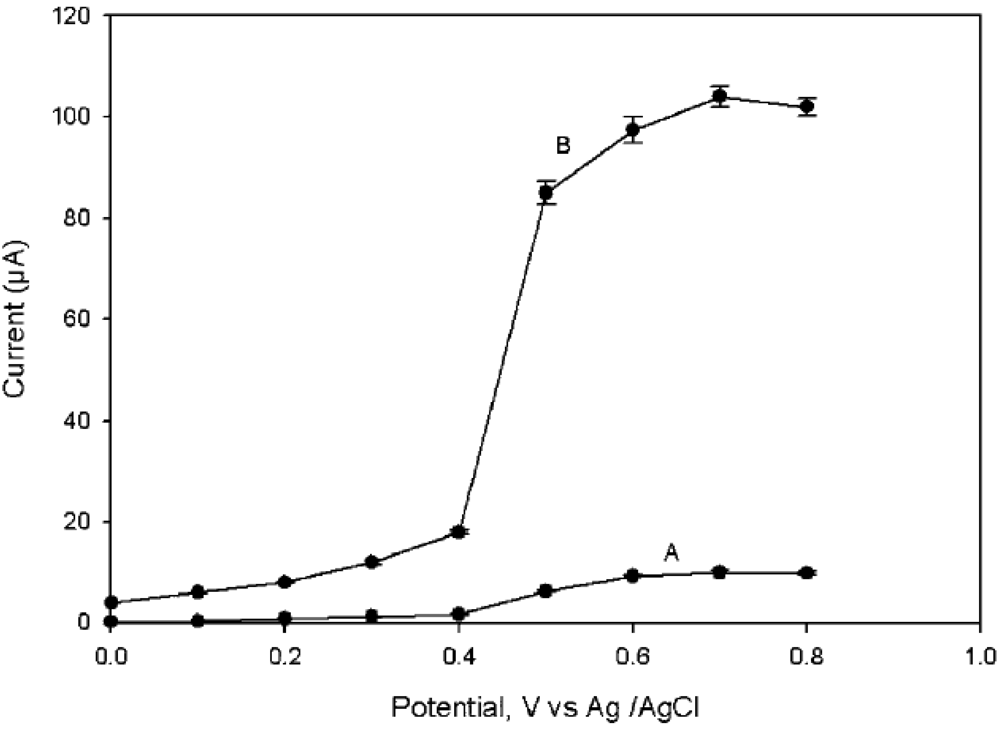
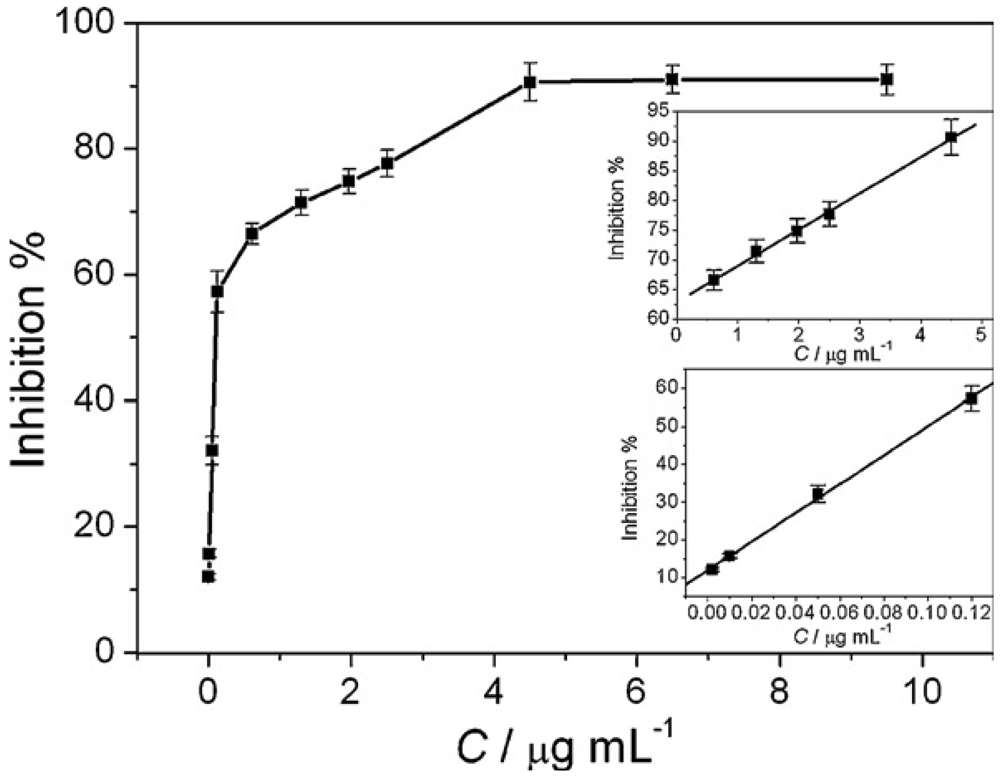
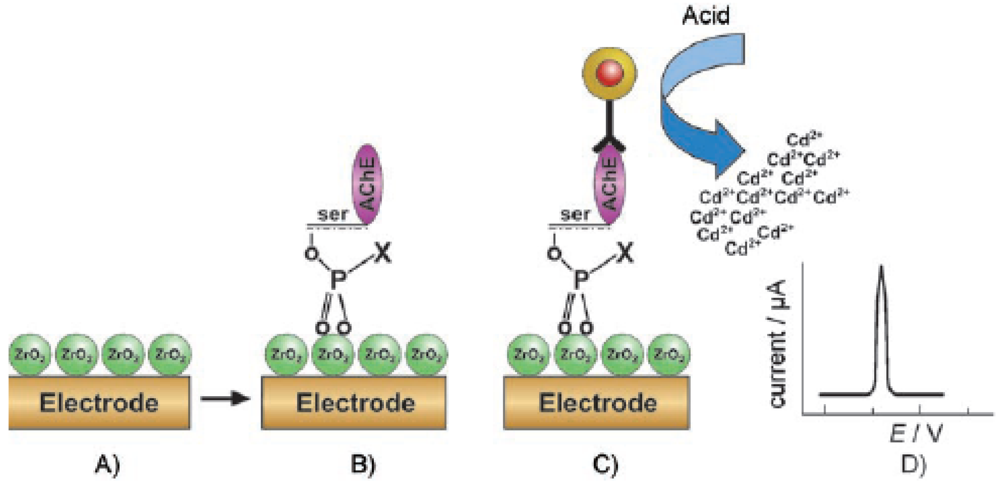
2.3. Zirconia Nanoparticles Based AChE immunosensors
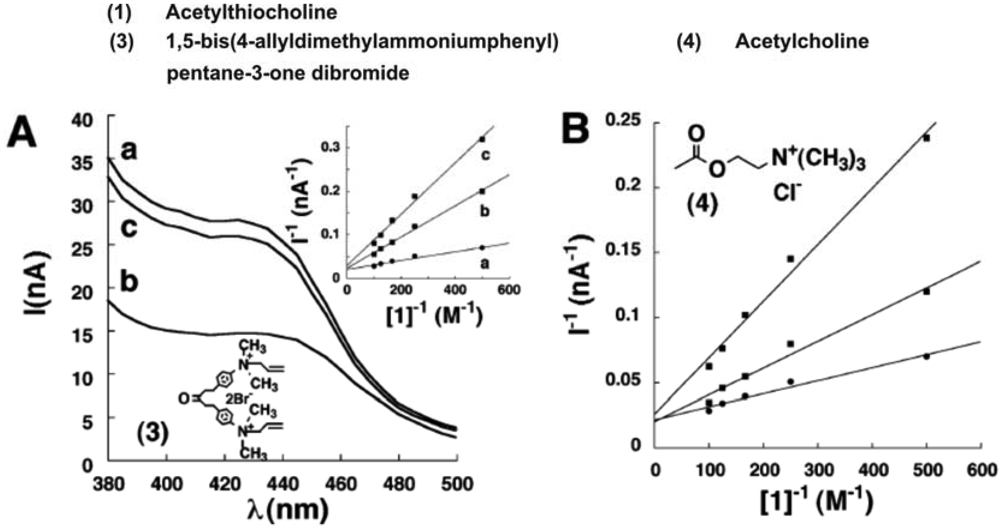
2.4. CdS Nanoparticles based Photoelectrochemical AChE Sensors
2.5. CdS Quantum Dots based AChE Sensors
3. Advantages of Using Nanomaterials for AChE Immobilization
3.1. Comparison of Real Sample Analysis of OP Pesticides at Nanomaterial Modified Electrodes and the Chromatographic Techniques
4. Conclusions
Acknowledgments
References
- Pandey, P.C.; Upadhyay, S.; Pathak, H.C.; Pandy, C.M.D.; Tiwari, I. Acetylthiocholine/acetylcholine and thiocholine/choline electrochemical biosensors/sensors based on an organically modified sol-gel glass enzyme reactor and graphite paste electrode. Sens. Actuat. B 2000, 62, 109–116. [Google Scholar]
- Stoytcheva, M.; Sharkova, V.; Magnin, J.P. Electrochemical approach in studying the inactivation of immobilized acetylcholinesterase by arsenate(III). Electroanalysis 1998, 10, 994–998. [Google Scholar]
- Shi, M.; Xu, J.; Zhang, S.; Liu, B.; Kong, J. A mediator-free screen-printed amperometric biosensor for screening of organophosphorus pesticides with flow-injection analysis (FIA) system. Talanta 2006, 68, 1089–1095. [Google Scholar]
- Neufeld, T.; Eshkenazi, I.; Cohen, E.; Rishpon, J. A micro flow injection electrochemical biosensor for organophosphorus pesticides. Biosens. Bioelectron. 2000, 15, 323–329. [Google Scholar]
- Corcia, A.D.; Marchetti, M. Multiresidue method for pesticides in drinking water using a graphitized carbon black cartridge extraction and liquid chromatographic analysis. Anal. Chem. 1991, 63, 580–585. [Google Scholar]
- Bakker, E.; Qin, Y. Electrochemical sensors. Anal. Chem. 2006, 78, 3965–3984. [Google Scholar]
- Anitha, K.; Mohan, S.V.; Reddy, S.J. Development of acetylcholinesterase silica sol-gel immobilized biosensor - an application towards oxydemeton methyl detection. Biosens. Bioelectron. 2004, 20, 848–856. [Google Scholar]
- Khayyami, M.; Pita, M.T.P.; Garcia, N.P.; Johansson, G.; Danielsson, B.; Larsson, P.O. Development of an amperometric biosensor based on acetylcholine esterase covalently bound to a new support material. Talanta 1998, 45, 557–563. [Google Scholar]
- Suprun, E.; Evtugyn, G.; Budnikov, H.; Ricci, F.; Moscone, D.; Palleschi, G. Acetylcholinesterase sensor based on screen-printed carbon electrode modified with prussian blue. Anal. Bioanal. Chem. 2005, 383, 597–604. [Google Scholar]
- Joshi, K.A.; Tang, J.; Haddon, R.; Joseph, W.; Chen, W.; Mulchandani, A. A disposable biosensor for organophosphorus nerve agents based on carbon nanotubes modified thick film strip electrode. Electroanalysis 2005, 17, 54–58. [Google Scholar]
- Liu, G.; Riechers, S.L.; Mellen, M.C.; Lin, Y. Sensitive electrochemical detection of enzymatically generated thiocholine at carbon nanotube modified glassy carbon electrode. Electrochem. Commun. 2005, 7, 1163–1169. [Google Scholar]
- Liu, G.; Lin, Y. Biosensor based on self-assembling acetylcholinesterase on carbon nanotubes for flow injection/amperometric detection of organophosphate pesticides and nerve agents. Anal. Chem. 2006, 78, 835–843. [Google Scholar]
- Du, D.; Huang, X.; Cai, J.; Zhang, A.; Ding, J.; Chen, S. An amperometric acetylthiocholine sensor based on immobilization of acetylcholinesterase on a multiwall carbon nanotube–cross-linked chitosan composite. Anal. Bioanal. Chem. 2007, 387, 1059–1065. [Google Scholar]
- Du, D.; Huang, X.; Cai, J.; Zhang, A. Amperometric detection of triazophos pesticide using acetylcholinesterase biosensor based on multiwall carbon nanotube-chitosan matrix. Sens. Actuat. B 2007, 127, 531–535. [Google Scholar]
- Du, D.; Cai, J.; Song, D.; Zhang, A. Rapid determination of triazophos using acetylcholinesterase biosensor based on sol-gel interface assembling multiwall carbon nanotubes. J. Appl. Electrochem. 2007, 37, 893–898. [Google Scholar]
- Lin, T.L.; Huang, K.T.; Liu, C.Y. Determination of organophosphorus pesticides by a novel biosensor based on localized surface plasmon resonance. Biosens. Bioelectron. 2006, 22, 513–518. [Google Scholar]
- Du, D.; Ding, J.; Cai, J.; Zhang, A. Electrochemical thiocholine inhibition sensor based on biocatalytic growth of Au nanoparticles using chitosan as template. Sens. Actuat. B 2007, 127, 317–322. [Google Scholar]
- Du, D.; Chen, S.; Cai, J.; Zhang, A. Immobilization of acetylcholinesterase on gold nanoparticles embedded in sol-gel film for amperometric detection of organophosphorus insecticide. Biosens. Bioelectron. 2007, 23, 130–134. [Google Scholar]
- Du, D.; Chen, S.; Songa, D.; Li, H.; Chen, X. Development of acetylcholinesterase biosensor based on CdTe quantum dots/gold nanoparticles modified chitosan microspheres interface. Biosens. Bioelectron. 2008, 24, 475–479. [Google Scholar]
- Gong, J.; Wang, L.; Zhang, L. Electrochemical biosensing of methyl parathion pesticide based on acetylcholinesterase immobilized onto Au–polypyrrole interlaced network-like nanocomposite. Biosens. Bioelectron. 2009, 24, 2285–2288. [Google Scholar]
- Pumera, M.; Sanchez, S.; Ichinose, I.; Tang, J. Electrochemical nanobiosensors. Sens. Actuat. B 2007, 123, 1195–1205. [Google Scholar]
- Guo, S.; Wang, E. Synthesis and electrochemical applications of gold nanoparticles. Anal. Chim. Acta 2007, 598, 181–192. [Google Scholar]
- Kerman, K.; Saito, M.; Yamamura, S.; Takamura, Y.; Tamiya, E. Nanomaterial-based electrochemical biosensors for medical applications. Trends Anal. Chem. 2008, 27, 585–592. [Google Scholar]
- Breton, Y.; Desarmot, G.; Salvetat, J.P.; Delpeux, S.; Sinturel, C.; Beguin, F.; Bonnamy, S. Mechanical properties of multiwall carbon nanotubes/epoxy composites: influence of network morphology. Carbon 2004, 42, 1027–1030. [Google Scholar]
- Xu, Z.; Wang, L.; Zheng, Q. Enhanced mechanical properties of prestressed multi-walled carbon nanotubes. Small 2008, 4, 733–737. [Google Scholar]
- Hobara, R.; Yoshimoto, S.; Ikuno, T.; Katayama, M.; Yamauchi, N.; Wongwiriyapan, W.; Honda, S.I.; Matsuda, I.; Hasegawa, S.; Kenjiro, O. Electronic transport in multiwalled carbon nanotubes contacted with patterned electrodes. Jap. J. Appl. Phys. 2004, 43, 1081–1084. [Google Scholar]
- Ulbricht, R.; Lee, S.; Jiang, X.; Inoue, K.; Zhang, M.; Fang, S.; Baughman, R.H.; Zakhidov, A.A. Transparent carbon nanotube sheets as 3-D charge collectors in organic solar cells. Sol. Energy Mater. Sol. Cells 2007, 91, 416–419. [Google Scholar]
- Allen, B.L.; Kichambare, P.D.; Star, A. Carbon nanotube field-effect-transistor-based biosensors. Adv. Mater. 2007, 19, 1439–1451. [Google Scholar]
- Kachoosangi, R.T.; Musameh, M.M.; Yousef, I.A.; Yousef, J.M.; Kanan, S.M.; Xiao, L.; Davies, S.G.; Russell, A.; Compton, R.G. Carbon nanotube-ionic liquid composite sensors and biosensors. Anal. Chem. 2009, 81, 435–442. [Google Scholar]
- Vairavapandian, D.; Vichchulada, P.; Lay, M.D. Preparation and modification of carbon nanotubes: Review of recent advances and applications in catalysis and sensing. Anal. Chim. Acta 2008, 62, 6119–129. [Google Scholar]
- Mauter, M.; Elimelech, M. Environmental applications of carbon-based nanomaterials. Environ. Sci. Technol. 2008, 42, 5843–5859. [Google Scholar]
- Valentini, F.; Palleschi, G. Nanomaterials and analytical chemistry. Anal. Lett. 2008, 41, 479–520. [Google Scholar]
- Agui, L.; Sedeno, P.L.; Pingarron, J.M. Role of carbon nanotubes in electroanalytical chemistry A review. Anal. Chim. Acta 2008, 622, 11–47. [Google Scholar]
- Liu, G.; Wang, J.; Barry, R.; Petersen, C.; Timchalk, C.; Gassman, P.L.; Lin, Y. Nanoparticle-based electrochemical immunosensor for the detection of phosphorylated acetylcholinesterase: an exposure biomarker of organophosphate pesticides and nerve agents. Chem. Eur. J. 2008, 14, 9951–9959. [Google Scholar]
- Pardo-Yissar, V.; Katz, E.; Wasserman, J.; Willner, I. Acetylcholine esterase-labeled CdS nanoparticles on electrodes: photoelectrochemical sensing of the enzyme inhibitors. J. Am. Chem. Soc. 2003, 125, 622–623. [Google Scholar]
- Li, X.H.; Xie, Z.; Min, H.; Li, C.; Liu, M.; Xian, Y.; Jin, L. Development of quantum dots modified acetylcholinesterase biosensor for the detection of trichlorfon. Electroanalysis 2006, 18, 2163–2167. [Google Scholar]
- Muccio, A.D.; Pelosi, P.; Camoni, I.; Barbini, D.A.; Dommarco, R.; Generali, T.; Ausil, A. Selective, solid-matrix dispersion extraction of organophosphate pesticide residues from milk. J. Chromatogr. A 1996, 754, 497–506. [Google Scholar]
- Lacorte, S.; Barcel, D. Determination of organophosphorus pesticides and their transformation products in river waters by automated on-line solid-phase extraction followed by thermospray liquid chromatography-mass spectrometry. J. Chrornatogr. A 1995, 712, 103–112. [Google Scholar]
- Juhler, R.K. Optimized method for the determination of organophosphorus pesticides in meat and fatty matrices. J. Chromatogr. A 1997, 786, 145–153. [Google Scholar]
- Fernandez, M.; Pico, Y.; Girotti, S.; Manes, J. Analysis of organophosphorus pesticides in honeybee by liquid chromatography-atmospheric pressure chemical ionization-mass spectrometry. J. Agric. Food Chem. 2001, 49, 3540–3547. [Google Scholar]
- Ruiz, T.P.; Lozano, C.M.; Sanz, A.; Bravo, E. Determination of organophosphorus pesticides in water, vegetables and grain by automated SPE and MEKCE. Chromatographia 2005, 61, 493–498. [Google Scholar]
- Albero, B.; Brunete, C.S; Tadeo, J.L. Determination of Organophosphorus Pesticides in Fruit Juices by Matrix Solid-Phase Dispersion and Gas Chromatography. J. Agric. Food Chem. 2003, 51, 6915–6921. [Google Scholar]
- Corcia, A.D.; Marchetti, M. Multiresidue method for pesticides in drinking water using a graphitized carbon black cartridge extraction and liquid chromatographic analysis. Anal. Chem. 1991, 63, 580–585. [Google Scholar]
- Campanella, L.; Lelo, D.; Martini, E.; Tomassetti, M. Organophosphorus and carbamate pesticide analysis using an inhibition tyrosinase organic phase enzyme sensor; comparison by butyrylcholinesterase + choline oxidase opee and application to natural waters. Anal.Chim. Acta 2007, 587, 22–32. [Google Scholar]
- Virel, A.; Saa, L.; Pavlov, V. Modulated growth of nanoparticles. Application for sensing nerve gases. Anal. Chem. 2009, 81, 268–272. [Google Scholar]
- Somerset, V.S.; Klink, M.J.; Baker, P.G.L.; Iwuoha, E.I. Acetylcholinesterase-polyaniline biosensor investigation of organophosphate pesticides in selected organic solvents. J. Environ. Sci. Health B 2007, 42, 297–304. [Google Scholar]
- Zourob, M.; Ong, K.G.; Zeng, K.; Mouffouk, F.; Grimes, C.A. A wireless magnetoelastic biosensor for the direct detection of organophosphorus pesticides. Analyst 2007, 132, 338–343. [Google Scholar]
- Campanella, L.; Bonanni, A.; Martini, E.; Todini, N.; Tomassetti, M. Determination of triazine pesticides using a new enzyme inhibition tyrosinase OPEE operating in chloroform. Sens. Actuat. B 2005, 111-112, 505–514. [Google Scholar]
- Campanella, L.; Dragone, R.; Lelo, D.; Martini, E.; Tomassetti, M. Tyrosinase inhibition organic phase biosensor for triazinic and benzotriazinic pesticide analysis (part two). Anal. Bioanal. Chem. 2006, 384, 915–921. [Google Scholar]
- Hu, S.Q.; Xie, J.W.; Xu, Q.H.; Rong, K.; Shen, G.L.; Yu, R. A label-free electrochemical immunosensor based on gold nanoparticles for detection of paraoxon. Talanta 2003, 61, 769–777. [Google Scholar]
- Jiang, X.; Li, D.; Xu, X.; Ying, Y.; Li, Y.; Ye, Z.; Wang, J. Immunosensors for detection of pesticide residues. Biosens. Bioelectron. 2008, 23, 1577–1587. [Google Scholar]
- Lopez, M.S.; Lopez-Cabarcos, E.; Lopez-Ruiz, B. Organic phase enzyme electrodes. Biomol. Eng. 2006, 23, 135–147. [Google Scholar]
- Campanella, L.; Lelo, D.; Martini, E.; Tomassetti, M. Investigation of interfering species in phytodrug analysis using an inhibition tyrosinase enzyme electrode working both in water and in organic solvent. Anal. Lett. 2008, 41, 1106–1127. [Google Scholar]
- Darko, G.; Akoto, O. Dietary intake of organophosphorus pesticide residues through vegetables from Kumasi, Ghana. Food Chem. Toxicol. 2008, 46, 3703–3706. [Google Scholar]
- Andreescu, S.; Marty, J.L. Twenty years research in cholinesterase biosensors: From basic research to practical applications. Biomol. Eng. 2006, 23, 1–15. [Google Scholar]


| Immobilization method | Electrode type | Techniques [Incubation time] | OP (Linear conc. in M) | Detection limit in M [correlation coefficient] | Ref. |
|---|---|---|---|---|---|
| Physical adsorption | MWCNTs/SPE | Amperometry [30 min] | Paraoxon (1.0 × 10-9 to 6.9 × 10-9) | 0.5 × 10-9 [0.9859] | [10] |
| LBL self assembling technique | PDDA/AChE/PDDA/MWCNT/GCE | FIA [6 min] | Paraoxon (1 × 10-12 to 0.1 × 10-9) | 0.4 × 10-12 | [12] |
| Covalent immobilization using glutaraldehyde as cross linking agent | AChE/MWCNTs-Chi/GCE | CV [10 min] | Triazophos (0.03 × 10-6 to 7.8 × 10-6 and 7.8 × 10-6 to 32 × 10-6) | 0.01 × 10-6 [0.9966, 0.9960] | [14] |
| Physical entrapment | MWCNTs/SiSG/GCE | CV [12 min] | Triazophos (0.02 × 10-6 to 1 × 10-6 and 5 × 10-6 to 30 × 10-6) | 5.0 × 10-9 [0.9957 and 0.9986] | [15] |
| Self assembled monolayer (SAM) | Optical fibers modified with self assembled AuNPs | LSPR [14 h] | Paraoxon (3.63 × 10-9 to 0.36 × 10-6) | 0.85 × 10-9 [0.996] | [16] |
| Physical adsorption | AChE/AuNPs/Chi | CV [10 min] | Malathion (0.30 × 10-9 to 1.51 × 10-6) | 0.06 × 10-9[0.9989] | [17] |
| Physical entrapment | AChE-AuNPs-SiSG | CV [10 min] | Monocrotophos (4.48 × 10-9 to 4.48 × 10-6 and 8.96 × 10-6 to 2.69 × 10-6) | 2.69 × 10-6 [0.9930 and 0.9985] | [18] |
| Covalent immobilization | AChE-CdTe-AuNPs-CM/GCE | CV[8 min] | Monocrotophos (5.0 × 10-9 to 4.48 × 10-6 and from 9.0 × 10-9 to 0.067 × 10-6) | 1.34 × 10-9 [0.9927 and 0.9945] | [19] |
| Physical adsorption | AChE–AuNPs–PPy/GCE | CV [12 min] | Methyl parathion (0.019 × 10-6 to 0.45 × 10-6 and 1.90 × 10-6 to 17.10 × 10-6) | 7.60 × 10-6 [0.9992 and 0.9989] | [20] |
| Affinity immobilization | ZrO2 NPs/SPE | Striping voltammetry | Phosphorylated AChE adducts (10 × 10-12 - 4 × 10-9) | 8.0 × 10-12 [0.9955] | [34] |
| Covalent immobilization using glutaraldehyde | AChE/CdS NPs | Photocurrent spectra | 1,5-bis(4- allyldimethylammoniumphenyl) pentane-3-one dibromide | - | [35] |
| Physical entrapment | AChE/ QCdS-PVP /GCE | Amperometry [5 min] | Trichlorfon (0.1 × 10-9 - 2 × 10-6) | 0.5 × 10-9 [0.9908] | [36] |
| - | - | Gas chromatography (GC) | Methyl parathion Malathion Triazophos | 0.04 × 10-6 0.03 × 10-6 0.08 × 10-6 | [37] |
| - | - | Automated on-line solid-phase extraction (OSP-2) and thermospray mass spectrometry (LC-TSP- MS) | Malathion (0.076 × 10-6 to 6.05 × 10-6 M) | 0.09 × 10-6 [0.9831] | [38] |
| - | - | GC | Malathion | 3.02 × 10-9 [0.997] | [39] |
| - | - | Liquid chromatography -atmospheric pressure chemical ionization-mass spectrometry (LC-APCI-MS) | Malathion Paraoxon Triazophos | 0.02 × 10-6 0.08 × 10-6 0. 02 × 10-6 | [40] |
| - | - | Automated solid phase extraction and micellar electrokinetic capillary chromatography (MEKC) | Malathion | 0.15 × 10-6 [0.9987] | [41] |
| - | - | Matrix Solid-Phase Dispersion (MSPD) and GC | Parathion methyl Malathion | 4.0 × 10-9 9.0 × 10-9 | [42] |
| - | - | High pressure Liquid chromatography (HPLC) | Paraoxon | 0.50 × 10-6 | [43] |
| - | Tyrosinase enzyme immobilized in kappa-carrageenan gel | Inhibition [15 min] | Paraoxon 0.01 × 10-6 to 0.1 × 10-6 Malathion 0.01 × 10-6 to 0.1 × 10-6 Parathion-ethyl 0.01 × 10-6 to 0.1 × 10-6 | 5.0 × 10-9 5.0 × 10-9 5.0 × 10-9 | [44] |
| - | Butyrylcholinesterase and choline oxidase immobilized in kappa-carrageenan gel | Inhibition [15 min] | Paraoxon 0.03 × 10-6 - 0.5 × 0-6 Malathion 0.03 × 10-6 to 0.025 ×10-6 Parathion-ethyl 0.02 × 10-6 to 0.025 ×10-6 | 15.0 × 10-9 15.0 × 10-9 2.0 ×10-9 | [44] |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Periasamy, A.P.; Umasankar, Y.; Chen, S.-M. Nanomaterials - Acetylcholinesterase Enzyme Matrices for Organophosphorus Pesticides Electrochemical Sensors: A Review. Sensors 2009, 9, 4034-4055. https://doi.org/10.3390/s90604034
Periasamy AP, Umasankar Y, Chen S-M. Nanomaterials - Acetylcholinesterase Enzyme Matrices for Organophosphorus Pesticides Electrochemical Sensors: A Review. Sensors. 2009; 9(6):4034-4055. https://doi.org/10.3390/s90604034
Chicago/Turabian StylePeriasamy, Arun Prakash, Yogeswaran Umasankar, and Shen-Ming Chen. 2009. "Nanomaterials - Acetylcholinesterase Enzyme Matrices for Organophosphorus Pesticides Electrochemical Sensors: A Review" Sensors 9, no. 6: 4034-4055. https://doi.org/10.3390/s90604034