Determination of Lactoferrin and Immunoglobulin G in Animal Milks by New Immunosensors
Abstract
:1. Introduction
2. Experimental Section
2.1 Apparatus
2.2. Materials
2.3. Sample analyzed
3. Methods
3.1. Lactoferrin biotinylation and extravidin-peroxidase conjugation
3.2. Anti-lactoferrin immobilization on Immobilon membrane
3.3. Immunosensor assembly
3.4. Determination of lactoferrin by immunosensor
3.5. IgG immobilization on Immobilon membrane
3.6. Construction of immunosensor for IgG measurements
3.7. Determination of IgG by new immunosensor
3.8. Determination of lactoferrin and immunoglobulin G in different milk samples
3.9. Determination of antioxidant capacity by SOD biosensor
4. Results and Discussion
5. Conclusions
Acknowledgments
References
- Wong, D.W.S.; Camirand, W.M.; Pavlath, A.E. Structures and functionalities of milk proteins. Crit. Rev. Food Sci 1996, 36, 807–844. [Google Scholar]
- Reiter, B. The biological significance of the non-immunoglobulin protective proteins in milk. Dev. Dairy Chem 1985, 3, 281–336. [Google Scholar]
- Steijns, J.M. Milk ingredients as nutraceuticals. Int. J. Dairy Technol 2001, 54, 81–88. [Google Scholar]
- Levay, P.F.; Viljoen, M. Lactoferrin: a general review. Haematologica 1995, 80, 252–267. [Google Scholar]
- Perraudin, J. Protéines à activités biologique: lactoferrin et lactoperoxydase. Connaissances recemment acquises et technologies d'obtention. Lait 1991, 71, 191–212. [Google Scholar]
- Ensminger, A.H.; Esminger, M.K.J. Food for Health: A Nutrition Encyclopedia; Pegus Press: Clovis, California, USA, 1986. [Google Scholar]
- Wood, R. The Whole Foods Encyclopedia; Prentice-Hall Press: New York, USA, 1988. [Google Scholar]
- Shimazaki, K.I. Lactoferrin: Structure, Function and Applications. Exerpta Medica International Congress Series 1195,. 2000. [Google Scholar]
- Shimazaki, K. Lactoferrin: a marvelous protein in milk? Anim. Sci. J 2000, 71, 329–347. [Google Scholar]
- Spik, G.; Legrand, D.; Mazurier, J.; Pierce, A.; Perraudin, J.P. Advances in lactoferrin research; Plenum Press: New York, N.Y, USA, 1998; Volume 443. [Google Scholar]
- Steijns, J.M.; van Hooijdonk, A.C. Occurrence, structure, biochemical properties and technological characteristics of lactoferrin. Brit. J. Nutr 2000, 84, S11–S17. [Google Scholar]
- Valenti, P. Antiviral activity of lactoferrin; Spik, G., Ed.; Plenum Press: New York, USA, 1998; pp. 199–203. [Google Scholar]
- Vorland, L.H. Lactoferrin: a multifunctional glycoprotein. Acta Path. Micro. Im. C 1999, 107, 971–981. [Google Scholar]
- Lonnerdal, B.; Iyer, S. Lactoferrin: molecular structure and biological function. Ann. Rev. Nutr 1995, 15, 93–110. [Google Scholar]
- Meulenbroek, A.J.; Zeijlemaker, W.P. Human IgG Subclasses: useful diagnostic markers for immunocompetence; CLB, Plesmanlaan: Amsterdam, Holland, 1996; Volume 125, p. 1066. [Google Scholar]
- Li-Chan, E.; Kummer, A.; Losso, J.N.; Nakai, S. Survey of immunoglobulin G content and antibody specificity in cow’s milk from British Columbia. Food Agric. Immunol 1994, 6, 443–449. [Google Scholar]
- Losso, J.N.; Dhar, J.; Kummer, A.; Li-Chan, E.; Nakai, S. Detection of antibody specificity of raw bovine and human milk to bacterial lipopolysaccharides using PCFIA. Food Agric. Immunol 1993, 5, 231–238. [Google Scholar]
- Yolken, R.H.; Losonsky, G.A.; Vonderfecht, S.; Leister, F.; Wee, S. Antibody to human rotavirus in cow’s milk. New Engl. J. Med 1985, 312, 605–612. [Google Scholar]
- Levieux, D.; Morgan, F.; Geneix, N.; Masle, I.; Bouvier, F. Caprine immunoglobulin G, β-lactoglobulin, α-lactoalbumin and serum albumin in colostrum and milk during the early post partum period. J. Dairy Res 2002, 69, 391–399. [Google Scholar]
- Cheng, J.B.; Wang, J.Q.; Bu, D.P.; Liu, G.L.; Zhang, C.G.; Wei, H.Y.; Zhou, L.Y.; Wang, J.Z. Factors affecting the lactoferrin concentration in bovine milk. J. Dairy Sci 2008, 91, 970–976. [Google Scholar]
- Hurley, W.L.; Grieve, R.C.J.; Magura, C.E.; Hegarty, H.M.; Zou, S. Electrophoretic comparisons of lactoferrin from bovine mammary secretions, milk neutrophils, and human milk. J. Dairy Sci 1993, 76, 377–387. [Google Scholar]
- Indyk, H.E.; Filonzi, E.L. Determination of lactoferrin in bovine milk, colostrum and infant formulas by optical biosensor analysis. Int. Dairy J 2005, 15, 429–438. [Google Scholar]
- Elagamy, E.I.; Ruppanner, R.; Ismail, A.; Champagne, C.P.; Assaf, R. Purification and characterization of lactoferrin, lactoperoxidase, lysozyme and immunoglobulins from camel's milk. Int. Dairy J 1996, 6, 129–145. [Google Scholar]
- Campanella, L.; Martini, E.; Tomassetti, M. Two new immunosensors and a biosensor for buffalo milk analysis. Proceeding of the 12th Int. Meeting on Chemical Sensors, Napoli, Italy, 12–14 February 2007; Word Scientific Publishing Co: Singapore, 2007. in press.. [Google Scholar]
- Campanella, L.; Lelo, D.; Martini, E.; Tomassetti, M. Immunoglobulin G determination in human serum and milk using an immunosensor of new conception fitted with an enzyme probe as transducer. Sensors 2008, 8, 6727–6746. [Google Scholar]
- Campanella, L.; Favero, G.; Tomassetti, M. Superoxide Dismutase biosensors for superoxide radical analysis. Anal. Lett 1999, 32, 2559–2581. [Google Scholar]
- Campanella, L.; Favero, G.; Persi, L.; Tomassetti, M. New biosensor of Superoxide radical used to evidence molecules of biomedical and pharmaceutical interest having radical scavenging properties. J. Pharmaceut. Biomed 2000, 23, 68–76. [Google Scholar]
- Campanella, L.; Favero, G.; Tomassetti, M. A modified amperometric electrode for the determination of free radicals. Sens. Act. B-Chem 1997, 44, 559–565. [Google Scholar]
- Duk, M.; Lisowska, E.; Wu, J.H.; Wu, A.M. The biotin/avidin-mediated microtiter plate lectin assay with the use of chemically modified glycoprotein ligand. Anal. Biochem 1994, 221, 266–272. [Google Scholar]
- Rao, S.V.; Anderson, K.W.; Bachas, L.G. Controlled layer-by-layer immobilization of horseradish peroxidase. Biotechnol. Bioeng 1999, 65, 389–396. [Google Scholar]
- Green, N.M. A spectrophotometric assay for avidin and biotin based on binding of dyes by avidin. Biochem. J 1965, 94, 23c–24c. [Google Scholar]
- Bartoli, M.; Monneron, A.; Ladantet, D. Interaction of calmodulin with striatin, a WD-repeat protein present in neuronal dendritic spines. J. Biol. Chem 1999, 273, 22248–22253. [Google Scholar]
- Hurley, I.P.; Coleman, R.C.; Ireland, H.E.; Williams, J.H.H. Measurement of bovine IgG by indirect competitive ELISA as a means of detecting milk adulteration. J. Dairy Sci 2004, 87, 543–549. [Google Scholar]
- Indyk, H.E.; Filonzi, E.L. Determination of immunoglobulin G in bovine colostrum and milk by direct biosensor SPR-immunoassay. J. AOAC Int 2003, 86, 386–393. [Google Scholar]
- Lebreton, J.P.; Joisel, F.; Boutleux, S.; Lannuzel, B.; Sauger, F. Dosage immunonéphélométrique des immunoglobulines G dans le lait de vache. Lait 1981, 61, 465–480. [Google Scholar]
- Montagne, C.; Gavriloff, G.; Humbert, M.L.; Cuillière, J.; Duheille, G. Linden Microparticle-enhanced nephelometric immunoassay for immunoglobulins G in cow's milk. Lait 1991, 71, 493–499. [Google Scholar]
- Copestake, D.E.J.; Indyk, H.E.; Otter, D.E. Affinity liquid chromatography method for the quantification of immunoglobulin G in bovine colostrum powders. J. AOAC Int 2006, 89, 1249–1256. [Google Scholar]
- Levieux, D. Dosage des IgG du lait de vache par immunodiffusion radiale seml-autornatlsée, pour la détection du colostrum, des laits de mammites ou de fin de gestation. 1.Mise au point du dosage. Lait 1991, 71, 327–328. [Google Scholar]
- Chen, P.W.; Mao, F.C. Detection of lactoferrin in bovine and goat milk by enzyme-linked immunosorbent assay. J. Food Drug Anal 2004, 12, 133–139. [Google Scholar]
- Bezwoda, W.R.; Baynes, R.D.; Khan, Q.; Mansoor, N. Enzyme linked immunosorbent assay for lactoferrin. Plasma and tissue measurements. Clin. Chim. Acta 1985, 151, 61–69. [Google Scholar]
- Palmano, K.P.; Elgar, D.F. Detection and quantitation of lactoferrin in bovine whey samples by reversed-phase high-performance liquid chromatography on polystyrene–divinylbenzene. J. Chromatogr. A 2002, 947, 307–311. [Google Scholar]
- Vojtech, A.; Zitka, O.; Dolezal, P.; Zeman, L.; Horna, A.; Hubalek, J.; Sileny, J.; Krizkova, S.; Trnkova, L.; Kizek, R. Lactoferrin isolation using monolithic column coupled with spectrometric or micro-amperometric detector. Sensors 2008, 8, 464–487. [Google Scholar]
- Tu, Y.Y.; Chen, C.C.; Chang, J.H.; Chang, H.M. Characterization of lactoferrin (lf) from colostral whey using anti-lf antibody immunoaffinity chromatography. J. Food Sci 2002, 67, 996–1001. [Google Scholar]
- Ribadeau-Dumas, B.; Grappin, R. Milk protein analysis. Lait 1989, 69, 357–416. [Google Scholar]
- Fleenor, W.A.; Stott, G.H. Single radial immunodiffusion analysis for quantitation of colostral immunoglobulin concentration. J. Dairy Sci 1981, 64, 740–747. [Google Scholar]
- Rong, R.; Shi, L.; Xua, Y.; Wanga, Z.; Yanga, R.J. Isolation of lactoferrin from bovine colostrum by ultrafiltration coupled with strong cation exchange chromatography on a production scale. J. Membr. Sci 2007, 297, 152–161. [Google Scholar]
- Chen, P.W.; Mao, F.C. Detection of lactoferrin in bovine and goat milk by enzyme-linked immunosorbent assay. J. Food Drug Anal 2004, 12, 133–139. [Google Scholar]
- Faye, G.; Loiseau, B.; Levieux, G. Lactoferrin and immunoglobulin contents in camel's milk (Camelus bactrianus, Camelus dromedarius, and hybrids) from Kazakhstan Konuspayeva. J. Dairy Sci 2007, 90, 38–46. [Google Scholar]
- Indyk, H.E.; McGrail, I.J.; Watene, G.A.; Filonzi, E.L. Optical biosensor analysis of the heat denaturation of bovine lactoferrin. Food Chem 2007, 101, 838–844. [Google Scholar]
- Johansson, B. Isolation of an iron-containing red protein from human milk. Acta Chem. Scan 1960, 14, 510–512. [Google Scholar]
- Lawrence, P.B. Breast milk. Best source of nutrition for term and preterm infants. Pediatr. Clin. N. Am 1994, 41, 925–941. [Google Scholar]
- Gauthier, S.F.; Pouliot, Y.Y.; Maubois, J.L. Growth factors from bovine milk and colostrum: composition, extraction and biological activities. Lait 2006, 86, 99–125. [Google Scholar]
- Van de Perre, P. Transfer of antibody via mother’s milk. Vaccine 2003, 21, 3374–3376. [Google Scholar]
- Rajala-Schultz, P.J.; Grohn, Y.T.; McCulloch, C.E.; Guard, C.L. Effects of clinical mastitis on milk yield in dairy cows. J.Dairy Sci. 1999, 82, 1213–1220. [Google Scholar]
- Carlsson, A.; Bjorck, L.; Persson, K. Lactoferrin and lysozyme in milk during acute mastitis and their inhibitory effect in Delvotest P. J. Dairy Sci 1989, 72, 3166–3175. [Google Scholar]
- Hagiwara, S.; Kawai, K.; Anri, A.; Nagahata, H. Lactoferrin concentrations in milk from normal and subclinical mastitic cows. J. Vet. Med. Sci 2003, 65, 319–323. [Google Scholar]
- Harmon, R.J.; Schanbacher, F.L.; Ferguson, L.C.; Smith, K.L. Changes in lactoferrin, immunoglobulin G, bovine serum albumin, and alpha-lactalbumin during acute experimental and natural coliform mastitis in cows. Inf. Immun 1976, 13, 533–542. [Google Scholar]
- Muller, L.D.; Ellinger, D.K. Colostral immunoglobulin concentrations among breeds of dairy cattle. J. Dairy Sci 1981, 64, 1727–1730. [Google Scholar]
- Konuspayeva, G.; Faye, B.; Loiseau, G.; Levieux, D. Lactoferrin and immunoglobulin contents in camel’s milk (Camelus bactrianus, Camelus dromedarius, and Hybrids) from Kazakhstan. J. Dairy Sci 2007, 90, 38–46. [Google Scholar]
- Conesa, C.; Lavilla, M.; Sánchez, L.; Pérez, M.D.; Mata, L.; Razquín, P.; Calvo, M. Determination of IgG levels in bovine bulk milk samples from different regions of Spain. Eur. Food Res. Technol 2005, 220, 222–225. [Google Scholar]
- Chen, P.W.; Chen, W.C.; Mao, C.H.F. Increase of lactoferrin concentration in mastitic goat milk. J. Vet. Med. Sci 2004, 66, 345–350. [Google Scholar]
- Britigan, B.E.; Serody, J.S.; Cohen, M.S. The role of lactoferrin as an anti-inflammatory molecule. Adv. Exp. Med. Biol 1994, 357, 143–156. [Google Scholar]
- Harmsen, M.C. Antiviral effects of plasma and milk proteins: lactoferrin shows a potent activity against both human immunodeficiency virus and human cytomegalovirus replication in vitro. J. Infect. Dis 1995, 172, 380–388. [Google Scholar]
- Vorland, L.H.; Ulvatne, H.; Andersen, J. Antibacterial effects of lactoferricin B. Scan. J. Infect. Dis 1999, 31, 179–184. [Google Scholar]
- Swart, P.J.; Kuipers, E.M.; Smit, C. Lactoferrin. Antiviral activity of lactoferrin. Adv. Exp. Med. Biol 1998, 4434, 205–213. [Google Scholar]
- Oria, R.; Ismail, M.; Sanchez, L.; Calvo, M.; Brock, J.H. Effect of heat treatment and other milk proteins on the interaction of lactoferrin with monocytes. J. Dairy Res 1993, 60, 363–369. [Google Scholar]
- Adamik, B.; Zimecki, M.; Wlaszczyk, A. Lactoferrin effects on the in vitro immune response in critically ill patients. Arch. Immunol. Ther. Exp. (Warcz) 1998, 46, 169–176. [Google Scholar]
- Schanbacher, F.L.; Talhouk, R.S.; Murray, F.A. Biology and origin of bioactive peptides in milk. Livest. Prod. Sci 1997, 50, 105–123. [Google Scholar]
- Schulz-Lell, G.; Dorner, K.; Oldigs, H.D.; Sievers, E.; Schaub, J. Iron availability from an infant formula supplemented with bovine lactoferrin. Acta Paed. Scan 1991, 80, 155–158. [Google Scholar]
- Arthington, J.D.; Cattell, M.B.; Quigley, J.D. Effect of dietary IgG source (colostrum, serum, or milk-derived supplement) on the efficiency of Ig absorption in newborn holstein calves. J. Dairy Sci 2000, 83, 1463–1467. [Google Scholar]
- Chena, J.; Lindmark-Månssona, H; Gortonc, L.; Åkesson, B. Antioxidant capacity of bovine milk as assayed by spectrophotometric and amperometric methods. Int. Dairy J 2003, 13, 927–935. [Google Scholar]
- Alberti-Fidanza, A.; Burini, G.; Perriello, G. Total antioxidant capacity of colostrum, and transitional and mature human milk. J. Mat. Fet. Neo. Med 2002, 11, 275–279. [Google Scholar]
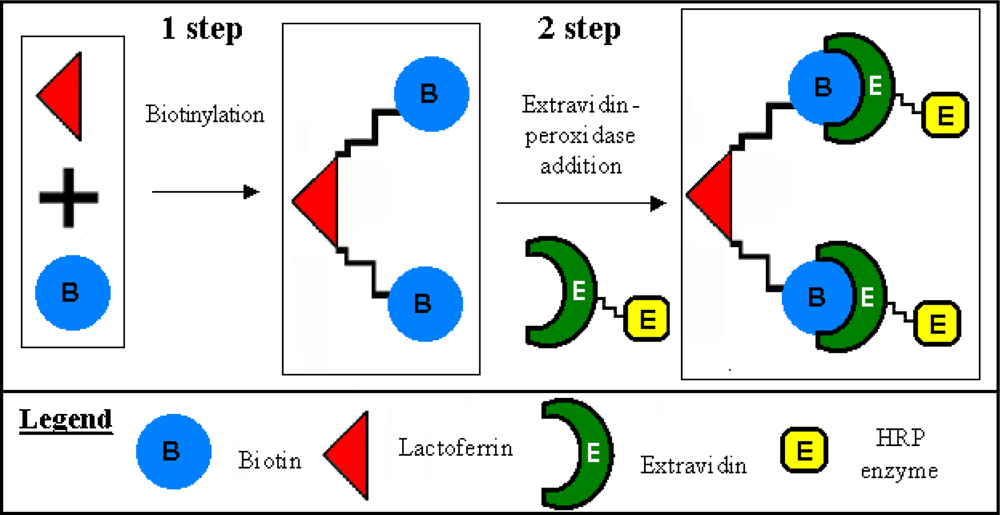
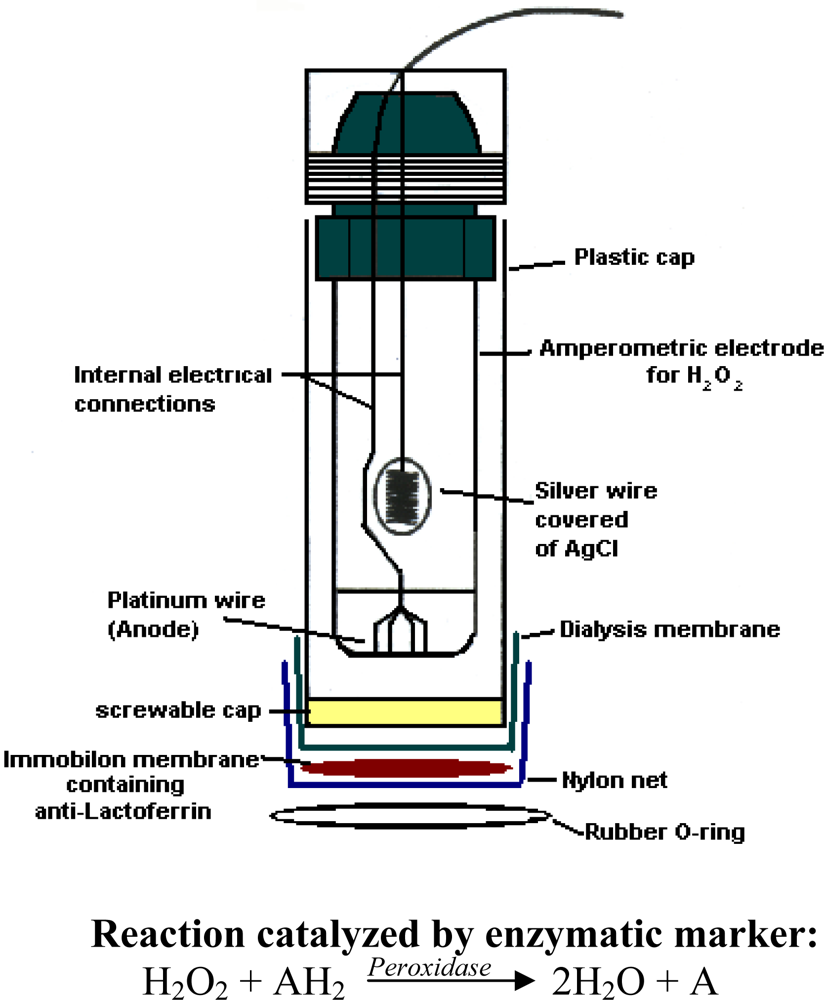
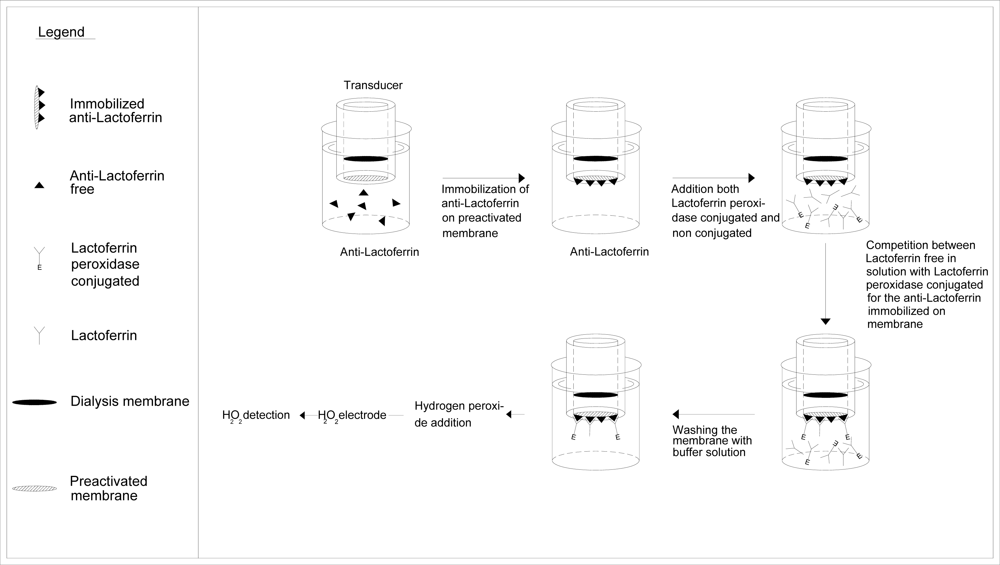

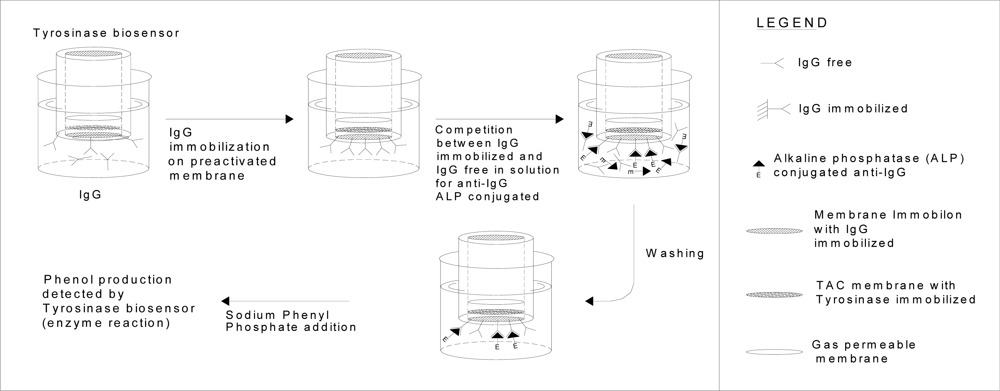
| Methods | Determination of lactoferrin by means of immunosensor. Test geometry: competition between lactoferrin biotin-avidin-peroxidase conjugated and lactoferrin free in solution for anti-lactoferrin immobilized in membraneA | Determination IgG by means of new immunosensor that uses as transducer a tyrosinase enzyme electrode. Test geometry: competition for the free in solution anti-IgG conjugated with the alkaline phosphatase, between the IgG immobilized on the membrane and IgG free in solutionB |
|---|---|---|
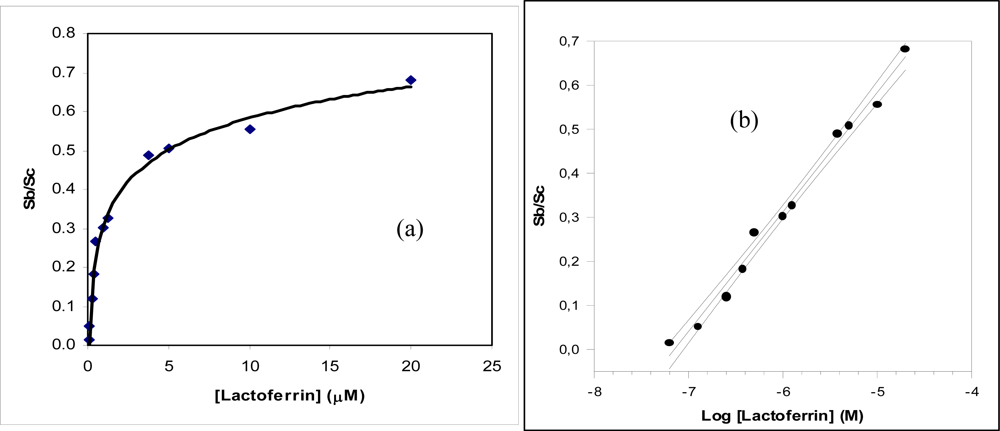
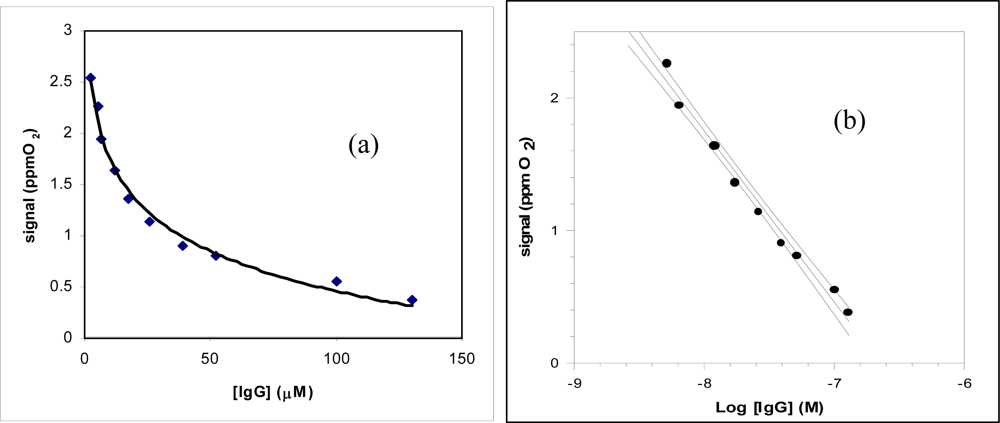
| Regression equation (Y = a.u., X = μM) confidence level (1- α) = 0.95; | Y = 0.27 (±0.04) log X + 0.31 (±0.02) (n – ν) = 9 ; (t = 2.26) | Y = −1.01 (±0.26) log X + 1.7 (±0.04) (n – ν) = 7 ; (t = 2.36) |
| Linear range (μM) | (0.7 – 100) × 10−1 | (2.6 – 130) × 10−3 |
| Correlation coefficient | 0.9891 | 0.9885 |
| Pooled SD% | ≤5.8 | ≤5.7 |
| Low detection limit (LOD) (μM) | 3.5 × 10−2 | 1.3 × 10−3 |
| Recovery of standard solution (% recovery values found in the linear range) | (99.2–100.3)% | (99.4–100.8)% |
| Repeatability of the measurement as relative standard deviation (RSD%) | ≤5.5 | ≤5.4 |
| Instrumental response time (min) | 5 | 10 |
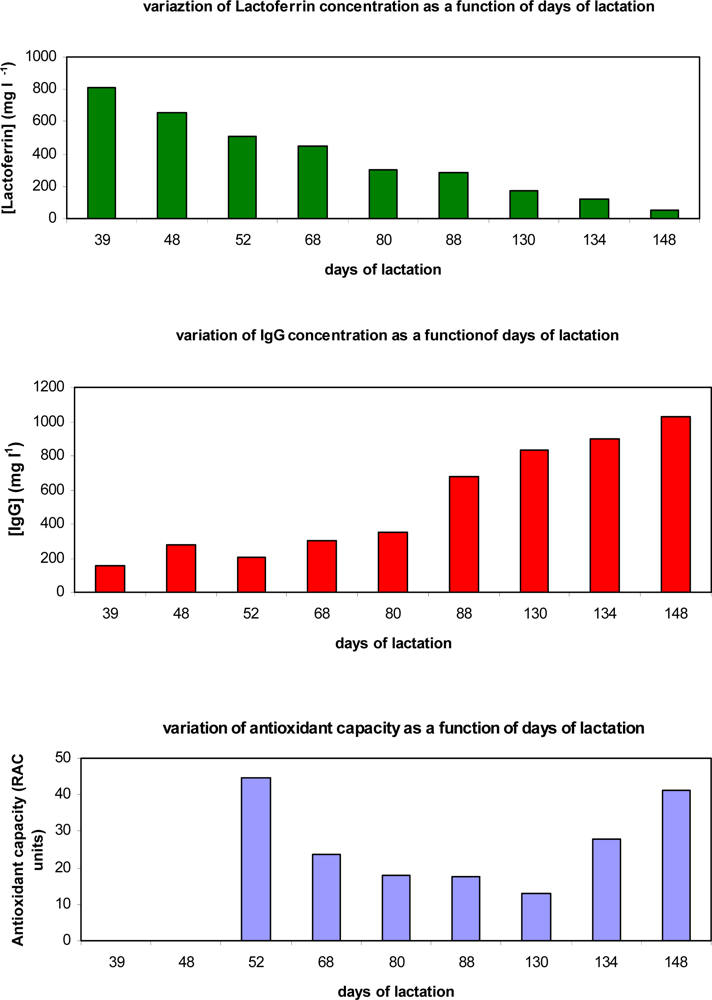
| Milk or dairy matrix | Found lactoferrin concentration (mg/L) n = 5; RSD% ≤ 5.5 | Found lactoferrin concentration (μM) n = 5; RSD% ≤ 5.5 | Found IgG concentration (mg/L) n = 5; RSD% ≤ 5.4 | Found IgG concentration (μM) n = 5; RSD% ≤ 5.4 |
|---|---|---|---|---|
| Raw Cow Milk | 182.4 | 2.28 | 772.5 | 5.15 |
| Cow Milk (UHT conservation) | 18.3 | 0.23 | 620.0 | 4.13 |
| Goat Milk (Partially skimmed) | 17.5 | 0.22 | 220.0 | 1.46 |
| Raw “Buffalo” milk | 232.0 | 2.90 | 675.0 | 4.50 |
| “Fruit of wood” Yogurth | 8.0 | 0.10 | 39.1 | 0.26 |
| “Pineapple” Yogurth | 7.8 | 0.09 | 42.5 | 0.28 |
| Milk Matrix | Found lactoferrin concentration (μM) (n=5); RSD% ≤ 5.5 | Added lactoferrin concentration (μM) | Experimental lactoferrin concentration (μM) (n=5); RSD% ≤ 5.5 | Recovery % lactoferrin concentration in milk matrix |
|---|---|---|---|---|
| Buffalo milk (Diluted 1:100) | 2.58 × 10−2 | 1.0 × 10−2 | 3.94 × 10−2 | 100.6 |
| Buffalo milk (Diluted 1:50) | 5.96 × 10−2 | 2.0 × 10−2 | 7.83 × 10−2 | 98.4 |
| Cow milk (UHT conservation) (Diluted 1:10) | 2.3 × 10−2 | 1.0 × 10−2 | 3.7 × 10−2 | 112.1 |
| Cow milk (UHT conservation) (Diluted 1:5) | 4.5 × 10−2 | 5.0 × 10−2 | 9.2 × 10−2 | 96.8 |
| Milk Matrix | Found IgG concentration (μM) (n = 5); RSD% ≤ 5.4 | Added Ig G concentration (μM) | Experimental IgG concentration (μM) (n = 5); RSD% ≤ 5.4 | Recovery % IgG concentration in milk matrix |
|---|---|---|---|---|
| Buffalo milk (Diluted 1:1,000) | 6.15 × 10−3 | 1.0 × 10−3 | 7.27 × 10−3 | 101.7 |
| Buffalo milk (Diluted 1:500) | 1.38 × 10−3 | 2.0 × 10−3 | 3.32 × 10−3 | 98.2 |
| Cow milk (UHT conservation) (Diluted 1:2,000) | 2.2 × 10−3 | 1.0 × 10−3 | 2.9 × 10−3 | 90.6 |
| Cow milk (UHT conservation) (Diluted 1:3,000) | 1.6 × 10−3 | 0.5 × 10−3 | 2.4 × 10−3 | 114.3 |
| IMMUNOGLOBULIN G | LACTOFERRIN | ||||||
|---|---|---|---|---|---|---|---|
| References | Method | Linear range (μg/mL) | LOD (μg/mL) | References | Method | Linear range (μg/mL) | LOD (μg/mL) |
| [this work] | immunosensor | 0.4 – 20 | 0.2 | [this work] | immunosensor | 5.6 – 800 | 2.8 |
| [33] | ELISA | 1–100 | 0.3 | [39] | ELISA | 0.05 – 10 | 0.01 |
| [34] | SPR–Immunoa ssay | 0.015–10 | 80 | [40] | ELISA | 3.12 – 200 | 1.0 |
| [35] | Immunonephelometry | 50–200 | 30 | [22] | Optical biosensor | 0.004 – 1.0 | 0.002 |
| [36] | Immunonephelometry | 0.05–0.8 | 0.008 | [41] | RP-HPLC method | 0.2 – 30 | 0.2 |
| [37] | Affinity LC | 10–150 | 0.5 | [42] | Spectrometric method | 10–100 | 1 |
| [38] | RID | 300–1400 | 120 | [43] | Immuno-affinity Chromatography | 20–200 | 12 |
© 2009 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Campanella, L.; Martini, E.; Pintore, M.; Tomassetti, M. Determination of Lactoferrin and Immunoglobulin G in Animal Milks by New Immunosensors. Sensors 2009, 9, 2202-2221. https://doi.org/10.3390/s90302202
Campanella L, Martini E, Pintore M, Tomassetti M. Determination of Lactoferrin and Immunoglobulin G in Animal Milks by New Immunosensors. Sensors. 2009; 9(3):2202-2221. https://doi.org/10.3390/s90302202
Chicago/Turabian StyleCampanella, Luigi, Elisabetta Martini, Manuela Pintore, and Mauro Tomassetti. 2009. "Determination of Lactoferrin and Immunoglobulin G in Animal Milks by New Immunosensors" Sensors 9, no. 3: 2202-2221. https://doi.org/10.3390/s90302202





