Cell Microarray Technologies for High-Throughput Cell-Based Biosensors
Abstract
:1. Introduction
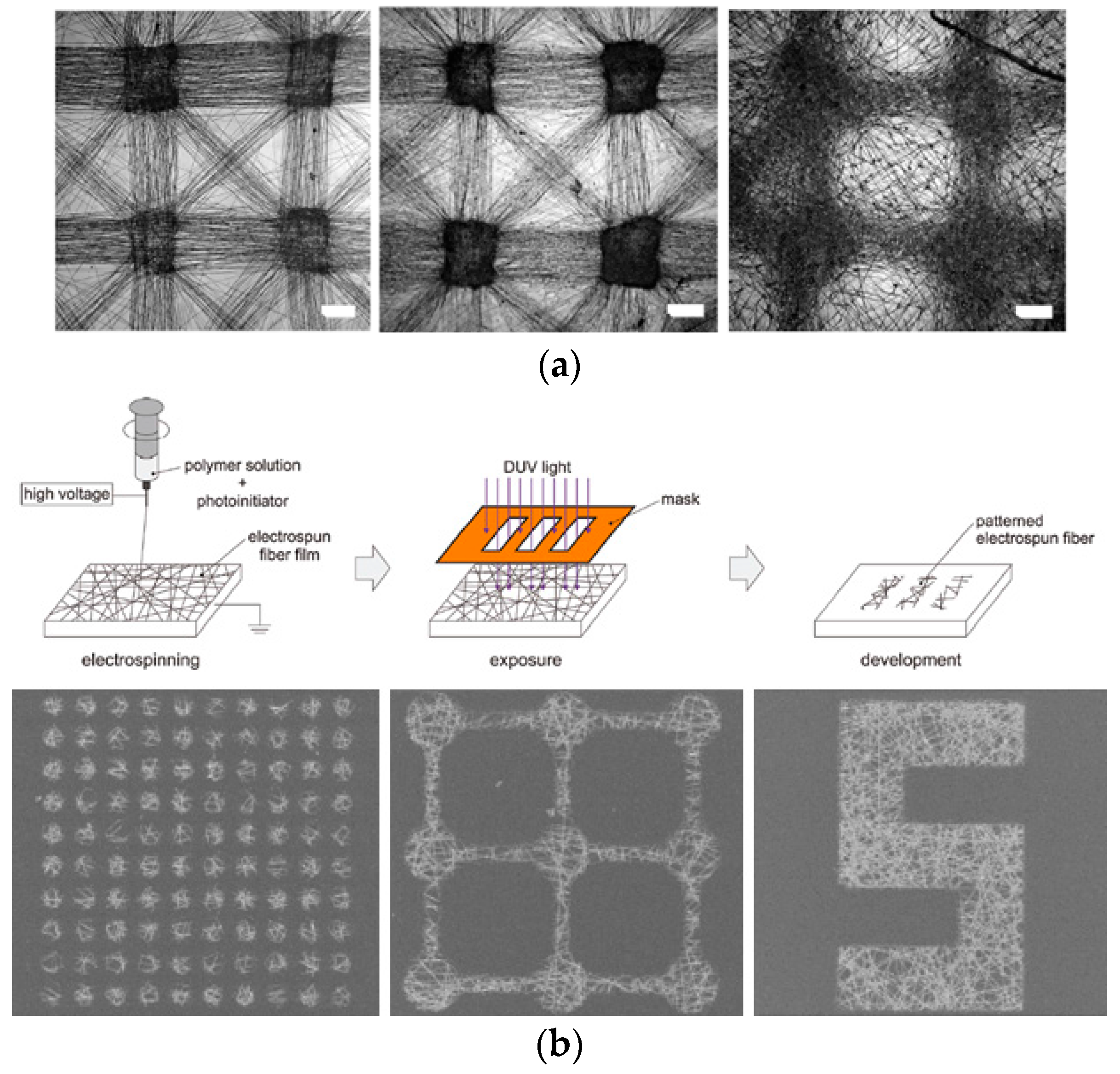
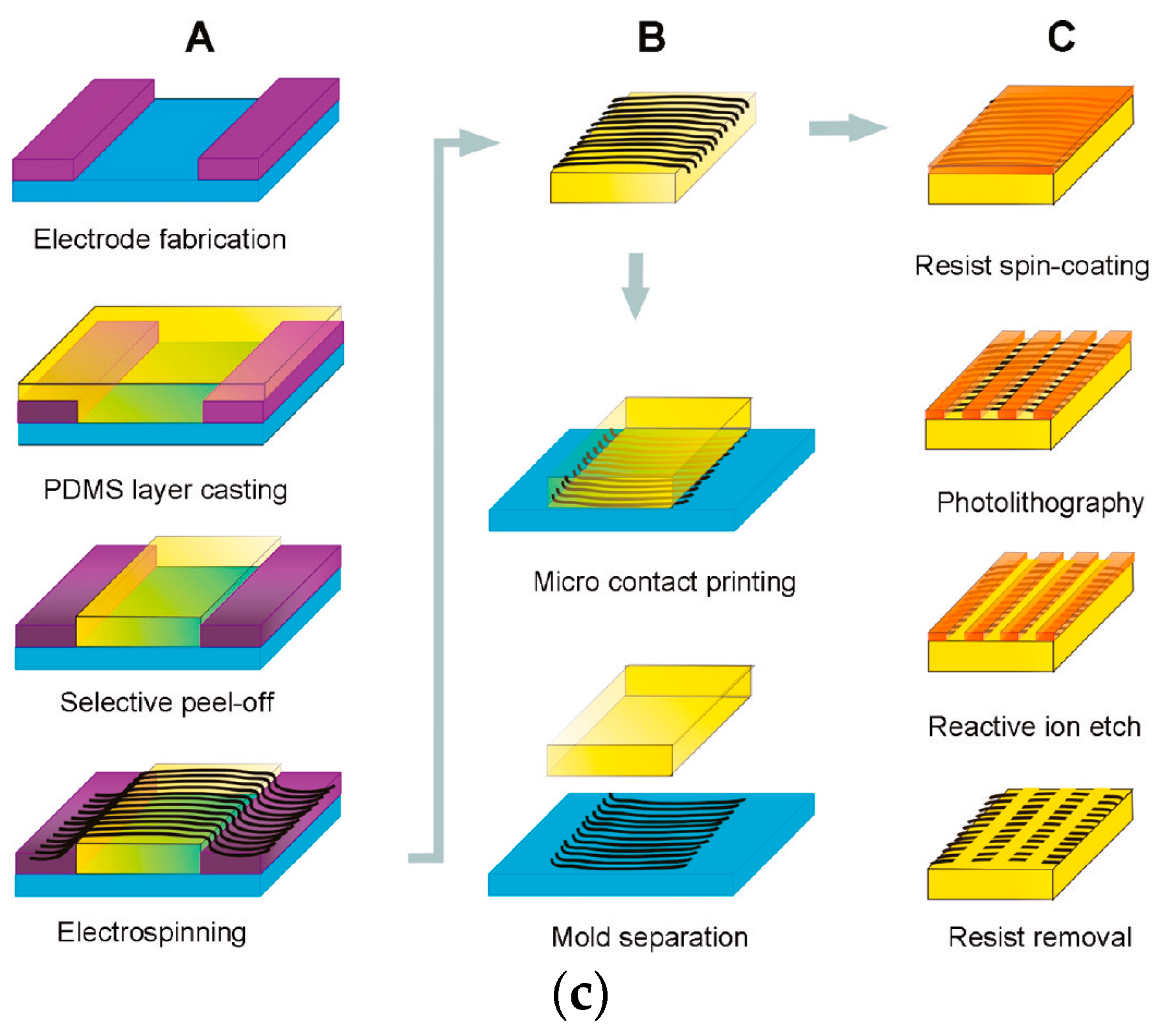
2. Preparation of Cell Microarray
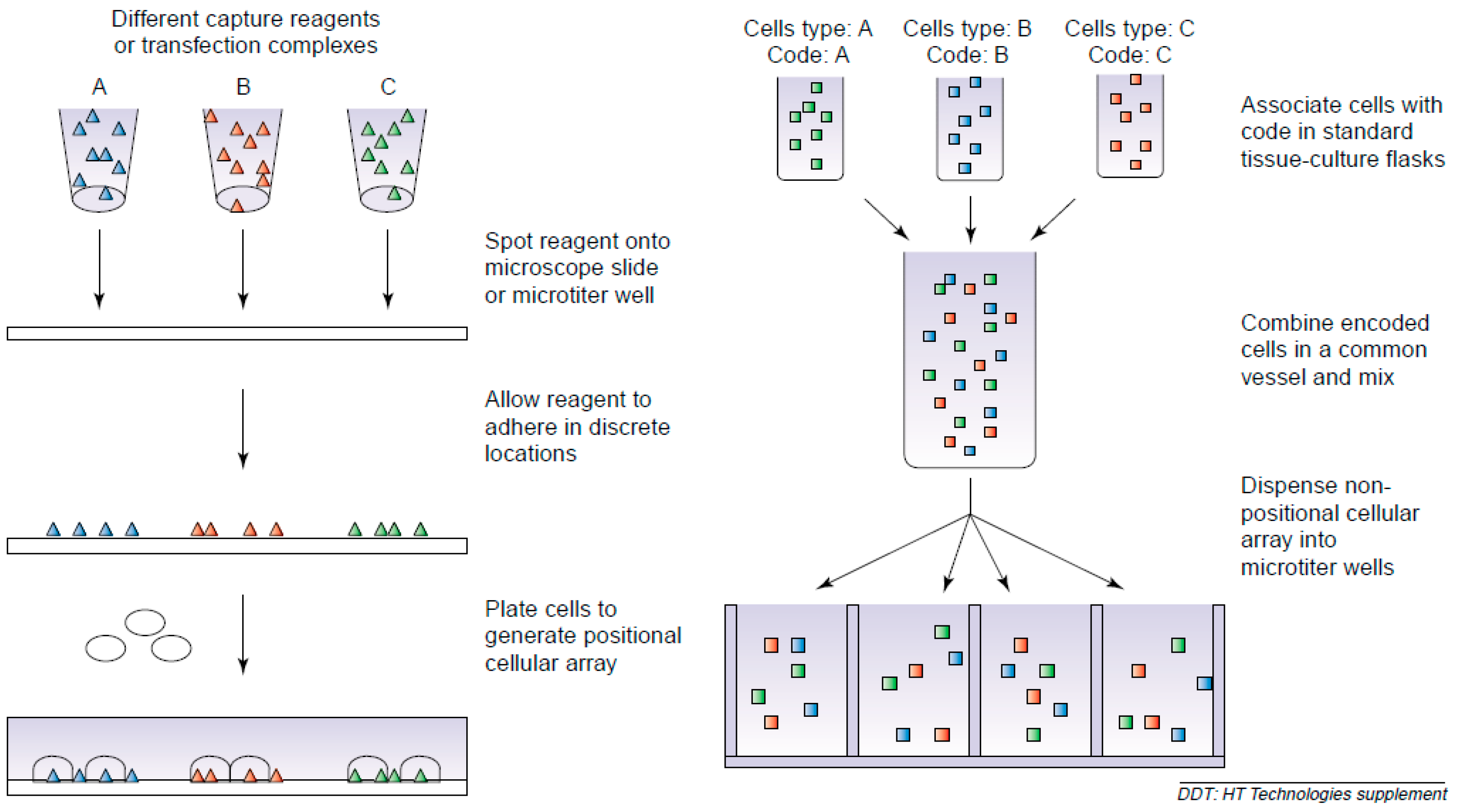
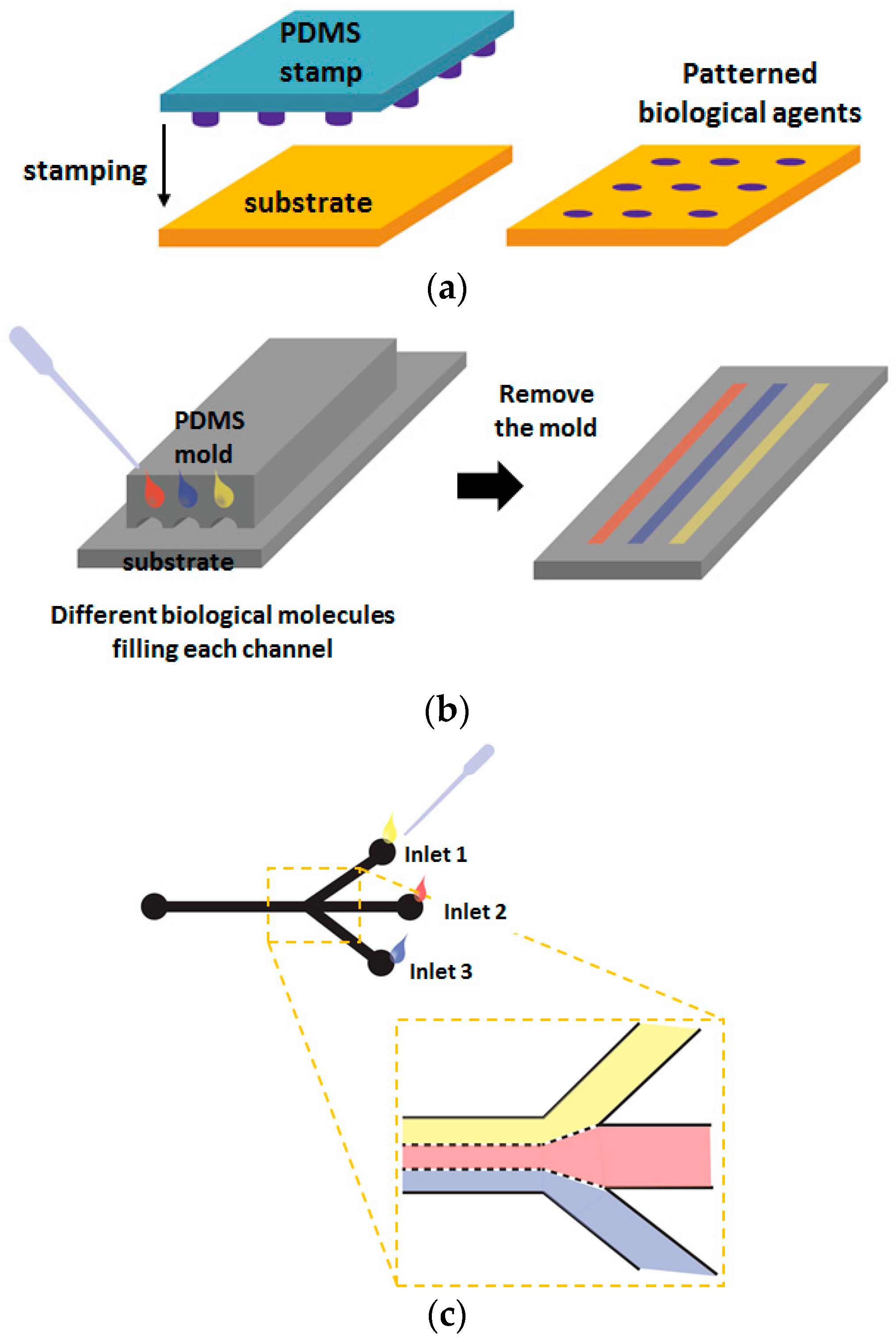
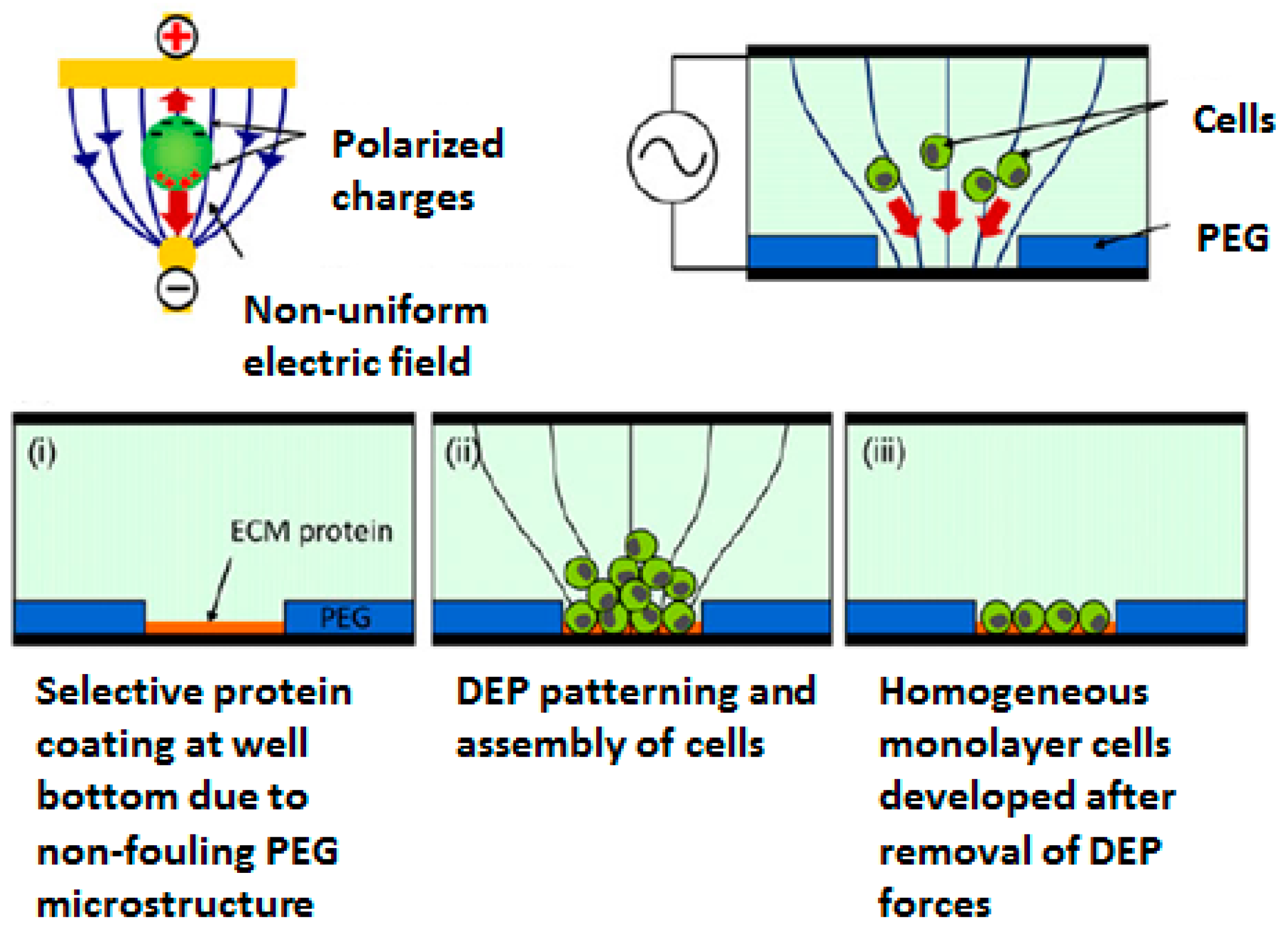
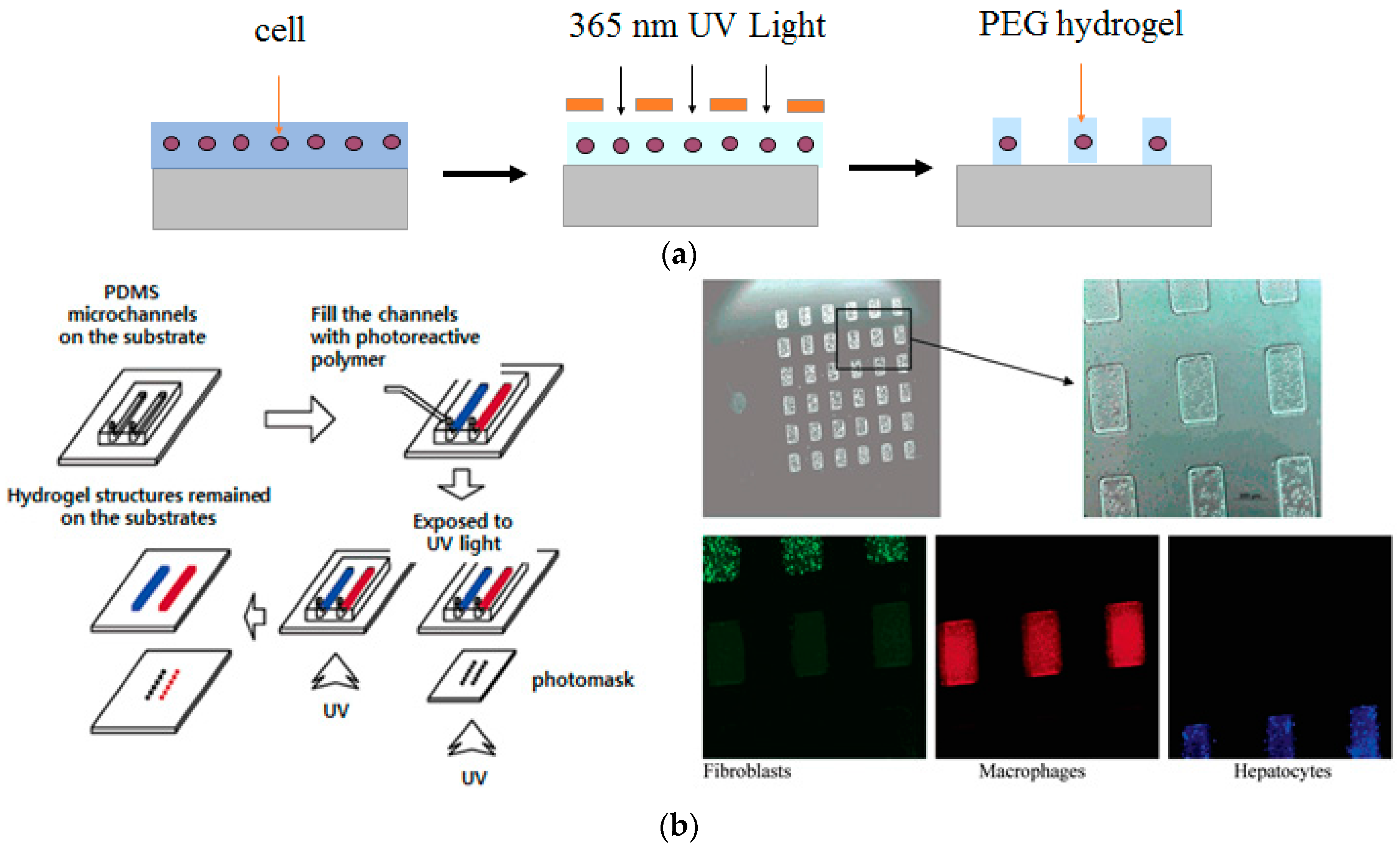
2.1. Positional Arrays
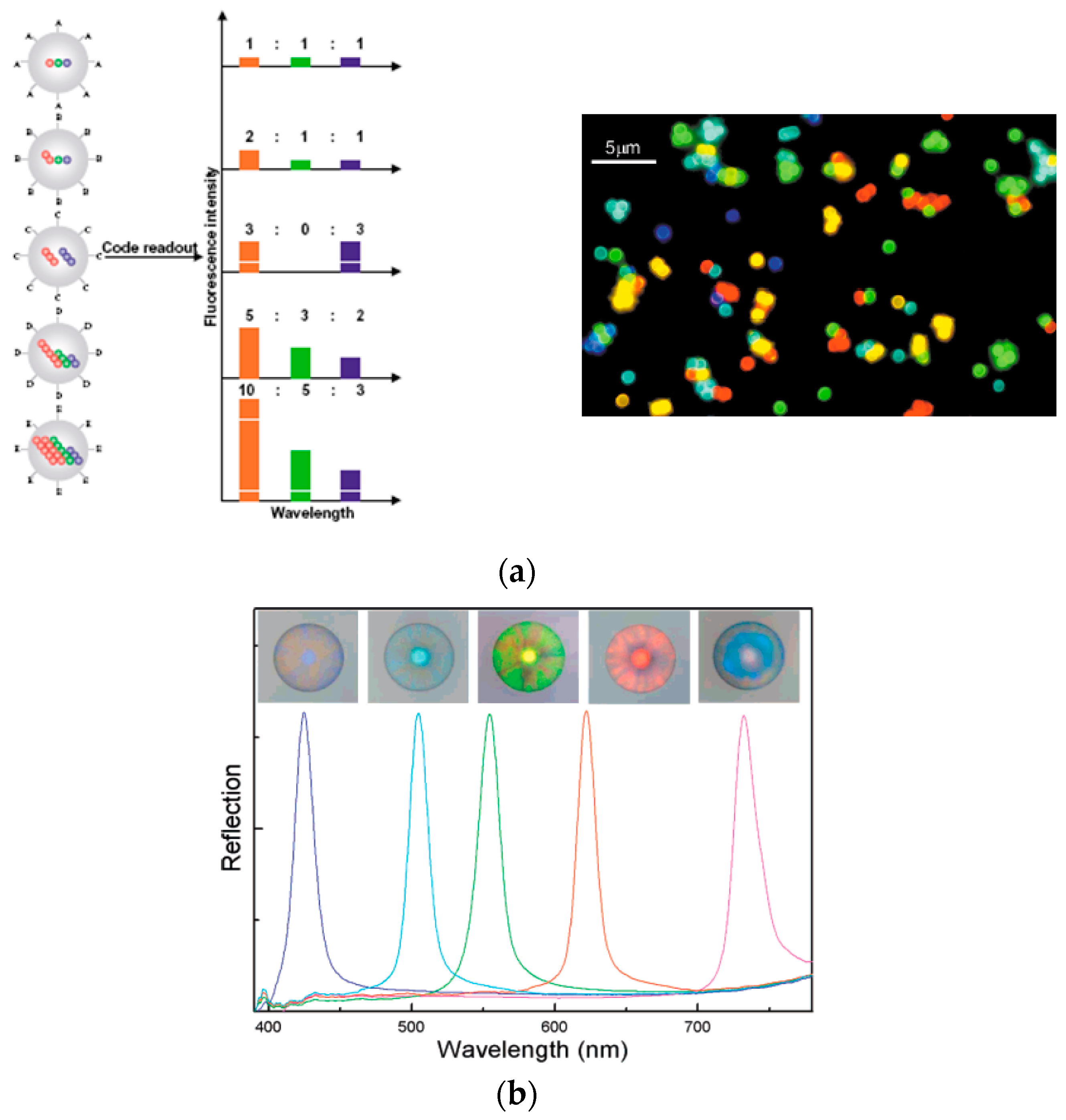
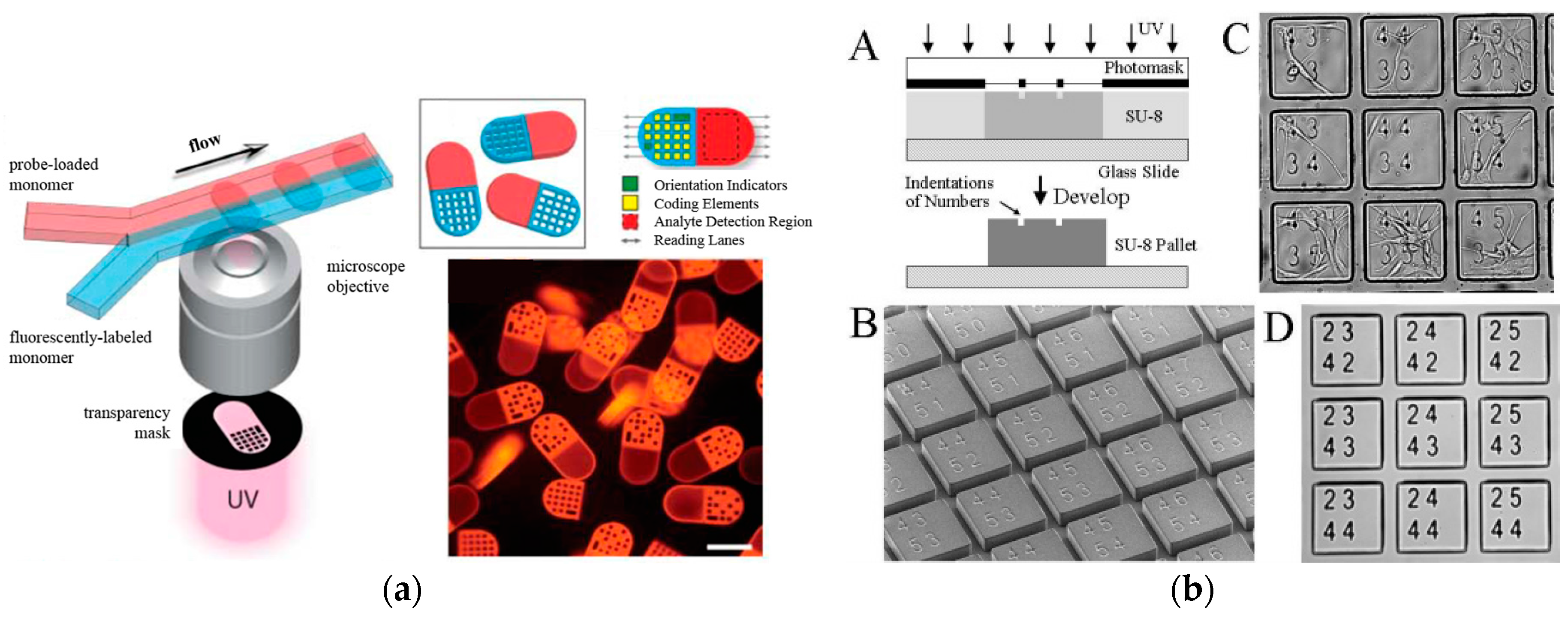
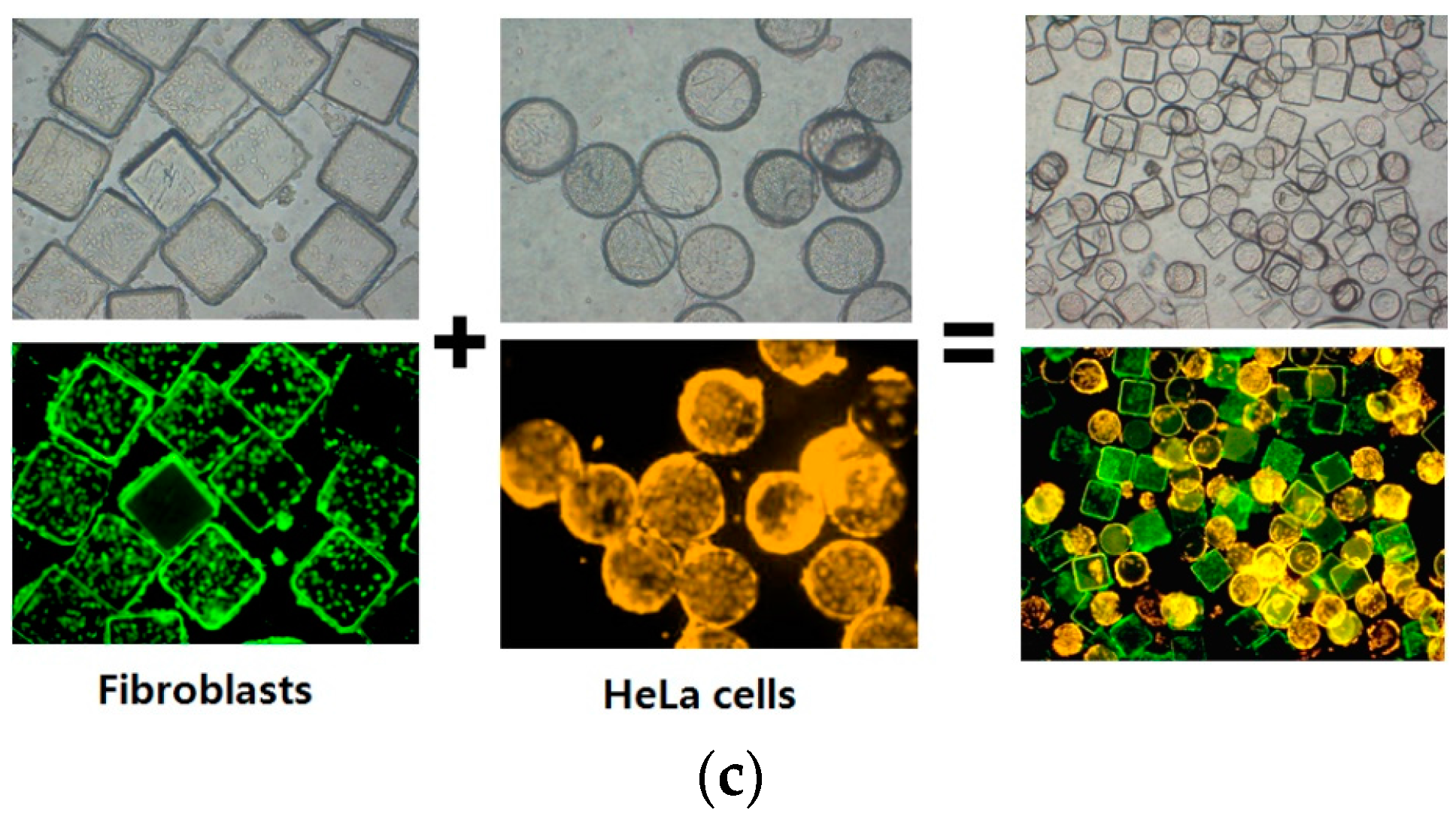
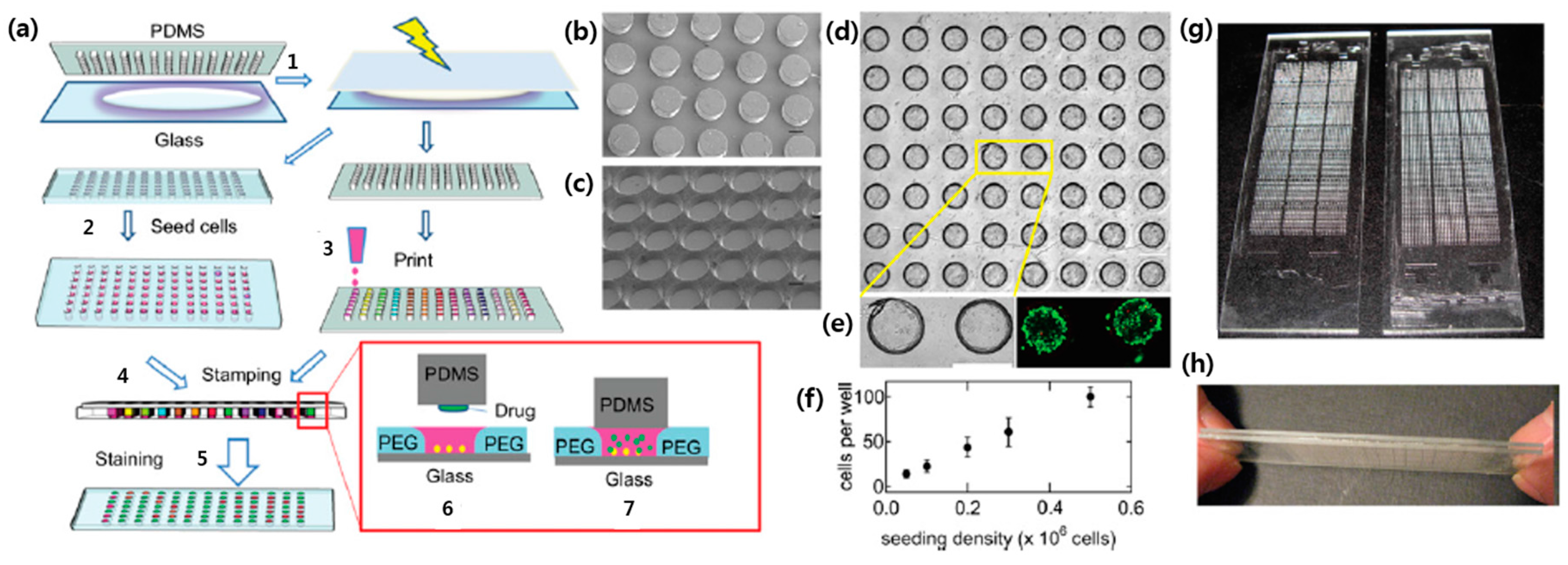
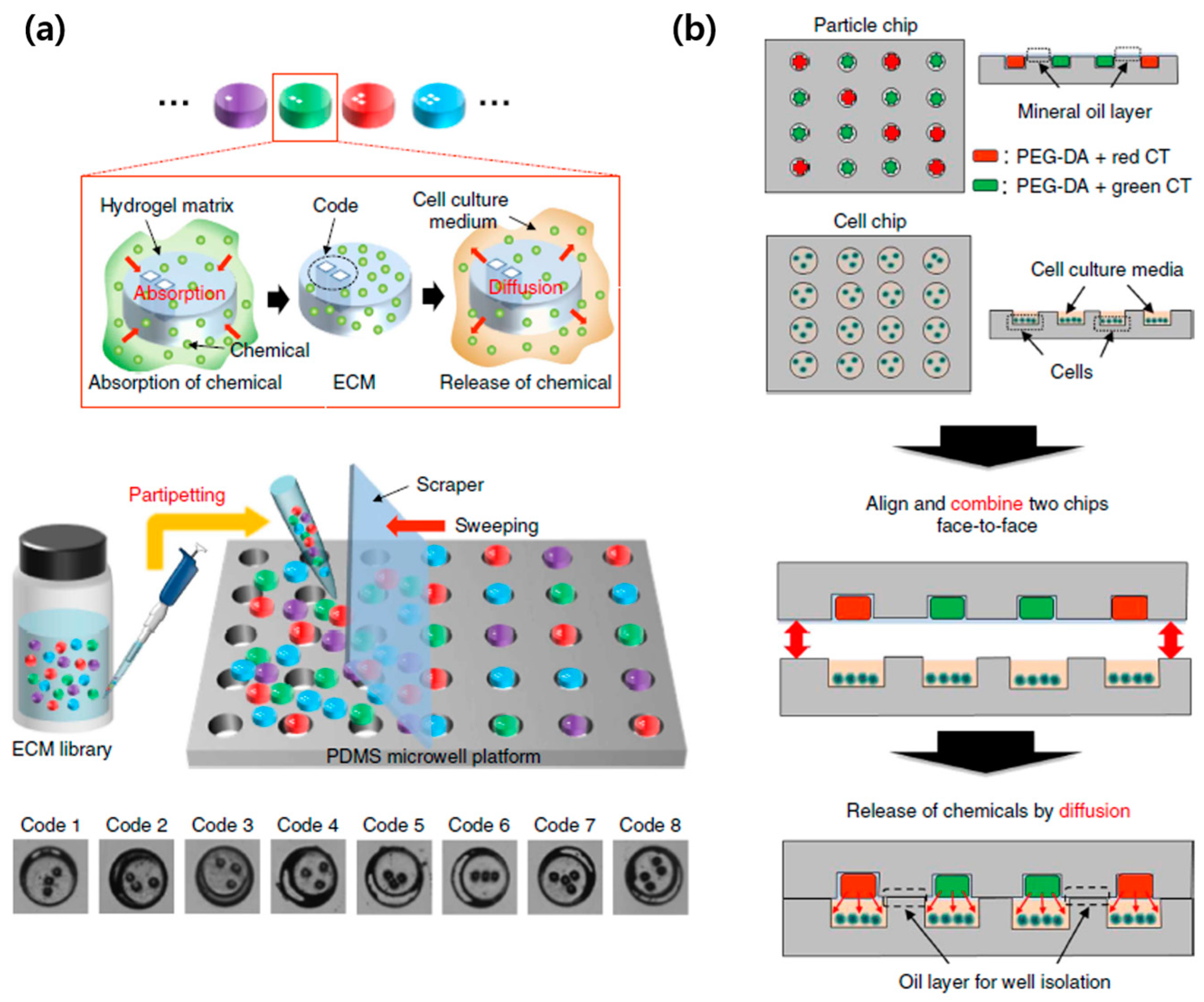
2.2. Suspension Arrays
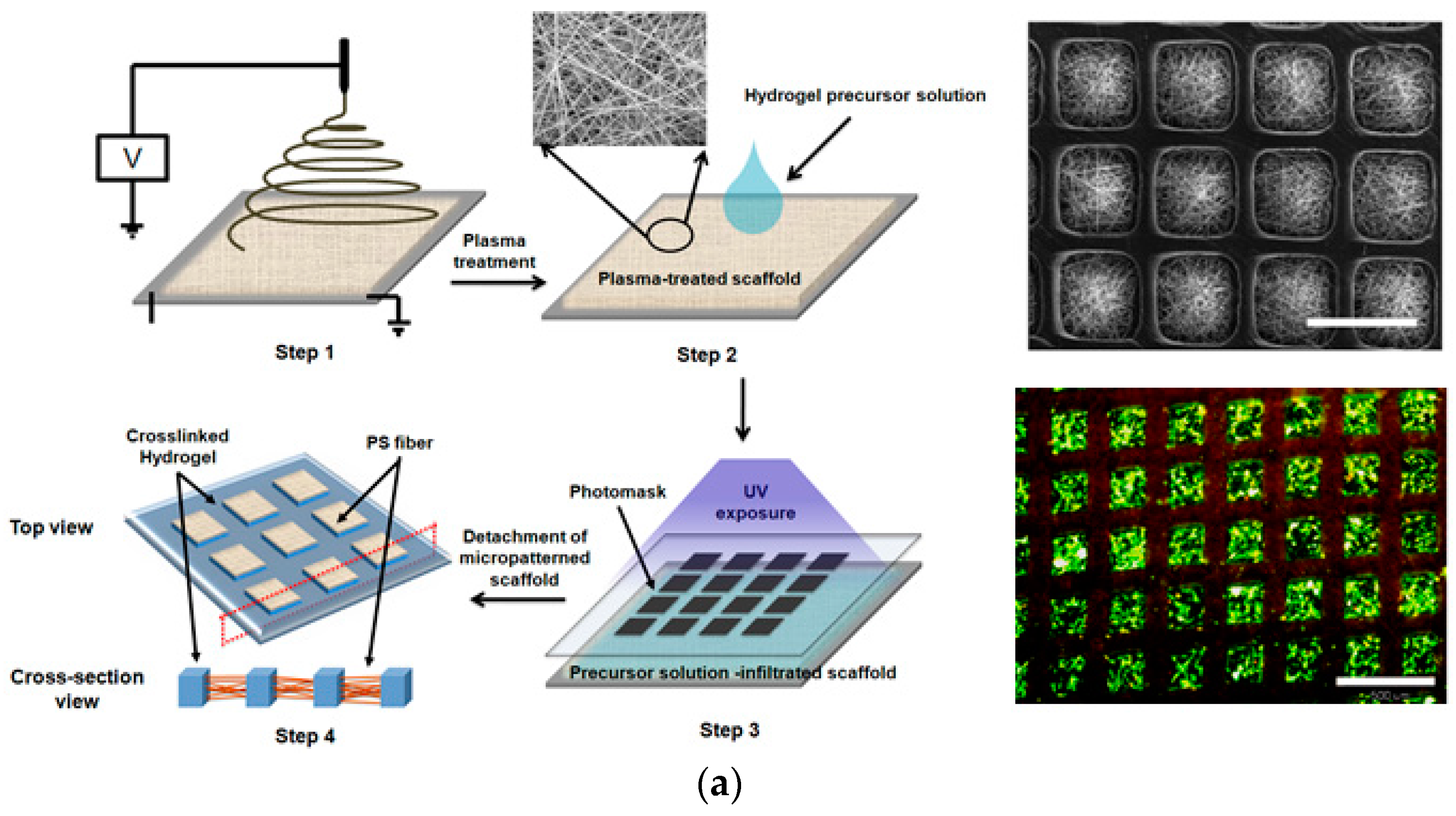
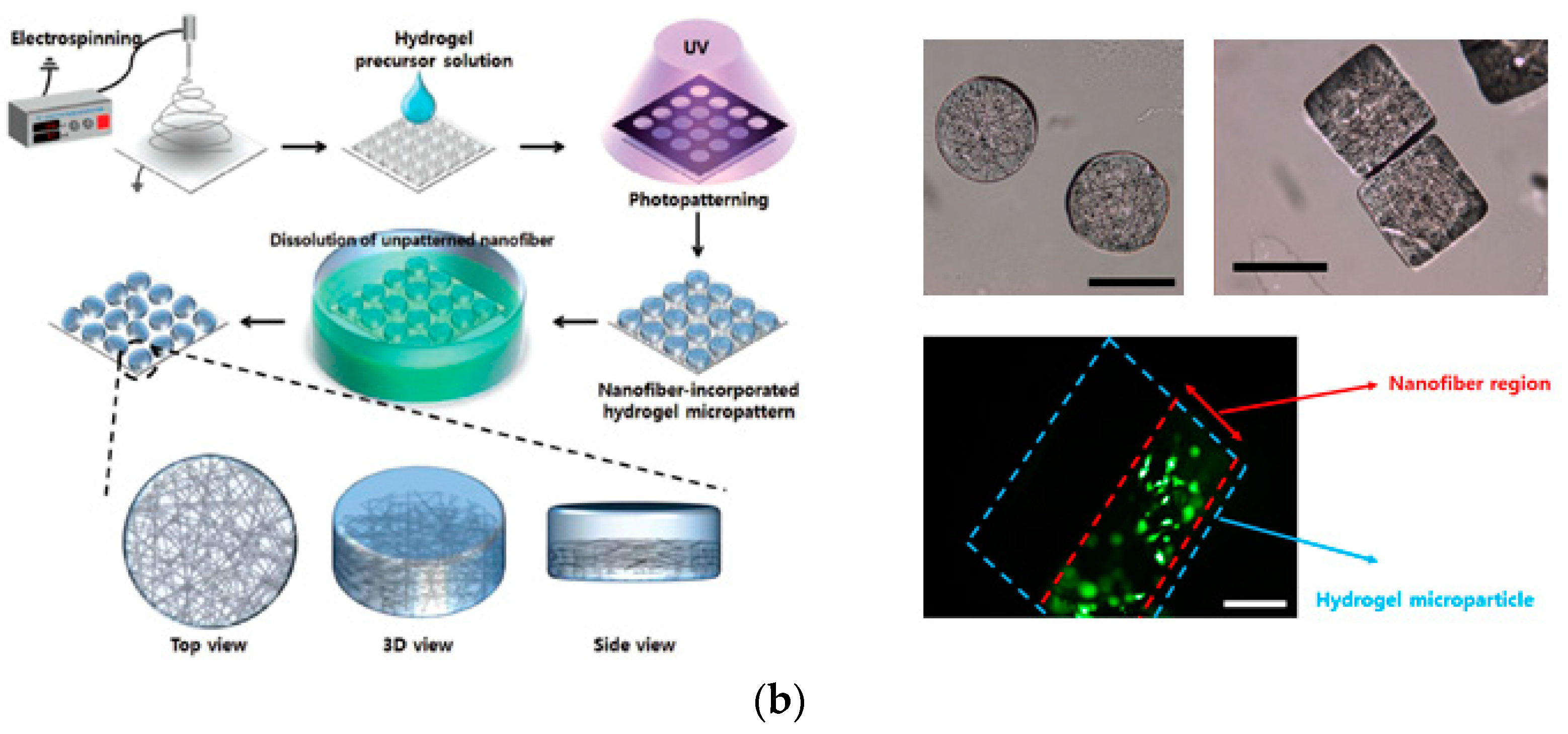
3. Cell Microarrays in a Biomimetic Environment
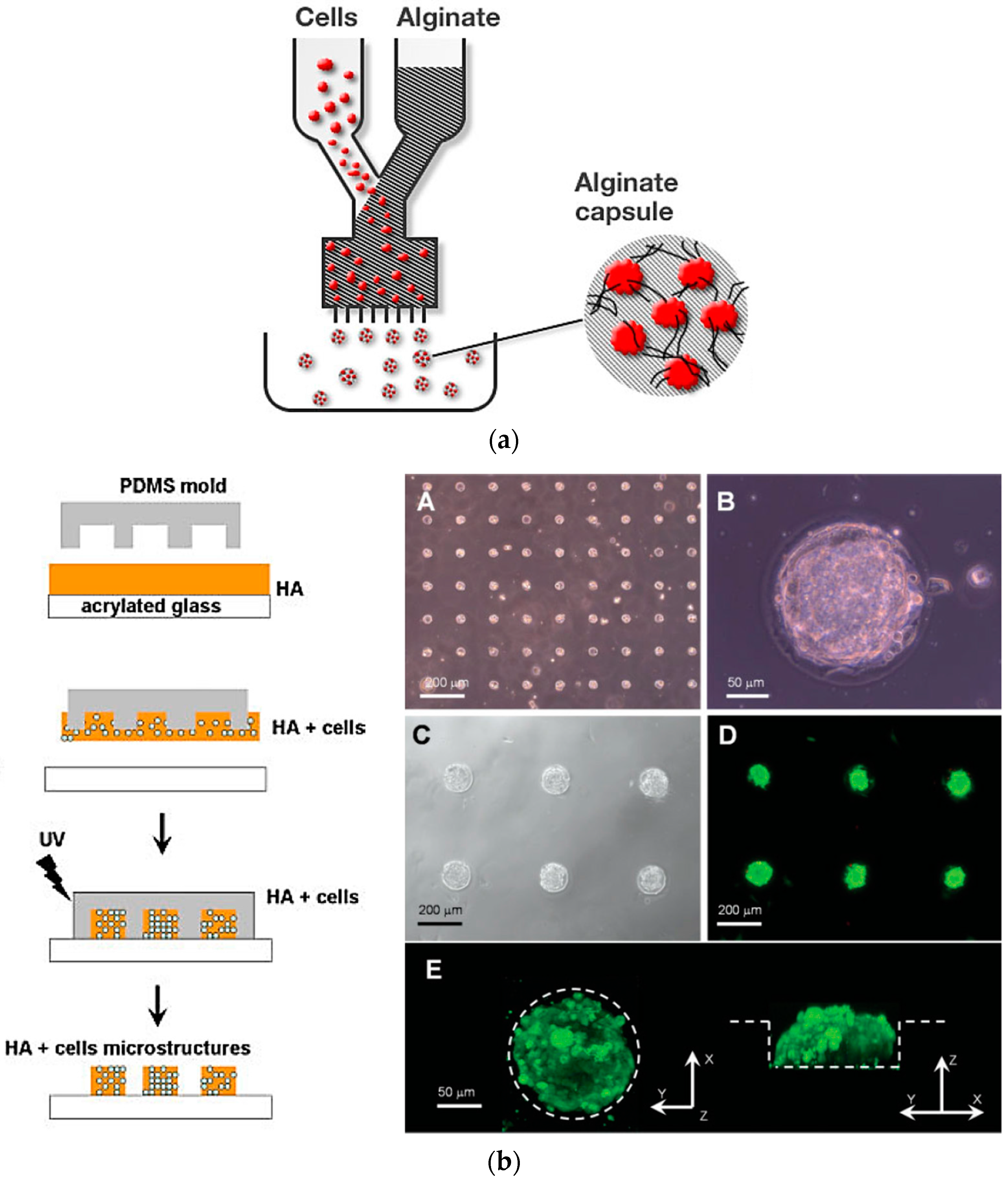
3.1. Hydrogel-Based 3D Cell Microarrays
3.2. Nanofiber-Based Microarrays
4. Applications of Cell Microarrays
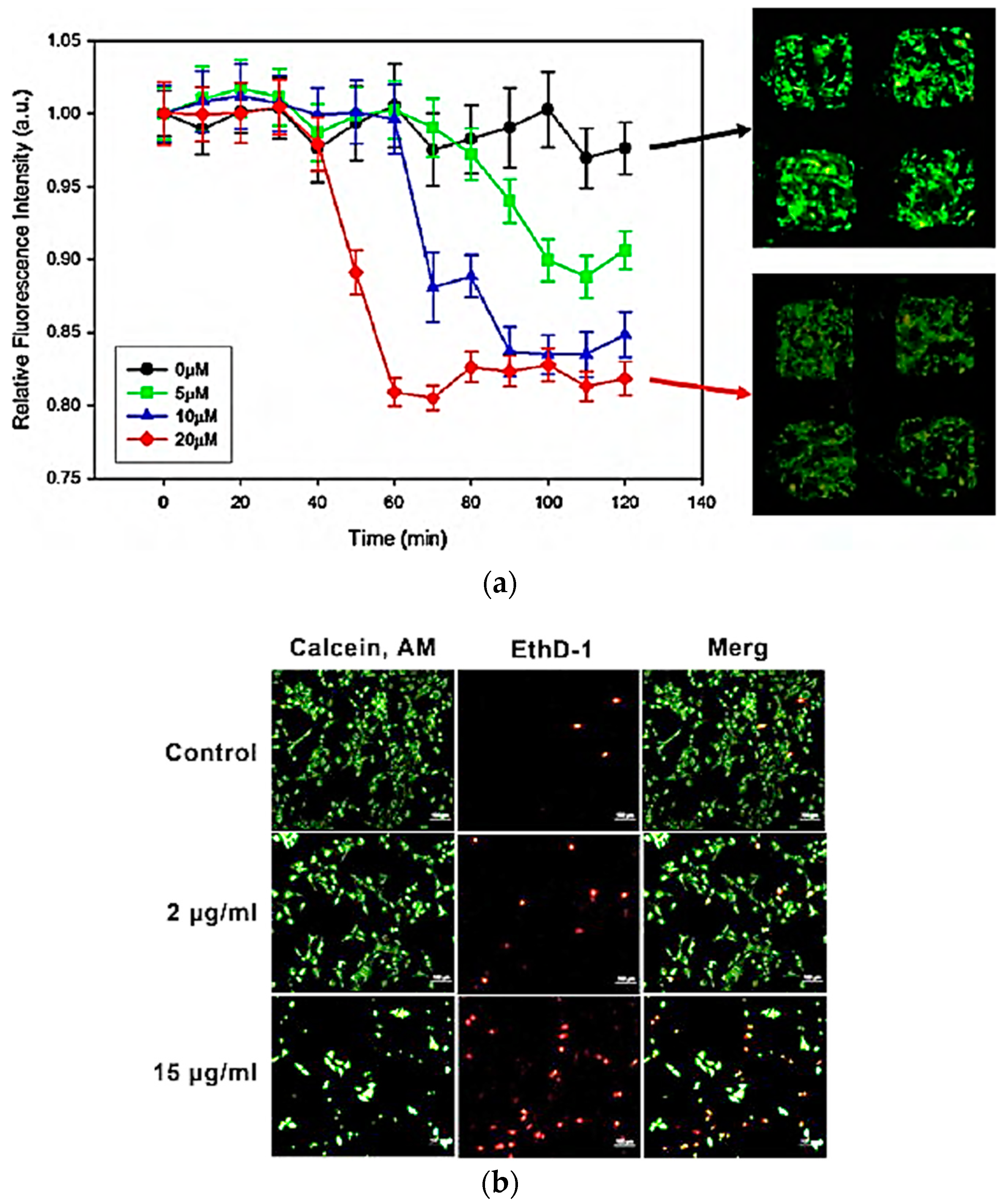
4.1. Toxicology
4.2. Drug Discovery
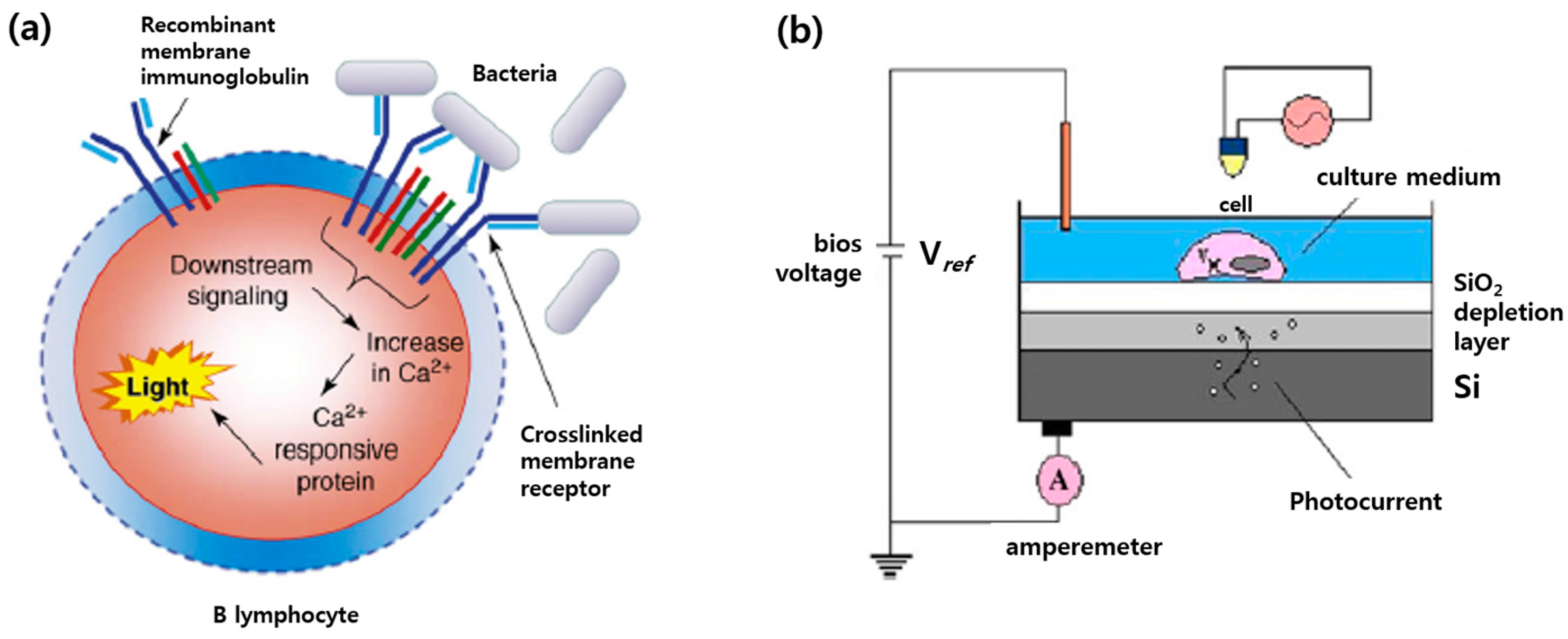
4.3. Biosensor for Pathogens and Toxins
5. Conclusions and Future Perspective
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Goode, J.; Rushworth, J.; Millner, P. Biosensor regeneration: A review of common techniques and outcomes. Langmuir 2014, 31, 6267–6276. [Google Scholar] [CrossRef] [PubMed]
- Berthuy, O.I.; Muldur, S.K.; Rossi, F.; Colpo, P.; Blum, L.J.; Marquette, C.A. Multiplex cell microarrays for high-throughput screening. Lab Chip 2016, 16, 4248–4262. [Google Scholar] [CrossRef] [PubMed]
- Pancrazio, J.; Whelan, J.; Borkholder, D.; Ma, W.; Stenger, D. Development and application of cell-based biosensors. Ann. Biomed. Eng. 1999, 27, 697–711. [Google Scholar] [CrossRef] [PubMed]
- Bousse, L. Whole cell biosensors. Sens. Actuators B 1996, 34, 270–275. [Google Scholar] [CrossRef]
- Khandurina, J.; Guttman, A. Microchip-based high-throughput screening analysis of combinatorial libraries. Curr. Opin. Chem. Biol. 2002, 6, 359–366. [Google Scholar] [CrossRef]
- El-Ali, J.; Sorger, P.K.; Jensen, K.F. Cells on chips. Nature 2006, 442, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.N.; Yarmush, M.L.; Toner, M. Controlling cell interactions by micropatterning in co-cultures: Hepatocytes and 3T3 fibroblasts. J. Biomed. Mater. Res. 1997, 34, 189–199. [Google Scholar] [CrossRef]
- Castel, D.; Pitaval, A.; Debily, M.-A.; Gidrol, X. Cell microarrays in drug discovery. Drug Discov. Today 2006, 11, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Folch, A.; Toner, M. Microengineering of cellular interactions. Annu. Rev. Biomed. Eng. 2000, 2, 227–256. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y. Surface micropatterning to regulate cell functions. Biomaterials 1999, 20, 2333–2342. [Google Scholar] [CrossRef]
- Kane, R.S.; Takayama, S.; Ostuni, E.; Ingber, D.E.; Whitesides, G.M. Patterning proteins and cells using soft lithography. Biomaterials 1999, 20, 2363–2376. [Google Scholar] [CrossRef]
- Beske, O.E.; Goldbard, S. High-throughput cell analysis using multiplexed array technologies. Drug Discov. Today 2002, 7, S131–S135. [Google Scholar] [CrossRef]
- Fernandes, T.G.; Diogo, M.M.; Clark, D.S.; Dordick, J.S.; Cabral, J.M. High-throughput cellular microarray platforms: Applications in drug discovery, toxicology and stem cell research. Trends Biotechnol. 2009, 27, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Yarmush, M.L.; King, K.R. Living-Cell Microarrays. Annu. Rev. Biomed. Eng. 2009, 11, 235–257. [Google Scholar] [CrossRef] [PubMed]
- Hanein, Y.; Pan, Y.V.; Ratner, B.D.; Denton, D.D.; Böhringer, K.F. Micromachining of non-fouling coatings for bio-MEMS applications. Sens. Actuators B 2001, 81, 49–54. [Google Scholar] [CrossRef]
- Khademhosseini, A.; Langer, R.; Borenstein, J.; Vacanti, J.P. Microscale technologies for tissue engineering and biology. Proc. Natl. Acad. Sci. USA 2006, 103, 2480–2487. [Google Scholar] [CrossRef] [PubMed]
- Allcock, H.R.; Phelps, M.V.; Barrett, E.W.; Pishko, M.V.; Koh, W.-G. Ultraviolet photolithographic development of polyphosphazene hydrogel microstructures for potential use in microarray biosensors. Chem. Mater. 2006, 18, 609–613. [Google Scholar] [CrossRef]
- Karp, J.M.; Yeo, Y.; Geng, W.; Cannizarro, C.; Yan, K.; Kohane, D.S.; Vunjak-Novakovic, G.; Langer, R.S.; Radisic, M. A photolithographic method to create cellular micropatterns. Biomaterials 2006, 27, 4755–4764. [Google Scholar] [CrossRef] [PubMed]
- Bouaidat, S.; Berendsen, C.; Thomsen, P.; Petersen, S.G.; Wolff, A.; Jonsmann, J. Micro patterning of cell and protein non-adhesive plasma polymerized coatings for biochip applications. Lab Chip 2004, 4, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Brétagnol, F.; Ceriotti, L.; Lejeune, M.; Papadopoulou-Bouraoui, A.; Hasiwa, M.; Gilliland, D.; Ceccone, G.; Colpo, P.; Rossi, F. Functional Micropatterned Surfaces by Combination of Plasma Polymerization and Lift-Off Processes. Plasma Processes Polym. 2006, 3, 30–38. [Google Scholar] [CrossRef]
- Martin, T.A.; Caliari, S.R.; Williford, P.D.; Harley, B.A.; Bailey, R.C. The generation of biomolecular patterns in highly porous collagen-GAG scaffolds using direct photolithography. Biomaterials 2011, 32, 3949–3957. [Google Scholar] [CrossRef] [PubMed]
- Myung, D.; Koh, W.; Bakri, A.; Zhang, F.; Marshall, A.; Ko, J.; Noolandi, J.; Carrasco, M.; Cochran, J.R.; Frank, C.W. Design and fabrication of an artificial cornea based on a photolithographically patterned hydrogel construct. Biomed. Microdevices 2007, 9, 911–922. [Google Scholar] [CrossRef] [PubMed]
- Dewez, J.-L.; Lhoest, J.-B.; Detrait, E.; Berger, V.; Dupont-Gillain, C.C.; Vincent, L.-M.; Schneider, Y.-J.; Bertrand, P.; Rouxhet, P. Adhesion of mammalian cells to polymer surfaces: from physical chemistry of surfaces to selective adhesion on defined patterns. Biomaterials 1998, 19, 1441–1445. [Google Scholar] [CrossRef]
- Willner, I.; Blonder, R. Patterning of surfaces by photoisomerizable antibody-antigen monolayers. Thin Solid Films 1995, 266, 254–257. [Google Scholar] [CrossRef]
- Hahn, M.S.; Taite, L.J.; Moon, J.J.; Rowland, M.C.; Ruffino, K.A.; West, J.L. Photolithographic patterning of polyethylene glycol hydrogels. Biomaterials 2006, 27, 2519–2524. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, D.N.; Park, S.; Lee, Y.; Koh, W.-G. Micropatterning of a nanoporous alumina membrane with poly (ethylene glycol) hydrogel to create cellular micropatterns on nanotopographic substrates. Acta Biomater. 2011, 7, 1281–1289. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.-G.; Revzin, A.; Simonian, A.; Reeves, T.; Pishko, M. Control of mammalian cell and bacteria adhesion on substrates micropatterned with poly (ethylene glycol) hydrogels. Biomed. Microdevices 2003, 5, 11–19. [Google Scholar] [CrossRef]
- Rettig, J.R.; Folch, A. Large-scale single-cell trapping and imaging using microwell arrays. Anal. Chem. 2005, 77, 5628–5634. [Google Scholar] [CrossRef] [PubMed]
- Caliari, S.R.; Burdick, J.A. A practical guide to hydrogels for cell culture. Nat. Methods 2016, 13, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Maitra, J.; Shukla, V.K. Cross-linking in hydrogels-a review. Am. J. Polym. Sci. 2014, 4, 25–31. [Google Scholar]
- Tseng, T.C.; Tao, L.; Hsieh, F.Y.; Wei, Y.; Chiu, I.M.; Hsu, S.H. An Injectable, Self-Healing Hydrogel to Repair the Central Nervous System. Adv. Mater. 2015, 27, 3518–3524. [Google Scholar] [CrossRef] [PubMed]
- Michel, B.; Bernard, A.; Bietsch, A.; Delamarche, E.; Geissler, M.; Juncker, D.; Kind, H.; Renault, J.-P.; Rothuizen, H.; Schmid, H. Printing meets lithography: Soft approaches to high-resolution patterning. IBM J. Res. Dev. 2001, 45, 697–719. [Google Scholar] [CrossRef]
- Hovis, J.S.; Boxer, S.G. Patterning and composition arrays of supported lipid bilayers by microcontact printing. Langmuir 2001, 17, 3400–3405. [Google Scholar] [CrossRef]
- Nie, Z.; Kumacheva, E. Patterning surfaces with functional polymers. Nat. Mater. 2008, 7, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Hauff, K.; Zambarda, C.; Dietrich, M.; Halbig, M.; Grab, A.L.; Medda, R.; Cavalcanti-Adam, E.A. Matrix-immobilized BMP-2 on microcontact printed fibronectin as an in vitro tool to study BMP-mediated signaling and cell migration. Front. Bioeng. Biotechnol. 2015, 3, 62. [Google Scholar] [CrossRef] [PubMed]
- Falconnet, D.; Csucs, G.; Grandin, H.M.; Textor, M. Surface engineering approaches to micropattern surfaces for cell-based assays. Biomaterials 2006, 27, 3044–3063. [Google Scholar] [CrossRef] [PubMed]
- Jung, D.; Kapur, R.; Adams, T.; Giuliano, K.; Mrksich, M.; Craighead, H.; Taylor, D. Topographical and physicochemical modification of material surface to enable patterning of living cells. Crit. Rev. Biotechnol. 2001, 21, 111–154. [Google Scholar] [CrossRef] [PubMed]
- Lan, M.A.; Gersbach, C.A.; Michael, K.E.; Keselowsky, B.G.; García, A.J. Myoblast proliferation and differentiation on fibronectin-coated self assembled monolayers presenting different surface chemistries. Biomaterials 2005, 26, 4523–4531. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Hyun, J.; Stiller, P.; Chilkoti, A. “Non-Fouling” Oligo (ethylene glycol)-Functionalized Polymer Brushes Synthesized by Surface-Initiated Atom Transfer Radical Polymerization. Adv. Mater. 2004, 16, 338–341. [Google Scholar] [CrossRef]
- Mrksich, M.; Dike, L.E.; Tien, J.; Ingber, D.E.; Whitesides, G.M. Using microcontact printing to pattern the attachment of mammalian cells to self-assembled monolayers of alkanethiolates on transparent films of gold and silver. Exp. Cell Res. 1997, 235, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Au, A.K.; Huynh, W.; Horowitz, L.F.; Folch, A. 3D-printed microfluidics. Angew. Chem. Int. Ed. 2016, 55, 3862–3881. [Google Scholar] [CrossRef] [PubMed]
- Breslauer, D.N.; Lee, P.J.; Lee, L.P. Microfluidics-based systems biology. Mol. Biosyst. 2006, 2, 97–112. [Google Scholar] [CrossRef] [PubMed]
- Khademhosseini, A.; Yeh, J.; Eng, G.; Karp, J.; Kaji, H.; Borenstein, J.; Farokhzad, O.C.; Langer, R. Cell docking inside microwells within reversibly sealed microfluidic channels for fabricating multiphenotype cell arrays. Lab Chip 2005, 5, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
- Rhee, S.W.; Taylor, A.M.; Tu, C.H.; Cribbs, D.H.; Cotman, C.W.; Jeon, N.L. Patterned cell culture inside microfluidic devices. Lab Chip 2005, 5, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Crozatier, C.; Le Berre, M.; Chen, Y. In situ bio-functionalization and cell adhesion in microfluidic devices. Microelectron. Eng. 2005, 78, 556–562. [Google Scholar] [CrossRef]
- Goto, M.; Tsukahara, T.; Sato, K.; Kitamori, T. Micro-and nanometer-scale patterned surface in a microchannel for cell culture in microfluidic devices. Anal. Bioanal. Chem. 2008, 390, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Lahann, J.; Balcells, M.; Lu, H.; Rodon, T.; Jensen, K.F.; Langer, R. Reactive polymer coatings: A first step toward surface engineering of microfluidic devices. Anal. Chem. 2003, 75, 2117–2122. [Google Scholar] [CrossRef] [PubMed]
- Chiu, D.T.; Jeon, N.L.; Huang, S.; Kane, R.S.; Wargo, C.J.; Choi, I.S.; Ingber, D.E.; Whitesides, G.M. Patterned deposition of cells and proteins onto surfaces by using three-dimensional microfluidic systems. Proc. Natl. Acad. Sci. USA 2000, 97, 2408–2413. [Google Scholar] [CrossRef] [PubMed]
- Nie, F.-Q.; Yamada, M.; Kobayashi, J.; Yamato, M.; Kikuchi, A.; Okano, T. On-chip cell migration assay using microfluidic channels. Biomaterials 2007, 28, 4017–4022. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yuan, B.; Ji, H.; Han, D.; Chen, S.; Tian, F.; Jiang, X. A Method for Patterning Multiple Types of Cells by Using Electrochemical Desorption of Self-Assembled Monolayers within Microfluidic Channels. Angew. Chem. 2007, 119, 1112–1114. [Google Scholar] [CrossRef]
- Ismagilov, R.F.; Rosmarin, D.; Kenis, P.J.; Chiu, D.T.; Zhang, W.; Stone, H.A.; Whitesides, G.M. Pressure-driven laminar flow in tangential microchannels: an elastomeric microfluidic switch. Anal. Chem. 2001, 73, 4682–4687. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.-S.; Kwon, K.; Kim, S.-I.; Han, H.; Sohn, J.; Lee, S.; Jung, H.-I. Continuous separation of breast cancer cells from blood samples using multi-orifice flow fractionation (MOFF) and dielectrophoresis (DEP). Lab Chip 2011, 11, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Takayama, S.; McDonald, J.C.; Ostuni, E.; Liang, M.N.; Kenis, P.J.; Ismagilov, R.F.; Whitesides, G.M. Patterning cells and their environments using multiple laminar fluid flows in capillary networks. Proc. Natl. Acad. Sci. USA 1999, 96, 5545–5548. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.-G.; Itle, L.J.; Pishko, M.V. Molding of hydrogel microstructures to create multiphenotype cell microarrays. Anal. Chem. 2003, 75, 5783–5789. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Zhu, X.; Futai, N.; Cho, B.S.; Takayama, S. Computerized microfluidic cell culture using elastomeric channels and Braille displays. Proc. Natl. Acad. Sci. USA 2004, 101, 15861–15866. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, M.N.; Houseman, B.T.; Mrksich, M. Using electroactive substrates to pattern the attachment of two different cell populations. Proc. Natl. Acad. Sci. USA 2001, 98, 5992–5996. [Google Scholar] [CrossRef] [PubMed]
- Voldman, J. Electrical forces for microscale cell manipulation. Annu. Rev. Biomed. Eng. 2006, 8, 425–454. [Google Scholar] [CrossRef] [PubMed]
- Voldman, J. A Microfabricated Dielectrophoretic Trapping Array for Cell-Based Biological Assays. Ph.D. Thesis, Massachusetts Institute of Technology, Cambridge, MA, USA, 2001. [Google Scholar]
- Gray, D.S.; Tan, J.L.; Voldman, J.; Chen, C.S. Dielectrophoretic registration of living cells to a microelectrode array. Biosens. Bioelectron. 2004, 19, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Joo, S.; Duhon, M.; Heller, M.; Wallace, B.; Xu, X. Dielectrophoretic cell separation and gene expression profiling on microelectronic chip arrays. Anal. Chem. 2002, 74, 3362–3371. [Google Scholar] [CrossRef] [PubMed]
- Voldman, J.; Gray, M.L.; Toner, M.; Schmidt, M.A. A microfabrication-based dynamic array cytometer. Anal. Chem. 2002, 74, 3984–3990. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, H.; Yu, E.; Marquina, S.; Valamehr, B.; Wong, I.; Wu, H.; Ho, C.-M. Efficient dielectrophoretic patterning of embryonic stem cells in energy landscapes defined by hydrogel geometries. Ann. Biomed. Eng. 2010, 38, 3777–3788. [Google Scholar] [CrossRef] [PubMed]
- Wilkins Stevens, P.; Wang, C.J.; Kelso, D.M. Immobilized particle arrays: coalescence of planar-and suspension-array technologies. Anal. Chem. 2003, 75, 1141–1146. [Google Scholar] [CrossRef]
- Nolan, J.P.; Sklar, L.A. Suspension array technology: evolution of the flat-array paradigm. Trends Biotechnol. 2002, 20, 9–12. [Google Scholar] [CrossRef]
- Gorris, H.H.; Wolfbeis, O.S. Photon-upconverting nanoparticles for optical encoding and multiplexing of cells, biomolecules, and microspheres. Angew. Chem. Int. Ed. 2013, 52, 3584–3600. [Google Scholar] [CrossRef] [PubMed]
- Beske, O.; Guo, J.; Li, J.; Bassoni, D.; Bland, K.; Marciniak, H.; Zarowitz, M.; Temov, V.; Ravkin, I.; Goldbard, S. A novel encoded particle technology that enables simultaneous interrogation of multiple cell types. J. Biomol. Screen. 2004, 9, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Martin, R.M.; Muzerelle, M.; Chitkul, N.; How, S.E.; Mittoo, S.; Bradley, M. Bead-Based Cellular Analysis, Sorting and Multiplexing. ChemBioChem 2005, 6, 1341–1345. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kim, J.; Kim, H.; Kim, J.; Kwon, S. Colour-barcoded magnetic microparticles for multiplexed bioassays. Nat. Mater. 2010, 9, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Birtwell, S.; Morgan, H. Microparticle encoding technologies for high-throughput multiplexed suspension assays. Integr. Biol. 2009, 1, 345–362. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Shim, J.W.; Yang, S.M. Microfluidic multicolor encoding of microspheres with nanoscopic surface complexity for multiplex immunoassays. Angew. Chem. 2011, 123, 1203–1206. [Google Scholar] [CrossRef]
- Braeckmans, K.; De Smedt, S.C.; Leblans, M.; Pauwels, R.; Demeester, J. Encoding microcarriers: Present and future technologies. Nat. Rev. Drug Discov. 2002, 1, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Stubbe, B.G.; Gevaert, K.; Derveaux, S.; Braeckmans, K.; De Geest, B.G.; Goethals, M.; Vandekerckhove, J.; Demeester, J.; De Smedt, S.C. Evaluation of Encoded Layer-by-Layer Coated Microparticles as Protease Sensors. Adv. Funct. Mater. 2008, 18, 1624–1631. [Google Scholar] [CrossRef]
- Han, S.W.; Jang, E.; Koh, W.-G. Microfluidic-based multiplex immunoassay system integrated with an array of QD-encoded microbeads. Sens. Actuators B 2015, 209, 242–251. [Google Scholar] [CrossRef]
- Han, M.; Gao, X.; Su, J.Z.; Nie, S. Quantum-dot-tagged microbeads for multiplexed optical coding of biomolecules. Nat. Biotechnol. 2001, 19, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhao, X.; Pei, X.; Hu, J.; Zhao, W.; Chen, B.; Gu, Z. Multiplex detection of tumor markers with photonic suspension array. Anal. Chim. Acta 2009, 633, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.; Xu, K.; Sun, Y.; Chen, Y.; Zheng, T.; Li, J. High sensitive immunoassay for multiplex mycotoxin detection with photonic crystal microsphere suspension array. Anal. Chem. 2013, 85, 2833–2840. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhao, X.; Sun, C.; Li, J.; Zhu, R.; Gu, Z. Encoded silica colloidal crystal beads as supports for potential multiplex immunoassay. Anal. Chem. 2008, 80, 1598–1605. [Google Scholar] [CrossRef] [PubMed]
- Appleyard, D.C.; Chapin, S.C.; Srinivas, R.L.; Doyle, P.S. Bar-coded hydrogel microparticles for protein detection: Synthesis, assay and scanning. Nat. Protoc. 2011, 6, 1761–1774. [Google Scholar] [CrossRef] [PubMed]
- Le Goff, G.C.; Srinivas, R.L.; Hill, W.A.; Doyle, P.S. Hydrogel microparticles for biosensing. Eur. Polym. J. 2015, 72, 386–412. [Google Scholar] [CrossRef] [PubMed]
- Suh, S.K.; Chapin, S.C.; Hatton, T.A.; Doyle, P.S. Synthesis of magnetic hydrogel microparticles for bioassays and tweezer manipulation in microwells. Microfluid. Nanofluidics 2012, 13, 665–674. [Google Scholar] [CrossRef]
- Dendukuri, D.; Pregibon, D.C.; Collins, J.; Hatton, T.A.; Doyle, P.S. Continuous-flow lithography for high-throughput microparticle synthesis. Nat. Mater. 2006, 5, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Bong, K.W.; Chapin, S.C.; Doyle, P.S. Magnetic barcoded hydrogel microparticles for multiplexed detection. Langmuir 2010, 26, 8008–8014. [Google Scholar] [CrossRef] [PubMed]
- Pregibon, D.C.; Toner, M.; Doyle, P.S. Multifunctional encoded particles for high-throughput biomolecule analysis. Science 2007, 315, 1393–1396. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Young, G.; Aoto, P.C.; Pai, J.H.; Bachman, M.; Li, G.; Sims, C.E.; Allbritton, N.L. Broadening cell selection criteria with micropallet arrays of adherent cells. Cytometry A 2007, 71, 866–874. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.H.; Lee, H.J.; Son, K.J.; Koh, W.-G. Non-positional cell microarray prepared by shape-coded polymeric microboards: A new microarray format for multiplex and high throughput cell-based assays. Biomicrofluidics 2011, 5, 032001. [Google Scholar] [CrossRef] [PubMed]
- To’a Salazar, G.; Wang, Y.; Young, G.; Bachman, M.; Sims, C.E.; Li, G.; Allbritton, N.L. Micropallet arrays for the separation of single, adherent cells. Anal. Chem. 2007, 79, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Young, G.; Bachman, M.; Sims, C.E.; Li, G.; Allbritton, N.L. Collection and expansion of single cells and colonies released from a micropallet array. Anal. Chem. 2007, 79, 2359–2366. [Google Scholar] [CrossRef] [PubMed]
- Matsusaki, M.; Case, C.P.; Akashi, M. Three-dimensional cell culture technique and pathophysiology. Adv. Drug Deliv. Rev. 2014, 74, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Griffith, L.G.; Swartz, M.A. Capturing complex 3D tissue physiology in vitro. Nat. Rev. Mol. Cell Biol. 2006, 7, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Fennema, E.; Rivron, N.; Rouwkema, J.; van Blitterswijk, C.; de Boer, J. Spheroid culture as a tool for creating 3D complex tissues. Trends Biotechnol. 2013, 31, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Gidrol, X.; Fouqué, B.; Ghenim, L.; Haguet, V.; Picollet-D’hahan, N.; Schaack, B. 2D and 3D cell microarrays in pharmacology. Curr. Opin. Pharmacol. 2009, 9, 664–668. [Google Scholar] [CrossRef] [PubMed]
- Godoy, P.; Hewitt, N.J.; Albrecht, U.; Andersen, M.E.; Ansari, N.; Bhattacharya, S.; Bode, J.G.; Bolleyn, J.; Borner, C.; Böttger, J. Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanisms of hepatotoxicity, cell signaling and ADME. Arch. Toxicol. 2013, 87, 1315–1530. [Google Scholar] [CrossRef] [PubMed]
- Larson, B. 3D Cell Culture: A Review of Current Techniques. Available online: http://mktg.biotek.com/news/2015/Fall/featured-application.html (accessed on 23 February 2017).
- Nisbet, D.; Forsythe, J.S.; Shen, W.; Finkelstein, D.; Horne, M.K. Review paper: a review of the cellular response on electrospun nanofibers for tissue engineering. J. Biomater. Appl. 2008, 24, 7–29. [Google Scholar] [CrossRef] [PubMed]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Augst, A.D.; Kong, H.J.; Mooney, D.J. Alginate hydrogels as biomaterials. Macromol. Biosci. 2006, 6, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Cushing, M.C.; Anseth, K.S. Hydrogel cell cultures. Science 2007, 316, 1133–1134. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.N.; Birkinshaw, C. Hyaluronic acid based scaffolds for tissue engineering—A review. Carbohydr. Polym. 2013, 92, 1262–1279. [Google Scholar] [CrossRef] [PubMed]
- Nichol, J.W.; Koshy, S.T.; Bae, H.; Hwang, C.M.; Yamanlar, S.; Khademhosseini, A. Cell-laden microengineered gelatin methacrylate hydrogels. Biomaterials 2010, 31, 5536–5544. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, G.D.; Bryant, S.J. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng. B Rev. 2008, 14, 149–165. [Google Scholar] [CrossRef] [PubMed]
- Jen, A.C.; Wake, M.C.; Mikos, A.G. Review: Hydrogels for cell immobilization. Biotechnol. Bioeng. 1996, 50, 357–364. [Google Scholar] [CrossRef]
- Gerecht, S.; Burdick, J.A.; Ferreira, L.S.; Townsend, S.A.; Langer, R.; Vunjak-Novakovic, G. Hyaluronic acid hydrogel for controlled self-renewal and differentiation of human embryonic stem cells. Proc. Natl. Acad. Sci. USA 2007, 104, 11298–11303. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Song, H.; Gong, Y.; Mao, Z.; Gao, C.; Shen, J. Covalently crosslinked chitosan hydrogel: properties of in vitro degradation and chondrocyte encapsulation. Acta Biomater. 2007, 3, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Kim, S.-H.; Ryoo, J.H.; Wong, S.F.; Lee, S.-H. Diffusion-mediated in situ alginate encapsulation of cell spheroids using microscale concave well and nanoporous membrane. Lab Chip 2011, 11, 1168–1173. [Google Scholar] [CrossRef] [PubMed]
- Andersen, T.; Auk-Emblem, P.; Dornish, M. 3D cell culture in alginate hydrogels. Microarrays 2015, 4, 133–161. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.H.; Takeuchi, S. Monodisperse alginate hydrogel microbeads for cell encapsulation. Adv. Mater. 2007, 19, 2696–2701. [Google Scholar] [CrossRef]
- Khademhosseini, A.; Eng, G.; Yeh, J.; Fukuda, J.; Blumling, J.; Langer, R.; Burdick, J.A. Micromolding of photocrosslinkable hyaluronic acid for cell encapsulation and entrapment. J. Biomed. Mater. Res. A 2006, 79, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Lee, J.-S.; Chansakul, T.; Yu, C.; Elisseeff, J.H.; Seungju, M.Y. Collagen mimetic peptide-conjugated photopolymerizable PEG hydrogel. Biomaterials 2006, 27, 5268–5276. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Anseth, K.S. PEG hydrogels for the controlled release of biomolecules in regenerative medicine. Pharm. Res. 2009, 26, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Selimović, Š.; Oh, J.; Bae, H.; Dokmeci, M.; Khademhosseini, A. Microscale strategies for generatingcell-encapsulating hydrogels. Polymers 2012, 4, 1554–1579. [Google Scholar] [CrossRef] [PubMed]
- Khademhosseini, A.; Eng, G.; Yeh, J.; Kucharczyk, P.A.; Langer, R.; Vunjak-Novakovic, G.; Radisic, M. Microfluidic patterning for fabrication of contractile cardiac organoids. Biomed. Microdevices 2007, 9, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Koh, W.-G.; Pishko, M. Photoreaction injection molding of biomaterial microstructures. Langmuir 2003, 19, 10310–10316. [Google Scholar] [CrossRef]
- Schmedlen, R.H.; Masters, K.S.; West, J.L. Photocrosslinkable polyvinyl alcohol hydrogels that can be modified with cell adhesion peptides for use in tissue engineering. Biomaterials 2002, 23, 4325–4332. [Google Scholar] [CrossRef]
- Primiceri, E.; Chiriacò, M.S.; Rinaldi, R.; Maruccio, G. Cell chips as new tools for cell biology–results, perspectives and opportunities. Lab Chip 2013, 13, 3789–3802. [Google Scholar] [CrossRef] [PubMed]
- Jen, C.-P.; Hsiao, J.-H.; Maslov, N.A. Single-cell chemical lysis on microfluidic chips with arrays of microwells. Sensors 2011, 12, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Lo, J.F.; Sinkala, E.; Eddington, D.T. Oxygen gradients for open well cellular cultures via microfluidic substrates. Lab Chip 2010, 10, 2394–2401. [Google Scholar] [CrossRef] [PubMed]
- Young, E.W.; Beebe, D.J. Fundamentals of microfluidic cell culture in controlled microenvironments. Chem. Soc. Rev. 2010, 39, 1036–1048. [Google Scholar] [CrossRef] [PubMed]
- Mehling, M.; Tay, S. Microfluidic cell culture. Curr. Opin. Biotechnol. 2014, 25, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Regehr, K.J.; Domenech, M.; Koepsel, J.T.; Carver, K.C.; Ellison-Zelski, S.J.; Murphy, W.L.; Schuler, L.A.; Alarid, E.T.; Beebe, D.J. Biological implications of polydimethylsiloxane-based microfluidic cell culture. Lab Chip 2009, 9, 2132–2139. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, A.S. Hydrogels for biomedical applications. Ann. N. Y. Acad. Sci. 2001, 944, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Illeperuma, W.R.K.; Suo, Z.; Vlassak, J.J. Hybrid hydrogels with extremely high stiffness and toughness. ACS Macro Lett. 2014, 3, 520–523. [Google Scholar] [CrossRef]
- Maguire, T.; Novik, E.; Schloss, R.; Yarmush, M. Alginate-PLL microencapsulation: effect on the differentiation of embryonic stem cells into hepatocytes. Biotechnol. Bioeng. 2006, 93, 581–591. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Yong, T.; Ma, Z.W.; Inai, R.; Teo, W.E.; Ramakrishna, S. Biodegradable polymer nanofiber mesh to maintain functions of endothelial cells. Tissue Eng. 2006, 12, 2457–2466. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.-S.; Kook, Y.-M.; Hong, H.J.; Kim, Y.-M.; Koh, W.-G.; Lim, J.-Y. Functional spheroid organization of human salivary gland cells cultured on hydrogel-micropatterned nanofibrous microwells. Acta Biomater. 2016, 45, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Wendorff, J.H.; Greiner, A. Use of electrospinning technique for biomedical applications. Polymer 2008, 49, 5603–5621. [Google Scholar] [CrossRef]
- Lim, D.-J.; Sim, M.; Heo, Y.; Jun, H.-W.; Park, H. Facile method for fabricating uniformly patterned and porous nanofibrous scaffolds for tissue engineering. Macromol. Res. 2015, 23, 1152–1158. [Google Scholar] [CrossRef]
- Lee, H.; Lim, S.; Birajdar, M.S.; Lee, S.-H.; Park, H. Fabrication of FGF-2 immobilized electrospun gelatin nanofibers for tissue engineering. Int. J. Biol. Macromol. 2016, 93, 1559–1566. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A review on polymer nanofibers by electrospinning and their applications in nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Cipitria, A.; Skelton, A.; Dargaville, T.; Dalton, P.; Hutmacher, D. Design, fabrication and characterization of PCL electrospun scaffolds—A review. J. Mater. Chem. 2011, 21, 9419–9453. [Google Scholar] [CrossRef]
- Shi, J.; Wang, L.; Chen, Y. Microcontact printing and lithographic patterning of electrospun nanofibers. Langmuir 2009, 25, 6015–6018. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, B.R.; Wang, T.; Liu, J. Direct photolithographic patterning of electrospun films for defined nanofibrillar microarchitectures. Langmuir 2010, 26, 2235–2239. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Salim, A.; Ziaie, B. Selective nanofiber deposition through field-enhanced electrospinning. Langmuir 2009, 25, 9648–9652. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Xie, Z.; Zhang, L.; Ma, X.; Wu, X.; Guo, Y.; Song, W.; Li, Z.; Cao, T. Microfluidic etching for fabrication of flexible and all-solid-state micro supercapacitor based on MnO2 nanoparticles. Nanoscale 2011, 3, 2703–2708. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xu, Y.; Xu, H.; Chang, J. Electrospun membranes: control of the structure and structure related applications in tissue regeneration and drug delivery. J. Mater. Chem. 2014, 2, 5492–5510. [Google Scholar] [CrossRef]
- Lee, H.J.; Park, Y.H.; Koh, W.G. Fabrication of nanofiber microarchitectures localized within hydrogel microparticles and their application to protein delivery and cell encapsulation. Adv. Funct. Mater. 2013, 23, 591–597. [Google Scholar] [CrossRef]
- Lee, H.J.; Kim, H.-S.; Kim, H.O.; Koh, W.-G. Micropatterns of double-layered nanofiber scaffolds with dual functions of cell patterning and metabolite detection. Lab Chip 2011, 11, 2849–2857. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Nam, S.H.; Son, K.J.; Koh, W.G. Micropatterned Fibrous Scaffolds Fabricated Using Electrospinning and Hydrogel Lithography: New Platforms to Create Cellular Micropatterns. Sens. Actuators B 2010, 148, 504–510. [Google Scholar] [CrossRef]
- Lee, H.W.; Kook, Y.-M.; Lee, H.J.; Park, H.; Koh, W.-G. A three-dimensional co-culture of HepG2 spheroids and fibroblasts using double-layered fibrous scaffolds incorporated with hydrogel micropatterns. RSC Adv. 2014, 4, 61005–61011. [Google Scholar] [CrossRef]
- Cho, K.; Lee, H.J.; Han, S.W.; Min, J.H.; Park, H.; Koh, W.G. Multi-Compartmental Hydrogel Microparticles Fabricated by Combination of Sequential Electrospinning and Photopatterning. Angew. Chem. Int. Ed. 2015, 54, 11511–11515. [Google Scholar] [CrossRef] [PubMed]
- Kook, Y.-M.; Kang, Y.M.; Moon, S.H.; Koh, W.-G. Bi-compartmental 3D scaffolds for the co-culture of intervertebral disk cells and mesenchymal stem cells. J. Ind. Eng. Chem. 2016, 38, 113–122. [Google Scholar] [CrossRef]
- Jiang, L.; Zhang, M.; Li, J.; Wen, W.; Qin, J. Simple Localization of Nanofiber Scaffolds via SU-8 Photoresist and Their Use for Parallel 3D Cellular Assays. Adv. Mater. 2012, 24, 2191–2195. [Google Scholar] [CrossRef] [PubMed]
- Han, S.W.; Koh, W.-G. Hydrogel-Framed Nanofiber Matrix Integrated with a Microfluidic Device for Fluorescence Detection of Matrix Metalloproteinases-9. Anal. Chem. 2016, 88, 6247–6253. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, C.; Cheng, Z.; Zhang, D.; Li, S.; Song, L.; Zhou, W.; Yang, M.; Wang, Z.; Zheng, Z. Spri determination of inter-peptide interaction by using 3D supramolecular co-assembly polyrotaxane film. Biosens. Bioelectron. 2015, 66, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Kurkuri, M.D.; Driever, C.; Johnson, G.; McFarland, G.; Thissen, H.; Voelcker, N.H. Multifunctional polymer coatings for cell microarray applications. Biomacromolecules 2009, 10, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Dalilottojari, A.; Delalat, B.; Harding, F.J.; Cockshell, M.P.; Bonder, C.S.; Voelcker, N.H. Porous Silicon-Based Cell Microarrays: Optimizing Human Endothelial Cell-Material Surface Interactions and Bioactive Release. Biomacromolecules 2016, 17, 3724–3731. [Google Scholar] [CrossRef] [PubMed]
- Stenger, D.A.; Gross, G.W.; Keefer, E.W.; Shaffer, K.M.; Andreadis, J.D.; Ma, W.; Pancrazio, J.J. Detection of physiologically active compounds using cell-based biosensors. Trends Biotechnol. 2001, 19, 304–309. [Google Scholar] [CrossRef]
- Prestwich, G.D. Evaluating drug efficacy and toxicology in three dimensions: Using synthetic extracellular matrices in drug discovery. Acc. Chem. Res. 2008, 41, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Wegener, J. Cell-based microarrays for in vitro toxicology. Annu. Rev. Anal. Chem. 2015, 8, 335–358. [Google Scholar] [CrossRef] [PubMed]
- Esch, M.B.; King, T.L.; Shuler, M.L. The Role of Body-on-a-Chip Devices in Drug and Toxicity Studies. Annu. Rev. Biomed. Eng. 2011, 13, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Fadeel, B.; Garcia-Bennett, A.E. Better safe than sorry: Understanding the toxicological properties of inorganic nanoparticles manufactured for biomedical applications. Adv. Drug Deliv. Rev. 2010, 62, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Laurent, S.; Shokrgozar, M.A.; Hosseinkhani, M. Toxicity Evaluations of Superparamagnetic Iron Oxide Nanoparticles: Cell "Vision" versus Physicochemical Properties of Nanoparticles. ACS Nano 2011, 5, 7263–7276. [Google Scholar] [CrossRef] [PubMed]
- Sung, J.H.; Shuler, M.L. A micro cell culture analog (mu CCA) with 3-D hydrogel culture of multiple cell lines to assess metabolism-dependent cytotoxicity of anti-cancer drugs. Lab Chip 2009, 9, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.W.; Lee, M.Y.; Ku, B.; Yi, S.H.; Ryu, J.H.; Jeon, R.; Yang, M. Application of the DataChip/MetaChip technology for the evaluation of ajoene toxicity in vitro. Arch. Toxicol. 2014, 88, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Hillegass, J.M.; Shukla, A.; Lathrop, S.A.; MacPherson, M.B.; Fukagawa, N.K.; Mossman, B.T. Assessing nanotoxicity in cells in vitro. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2010, 2, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Montanez-Sauri, S.I.; Sung, K.E.; Puccinelli, J.P.; Pehlke, C.; Beebe, D.J. Automation of Three-Dimensional Cell Culture in Arrayed Microfluidic Devices. J. Lab. Autom. 2011, 16, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Fritzsche, M.; Mandenius, C.F. Fluorescent cell-based sensing approaches for toxicity testing. Anal. Bioanal. Chem. 2010, 398, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Cheng, F.Y.; Chiu, H.W.; Tsai, J.C.; Fang, C.Y.; Chen, C.W.; Wang, Y.J. Cytotoxicity, oxidative stress, apoptosis and the autophagic effects of silver nanoparticles in mouse embryonic fibroblasts. Biomaterials 2014, 35, 4706–4715. [Google Scholar] [CrossRef] [PubMed]
- Fotakis, G.; Timbrell, J.A. In vitro cytotoxicity assays: Comparison of LDH, neutral red, MTT and protein assay in hepatoma cell lines following exposure to cadmium chloride. Toxicol. Lett. 2006, 160, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.M.; Lalwani, G.; Zhang, K.V.; Yang, J.Y.; Neville, K.; Sitharaman, B. Cell specific cytotoxicity and uptake of graphene nanoribbons. Biomaterials 2013, 34, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Chabot, V.; Miron, Y.; Grandbois, M.; Charette, P.G. Long range surface plasmon resonance for increased sensitivity in living cell biosensing through greater probing depth. Sens. Actuators B 2012, 174, 94–101. [Google Scholar] [CrossRef]
- Fang, Y.; Ferrie, A.M.; Fontaine, N.H.; Mauro, J.; Balakrishnan, J. Resonant waveguide grating biosensor for living cell sensing. Biophys. J. 2006, 91, 1925–1940. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y. Live Cell Optical Sensing for High Throughput Applications. Adv. Biochem. Eng. Biotechnol. 2010, 118, 153–163. [Google Scholar]
- Muller, J.; Thirion, C.; Pfaffl, M.W. Electric cell-substrate impedance sensing (ECIS) based real-time measurement of titer dependent cytotoxicity induced by adenoviral vectors in an IPI-2I cell culture model. Biosens. Bioelectron. 2011, 26, 2000–2005. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.B.; Cho, S.; Min, J. Hydrogel-based diffusion chip with Electric Cell-substrate Impedance Sensing (ECIS) integration for cell viability assay and drug toxicity screening. Biosens. Bioelectron. 2013, 50, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Kojima, K.; Yasuda, K. An on-chip cardiomyocyte cell network assay for stable drug screening regarding community effect of cell network size. Analyst 2007, 132, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, A.F.M.; Gross, G.W.; Weiss, D.G.; Schroeder, O.H.U.; Gramowski, A.; Shafer, T.J. Microelectrode arrays: A physiologically based neurotoxicity testing platform for the 21st century. Neurotoxicology 2010, 31, 331–350. [Google Scholar] [CrossRef] [PubMed]
- Blau, A.; Murr, A.; Wolff, S.; Sernagor, E.; Medini, P.; Iurilli, G.; Ziegler, C.; Benfenati, F. Flexible, all-polymer microelectrode arrays for the capture of cardiac and neuronal signals. Biomaterials 2011, 32, 1778–1786. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.N.; Wu, R.Z.; Sabatini, D.M. Applications of transfected cell microarrays in high-throughput drug discovery. Drug Discov. Today 2002, 7, S113–S118. [Google Scholar] [CrossRef]
- Chan, C.Y.; Huang, P.-H.; Guo, F.; Ding, X.; Kapur, V.; Mai, J.D.; Yuen, P.K.; Huang, T.J. Accelerating drug discovery via organs-on-chips. Lab Chip 2013, 13, 4697–4710. [Google Scholar] [CrossRef] [PubMed]
- Kapur, R.; Giuliano, K.A.; Campana, M.; Adams, T.; Olson, K.; Jung, D.; Mrksich, M.; Vasudevan, C.; Taylor, D.L. Streamlining the drug discovery process by integrating miniaturization, high throughput screening, high content screening, and automation on the cellchip™ system. Biomed. Microdevices 1999, 2, 99–109. [Google Scholar] [CrossRef]
- Bhise, N.S.; Ribas, J.; Manoharan, V.; Zhang, Y.S.; Polini, A.; Massa, S.; Dokmeci, M.R.; Khademhosseini, A. Organ-on-a-chip platforms for studying drug delivery systems. J. Control. Release 2014, 190, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.B.; Sung, J.H. Organ-on-a-chip technology and microfluidic whole-body models for pharmacokinetic drug toxicity screening. Biotechnol. J. 2013, 8, 1258–1266. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Wu, J.H.; Wang, S.Q.; Durmus, N.G.; Gurkan, U.A.; Demirci, U. Microengineering methods for cell-based microarrays and high-throughput drug-screening applications. Biofabrication 2011, 3, 034101. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.N.; Sabatini, D.M.; Stockwell, B.R. Microarrays of small molecules embedded in biodegradable polymers for use in mammalian cell-based screens. Proc. Natl. Acad. Sci. USA 2004, 101, 16144–16149. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.; Wheeldon, I.; Guo, Y.Q.; Lu, T.L.; Du, Y.N.; Wang, B.; He, J.K.; Hu, Y.Q.; Khademhosseini, A. A sandwiched microarray platform for benchtop cell-based high throughput screening. Biomaterials 2011, 32, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.E.; Kim, J.; Oh, D.Y.; Song, Y.; Lee, S.H.; Min, S.; Kwon, S. One-step pipetting and assembly of encoded chemical-laden microparticles for high-throughput multiplexed bioassays. Nat. Commun. 2014, 5, 3468. [Google Scholar] [CrossRef] [PubMed]
- Delamarche, E.; Bernard, A.; Schmid, H.; Bietsch, A.; Michel, B.; Biebuyck, H. Microfluidic networks for chemical patterning of substrates: Design and application to bioassays. J. Am. Chem. Soc. 1998, 120, 500–508. [Google Scholar] [CrossRef]
- Kim, J.; Taylor, D.; Agrawal, N.; Wang, H.; Kim, H.; Han, A.; Rege, K.; Jayaraman, A. A programmable microfluidic cell array for combinatorial drug screening. Lab Chip 2012, 12, 1813–1822. [Google Scholar] [CrossRef] [PubMed]
- Toh, Y.C.; Lim, T.C.; Tai, D.; Xiao, G.F.; van Noort, D.; Yu, H.R. A microfluidic 3D hepatocyte chip for drug toxicity testing. Lab Chip 2009, 9, 2026–2035. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.B.; Mitchell, R.J.; Kim, B.C. Whole-cell-based biosensors for environmental biomonitoring and application. Adv. Biochem. Eng. 2004, 87, 269–305. [Google Scholar]
- Bhunia, A.K.; Banada, P.; Banerjee, P.; Valadez, A.; Hirleman, E.D. Light Scattering, Fiber Optic- and Cell-Based Sensors for Sensitive Detection of Foodborne Pathogens. J. Rapid Meth. Autom. Microbiol. 2007, 15, 121–145. [Google Scholar] [CrossRef]
- Rawson, D.M.; Willmer, A.J.; Turner, A.P. Whole-cell biosensors for environmental monitoring. Biosensors 1989, 4, 299–311. [Google Scholar] [CrossRef]
- Banerjee, P.; Bhunia, A.K. Mammalian cell-based biosensors for pathogens and toxins. Trends Biotechnol. 2009, 27, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Han, S.W.; Chung, U.S.; Koh, W.-G. Application of cellular micropatterns to miniaturized cell-based biosensor. Biomed. Eng. Lett. 2013, 3, 117–130. [Google Scholar] [CrossRef]
- Liu, Q.J.; Wu, C.S.; Cai, H.; Hu, N.; Zhou, J.; Wang, P. Cell-Based Biosensors and Their Application in Biomedicine. Chem. Rev. 2014, 114, 6423–6461. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.L.; Horsburgh, A.; Paton, G.I.; Killham, K.; Meharg, A.; Primrose, S.; Strachan, N.J. A novel toxicity fingerprinting method for pollutant identification with lux-marked biosensors. Environ. Toxicol. Chem. 2001, 20, 2456–2461. [Google Scholar] [CrossRef] [PubMed]
- Ganjoo, A.; Jain, H.; Yu, C.; Irudayaraj, J.; Pantano, C. Detection and fingerprinting of pathogens: Mid-IR biosensor using amorphous chalcogenide films. J. Non-Cryst. Solids 2008, 354, 2757–2762. [Google Scholar] [CrossRef]
- Rider, T.H.; Petrovick, M.S.; Nargi, F.E.; Harper, J.D.; Schwoebel, E.D.; Mathews, R.H.; Blanchard, D.J.; Bortolin, L.T.; Young, A.M.; Chen, J.Z.; et al. A B cell-based sensor for rapid identification of pathogens. Science 2003, 301, 213–215. [Google Scholar] [CrossRef] [PubMed]
- Moschopoulou, G.; Vitsa, K.; Bem, F.; Vassilakos, N.; Perdikaris, A.; Blouhos, P.; Yialouris, C.; Frosyniotis, D.; Anthopoulos, I.; Mangana, O.; et al. Engineering of the membrane of fibroblast cells with virus-specific antibodies: A novel biosensor tool for virus detection. Biosens. Bioelectron. 2008, 24, 1027–1030. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.E.; Laane, M.M.; Haugarvoll, E.; Giaever, I. Monitoring viral-induced cell death using electric cell-substrate impedance sensing. Biosens. Bioelectron. 2007, 23, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, P.; Lenz, D.; Robinson, J.P.; Rickus, J.L.; Bhunia, A.K. A novel and simple cell-based detection system with a collagen-encapsulated B-lymphocyte cell line as a biosensor for rapid detection of pathogens and toxins. Lab. Investig. 2008, 88, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.J.; Cai, H.; Xu, Y.; Xiao, L.D.; Yang, M.; Wang, P. Detection of heavy metal toxicity using cardiac cell-based biosensor. Biosens. Bioelectron. 2007, 22, 3224–3229. [Google Scholar] [CrossRef] [PubMed]
- Abassi, Y.A.; Xi, B.A.; Li, N.; Ouyang, W.; Seiler, A.; Watzele, M.; Kettenhofen, R.; Bohlen, H.; Ehlich, A.; Kolossov, E.; et al. Dynamic monitoring of beating periodicity of stem cell-derived cardiomyocytes as a predictive tool for preclinical safety assessment. Brit. J. Pharmacol. 2012, 165, 1424–1441. [Google Scholar] [CrossRef] [PubMed]
- Shamah, S.M.; Cunningham, B.T. Label-free cell-based assays using photonic crystal optical biosensors. Analyst 2011, 136, 1090–1102. [Google Scholar] [CrossRef] [PubMed]
- Hogberg, H.T.; Sobanski, T.; Novellino, A.; Whelan, M.; Weiss, D.G.; Bal-Price, A.K. Application of micro-electrode arrays (MEAs) as an emerging technology for developmental neurotoxicity: Evaluation of domoic acid-induced effects in primary cultures of rat cortical neurons. Neurotoxicology 2011, 32, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Wallace, K.; Strickland, J.D.; Valdivia, P.; Mundy, W.R.; Shafer, T.J. A multiplexed assay for determination of neurotoxicant effects on spontaneous network activity and viability from microelectrode arrays. Neurotoxicology 2015, 49, 79–85. [Google Scholar] [CrossRef] [PubMed]


| Advantages | Disadvantages | |||
|---|---|---|---|---|
| 2D | Positional array | Photolithography |
|
|
| Soft lithography |
|
| ||
| DEP |
|
| ||
| Suspension array | Optical-encoding |
|
| |
| Graphical-encoding |
|
| ||
| 3D | Hydrogel-based |
|
| |
| Nanofiber-based |
|
| ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, H.J.; Koom, W.S.; Koh, W.-G. Cell Microarray Technologies for High-Throughput Cell-Based Biosensors. Sensors 2017, 17, 1293. https://doi.org/10.3390/s17061293
Hong HJ, Koom WS, Koh W-G. Cell Microarray Technologies for High-Throughput Cell-Based Biosensors. Sensors. 2017; 17(6):1293. https://doi.org/10.3390/s17061293
Chicago/Turabian StyleHong, Hye Jin, Woong Sub Koom, and Won-Gun Koh. 2017. "Cell Microarray Technologies for High-Throughput Cell-Based Biosensors" Sensors 17, no. 6: 1293. https://doi.org/10.3390/s17061293






