Fabrication and Characterization of Polyaniline/PVA Humidity Microsensors
Abstract
: This study presents the fabrication and characterization of a humidity microsensor that consists of interdigitated electrodes and a sensitive film. The area of the humidity microsensor is about 2 mm2. The sensitive film is polyaniline doping polyvinyl alcohol (PVA) that is prepared by the sol-gel method, and the film has nanofiber and porous structures that help increase the sensing reaction. The commercial 0.35 μm Complimentary Metal Oxide Semiconductor (CMOS) process is used to fabricate the humidity microsensor. The sensor needs a post-CMOS process to etch the sacrificial layer and to coat the sensitive film on the interdigitated electrodes. The sensor produces a change in resistance as the polyaniline/PVA film absorbs or desorbs vapor. Experimental results show that the sensitivity of the humidity sensor is about 12.6 kΩ/%RH at 25 °C.1. Introduction
Humidity sensors are important devices that can be used to measure and monitor environmental humidity. Recently, various microsensors have been manufactured using microelectromechanical system (MEMS) technology [1–4]. Humidity sensors fabricated by this technology have the benefits of small size, low cost, high performance and easy mass-production [5,6]. For instance, Li et al. [7] presented a resistive humidity sensor fabricated by the multi-user MEMS process. Organic conductive polymer—poly(3,4-ethylenedioxythiophene) synthesized by a electrochemical deposition was adopted as the sensitive material of the sensor, and the sensitive material filled a narrow air gap between two nickel electrodes. The resistance of the sensor changed from 37 to 62 Ω as relative humidity increased from 22 to 99.9 %RH at room temperature. A resistive humidity sensor, proposed by Chen et al. [8], contained a freestanding cantilever that was a composite structure comprising a layer of platinum deposited on a silicon nitride layer and covered with a polyimide sensing layer. The cantilever generated a deflection as the polyimide sensing layer absorbed vapor, resulting in the platinum layer produced a change in resistance. It was showed experimentally that the resistance of the sensor decreased from 210 to 70 kΩ as relative humidity increased from 40 to 85 %RH at 40 °C. Kang and Wise [9] developed a capacitive humidity microsensor integrated with a polysilicon heater. Polyimide was the sensitive material of the sensor. The polysilicon heater was used to reduce the recovery time after wetting. The humidity sensor contained thousands of polyimide columns, which all connected in parallel by the suspended upper electrode to form a capacitor, and the columns were located on top of lower electrode. The sensor exhibited a sensitivity of 30 fF/%RH. Su et al. [10] utilized MEMS and thick-film technologies to fabricate a resistive humidity microsensor. The sensor consisted of a suspended planar membrane bridged to the silicon substrate with two beams. The sensitive material was a composite film of poly-[3-(methacryloylamino)propyl]trimethyl ammonium chloride and SiO2 that was coated on the electrodes. The conductivity of the composite film changed upon adsorbing or desorbing. A capacitive humidity microsensor was presented by Kim et al. [11], and the sensor was composed of a substrate with a cavity, a bottom electrode, a polyimide sensing layer and a comb-shaped top electrode. In order to enhance the performance of the sensor, the polyimide sensing layer was etched by an O2 plasma asher in accordance with the top electrode passivation. The humidity sensor had a sensitivity of 506 fF/%RH. Lee et al. [12] made a capacitive humidity microsensor with a micro-bridge structure using front-side etching with XeF2 gas. The sensitive film of the sensor was polyimide, and the film locally cured at a temperature over 350 °C for 1 h by the MEMS microhotplate. The sensitivity of the humidity sensor was 0.77 pF/%RH, and its hysteresis was about 0.61 %RH. Sensitivity is an important characteristic for humidity sensors. Polyaniline/PVA is a material that is highly sensitive to water vapor. Thereby, this study employed polyaniline/PVA to develop a humidity microsensor, and its sensitivity exceeds that of Li et al. [7] and Chen et al. [8].
Fabrication of MEMS devices using the commercial CMOS process is called the CMOS-MEMS technique [13–15]. This technique has a potential to integrate MEMS devices with circuitry on-a-chip. Micro devices manufactured by the CMOS-MEMS technique usually need a post-CMOS process to coat the functional films [16] or to release the suspended structures [17,18]. In this work, the fabrication of a humidity microsensor using the CMOS-MEMS technique was investigated. The sensor in this case required a post-process to coat a sensitive film of polyaniline/PVA. The post-process utilizes a wet etching to etch the sacrificial layer and the polyaniline/PVA is coated on the humidity sensor. The sensor is a resistive type, and it produces a change in resistance when the sensitive film absorbs or desorbs vapor.
2. Preparation of the Humidity Sensor
In our humidity sensor, polyaniline/PVA prepared by sol-gel method was adopted as the humidity sensitive material [19,20]. Preparation steps for the polyaniline/PVA included: (1) aniline (C6H5NH2, 0.68 mL) was dissolved in HCl (105 mL) with vigorous stirring to form a homogenous solution; (2) ammonium persulfate (N2H8S2O8, 1.67 g) was dissolved in HCl (40 mL) and added to the aniline solution with stirring for 8 h at room temperature; (3) the mixed N2H8S2O8/C6H5NH2/HCl solution was aged at room temperature for 120 h, producing a blackish green sediment; (4) PVA (100 mg) was dissolved in dimethyl sulfoxide (DMSO, 20 mL) with stirring at 80 °C until the mixed solution became homogenous; (5) the PVA/DMSO solution was added to the C6H5NH2/N2H8S2O8/HCl residue and stirred at room temperature until a homogeneous solution was obtained; (6) the resulting product was filtered, followed by storage in air at 80 °C for 1 h.
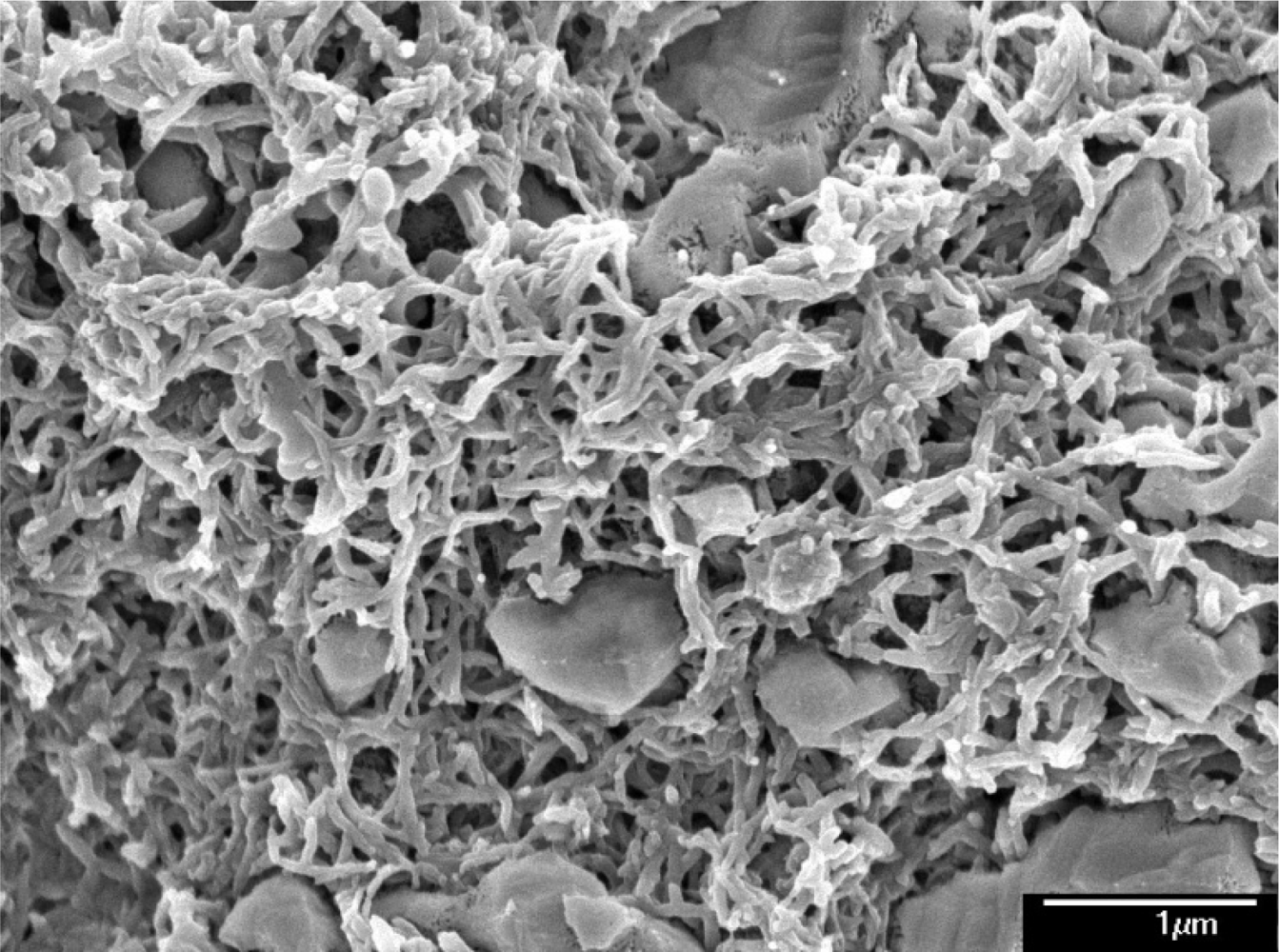
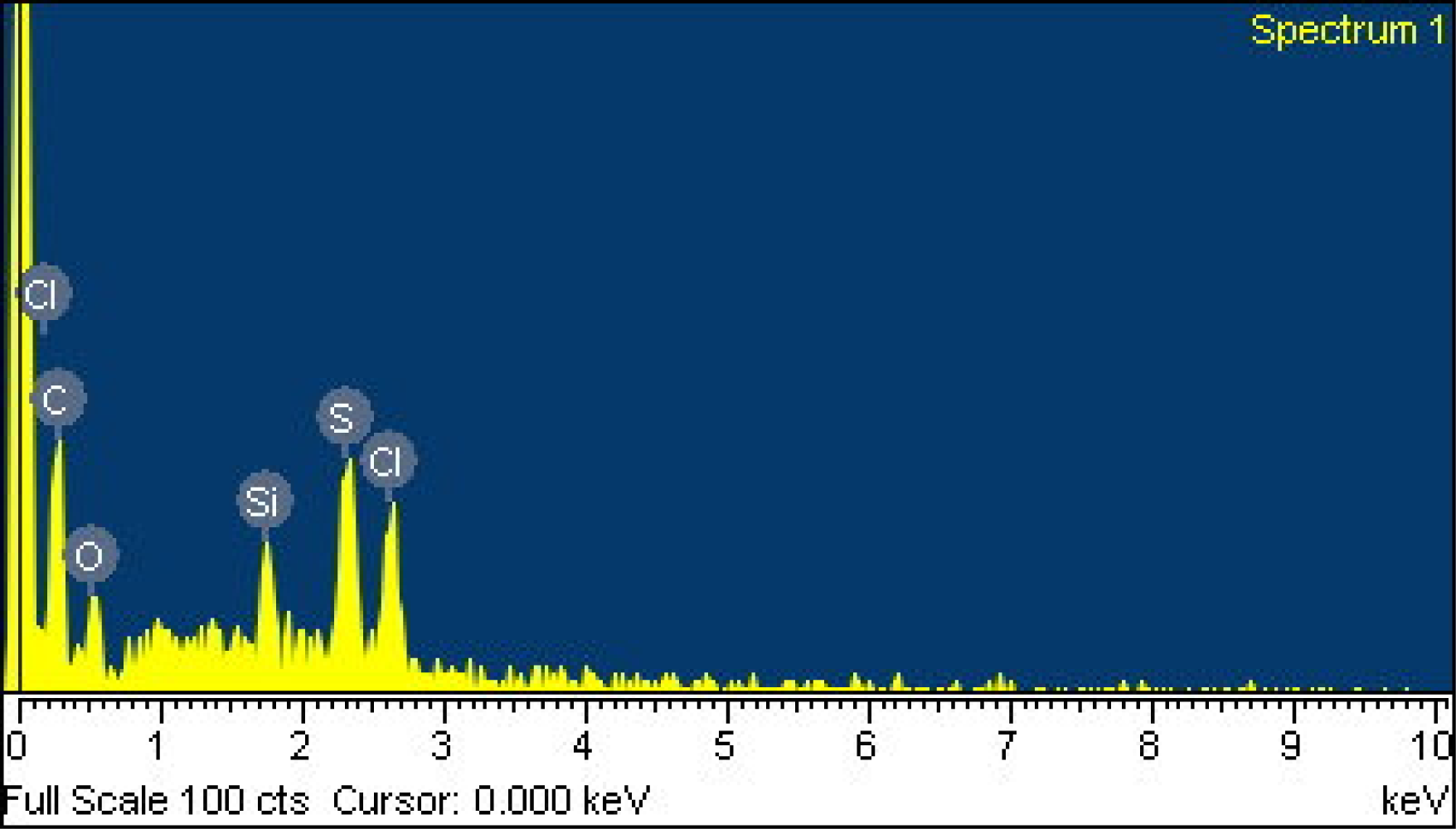
Scanning electron microscopy (SEM, JEOL JSM-6700F) was employed to measure the surface morphology of the polyaniline/PVA film. Figure 1 presents a scanning electron microscopy image of the polyaniline/PVA film. The sensitive film exhibits micro-porous and nanofiber structures that help increase the sensing reaction because the film has a large surface area. An energy dispersive spectrometer (OXFORD INCA ENERGY 400) was used to detect elements of the polyaniline/PVA. Figure 2 shows the elements of the polyaniline/PVA film measured by energy dispersive spectrometer. Table 1 summarizes composition of the polyaniline/PVA film. The results showed that the polyaniline/PVA film contained 63.68 wt% C, 15.87 wt% O, 8.58 wt% S, 8.06 wt% Cl and 3.80 wt% Si. The Si, Cl and S elements resulted from substrate, DMSO and HCl, respectively.
3. Fabrication of the Humidity Sensor
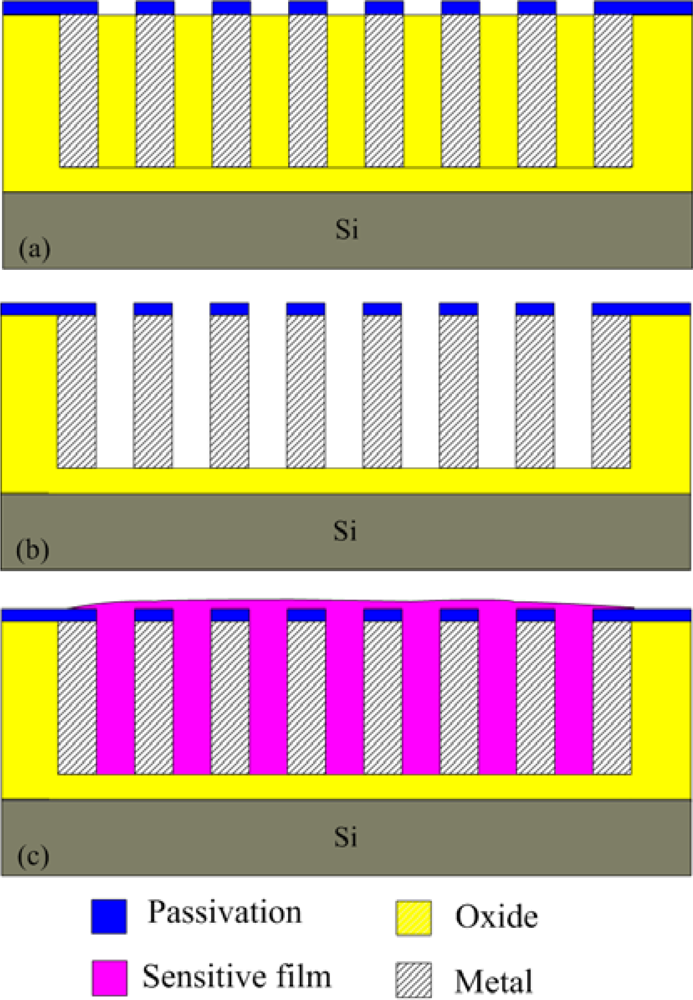
Figure 3 illustrates the schematic structure of the humidity microsensor. The humidity sensor is constructed of interdigitated electrodes and a sensitive film. The sensitive film of the sensor is polyaniline/PVA, and the film is located on the interdigitated electrodes. The material of the interdigitated electrodes is the stacked metals of the CMOS process. Length, thickness and width of the interdigital electrodes are about 960 μm, 7 μm and 5 μm, respectively, and the gap between the electrodes is 5 μm. The humidity sensor is a resistive type. The resistance variation of the sensor depends on its sensitive film. The humidity sensor generates a change in resistance as the polyaniline/PVA film absorbs or desorbs vapor.
The humidity microsensor was fabricated using the commercial 0.35 μm CMOS process of Taiwan Semiconductor Manufacturing Company (TSMC). Figure 4 illustrates the fabrication flow of the humidity microsensor. Figure 4(a) shows the humidity microsensor after completion of the CMOS process. The metal layers were used as the interdigitated electrodes. The sacrificial layer was silicon dioxide that was located between the interdigitated electrodes. In order to coat the sensitive film between the electrodes, the sacrificial oxide layer must be removed. The sensor needed a post-process to etch the sacrificial layer and to coat the polyaniline/PVA film. Figure 4(b) displays that the sensor is immersed in Silox to etch the silicon dioxide and to obtain the interdigital electrode gap [21]. Figure 5 shows an SEM image of the humidity microsensor after the wet etching process. Figure 6 presents an SEM image of the interdigitated electrodes for the sensor. Figure 4(c) shows polyaniline/PVA to be dropped using a precision-control micro-dropper. Finally, the polyaniline/PVA was kept in air at 80 °C for 1 h.
4. Results and Discussion
A test chamber (GTH-099-40-1P, Giant Force Instruments Enterprise Co.) and an LCR meter were used to measure the performance of the humidity microsensor. The test chamber was able to provide a humidity range of 25–90 %RH and a temperature range of 0–100 °C. Humidity and temperature in the test chamber could be tuned separately and maintained at constant levels. An LCR meter was adopted to measure the resistance of the humidity sensor.
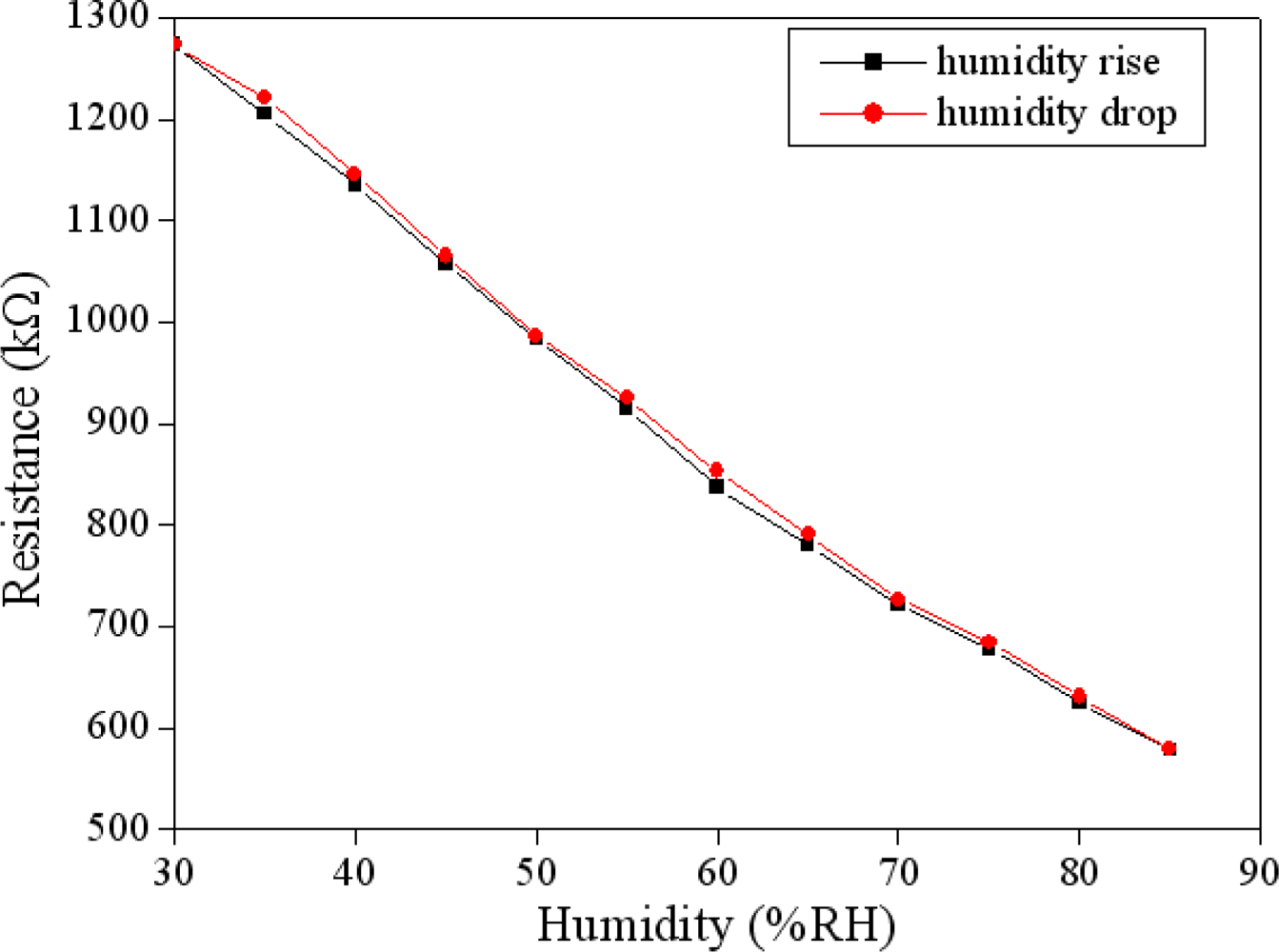
The humidity sensor was set in the test chamber. The test chamber provided different humidity to the humidity sensor. The resistance of the humidity sensor changed as humidity in the test chamber rose or dropped. The resistance variation of the sensor was recorded by the LCR meter. Figure 7 shows the measured results of the humidity sensor. In this measurement, the temperature kept constant at 25 °C and the humidity increased from 25 %RH to 85 %RH in 35 min and then dehumidified to 25 %RH at the same rate. The experimental results showed that the humidity sensor almost had no humidity hysteresis. As shown in Figure 7, the resistance of the sensor decreased from 1,285 to 590 kΩ as humidity increased from 25 to 85 %RH, and the sensitivity of the sensor was about 12.6 kΩ/%RH.
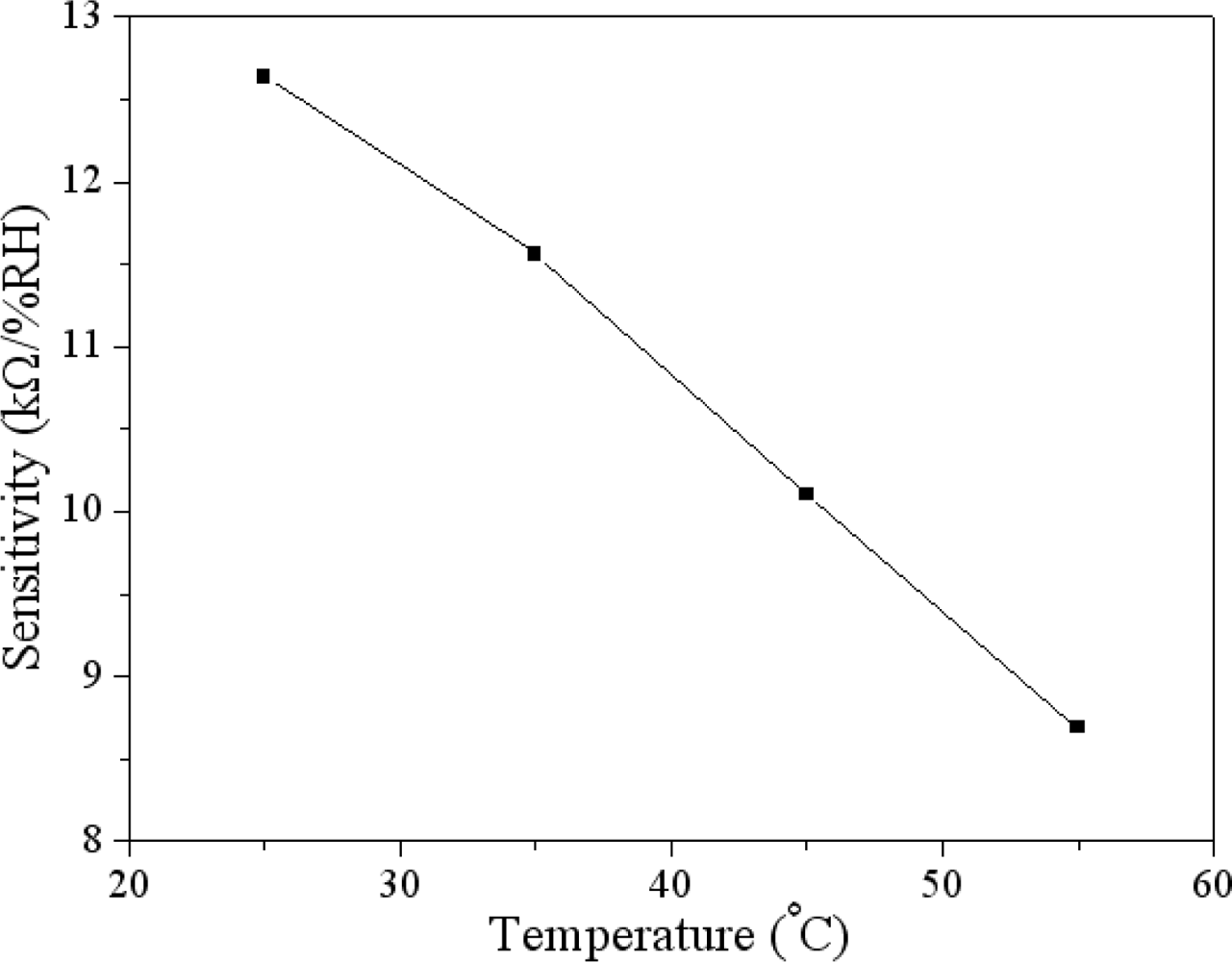
The humidity sensor was tested under different temperatures in order to characterize the influence of temperature by recording the resistance variation of the sensor. Figure 8 shows the measured resistance of the humidity sensor at different temperatures. The curves in Figure 8 are linear in the range of 30–75 %RH. The sensitivity of the sensor can be obtained by the linear fitting to the data in Figure 8. The evaluated results showed that the sensor had a sensitivity of 12.6 kΩ/%RH at 25 °C and a sensitivity of 8.7 kΩ/%RH at 55 °C. In accordance with the results in Figure 8, the relation between the humidity sensitivity and temperature can be obtained, and is shown in Figure 9. The results revealed that the sensitivity of the sensor decreased as the temperature increased, and the sensor had a high sensitivity at room temperature.
Li et al. [7] used organic conductive polymer—poly(3,4-ethylenedioxythiophene) to manufacture a resistive humidity microsensor, and its resistance changed from 37 to 62 Ω as relative humidity increased from 22 to 99.9 %RH at room temperature, which the sensitivity of the sensor was about 0.32 Ω/%RH. Chen et al. [8] developed a resistive humidity microsensor that the sensitive material was polyimide, and the resistance of the sensor decreased from 210 to 70 kΩ as relative humidity increased from 40 to 85 %RH. The sensor had a sensitivity of about 3.1 kΩ/%RH. This work adopted polyaniline/PVA as a humidity sensitive material, and the sensitivity of the sensor was about 12.6 kΩ/%RH. A comparison with Li et al. [7] and Chen et al. [8], indicates that the sensitivity of this work exceeds that of Li et al. [7] and Chen et al. [8].
5. Conclusions
A humidity microsensor has been manufactured using the commercial 0.35 μm CMOS process and an appropriate post-process. The humidity sensor had the advantages of small area and high sensitivity. The sensor was composed of interdigitated electrodes and a sensing film. The sensing film was polyaniline/PVA that was synthesized by the sol-gel method, and the film had nanofiber and porous structures that helps to increase the sensor’s sensitivity. The interdigitated electrodes were constructed by the stacked metals of the CMOS process. The post-process used a wet etching to etch the sacrificial oxide layer, and then the polyaniline/PVA was coated on the interdigitated electrodes. The humidity sensor was tested under different temperatures. The results showed that the sensitivity of the sensor decreased as the temperature rose, and the sensor had a high sensitivity (12.6 kΩ/%RH) at room temperature (25 °C).
Acknowledgments
The authors would like to thank National Center for High-performance Computing (NCHC) for chip simulation, National Chip Implementation Center (CIC) for chip fabrication and the National Science Council of the Republic of China for financially supporting this research under Contract No NSC 99-2221-E-005-082-MY2.
References
- Zribi, A; Knobloch, A; Tian, WC; Goodwin, S. Micromachined resonant multiple gas sensor. Sens. Actuat. A 2005, 122, 31–38. [Google Scholar]
- Hautefeuille, M; O’Flynn, B; Peters, F; O’Mahony, C. Miniaturised multi-MEMS sensor development. Microelectron. Reliab 2009, 49, 621–626. [Google Scholar]
- Dai, CL; Tai, YW; Kao, PH. Modeling and fabrication of micro FET pressure sensor with circuits. Sensors 2007, 7, 3386–3398. [Google Scholar]
- Liu, MC; Dai, CL; Chan, CH; Wu, CC. Manufacture of a polyaniline nanofiber ammonia sensor integrated with a readout circuit using the CMOS-MEMS technique. Sensors 2009, 9, 869–880. [Google Scholar]
- Gardner, JW; Varadan, VK; Awadelkarim, OO. Microsensors MEMS and Smart Devices; John Wiley & Sons: Chichester, UK, 2001. [Google Scholar]
- Fang, Z; Zhao, Z; Wu, Y; Zhang, B; Wang, YZ. Integrated temperature and humidity sensor based MEMS. Proceedings of International Conference on Information Acquisition, Hefei, China, 21–25 June 2004; pp. 84–87.
- Li, L; Vilela, F; Forgie, J; Skabara, PJ; Uttamchandani, D. Miniature humidity micro-sensor based on organic conductive polymer—poly(3,4-ethylenedioxythiophene). Micro Nano Lett 2009, 4, 84–87. [Google Scholar]
- Chen, LT; Lee, CY; Cheng, WH. MEMS-based humidity sensor with integrated temperature compensation mechanism. Sens. Actuat. A 2008, 147, 522–528. [Google Scholar]
- Kang, U; Wise, KD. High-speed capacitive humidity sensor with on-chip thermal reset. IEEE Trans. Electron. Devices 2000, 47, 702–710. [Google Scholar]
- Su, PG; Ho, CJ; Sun, YL; Chen, IC. A micromachined resistive-type humidity sensor with a composite material as sensitive film. Sens. Actuat. B 2006, 113, 837–842. [Google Scholar]
- Kim, JH; Hong, SM; Moon, BM; Kim, K. High-performance capacitive humidity sensor with novel electrode and polyimide layer based on MEMS technology. Microsyst. Technol 2010, 16, 2017–2021. [Google Scholar]
- Lee, MJ; Min, NK; Yoo, KP; Kwak, KY; Kwon, KH. Microhotplate-based high-speed polyimide capacitive humidity sensors. Sens. Lett 2009, 7, 517–522. [Google Scholar]
- Dai, CL; Chiou, JH; Lu, SC. A maskless post-CMOS bulk micromachining process and its application. J. Micromech. Microeng 2005, 15, 2366–2371. [Google Scholar]
- Dai, CL; Xiao, FY; Juang, YZ; Chiu, CF. An approach to fabricating microstructures that incorporate circuits using a post-CMOS process. J. Micromech. Microeng 2005, 15, 98–103. [Google Scholar]
- Fedder, GK; Howe, RT; Liu, TJK; Quévy, EP. Technologies for cofabricating MEMS and electronics. Proc. IEEE 2008, 96, 306–322. [Google Scholar]
- Yang, MZ; Dai, CL; Lu, DH. Polypyrrole porous micro humidity sensor integrated with a ring oscillator circuit on chip. Sensors 2010, 10, 10095–10104. [Google Scholar]
- Dai, CL; Chen, YL. Modeling and manufacturing of micromechanical RF switch with inductors. Sensors 2007, 7, 2660–2670. [Google Scholar]
- Dai, CL; Tsai, CH. Fabrication of integrated chip with microinductors and micro-tunable capacitors by complementary metal-oxide-semiconductor postprocess. Jpn. J. Appl. Phys 2005, 44, 2030–2036. [Google Scholar]
- Huang, J. Syntheses and applications of conducting polymer polyaniline nanofibers. Pure Appl. Chem 2006, 78, 15–27. [Google Scholar]
- Xing, S; Zhao, C; Jing, S; Wang, Z. Morphology and conductivity of polyaniline nanofibers prepared by ‘seeding’ polymerization. Polymer 2006, 47, 2305–2313. [Google Scholar]
- Dai, CL. A maskless wet etching silicon dioxide post-CMOS process and its application. Microelectron. Eng 2006, 83, 2543–2550. [Google Scholar]




| Elements | wt% |
|---|---|
| C | 63.68 |
| O | 15.87 |
| S | 8.58 |
| Cl | 8.06 |
| Si | 3.80 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, M.-Z.; Dai, C.-L.; Lin, W.-Y. Fabrication and Characterization of Polyaniline/PVA Humidity Microsensors. Sensors 2011, 11, 8143-8151. https://doi.org/10.3390/s110808143
Yang M-Z, Dai C-L, Lin W-Y. Fabrication and Characterization of Polyaniline/PVA Humidity Microsensors. Sensors. 2011; 11(8):8143-8151. https://doi.org/10.3390/s110808143
Chicago/Turabian StyleYang, Ming-Zhi, Ching-Liang Dai, and Wei-Yi Lin. 2011. "Fabrication and Characterization of Polyaniline/PVA Humidity Microsensors" Sensors 11, no. 8: 8143-8151. https://doi.org/10.3390/s110808143




