Characteristics of Ambient Volatile Organic Compounds (VOCs) Measured in Shanghai, China
Abstract
:1. Introduction
2. Experimental Method
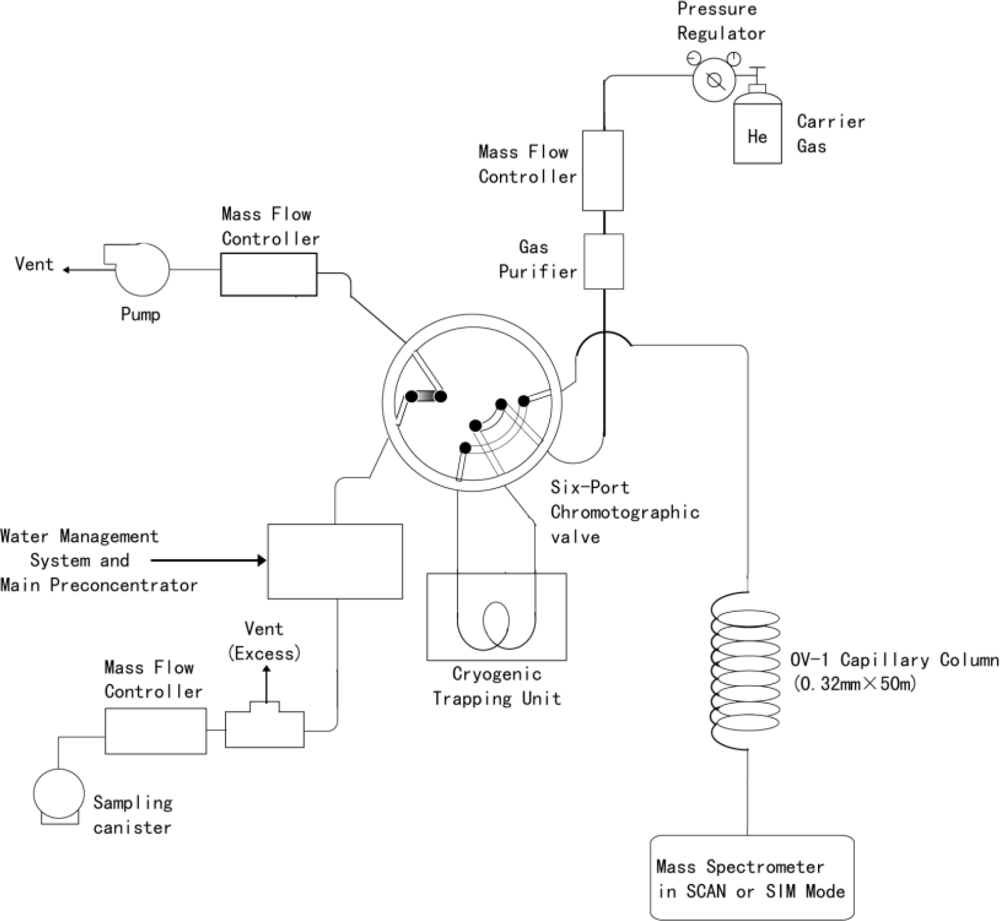
2.1. GC/MSD Measurement System
2.2. Calibration
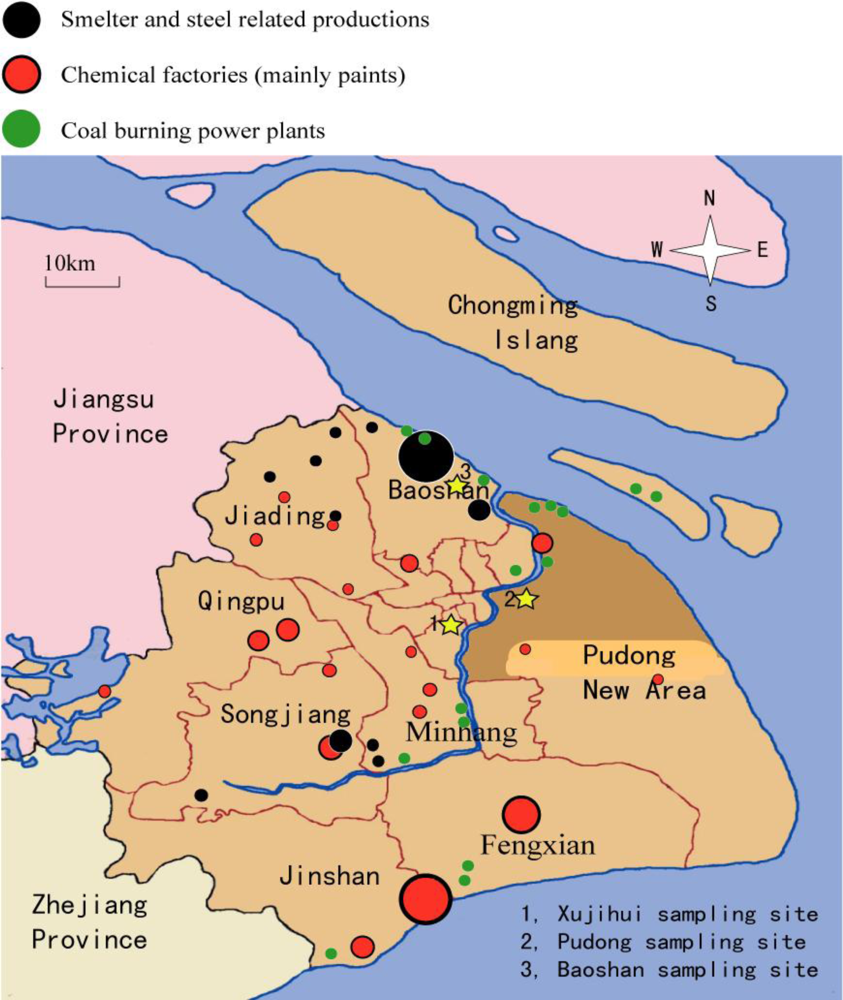
2.3. Sampling Site
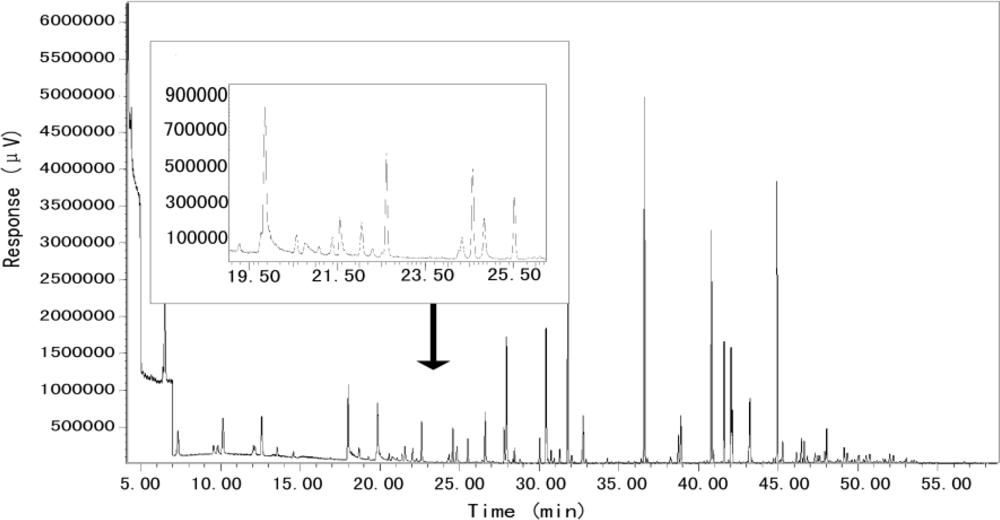
2.4. Analysis Method
3. Results and Discussion
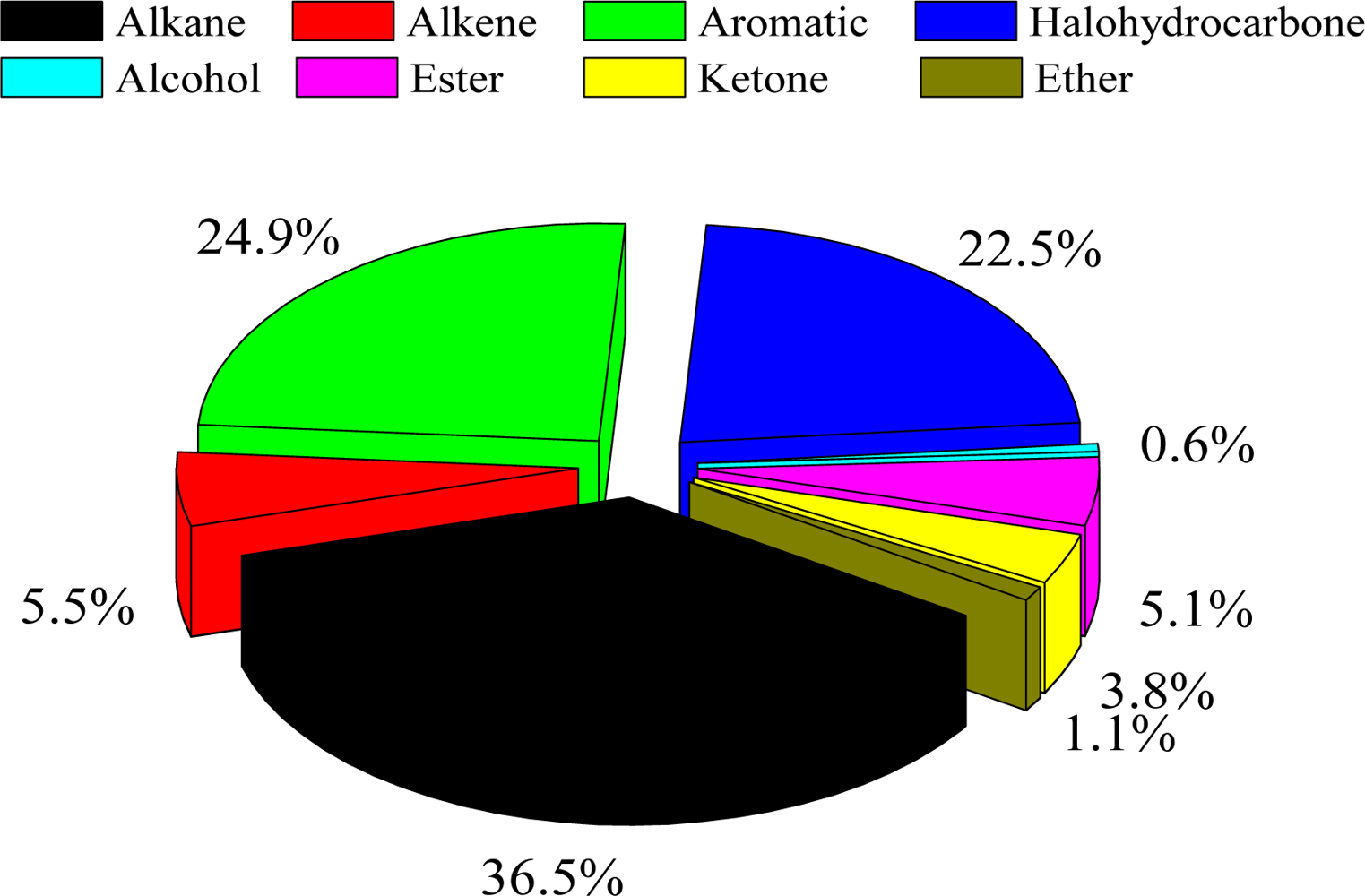
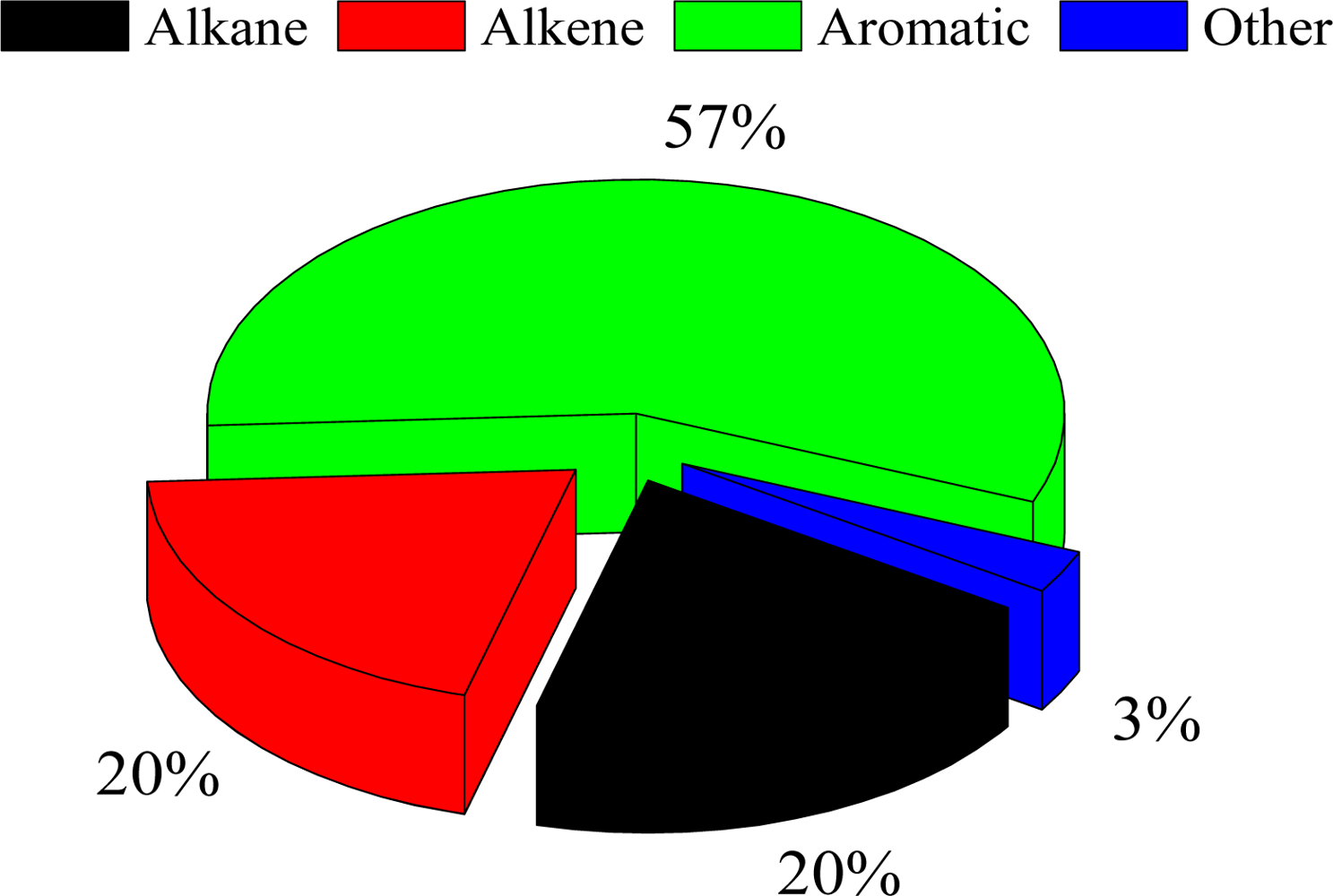
3.1. Characteristics of Ambient VOC Concentrations in Shanghai
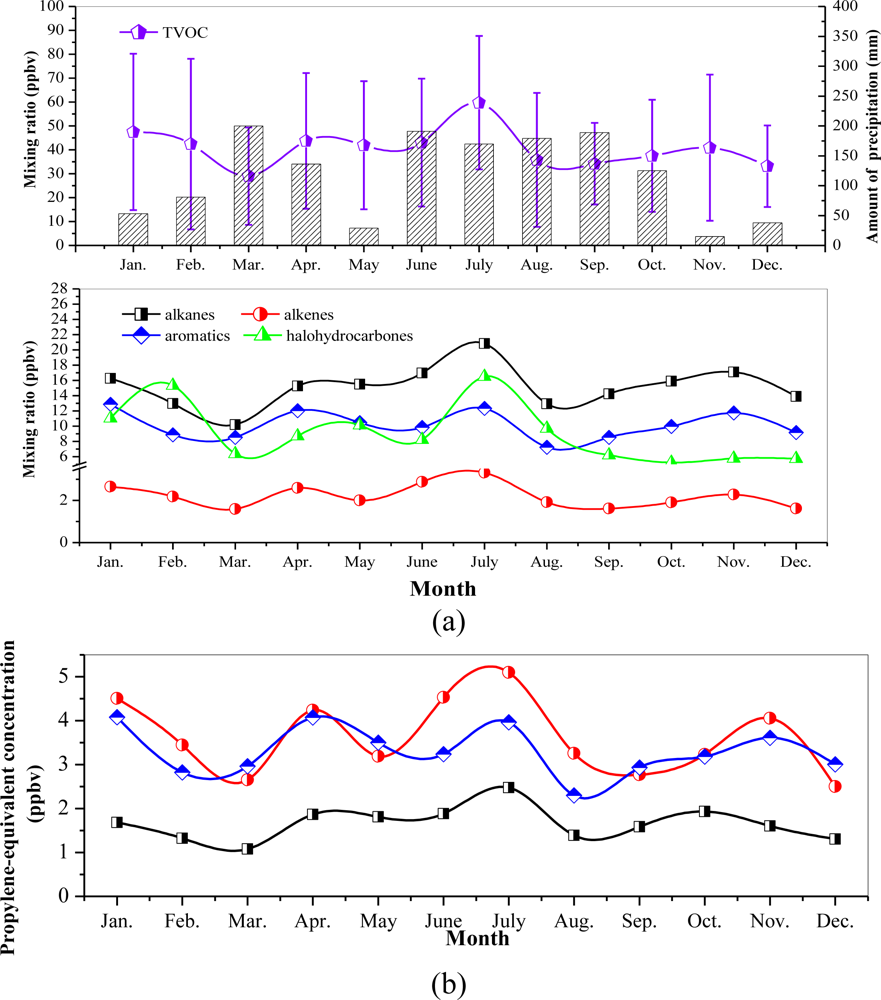
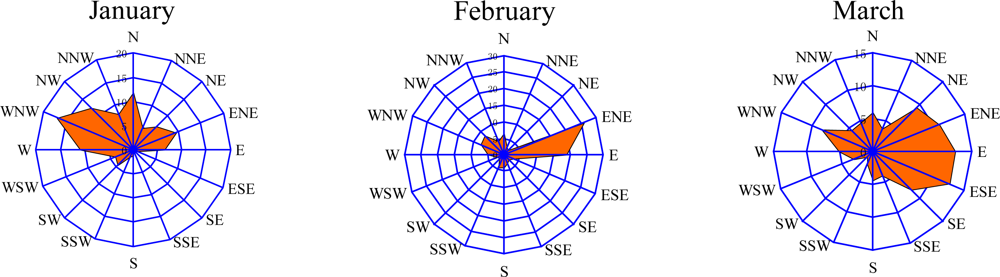
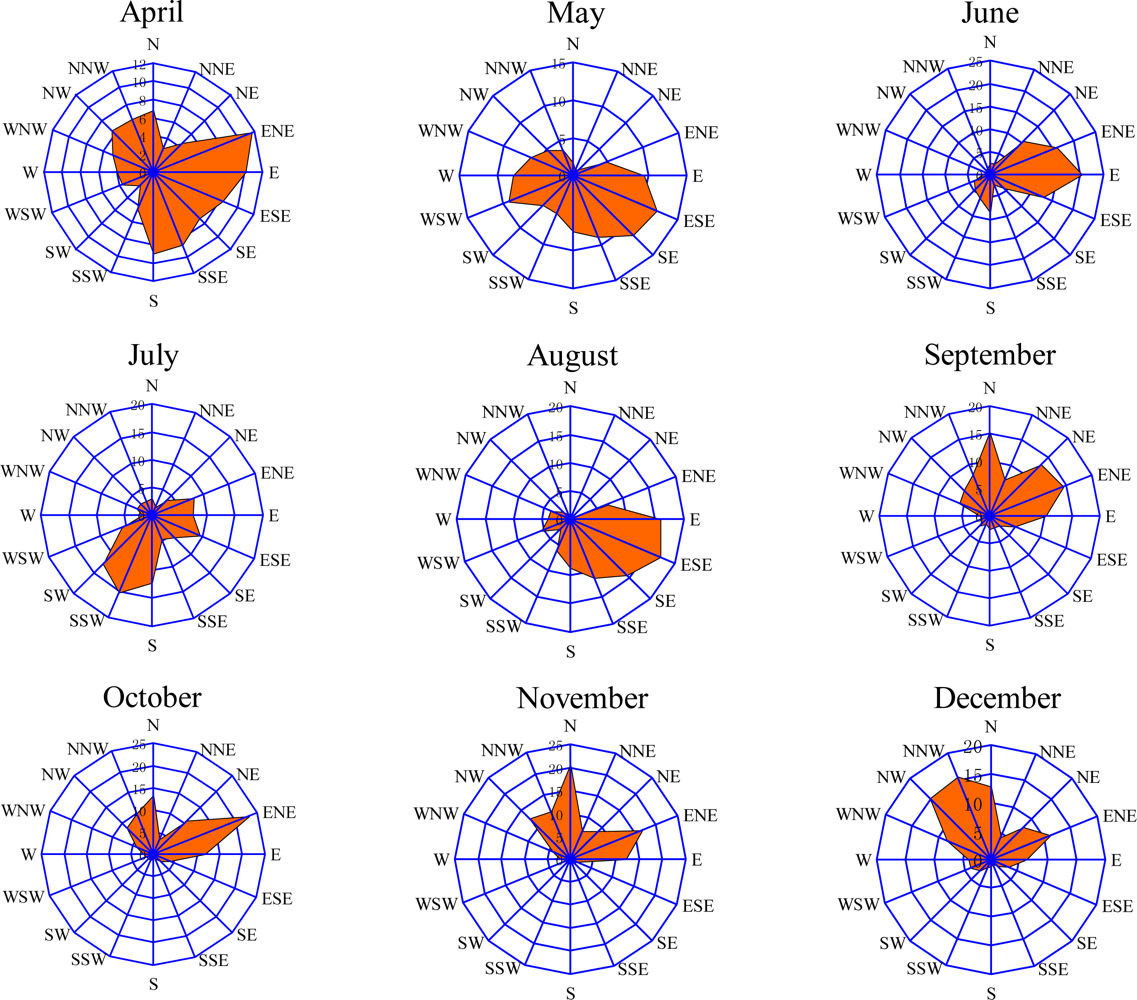
3.2. Seasonal Variations and Ozone Formation Potential
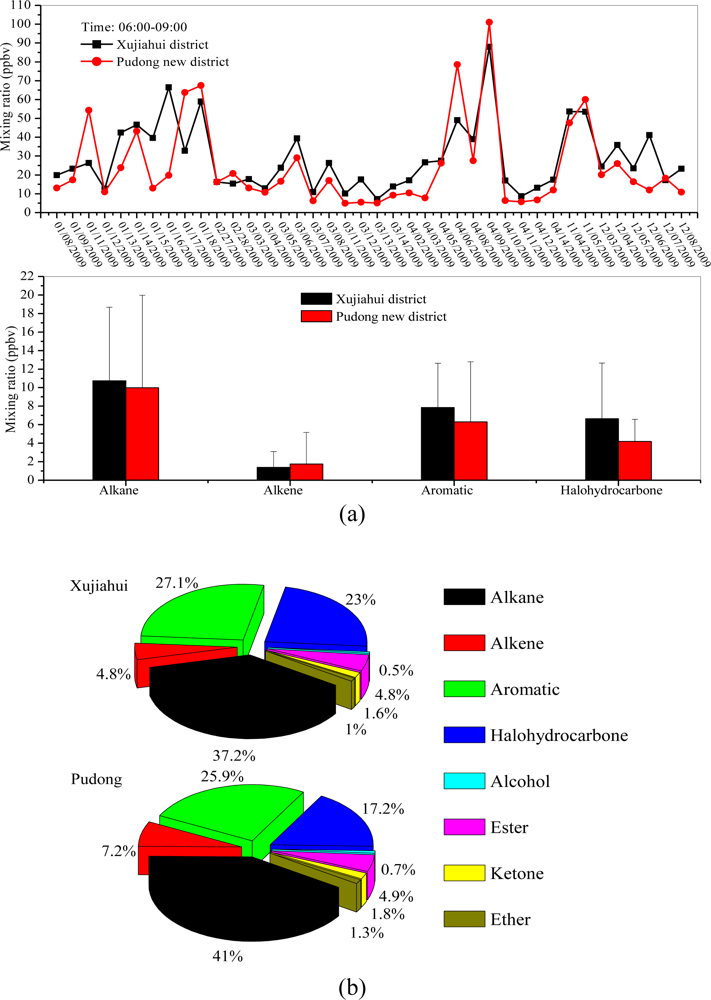
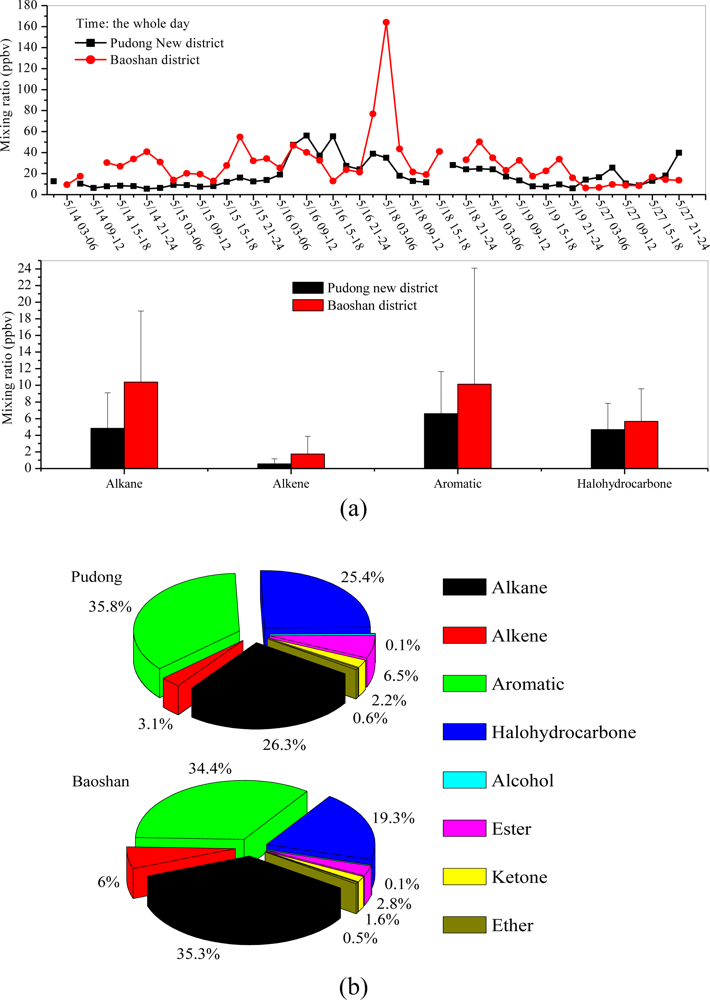
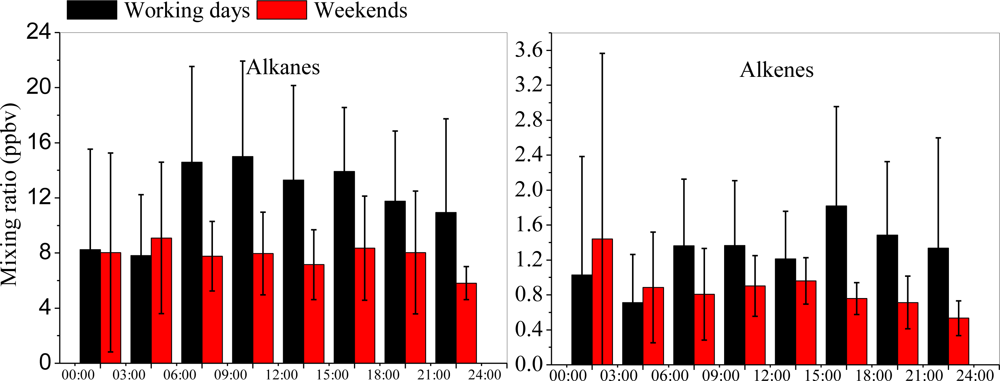
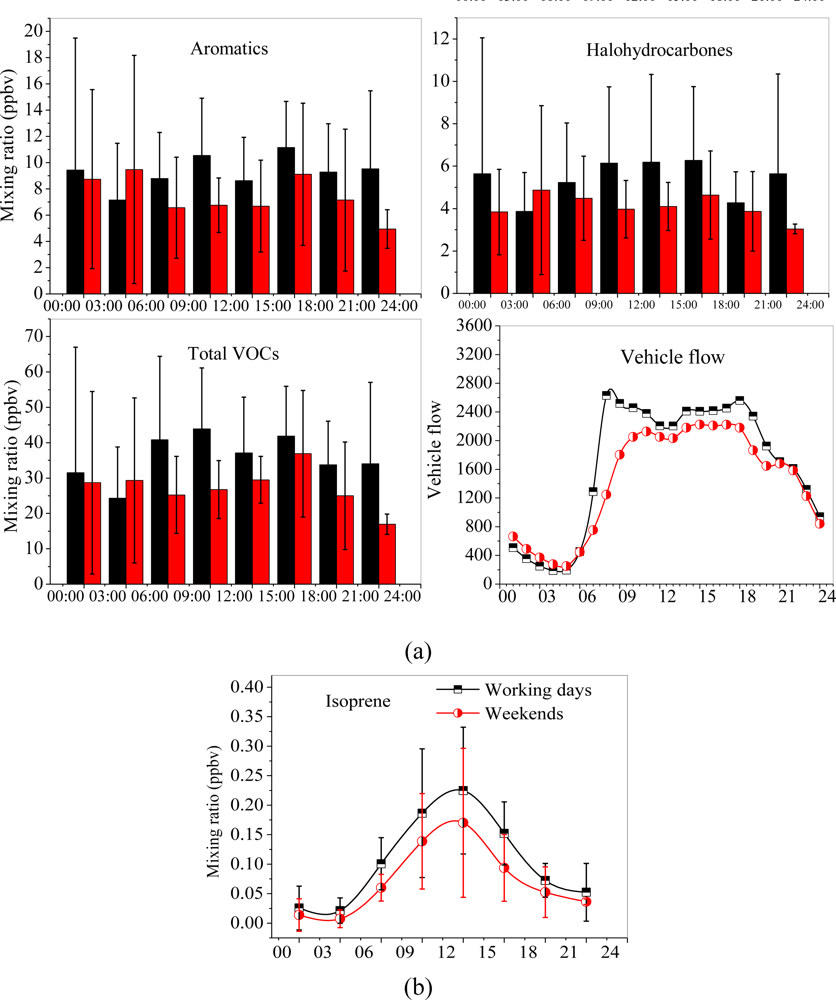
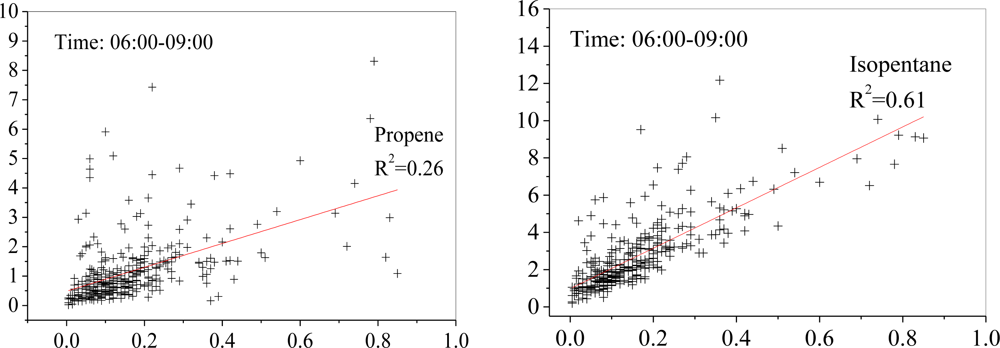
3.3. Diurnal Variations and Weekend Effect
4. Conclusions
Acknowledgments
References
- Shanghai Municipal Statistics Bureau (SMSB). Shanghai Statistical Yearbook; China Statistical Press: Shanghai, China, 1997. [Google Scholar]
- Shanghai Municipal Statistics Bureau (SMSB). Shanghai Statistical Yearbook; China Statistical Press: Shanghai, China, 2009. [Google Scholar]
- Tie, X; Brasseur, C; Zhao, C; Granier, S; Massie, Y; Qin, PC; Wang, GL; Wang, PC; Yang, PC. Chemical characterization of air pollution in Eastern China and the Eastern United States. Atmos. Environ 2006, 40, 2607–2625. [Google Scholar]
- Streets, DG; Yu, C; Wu, Y; Chin, M; Zhao, Z; Hayasaka, T; Shi, G. Aerosol trends over China. Atmos. Res 2008, 88, 174–182. [Google Scholar]
- Geng, F; Zhao, C; Tang, X; Lu, GL; Tie, X. Analysis of ozone and VOCs measured in Shanghai: A case study. Atmos. Environ 2007, 41, 989–1001. [Google Scholar]
- Geng, F; Tie, X; Xu, J; Zhou, G; Peng, L; Gao, W; Tang, X; Zhao, C. Characterization of O3, NOx and VOCs measured in Shanghai, China. Atmos. Environ 2008, 42, 6873–6883. [Google Scholar]
- Tie, X; Geng, F; Peng, L; Gao, W; Zhao, C. Measurement and modeling of O3 variability in Shanghai, China: Application of the WRF-Chem model. Atmos. Environ 2009, 43, 4289–4302. [Google Scholar]
- Geng, F; Zhang, Q; Tie, X; Huang, M; Ma, X; Deng, Z; Yu, Q; Quan, J; Zhao, C. Aircraft measurements of O3, NOx, CO, VOCs, and SO2 in the Yangtze River Delta region. Atmos. Environ 2009, 43, 584–593. [Google Scholar]
- Geng, F; Cai, C; Tie, X; Yu, Q; An, J; Peng, L; Zhou, G; Xu, J. Analysis of VOC emissions using PCA/APCS receptor model at city of Shanghai, China. J Atmos Chem 2010. [Google Scholar] [CrossRef]
- Tang, WY; Zhao, C; Geng, F; Tie, X. Study of ozone “weekend effect” in Shanghai. Sci. China 2008, 51, 1354–1360. [Google Scholar]
- Chameides, WL; Fehsenfeld, F; Rodgers, MO; Cardelino, C; Martines, J; Parrish, D; Lonneman, W; Lawson, DR; Rasmussen, RA; Zimmerman, P; Greenberg, J; Middleton, P; Wang, T. Ozone precursor relationships in the ambient atmosphere. J. Geophys. Res 1992, 97, 6037–6055. [Google Scholar]
- Ran, L; Zhao, C; Geng, F; Tie, X; Tang, X; Peng, L; Zhou, G; Yu, Q; Xu, J; Guenther, A. Ozone photochemical production in urban Shanghai, China: Analysis based on ground level observations. J Geophys Res 2009. [Google Scholar] [CrossRef]
- Tie, X; Madronich, S; Li, G; Ying, Z; Weinheimer, A; Apel, E; Campos, T. Simulation of Mexico city plumes during the MIRAGE-Mex field campaign using the WRE-Chem model. Atmos. Chem. Phys 2009, 9, 4621–4638. [Google Scholar]
- Carter, WPL. Development of ozone reactivity scales for volatile organic compounds. J. Air Waste Manag. Assoc 1994, 44, 881–899. [Google Scholar]
- Atkinson, R; Arey, J. Atmospheric degradation of volatile organic compounds. Chem. Rev 2003, 103, 4605–4638. [Google Scholar]
- Liu, Y; Shao, M; Fu, L; Lu, S; Zeng, L; Tang, D. Source profiles of volatile organic compounds (VOCs) measured in China: Part I. Atmos. Environ 2008, 42, 6247–6260. [Google Scholar]
- Song, Y; Dai, W; Shao, M; Liu, Y; Lu, SH; Kuster, W; Goldan, P. Comparison of receptor models for source apportionment of volatile organic compounds in Beijing, China. Environ. Pollution 2008, 156, 174–183. [Google Scholar]
- Tang, JH; Chan, LY; Chan, CY; Li, YS; Chang, CC; Wang, XM; Zou, SC; Barletta, B; Blake, DR; Wu, D. Implication of changing urban and rural emissions on non-methane hydrocarbons in the Pearl River Delta region of China. Atmos. Environ 2008, 42, 3780–3794. [Google Scholar]
- Guo, H; Wang, T; Louie, PKK. Source apportionment of ambient non-methane hydrocarbons in Hong Kong: Application of a principal component analysis/absolute principal component scores (PCA/APCS) receptor model. Atmos. Environ 2004, 129, 489–498. [Google Scholar]
- Nguyen, HT; Kim, KH; Kim, MY. Volatile organic compounds at an urban monitoring station in Korea. J. Hazard. Mat 2009, 161, 163–174. [Google Scholar]
- Saito, S; Nagao, I; Kanzawa, H. Characteristics of ambient C2-C11 non-methane hydrocarbons in metropolitan Nagoya, Japan. Atmos. Environ 2009, 43, 4384–4395. [Google Scholar]
- Lu, S; Bai, Y; Zhang, G; Li, T. Source apportionment of anthropogenic emissions of volatile organic compounds. Acta Scient. Circumstant 2006, 26, 757–763. [Google Scholar]
- Guenther, A; Zimmerman, P; Harley, P; Monson, R; Fall, R. Isoprene and monoterpene emission rate variability: model evaluation and sensitivity analysis. J. Geophys. Res 1993, 98, 12609–12617. [Google Scholar]
- Tie, X; Li, G; Ying, Z; Guenther, A; Madronich, S. Biogenic emissions of VOCs and NO in China and comparison to anthropogenic emissions. Sci. Total Environ 2006, 371, 238–251. [Google Scholar]
| Number | Name | Chemical group | *Correlation coefficient | *DL (ng) | Mixing ratio (ppbv) | |
|---|---|---|---|---|---|---|
| Mean ± S.D. | Rang | |||||
| 1 | Propene | Alkene | 0.999440 | 0.33 | 0.96 ± 1.03 | 0.00–8.31 |
| 2 | 1-Butene | Alkene | 0.999550 | 0.38 | 0.27 ± 0.27 | 0.00–2.30 |
| 3 | 1,3-Butadiene | Alkene | 0.998762 | 0.26 | 0.16 ± 0.45 | 0.00–8.32 |
| 4 | cis-2-Butene | Alkene | 0.999607 | 0.44 | 0.22 ± 0.25 | 0.00–1.75 |
| 5 | trans-2-Butene | Alkene | 0.999834 | 0.38 | 0.24 ± 0.29 | 0.00–2.08 |
| 6 | 1-Pentene | Alkene | 0.999619 | 0.71 | 0.14 ± 0.11 | 0.00–0.70 |
| 7 | Isoprene | Alkene | 0.999373 | 0.18 | 0.13 ± 0.14 | 0.00–1.00 |
| 8 | 2-Pentene | Alkene | 0.999566 | 0.23 | 0.12 ± 0.14 | 0.00–1.69 |
| 9 | 1-Hexene | Alkene | 0.999467 | 0.66 | 0.03 ± 0.07 | 0.00–0.80 |
| 10 | Propane | Alkane | 0.999509 | 0.20 | 4.56 ± 2.71 | 0.00–21.88 |
| 11 | Isobutane | Alkane | 0.999797 | 0.27 | 1.41 ± 1.09 | 0.07–6.74 |
| 12 | Butane | Alkane | 0.999780 | 0.17 | 2.08 ± 1.40 | 0.23–9.28 |
| 13 | Isopentane | Alkane | 0.999637 | 0.56 | 2.36 ± 1.77 | 0.13–12.17 |
| 14 | 2,2-Dimethylbutane | Alkane | 0.999736 | 0.45 | 0.03 ± 0.06 | 0.00–0.95 |
| 15 | Cyclopentane | Alkane | 0.998352 | 0.34 | 0.10 ± 0.17 | 0.00–3.27 |
| 16 | 2,3-Dimethylbutane | Alkane | 0.999659 | 0.39 | 0.14 ± 0.18 | 0.00–2.48 |
| 17 | 2-Methylpentane | Alkane | 0.999641 | 0.48 | 0.80 ± 1.18 | 0.04–15.57 |
| 18 | 3-Methylpentane | Alkane | 0.999741 | 0.48 | 0.60 ± 1.12 | 0.02–15.57 |
| 19 | n-Hexane | Alkane | 0.999956 | 0.76 | 0.97 ± 1.66 | 0.03–24.25 |
| 20 | 2,4-Dimethylpentane | Alkane | 0.999749 | 0.42 | 0.23 ± 0.27 | 0.01–3.50 |
| 21 | Methylcyclopentane | Alkane | 0.999516 | 0.38 | 0.28 ± 0.34 | 0.00–4.72 |
| 22 | Cyclohexane | Alkane | 0.999238 | 0.41 | 0.11 ± 0.11 | 0.00–0.93 |
| 23 | 2-Methylhexane | Alkane | 0.997083 | 0.42 | 0.20 ± 0.20 | 0.00–1.92 |
| 24 | 2,3-Dimethylpentane | Alkane | 0.999302 | 0.49 | 0.09 ± 0.09 | 0.00–0.69 |
| 25 | 3-Methylhexane | Alkane | 0.998441 | 0.46 | 0.21 ± 0.22 | 0.00–1.86 |
| 26 | 2,2,4-Trimethylpentane | Alkane | 0.997326 | 0.63 | 0.03 ± 0.06 | 0.00–0.96 |
| 27 | n-Heptane | Alkane | 0.994699 | 0.59 | 0.22 ± 0.20 | 0.00–1.42 |
| 28 | Methylcyclohexane | Alkane | 0.999567 | 0.29 | 0.09 ± 0.08 | 0.00–0.79 |
| 29 | 2,3,4-Trimethylpentane | Alkane | 0.997527 | 0.30 | 0.01 ± 0.03 | 0.00–0.71 |
| 30 | 2-Methylheptane | Alkane | 0.997880 | 0.34 | 0.05 ± 0.04 | 0.00–0.30 |
| 31 | 3-Methylheptane | Alkane | 0.995449 | 0.37 | 0.08 ± 0.08 | 0.00–0.60 |
| 32 | n-Octane | Alkane | 0.996870 | 0.34 | 0.10 ± 0.23 | 0.00–4.75 |
| 33 | Nonane | Alkane | 0.985404 | 0.54 | 0.10 ± 0.10 | 0.00–1.08 |
| 34 | n-Decane | Alkane | 0.994657 | 0.83 | 0.10 ± 0.10 | 0.00–2.49 |
| 35 | n-Dodecane | Alkane | 0.995704 | 2.11 | 0.09 ± 0.14 | 0.00–1.11 |
| 36 | n-Undecane | Alkane | 0.999883 | 2.09 | 0.09 ± 0.18 | 0.00–3.53 |
| 37 | Styrene | Aromatic | 0.999182 | 0.65 | 0.15 ± 0.19 | 0.00–2.86 |
| 38 | Benzene | Aromatic | 0.999608 | 0.46 | 1.76 ± 1.38 | 0.29–12.56 |
| 39 | Toluene | Aromatic | 0.998413 | 0.57 | 4.62 ± 4.52 | 0.35–42.35 |
| 40 | Ethylbenzene | Aromatic | 0.998121 | 0.69 | 1.29 ± 1.20 | 0.06–9.14 |
| 41 | m-Xylene | Aromatic | 0.995785 | 0.62 | 1.36 ± 1.21 | 0.11–8.79 |
| 42 | p-Xylene | Aromatic | 0.66 | |||
| 43 | o-Xylene | Aromatic | 0.993700 | 0.66 | 0.55 ± 0.64 | 0.05–8.36 |
| 44 | Isopropylbenzene | Aromatic | 0.999219 | 0.35 | 0.08 ± 0.25 | 0.00–4.45 |
| 45 | n-Propylbenzene | Aromatic | 0.999922 | 0.31 | 0.07 ± 0.07 | 0.00–0.83 |
| 46 | 1,3,5-Trimethylbenzene | Aromatic | 0.995870 | 0.86 | 0.06 ± 0.06 | 0.00–0.91 |
| 47 | m-Ethyltoluene | Aromatic | 0.999164 | 1.35 | 0.19 ± 0.19 | 0.02–1.19 |
| 48 | p-Ethyltoluene | Aromatic | 0.995702 | 0.77 | 0.11 ± 0.08 | 0.01–0.74 |
| 49 | o-Ethyltoluene | Aromatic | 0.999347 | 0.89 | 0.06 ± 0.05 | 0.00–0.84 |
| 50 | m-Diethylbenzene | Aromatic | 0.999796 | 0.96 | 0.02 ± 0.03 | 0.00–0.43 |
| 51 | Vinyl chloride | Halohydrocabone | 0.996365 | 0.39 | 1.07 ± 3.51 | 0.00–33.83 |
| 52 | 1,1-Dichloroethene | Halohydrocabone | 0.999456 | 0.54 | 0.01 ± 0.06 | 0.00–1.12 |
| 53 | Allyl chloride | Halohydrocabone | 0.999718 | 0.25 | 0.03 ± 0.10 | 0.00–2.11 |
| 54 | trans-1,2-Dichloroethene | Halohydrocabone | 0.999731 | 0.57 | 0.01 ± 0.03 | 0.00–0.58 |
| 55 | cis-1,2-Dichloroethene | Halohydrocabone | 0.999986 | 0.54 | 0.00 ± 0.03 | 0.00–0.52 |
| 56 | cis-1,3-Dichloropropene | Halohydrocabone | 0.999962 | 0.51 | 0.00 ± 0.03 | 0.00–0.46 |
| 57 | trans-1,3-Dichloropropene | Halohydrocabone | 0.999383 | 0.62 | 0.08 ± 0.09 | 0.00–0.76 |
| 58 | Tetrachloroethylene | Halohydrocabone | 0.999676 | 0.87 | 0.19 ± 1.44 | 0.00–30.95 |
| 59 | Hexachloro-1,3-butadiene | Halohydrocabone | 0.999645 | 2.04 | 0.08 ± 0.16 | 0.00–0.88 |
| 60 | Trichloroethylene | Halohydrocabone | 0.999693 | 0.69 | 0.15 ± 0.31 | 0.00–3.65 |
| 61 | Freon-12 | Halohydrocabone | 0.999827 | 0.63 | 0.58 ± 0.13 | 0.00–1.95 |
| 62 | Chloromethane | Halohydrocabone | 0.995610 | 0.38 | 1.76 ± 2.64 | 0.14–23.31 |
| 63 | Freon-114 | Halohydrocabone | 0.997958 | 1.06 | 0.00 ± 0.00 | 0.00–0.02 |
| 64 | Bromomethane | Halohydrocabone | 0.998258 | 0.47 | 1.26 ± 4.41 | 0.00–17.95 |
| 65 | Chloroethane | Halohydrocabone | 0.998785 | 0.38 | 0.05 ± 0.10 | 0.00–1.95 |
| 66 | Freon-11 | Halohydrocabone | 0.999825 | 0.81 | 0.31 ± 0.10 | 0.00–2.00 |
| 67 | Freon-113 | Halohydrocabone | 0.997887 | 1.59 | 0.08 ± 0.03 | 0.00–0.44 |
| 68 | Methylene chloride | Halohydrocabone | 0.999810 | 0.67 | 0.95 ± 0.96 | 0.00–13.93 |
| 69 | 1,1-Dichloroethane | Halohydrocabone | 0.999859 | 0.52 | 0.12 ± 0.58 | 0.00–8.92 |
| 70 | Chloroform | Halohydrocabone | 0.999752 | 0.62 | 0.15 ± 0.18 | 0.00–1.69 |
| 71 | 1,2-Dichloroethane | Halohydrocabone | 0.999767 | 0.55 | 1.53 ± 2.25 | 0.08–23.02 |
| 72 | Carbon tetrachloride | Halohydrocabone | 0.999621 | 0.75 | 0.22 ± 0.94 | 0.00–20.45 |
| 73 | 1,2-Dichloropropane | Halohydrocabone | 0.999582 | 0.55 | 0.18 ± 0.71 | 0.00–11.07 |
| 74 | Bromodichloromethane | Halohydrocabone | 0.999257 | 0.86 | 0.01 ± 0.01 | 0.00–0.03 |
| 75 | 1,1,2-Trichloroethane | Halohydrocabone | 0.999925 | 0.74 | 0.04 ± 0.05 | 0.00–0.55 |
| 76 | Dibromochloromethane | Halohydrocabone | 0.999857 | 1.02 | 0.09 ± 0.91 | 0.00–19.62 |
| 77 | 1,2-Dibromoethane | Halohydrocabone | 0.999775 | 0.98 | 0.00 ± 0.01 | 0.00–0.05 |
| 78 | Bromoform | Halohydrocabone | 0.999508 | 1.32 | 0.01 ± 0.02 | 0.00–0.09 |
| 79 | 1,1,1-Trichloroethane | Halohydrocabone | 0.999943 | 0.57 | 0.02 ± 0.02 | 0.00–0.15 |
| 80 | Tetrachloroethane | Halohydrocabone | 0.999898 | 1.10 | 0.01 ± 0.03 | 0.00–0.20 |
| 81 | Chlorobenzene | Halohydrocabone | 0.999950 | 0.66 | 0.07 ± 0.08 | 0.00–0.54 |
| 82 | 1,3-Dichlorobenzene | Halohydrocabone | 0.998193 | 0.82 | 0.14 ± 0.18 | 0.00–1.19 |
| 83 | Benzyl chloride | Halohydrocabone | 0.999574 | 0.66 | 0.03 ± 0.03 | 0.00–0.14 |
| 84 | 1,2-Dichlorobenzene | Halohydrocabone | 0.999291 | 0.96 | 0.04 ± 0.07 | 0.00–0.50 |
| 85 | 1,2,4-Trichlorobenzene | Halohydrocabone | 0.997771 | 1.90 | 0.06 ± 0.13 | 0.00–0.67 |
| 86 | Isopropyl alcohol | Alcohol | 0.999995 | 0.63 | 0.27 ± 1.08 | 0.00–14.32 |
| 87 | Vinyl acetate | Ester | 0.998669 | 0.48 | 0.12 ± 0.28 | 0.00–2.83 |
| 88 | Ethyl acetate | Ester | 0.998476 | 0.83 | 2.01 ± 2.59 | 0.00–15.48 |
| 89 | 2-Butanone | Ketone | 0.999973 | 2.05 | 1.45 ± 1.80 | 0.00–12.42 |
| 90 | Methyl isobutyl ketone | Ketone | 0.998836 | 0.79 | 0.10 ± 0.12 | 0.00–0.83 |
| 91 | Methyl butyl ketone | Ketone | 0.995532 | 0.72 | 0.02 ± 0.02 | 0.00–0.14 |
| 92 | Methyl tert-butyl ether | Ether | 0.995313 | 0.46 | 0.33 ± 0.33 | 0.00–2.39 |
| 93 | Tetrahydrofuran | Ether | 0.999881 | 0.40 | 0.12 ± 0.14 | 0.00–1.25 |
| TVOCs | 41.43 ± 30.13 | 7.12–210.87 | ||||
| Compounds | Xujiahui site (40 samples) | Pudong site (40 samples) | ||||
|---|---|---|---|---|---|---|
| Mean ± S.D. | Median | Range | Mean ± S.D. | Median | Range | |
| Propene | 0.66 ± 0.67 | 0.40 | 0.04–3.14 | 0.65 ± 1.17 | 0.36 | 0.00–7.23 |
| 1-Butene | 0.14 ± 0.16 | 0.07 | 0.00–0.60 | 0.19 ± 0.33 | 0.08 | 0.00–1.42 |
| cis-2-Butene | 0.12 ± 0.16 | 0.06 | 0.00–0.95 | 0.20 ± 0.36 | 0.06 | 0.00–1.63 |
| trans-2-Butene | 0.15 ± 0.24 | 0.07 | 0.00–1.42 | 0.21 ± 0.40 | 0.05 | 0.00–1.72 |
| 1-Pentene | 0.07 ± 0.08 | 0.05 | 0.00–0.41 | 0.07 ± 0.11 | 0.03 | 0.00–0.48 |
| 2-Pentene | 0.06 ± 0.09 | 0.03 | 0.00–0.50 | 0.09 ± 0.16 | 0.02 | 0.00–0.76 |
| Isoprene | 0.05 ± 0.13 | 0.02 | 0.00–0.82 | 0.03 ± 0.05 | 0.00 | 0.00–0.18 |
| Propane | 3.98 ± 2.41 | 3.75 | 1.05–14.67 | 3.07 ± 2.76 | 2.67 | 0.07–13.49 |
| Isobutane | 1.01 ± 0.86 | 0.84 | 0.07–4.46 | 0.95 ± 1.15 | 0.60 | 0.00–4.36 |
| Butane | 1.62 ± 1.26 | 1.35 | 0.23–7.30 | 1.43 ± 1.36 | 1.04 | 0.08–5.00 |
| Isopentane | 1.53 ± 1.13 | 1.22 | 0.13–5.70 | 1.90 ± 2.31 | 1.10 | 0.17–9.23 |
| 2-Methyl pentane | 0.43 ± 0.45 | 0.29 | 0.07–2.40 | 0.46 ± 0.62 | 0.21 | 0.02–3.25 |
| 3-Methyl pentane | 0.33 ± 0.42 | 0.19 | 0.04–2.39 | 0.35 ± 0.62 | 0.13 | 0.02–3.60 |
| n-Hexane | 0.58 ± 0.85 | 0.30 | 0.07–5.09 | 0.64 ± 1.42 | 0.22 | 0.00–7.98 |
| Styrene | 0.12 ± 0.12 | 0.08 | 0.03–0.73 | 0.12 ± 0.19 | 0.06 | 0.00–1.15 |
| Benzene | 1.50 ± 0.99 | 1.22 | 0.40–5.03 | 1.13 ± 0.89 | 0.87 | 0.20–4.19 |
| Toluene | 3.21 ± 2.29 | 2.76 | 0.63–10.15 | 2.64 ± 3.03 | 1.48 | 0.26–11.22 |
| Ethylbenzene | 0.89 ± 0.89 | 0.71 | 0.15–4.97 | 0.64–0.79 | 0.29 | 0.08–3.04 |
| m/p-Xylene | 1.32 ± 1.05 | 0.96 | 0.13–4.43 | 1.08 ± 1.30 | 0.53 | 0.12–5.30 |
| o-Xylene | 0.36 ± 0.24 | 0.27 | 0.08–0.95 | 0.29 ± 0.33 | 0.16 | 0.06 ± 1.37 |
| Chloromethane | 1.62 ± 2.89 | 0.92 | 0.14–18.39 | 1.02 ± 0.80 | 0.84 | 0.19–4.13 |
| Freon-12 | 0.56 ± 0.14 | 0.59 | 0.00–0.74 | 0.52 ± 0.16 | 0.58 | 0.02–0.91 |
| Freon-11 | 0.28 ± 0.05 | 0.28 | 0.00–0.36 | 0.27 ± 0.04 | 0.28 | 0.04–0.33 |
| Chloroform | 0.12 ± 0.13 | 0.09 | 0.00–0.77 | 0.09 ± 0.07 | 0.07 | 0.03–0.39 |
| 1,2-Dichloroethane | 1.51 ± 1.80 | 0.83 | 0.13–9.39 | 0.67 ± 0.79 | 0.36 | 0.10–0.45 |
| Carbon tetrachloride | 0.20 ± 0.17 | 0.13 | 0.00–0.85 | 0.11 ± 0.02 | 0.11 | 0.02–0.18 |
| Ethyl acetate | 1.32 ± 1.49 | 0.92 | 0.04–7.38 | 1.13 ± 1.80 | 0.38 | 0.02–7.49 |
| 2-Butanone | 0.41 ± 0.54 | 0.13 | 0.00–2.34 | 0.40 ± 0.60 | 0.09 | 0.00–2.74 |
| Methyl tert-butyl ether | 0.22 ± 0.39 | 0.11 | 0.02–2.39 | 0.24 ± 0.42 | 0.06 | 0.00–1.96 |
| Total VOCs | 28.90 ± 17.83 | 23.66 | 7.14–87.96 | 24.35 ± 22.68 | 16.49 | 4.98–100.97 |
| Compounds | Baoshan site (45 samples) | Pudong site (46 samples) | ||||
|---|---|---|---|---|---|---|
| Mean ± S.D. | Median | Range | Mean ± S.D. | Median | Range | |
| Propene | 0.99 ± 1.31 | 0.50 | 0.03–5.86 | 0.31 ± 0.38 | 0.13 | 0.03–1.97 |
| 1-Butene | 0.19 ± 0.23 | 0.11 | 0.00–1.12 | 0.07 ± 0.09 | 0.03 | 0.00–0.35 |
| cis-2-Butene | 0.13 ± 0.23 | 0.05 | 0.00–0.98 | 0.03 ± 0.05 | 0.00 | 0.00–0.24 |
| trans-2-Butene | 0.14 ± 0.24 | 0.03 | 0.00–0.95 | 0.02 ± 0.04 | 0.00 | 0.00–0.23 |
| 1-Pentene | 0.06 ± 0.06 | 0.04 | 0.00–0.25 | 0.03 ± 0.03 | 0.02 | 0.00–0.10 |
| 2-Pentene | 0.02 ± 0.04 | 0.01 | 0.00–0.22 | 0.01 ± 0.01 | 0.00 | 0.00–0.04 |
| Isoprene | 0.06 ± 0.06 | 0.05 | 0.00–0.22 | 0.07 ± 0.09 | 0.04 | 0.00–0.42 |
| Propane | 4.21 ± 4.46 | 3.12 | 0.65–29.95 | 1.63 ± 1.43 | 1.04 | 0.12–5.62 |
| Isobutane | 1.00 ± 0.93 | 0.76 | 0.05–3.97 | 0.46 ± 0.56 | 0.24 | 0.00–2.14 |
| Butane | 1.74 ± 1.63 | 1.35 | 0.14–8.16 | 0.66 ± 0.68 | 0.37 | 0.06–2.72 |
| Isopentane | 1.30 ± 1.05 | 1.03 | 0.10–4.52 | 0.74 ± 0.65 | 0.47 | 0.10–2.81 |
| 2-Methyl pentane | 0.34 ± 0.31 | 0.27 | 0.01–1.50 | 0.21 ± 0.22 | 0.13 | 0.04–1.05 |
| 3-Methyl pentane | 0.25 ± 0.29 | 0.16 | 0.01–1.52 | 0.15 ± 0.21 | 0.09 | 0.02–1.25 |
| n-Hexane | 0.54 ± 0.67 | 0.34 | 0.02–3.28 | 0.31 ± 0.49 | 0.15 | 0.03–2.90 |
| Styrene | 0.10 ± 0.14 | 0.05 | 0.01–0.72 | 0.09 ± 0.13 | 0.05 | 0.02–0.88 |
| Benzene | 1.16 ± 1.03 | 0.82 | 0.15–4.65 | 0.77 ± 0.73 | 0.52 | 0.17–3.17 |
| Toluene | 5.83 ± 12.15 | 1.98 | 0.25–76.25 | 2.71 ± 2.43 | 1.94 | 0.43–10.15 |
| Ethylbenzene | 0.96 ± 0.67 | 0.81 | 0.14–2.68 | 0.98 ± 1.06 | 0.72 | 0.16–6.70 |
| m/p-Xylene | 1.29 ± 0.89 | 1.10 | 0.18–3.57 | 1.37 ± 1.58 | 0.94 | 0.19–9.38 |
| o-Xylene | 0.34 ± 0.25 | 0.29 | 0.06–1.40 | 0.35 ± 0.37 | 0.24 | 0.07–2.05 |
| Chloromethane | 1.29 ± 0.77 | 1.07 | 0.66–4.40 | 1.21 ± 1.26 | 0.85 | 0.26–7.68 |
| Freon-12 | 0.74 ± 0.23 | 0.68 | 0.23–1.51 | 0.61 ± 0.15 | 0.62 | 0.15–0.90 |
| Freon-11 | 0.30 ± 0.05 | 0.30 | 0.05–0.46 | 0.31 ± 0.06 | 0.31 | 0.06–0.51 |
| Chloroform | 0.14 ± 0.13 | 0.07 | 0.03–0.58 | 0.11 ± 0.11 | 0.05 | 0.03–0.55 |
| 1,2-Dichloroethane | 1.15 ± 2.18 | 0.85 | 0.14–15.03 | 0.71 ± 0.70 | 0.49 | 0.11–3.41 |
| Carbon tetrachloride | 0.12 ± 0.02 | 0.12 | 0.02–0.20 | 0.12 ± 0.02 | 0.12 | 0.02–0.19 |
| Ethyl acetate | 0.78 ± 0.58 | 0.75 | 0.02–2.51 | 1.17 ± 1.16 | 0.77 | 0.13–4.38 |
| 2-Butanone | 0.42 ± 0.35 | 0.36 | 0.01–1.51 | 0.35 ± 0.45 | 0.22 | 0.01–1.94 |
| Methyl tert-butyl ether | 0.11 ± 0.10 | 0.08 | 0.00–0.43 | 0.08 ± 0.09 | 0.05 | 0.00–0.32 |
| Total VOCs | 28.78 ± 25.14 | 23.42 | 6.44–164.04 | 18.40 ± 13.03 | 13.28 | 5.58–56.22 |
| VOC species | Shanghai (XJH) China | Beijing China | Guangzhou China | Hong Kong (CW) China | Seoul Korea | Nagoya Japan |
|---|---|---|---|---|---|---|
| This work | [17] | [18] | [19] | [20] | [21] | |
| Propene | 0.84 | 1.97 | 1.79 | 1.31 | 2.12 | 0.705 |
| 1-Butene | 0.26 | 2.21 | 0.60 | - | 0.22 | 0.214 |
| cis-2-Butene | 0.22 | 0.75 | 0.43 | - | 0.22 | 0.141 |
| trans-2-Butene | 0.24 | 0.86 | 0.54 | - | 0.36 | 0.143 |
| 1-Pentene | 0.13 | 0.34 | 0.33 | 0.52 | 0.11 | 0.147 |
| Isoprene | 0.12 | 1.12 | 0.26 | 0.86 | 0.34 | 0.656 |
| Propane | 4.81 | 6.24 | 5.39 | 3.57 | 9.57 | 3.339 |
| Isobutane | 1.43 | 5.36 | 3.11 | 2.75 | 3.16 | 1.404 |
| Butane | 2.03 | 6.36 | 4.60 | 5.55 | 5.37 | 2.661 |
| Isopentane | 2.29 | 11.84 | 3.00 | 3.05 | 2.24 | 1.331 |
| 2-Methyl pentane | 0.67 | - | 1.28 | - | 0.14 | 0.371 |
| 3-Methyl pentane | 0.48 | - | 1.02 | - | 0.13 | 0.290 |
| n-Hexane | 0.84 | 2.22 | 1.13 | - | 3.30 | 0.555 |
| 2,4-Dimethyl pentane | 0.21 | - | - | - | 6.98 | 0.058 |
| Methyl cyclopentane | 0.27 | - | 0.54 | - | 4.38 | 0.128 |
| 2-Methyl hexane | 0.18 | - | - | - | 0.43 | 0.121 |
| 3-Methyl hexane | 0.21 | - | - | - | 0.44 | 0.143 |
| n-Heptane | 0.23 | - | 1.22 | - | 0.60 | 0.156 |
| n-Nonane | 0.09 | - | 0.22 | - | 1.52 | 0.129 |
| n-Decane | 0.09 | 1.50 | - | - | 2.31 | 0.187 |
| Styrene | 0.14 | - | - | 0.88 | 0.51 | 0.133 |
| Benzene | 1.81 | 5.43 | 2.80 | 2.11 | 0.84 | 0.519 |
| Toluene | 4.70 | 11.14 | 14.09 | 13.45 | 39.80 | 2.544 |
| Ethyl benzene | 1.23 | 4.08 | 2.21 | 1.34 | 4.35 | 0.524 |
| m/p-Xylene | 1.40 | 8.54 | 5.16 | 1.56 | 5.25 | 0.675 |
| o-Xylene | 0.49 | 3.91 | 2.63 | 0.53 | 2.08 | 0.253 |
| MTBE | 0.29 | 3.07 | - | - | - | - |
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cai, C.-J.; Geng, F.-H.; Tie, X.-X.; Yu, Q.; Peng, L.; Zhou, G.-Q. Characteristics of Ambient Volatile Organic Compounds (VOCs) Measured in Shanghai, China. Sensors 2010, 10, 7843-7862. https://doi.org/10.3390/s100807843
Cai C-J, Geng F-H, Tie X-X, Yu Q, Peng L, Zhou G-Q. Characteristics of Ambient Volatile Organic Compounds (VOCs) Measured in Shanghai, China. Sensors. 2010; 10(8):7843-7862. https://doi.org/10.3390/s100807843
Chicago/Turabian StyleCai, Chang-Jie, Fu-Hai Geng, Xue-Xi Tie, Qiong Yu, Li Peng, and Guang-Qiang Zhou. 2010. "Characteristics of Ambient Volatile Organic Compounds (VOCs) Measured in Shanghai, China" Sensors 10, no. 8: 7843-7862. https://doi.org/10.3390/s100807843




