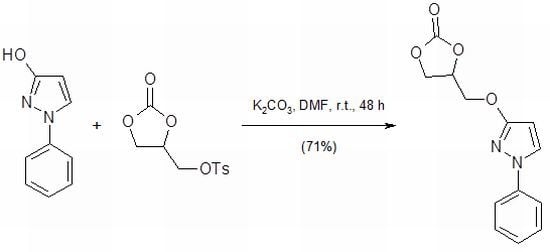
4-{[(1-Phenyl-1H-pyrazol-3-yl)oxy]methyl}-1,3-dioxolan-2-one
Abstract
:
Experimental
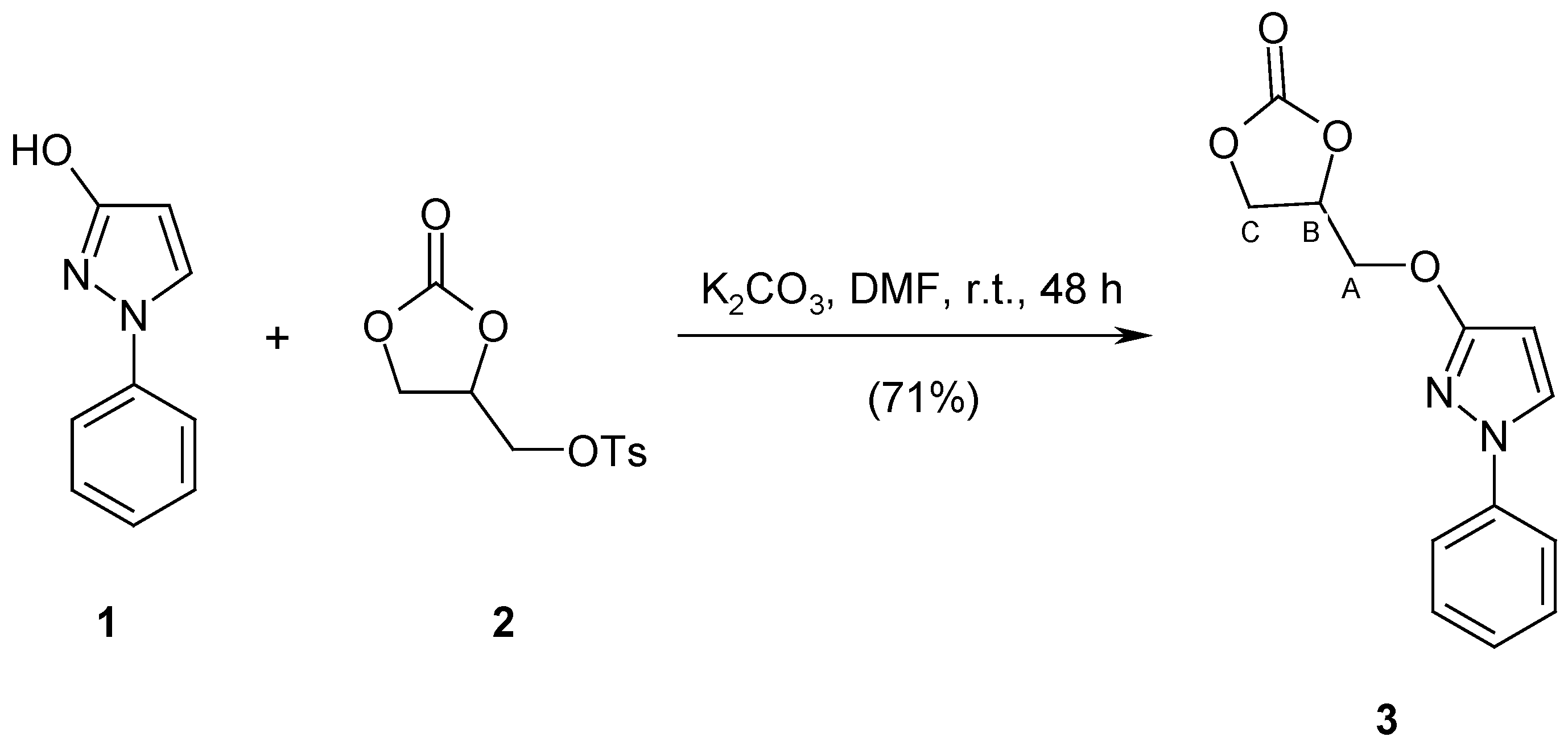
4-{[(1-Phenyl-1H-pyrazol-3-yl)oxy]methyl}-1,3-dioxolan-2-one (3)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3References and Notes
- Ha-Duong, N.-T.; Dijols, S.; Marques-Soares, C.; Minoletti, C.; Dansette, P.M.; Mansuy, D. Synthesis of Sulfaphenazole Derivatives and Their Use as Inhibitors and Tools for Comparing the Active Sites of Human Liver Cytochromes P450 of the 2C Subfamily. J. Med. Chem. 2001, 44, 3622–3631. [Google Scholar] [CrossRef] [PubMed]
- de Paulis, T.; Hemstapat, K.; Chen, Y.; Zhang, Y.; Saleh, S.; Alagille, D.; Baldwin, R.M.; Tamagnan, G.D.; Conn, P.J. Substituent Effects of N-(1,3-Diphenyl-1H-pyrazol-5-yl)benzamides on Positive Allosteric Modulation of the Metabotropic Glutamate-5 Receptor in Rat Cortical Astrocytes. J. Med. Chem. 2006, 49, 3332–3344. [Google Scholar] [CrossRef] [PubMed]
- Bebernitz, G.R.; Argentieri, G.; Battle, B.; Brennan, C.; Balkan, B.; Burkey, B.F.; Eckhardt, M.; Gao, J.; Kapa, P.; Strohschein, R.J.; et al. The Effect of 1,3-Diaryl-[1H]-pyrazole-4-acetamides on Glucose Utilization in ob/ob Mice. J. Med. Chem. 2001, 44, 2601–2611. [Google Scholar] [CrossRef] [PubMed]
- Raufl, M.; König, W. Effects of the nonsteroidal anti-inflammatory compounds Lonazolac Ca, indomethacin and NDGA on inflammatory mediator generation and release from various cells. Immunopharmacology 1990, 19, 103–111. [Google Scholar]
- Ahrens, H.; Koch, H.; Schröder, E.; Biere, H.; Kapp, J.-F. Novel Anti-Inflammatory Pyrazole Derivatives and Preparation Thereof. U.S. Patent 4,042,706, 16 August 1977. [Google Scholar]
- Oh, L.M. Synthesis of celecoxib via 1,3-dipolar cycloaddition. Tetrahedron Lett. 2006, 47, 7943–7946. [Google Scholar] [CrossRef]
- Arbačiauskienė, E.; Vilkauskaitė, G.; Eller, G.A.; Holzer, W.; Šačkus, A. Pd-catalyzed cross-coupling reactions of halogenated 1-phenylpyrazol-3-ols and related triflates. Tetrahedron 2009, 65, 7817–7824. [Google Scholar] [CrossRef]
- Arbačiauskienė, E.; Martynaitis, V.; Krikštolaitytė, S.; Holzer, W.; Šačkus, A. Synthesis of 3-substituted 1-phenyl-1H-pyrazole-4-carbaldehydes and the corresponding ethanones by Pd-catalysed cross-coupling reactions. ARKIVOC 2011, xi, 1–21. [Google Scholar]
- Giardi, C.; Lapinte, V.; Nielloud, F.; Devoisselle, J.-M.; Robin, J.-J. Synthesis of polyoxazolines using glycerol carbonate derivative and end chains functionalization via carbonate and isocyanate routes. J. Polym. Sci. Pol. Chem. 2010, 48, 4027–4035. [Google Scholar] [CrossRef]
- Simão, A.-C.; Lynikaitė-Puklevičienė, B.; Rousseau, C.; Tatibouët, A.; Cassel, S.; Šačkus, A.; Rauter, A.P.; Rollin, P. 1,2-Glycerol Carbonate: A Versatile Renewable Synthon. Lett. Org. Chem. 2006, 3, 744–748. [Google Scholar]
- Rousseau, J.; Rousseau, C.; Lynikaitė, B.; Šačkus, A.; de Leon, C.; Rollin, P.; Tatibouët, A. Tosylated glycerol carbonate, a versatile bis-electrophile to access new functionalized glycidol derivatives. Tetrahedron 2009, 65, 8571–8581. [Google Scholar] [CrossRef]
- Bai, R.; Wang, Y.; Wang, S.; Mei, F.; Li, T.; Li, G. Synthesis of glycerol carbonate from glycerol and dimethyl carbonate catalyzed by NaOH/γ-Al2O3. Fuel Process. Technol. 2012, in press. Available online: http://dx.doi.org/10.1016/j.fuproc.2012.07.027 (accessed on 7 November 2012). [Google Scholar] and references cited therein.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Vilkauskaitė, G.; Holzer, W.; Šačkus, A. 4-{[(1-Phenyl-1H-pyrazol-3-yl)oxy]methyl}-1,3-dioxolan-2-one. Molbank 2012, 2012, M786. https://doi.org/10.3390/M786
Vilkauskaitė G, Holzer W, Šačkus A. 4-{[(1-Phenyl-1H-pyrazol-3-yl)oxy]methyl}-1,3-dioxolan-2-one. Molbank. 2012; 2012(4):M786. https://doi.org/10.3390/M786
Chicago/Turabian StyleVilkauskaitė, Gytė, Wolfgang Holzer, and Algirdas Šačkus. 2012. "4-{[(1-Phenyl-1H-pyrazol-3-yl)oxy]methyl}-1,3-dioxolan-2-one" Molbank 2012, no. 4: M786. https://doi.org/10.3390/M786