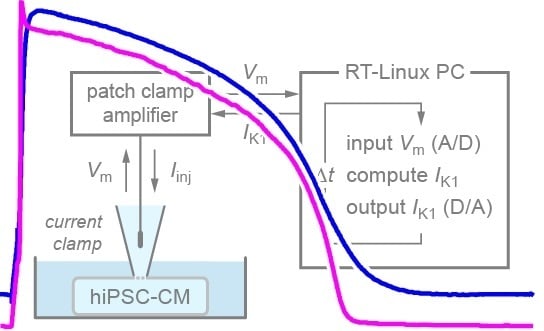
Patch-Clamp Recording from Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: Improving Action Potential Characteristics through Dynamic Clamp
Abstract
:1. Introduction
2. Results
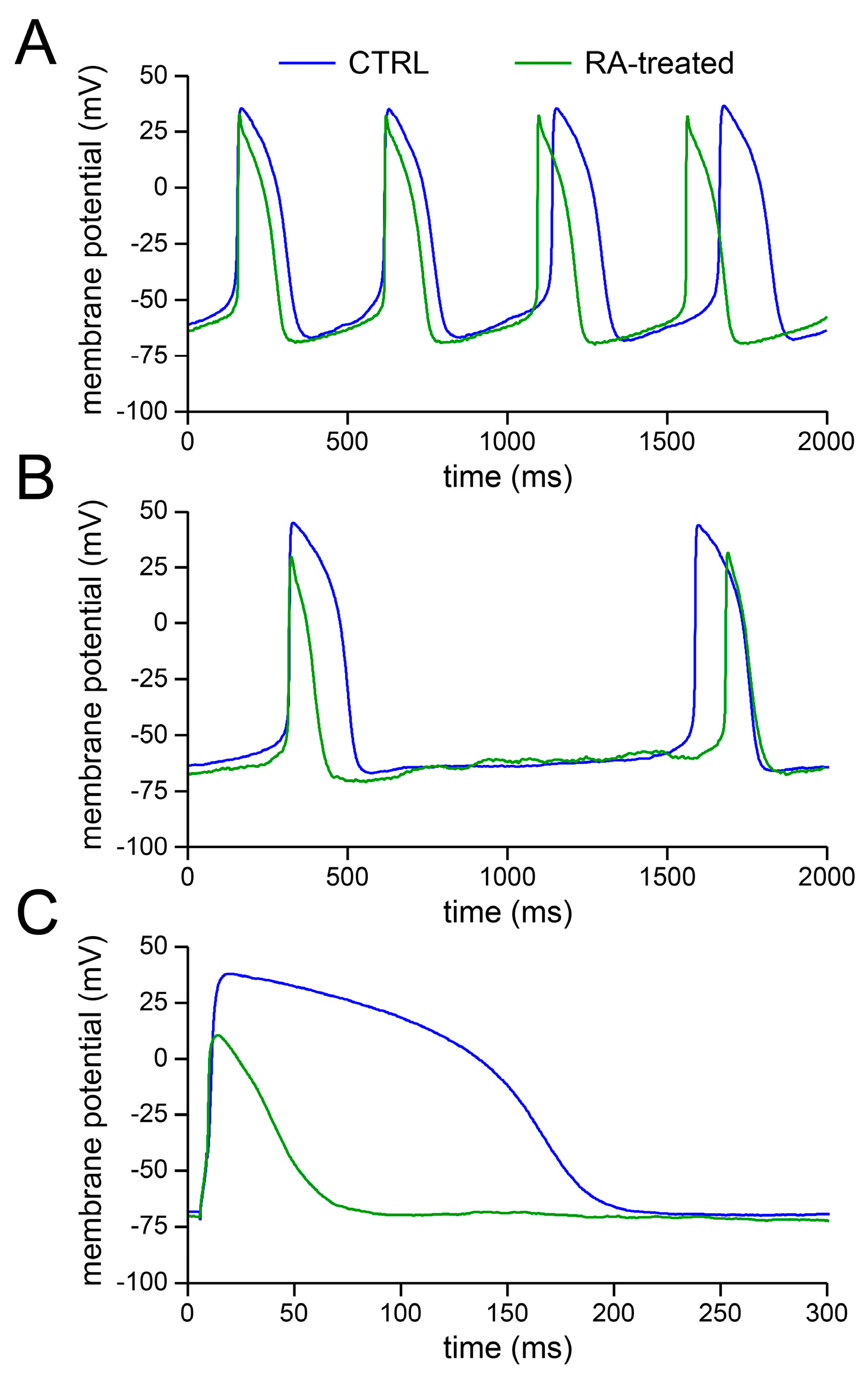
2.1. Basic Action Potential Characterisation of Control and Retinoic Acid-Treated Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes (hiPSC-CMs)
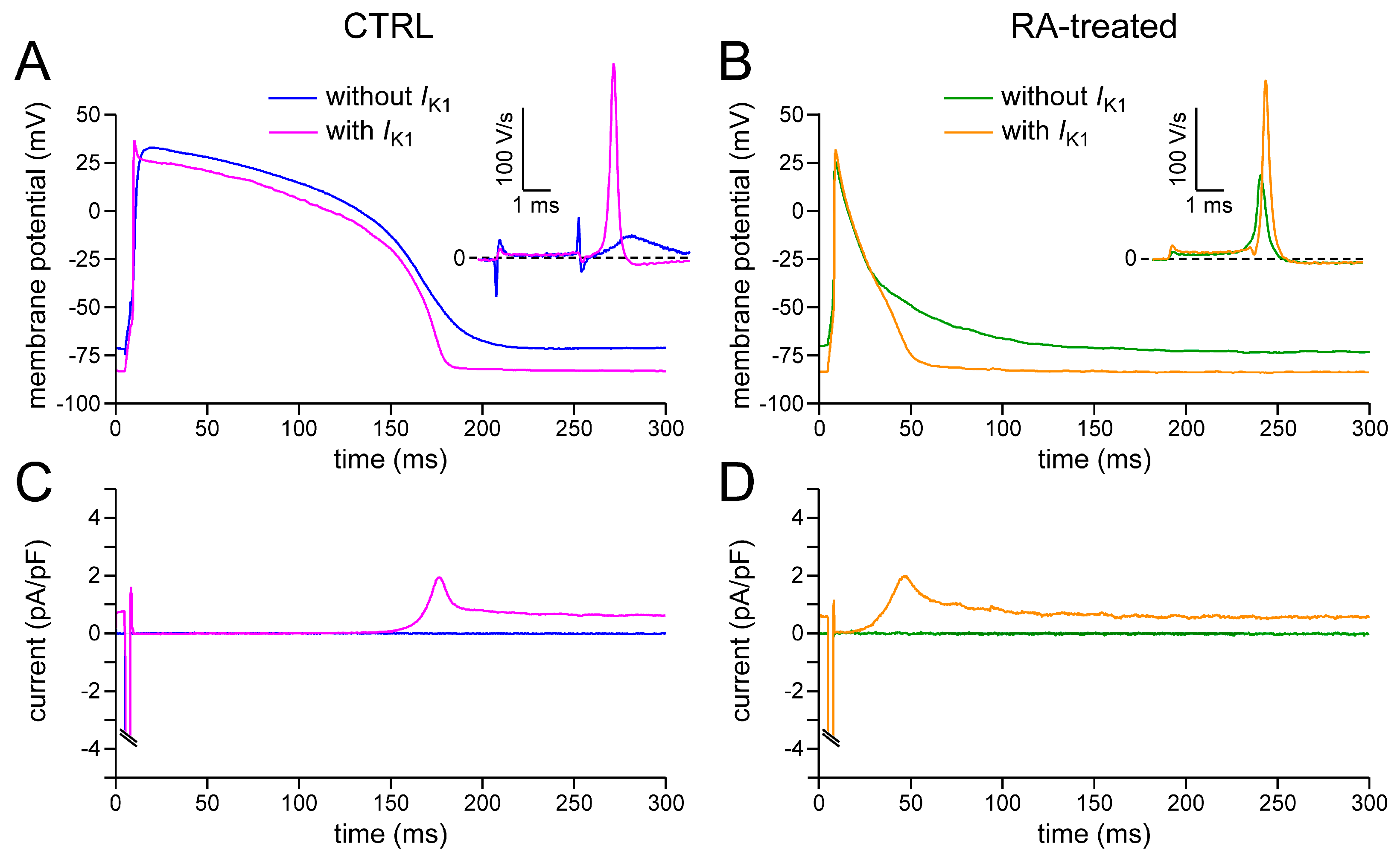
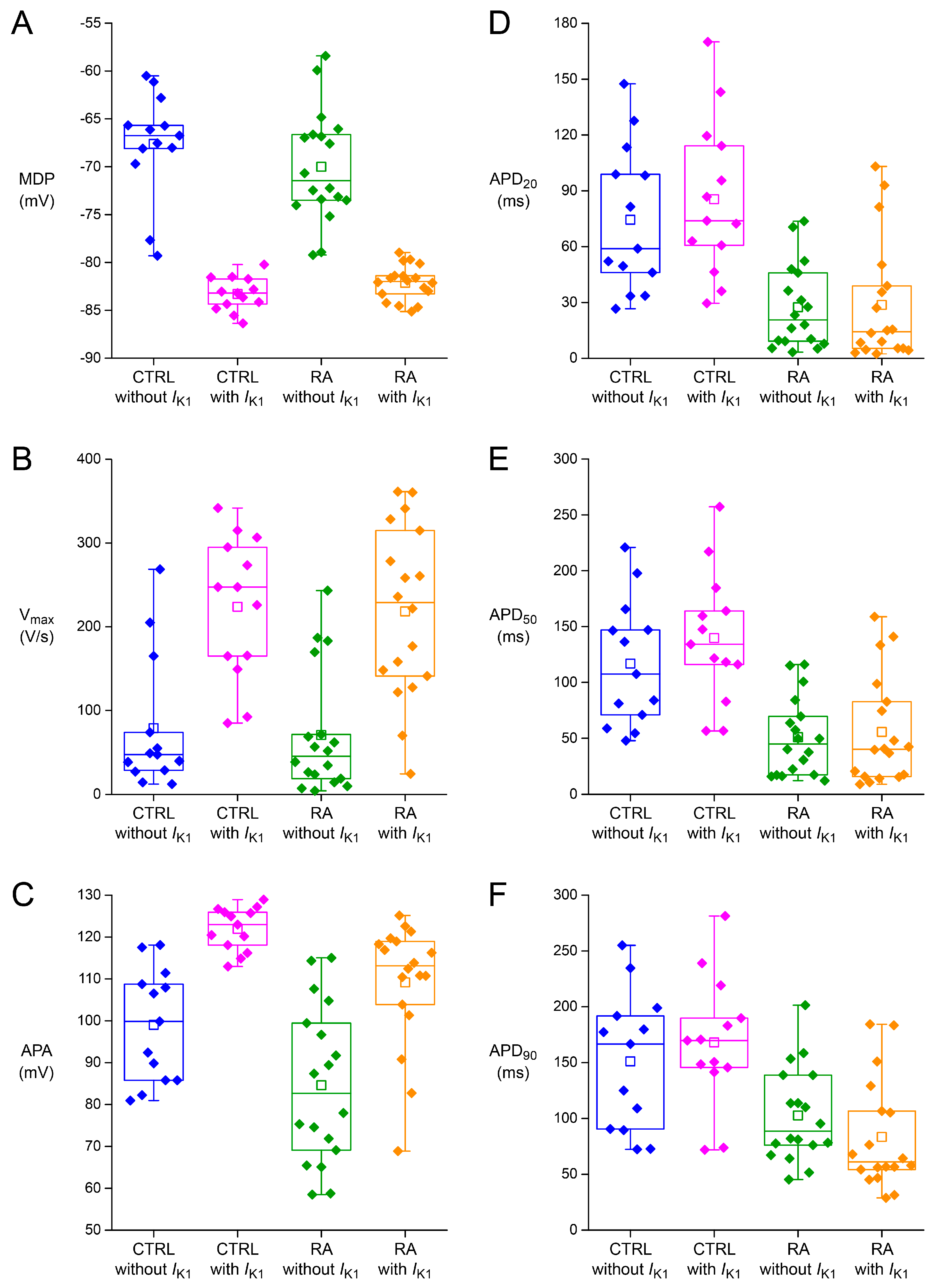
2.2. Effects of IK1 Injection on Action Potential Morphology
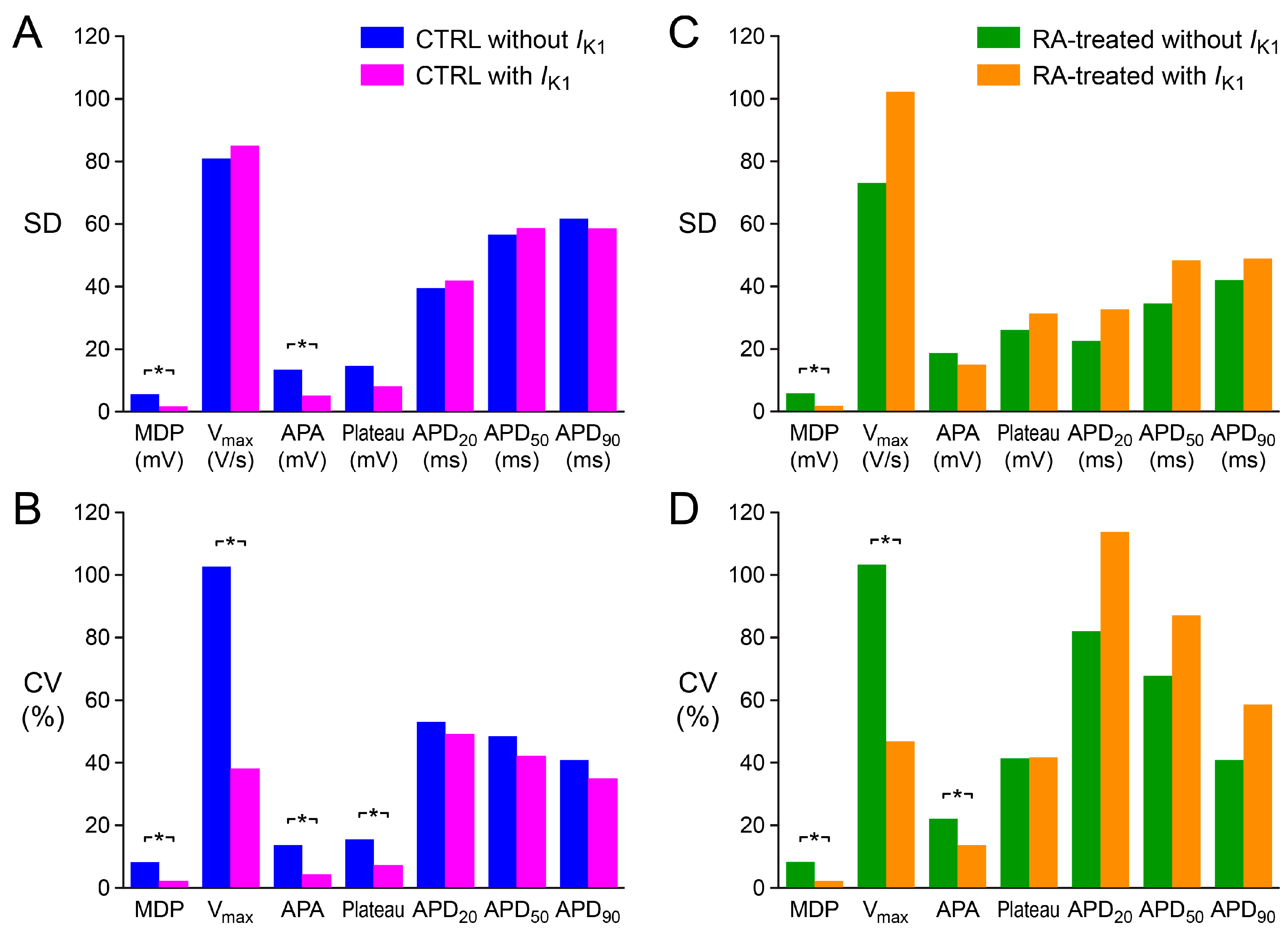
2.3. Variability in Action Potential Parameters
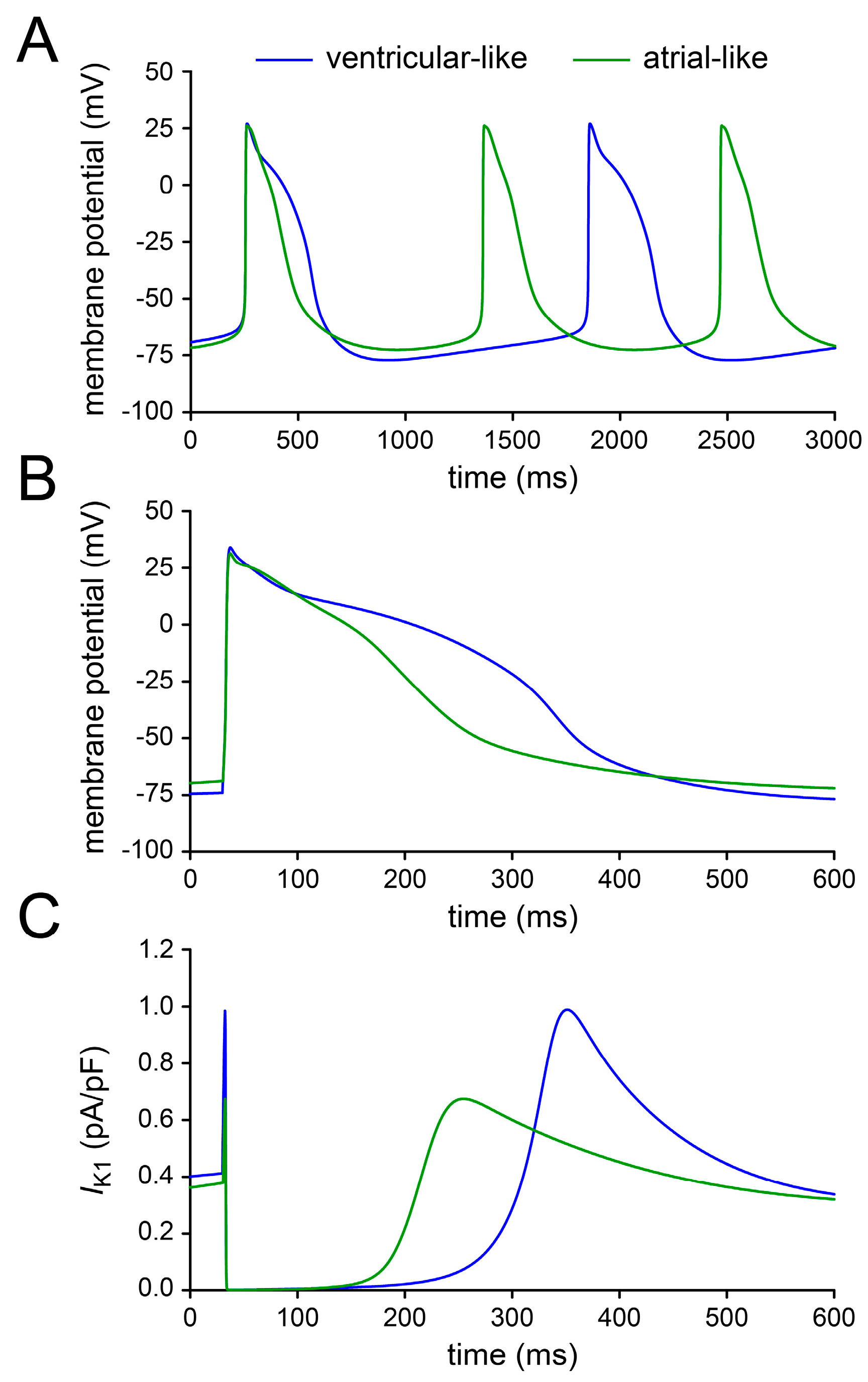
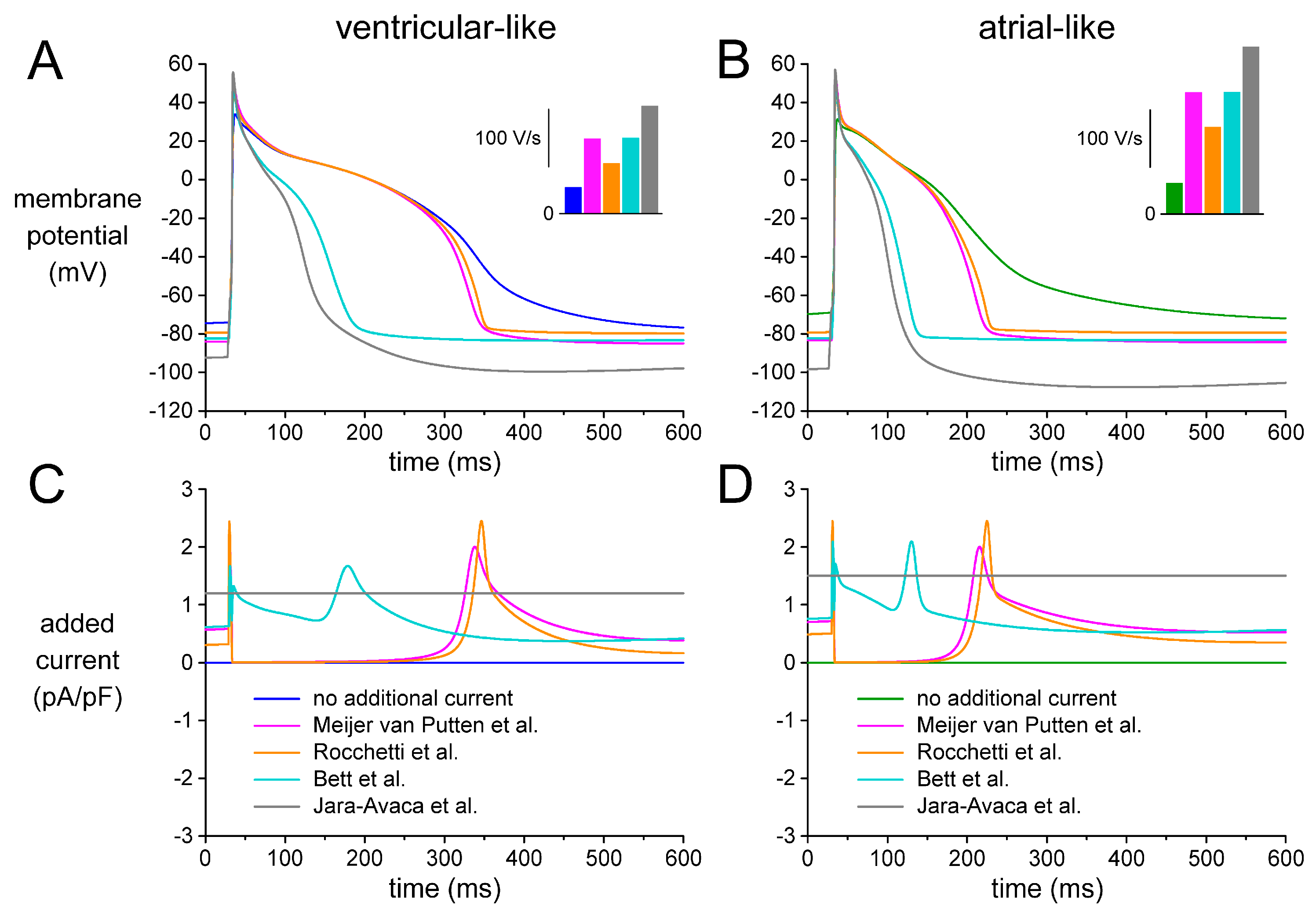
2.4. Assessing Dynamic Clamp Approach in Models of Ventricular-Like and Atrial-Like hiPSC-CMs
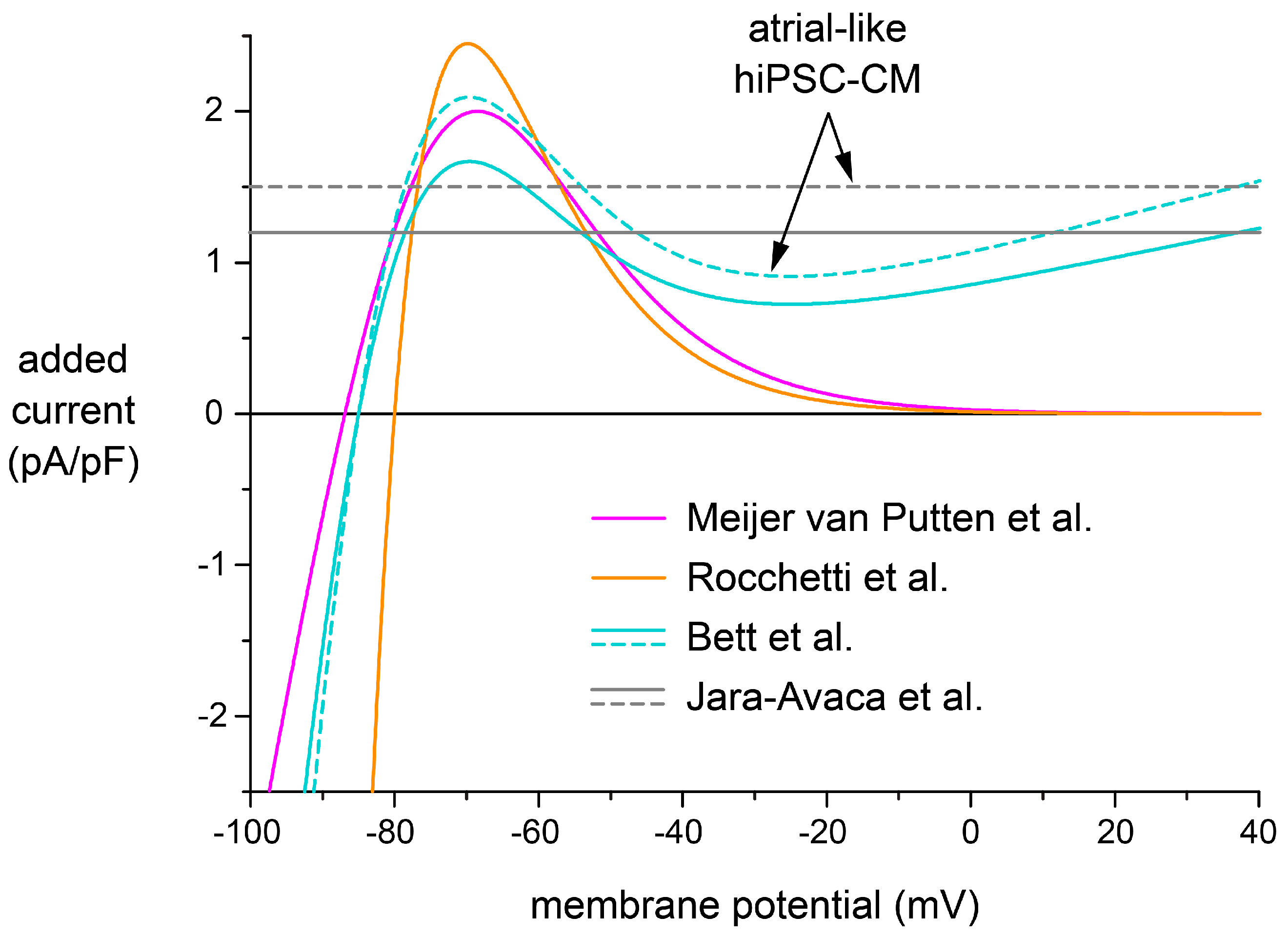
2.5. Effects of Different IK1 Formulations on Action Potential Morphology
3. Discussion
3.1. Action Potential Morphology of CTRL and RA-Treated hiPSC-CMs and Underlying Ionic Currents
3.2. Variability of Action Potential Parameters
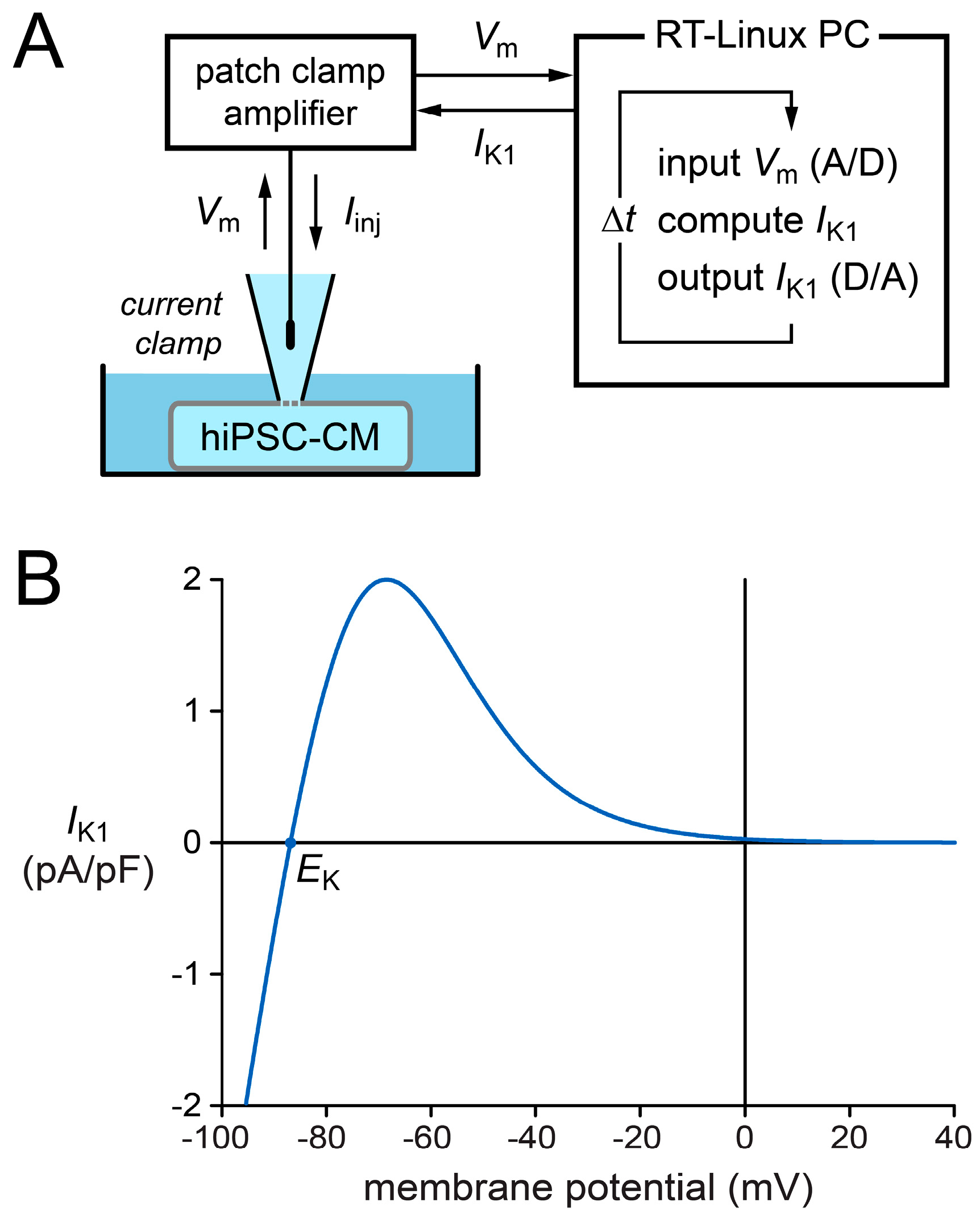
3.3. Selection of a Synthetic IK1
3.4. Future Directions
3.5. Limitations
4. Materials and Methods
4.1. Differentiation to hiPSC-CMs
4.2. Dissociation of hiPSC-CMs
4.3. Action Potential Measurements
4.4. Dynamic Clamp
4.5. Computer Simulations
4.6. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 4-AP | 4-aminopyridine |
| AF | Atrial fibrillation |
| AP | Action potential |
| APA | Action potential amplitude |
| APD | Action potential duration |
| APD20 | Action potential duration at 20% repolarisation |
| APD50 | Action potential duration at 50% repolarisation |
| APD90 | Action potential duration at 90% repolarisation |
| CTRL | Control |
| CV | Coefficient of variation |
| EK | Potassium equilibrium potential |
| hESC-CM | Human embryonic stem cell-derived cardiomyocyte |
| HEK-293 | Human embryonic kidney 293 |
| hiPSC | Human induced pluripotent stem cell |
| hiPSC-CM | Human induced pluripotent stem cell-derived cardiomyocyte |
| ICa,L | L-type calcium current |
| If | Hyperpolarisation-activated cyclic nucleotide-gated funny current |
| IK,ACh | Acetylcholine-activated potassium current |
| IK1 | Inward rectifier potassium current |
| IKur | Ultrarapid delayed rectifier potassium current |
| Ito1 | Transient outward potassium current |
| INa | Fast sodium current |
| Kir2.1 | Inward rectifier potassium channel subfamily 2 member 1 |
| MDP | Maximum diastolic potential |
| MEA | Multi-electrode array |
| RA | All-trans retinoic acid |
| RMP | Resting membrane potential |
| RT-Linux | Real-Time Linux |
| Vm | Membrane potential |
| Vmax | Maximum upstroke velocity |
References
- Davis, R.P.; van den Berg, C.W.; Casini, S.; Braam, S.R.; Mummery, C.L. Pluripotent stem cell models of cardiac disease and their implication for drug discovery and development. Trends Mol. Med. 2011, 17, 475–484. [Google Scholar] [CrossRef] [PubMed]
- Hoekstra, M.; Mummery, C.L.; Wilde, A.A.M.; Bezzina, C.R.; Verkerk, A.O. Induced pluripotent stem cell derived cardiomyocytes as models for cardiac arrhythmias. Front. Physiol. 2012, 3, 346. [Google Scholar] [CrossRef] [PubMed]
- Kane, C.; Terracciano, C.M.N. Concise reviews: Criteria for chamber-specific categorization of human cardiac myocytes derived from pluripotent stem cells. Stem Cells 2017, 35, 1881–1897. [Google Scholar] [CrossRef] [PubMed]
- Casini, S.; Verkerk, A.O.; Remme, C.A. Human iPSC-derived cardiomyocytes for investigation of disease mechanisms and therapeutic strategies in inherited arrhythmia syndromes: Strengths and limitations. Cardiovasc. Drugs Ther. 2017, 31, 325–344. [Google Scholar] [CrossRef] [PubMed]
- Meijer van Putten, R.M.E.; Mengarelli, I.; Guan, K.; Zegers, J.G.; van Ginneken, A.C.G.; Verkerk, A.O.; Wilders, R. Ion channelopathies in human induced pluripotent stem cell derived cardiomyocytes: A dynamic clamp study with virtual IK1. Front. Physiol. 2015, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Veerman, C.C.; Kosmidis, G.; Mummery, C.L.; Casini, S.; Verkerk, A.O.; Bellin, M. Immaturity of human stem-cell-derived cardiomyocytes in culture: Fatal flaw or soluble problem? Stem Cells Dev. 2015, 24, 1035–1052. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, Y.; Furukawa, T.; Singer, D.H.; Jia, H.; Backer, C.L.; Arentzen, C.E.; Wasserstrom, J.A. Sodium current in isolated human ventricular myocytes. Am. J. Physiol. 1993, 265, H1301–H1309. [Google Scholar] [PubMed]
- Pu, J.; Boyden, P.A. Alterations of Na+ currents in myocytes from epicardial border zone of the infarcted heart: A possible ionic mechanism for reduced excitability and postrepolarization refractoriness. Circ. Res. 1997, 81, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, J.M.; Nesterenko, V.V.; Sicouri, S.; Goodrow, R.J.; Treat, J.A.; Desai, M.; Wu, Y.; Doss, M.X.; Antzelevitch, C.; Di Diego, J.M. Identification and characterization of a transient outward K+ current in human induced pluripotent stem cell-derived cardiomyocytes. J. Mol. Cell. Cardiol. 2013, 60, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Benitah, J.P.; Alvarez, J.L.; Gómez, A.M. L-type Ca2+ current in ventricular cardiomyocytes. J. Mol. Cell. Cardiol. 2010, 48, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Lemoine, M.D.; Mannhardt, I.; Breckwoldt, K.; Prondzynski, M.; Flenner, F.; Ulmer, B.; Hirt, M.N.; Neuber, C.; Horváth, A.; Kloth, B.; et al. Human iPSC-derived cardiomyocytes cultured in 3D engineered heart tissue show physiological upstroke velocity and sodium current density. Sci. Rep. 2017, 7, 5464. [Google Scholar] [CrossRef] [PubMed]
- Lieu, D.K.; Fu, J.D.; Chiamvimonvat, N.; Tung, K.C.; McNerney, G.P.; Huser, T.; Keller, G.; Kong, C.W.; Li, R.A. Mechanism-based facilitated maturation of human pluripotent stem cell-derived cardiomyocytes. Circ. Arrhythm. Electrophysiol. 2013, 6, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, R.; Markandeya, Y.S.; Kamp, T.J.; Makielski, J.C.; January, C.T.; Eckhardt, L.L. IK1-enhanced human-induced pluripotent stem cell-derived cardiomyocytes: An improved cardiomyocyte model to investigate inherited arrhythmia syndromes. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H1611–H1621. [Google Scholar] [CrossRef] [PubMed]
- Bett, G.C.L.; Kaplan, A.D.; Lis, A.; Cimato, T.R.; Tzanakakis, E.S.; Zhou, Q.; Morales, M.J.; Rasmusson, R.L. Electronic “expression” of the inward rectifier in cardiocytes derived from human-induced pluripotent stem cells. Heart Rhythm 2013, 10, 1903–1910. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, M.; Sala, L.; Dreizehnter, L.; Crotti, L.; Sinnecker, D.; Mura, M.; Pane, L.S.; Altomare, C.; Torre, E.; Mostacciuolo, G.; et al. Elucidating arrhythmogenic mechanisms of long-QT syndrome CALM1-F142L mutation in patient-specific induced pluripotent stem cell-derived cardiomyocytes. Cardiovasc. Res. 2017, 113, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Jara-Avaca, M.; Kempf, H.; Rückert, M.; Robles-Diaz, D.; Franke, A.; de la Roche, J.; Fischer, M.; Malan, D.; Sasse, P.; Solodenko, W.; et al. EBIO does not induce cardiomyogenesis in human pluripotent stem cells but modulates cardiac subtype enrichment by lineage-selective survival. Stem Cell Rep. 2017, 8, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Krijthe, B.P.; Kunst, A.; Benjamin, E.J.; Lip, G.Y.H.; Franco, O.H.; Hofman, A.; Witteman, J.C.M.; Stricker, B.H.; Heeringa, J. Projections on the number of individuals with atrial fibrillation in the European Union, from 2000 to 2060. Eur. Heart J. 2013, 34, 2746–2751. [Google Scholar] [CrossRef] [PubMed]
- Colilla, S.; Crow, A.; Petkun, W.; Singer, D.E.; Simon, T.; Liu, X. Estimates of current and future incidence and prevalence of atrial fibrillation in the U.S. adult population. Am. J. Cardiol. 2013, 112, 1142–1147. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Sun, H.; Levesque, P. Antiarrhythmic drug therapy for atrial fibrillation: Focus on atrial selectivity and safety. Cardiovasc. Hematol. Agents Med. Chem. 2009, 7, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Dobrev, D.; Nattel, S. New antiarrhythmic drugs for treatment of atrial fibrillation. Lancet 2010, 375, 1212–1223. [Google Scholar] [CrossRef]
- Devalla, H.D.; Schwach, V.; Ford, J.W.; Milnes, J.T.; El-Haou, S.; Jackson, C.; Gkatzis, K.; Elliott, D.A.; Chuva de Sousa Lopes, S.M.; Mummery, C.L.; et al. Atrial-like cardiomyocytes from human pluripotent stem cells are a robust preclinical model for assessing atrial-selective pharmacology. EMBO Mol. Med. 2015, 7, 394–410. [Google Scholar] [CrossRef] [PubMed]
- Marczenke, M.; Piccini, I.; Mengarelli, I.; Fell, J.; Röpke, A.; Seebohm, G.; Verkerk, A.O.; Greber, B. Cardiac subtype-specific modeling of Kv1.5 ion channel deficiency using human pluripotent stem cells. Front. Physiol. 2017, 8, 469. [Google Scholar] [CrossRef] [PubMed]
- Nerbonne, J.M.; Kass, R.S. Molecular physiology of cardiac repolarization. Physiol. Rev. 2005, 85, 1205–1253. [Google Scholar] [CrossRef] [PubMed]
- Chiamvimonvat, N.; Chen-Izu, Y.; Clancy, C.E.; Deschenes, I.; Dobrev, D.; Heijman, J.; Izu, L.; Qu, Z.; Ripplinger, C.M.; Vandenberg, J.I.; et al. Potassium currents in the heart: Functional roles in repolarization, arrhythmia and therapeutics. J. Physiol. 2017, 595, 2229–2252. [Google Scholar] [CrossRef] [PubMed]
- Dhamoon, A.S.; Pandit, S.V.; Sarmast, F.; Parisian, K.R.; Guha, P.; Li, Y.; Bagwe, S.; Taffet, S.M.; Anumonwo, J.M.B. Unique Kir2.x properties determine regional and species differences in the cardiac inward rectifier K+ current. Circ. Res. 2004, 94, 1332–1339. [Google Scholar] [CrossRef] [PubMed]
- Veerman, C.C.; Mengarelli, I.; Guan, K.; Stauske, M.; Barc, J.; Tan, H.L.; Wilde, A.A.M.; Verkerk, A.O.; Bezzina, C.R. hiPSC-derived cardiomyocytes from Brugada Syndrome patients without identified mutations do not exhibit clear cellular electrophysiological abnormalities. Sci. Rep. 2016, 6, 30967. [Google Scholar] [CrossRef] [PubMed]
- Portero, V.; Casini, S.; Hoekstra, M.; Verkerk, A.O.; Mengarelli, I.; Belardinelli, L.; Rajamani, S.; Wilde, A.A.M.; Bezzina, C.R.; Veldkamp, M.W.; et al. Anti-arrhythmic potential of the late sodium current inhibitor GS-458967 in murine Scn5a-1798insD+/− and human SCN5A-1795insD+/− iPSC-derived cardiomyocytes. Cardiovasc. Res. 2017, 113, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Jost, N.; Virág, L.; Comtois, P.; Ördög, B.; Szuts, V.; Seprényi, G.; Bitay, M.; Kohajda, Z.; Koncz, I.; Nagy, N.; et al. Ionic mechanisms limiting cardiac repolarization reserve in humans compared to dogs. J. Physiol. 2013, 591, 4189–4206. [Google Scholar] [CrossRef] [PubMed]
- Berecki, G.; Wilders, R.; de Jonge, B.; van Ginneken, A.C.G.; Verkerk, A.O. Re-evaluation of the action potential upstroke velocity as a measure of the Na+ current in cardiac myocytes at physiological conditions. PLoS ONE 2010, 5, e15772. [Google Scholar] [CrossRef] [PubMed]
- Paci, M.; Hyttinen, J.; Aalto-Setälä, K.; Severi, S. Computational models of ventricular- and atrial-like human induced pluripotent stem cell derived cardiomyocytes. Ann. Biomed. Eng. 2013, 41, 2334–2348. [Google Scholar] [CrossRef] [PubMed]
- Paci, M.; Hyttinen, J.; Rodriguez, B.; Severi, S. Human induced pluripotent stem cell-derived versus adult cardiomyocytes: An in silico electrophysiological study on effects of ionic current block. Br. J. Pharmacol. 2015, 172, 5147–5160. [Google Scholar] [CrossRef] [PubMed]
- Li, G.R.; Feng, J.; Yue, L.; Carrier, M. Transmural heterogeneity of action potentials and Ito1 in myocytes isolated from the human right ventricle. Am. J. Physiol. 1998, 275, H369–H377. [Google Scholar] [PubMed]
- Wang, Z.; Yue, L.; White, M.; Pelletier, G.; Nattel, S. Differential distribution of inward rectifier potassium channel transcripts in human atrium versus ventricle. Circulation 1998, 98, 2422–2428. [Google Scholar] [CrossRef] [PubMed]
- Bailly, P.; Mouchonière, M.; Bénitah, J.P.; Camilleri, L.; Vassort, G.; Lorente, P. Extracellular K+ dependence of inward rectification kinetics in human left ventricular cardiomyocytes. Circulation 1998, 98, 2753–2759. [Google Scholar] [CrossRef] [PubMed]
- Koumi, S.I.; Backer, C.L.; Arentzen, C.E. Characterization of inwardly rectifying K+ channel in human cardiac myocytes: Alterations in channel behavior in myocytes isolated from patients with idiopathic dilated cardiomyopathy. Circulation 1995, 92, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.P.; Casini, S.; van den Berg, C.W.; Hoekstra, M.; Remme, C.A.; Dambrot, C.; Salvatori, D.; Ward-van Oostwaard, D.; Wilde, A.A.M.; Bezzina, C.R.; et al. Cardiomyocytes derived from pluripotent stem cells recapitulate electrophysiological characteristics of an overlap syndrome of cardiac sodium channel disease. Circulation 2012, 125, 3079–3091. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; D’Aniello, C.; Verkerk, A.O.; Wrobel, E.; Frank, S.; Ward-van Oostwaard, D.; Piccini, I.; Freund, C.; Rao, J.; Seebohm, G.; et al. Recessive cardiac phenotypes in induced pluripotent stem cell models of Jervell and Lange-Nielsen syndrome: Disease mechanisms and pharmacological rescue. Proc. Natl. Acad. Sci. USA 2014, 111, E5383–E5392. [Google Scholar] [CrossRef] [PubMed]
- Birket, M.J.; Ribeiro, M.C.; Verkerk, A.O.; Ward, D.; Leitoguinho, A.R.; den Hartogh, S.C.; Orlova, V.V.; Devalla, H.D.; Schwach, V.; Bellin, M.; et al. Expansion and patterning of cardiovascular progenitors derived from human pluripotent stem cells. Nat. Biotechnol. 2015, 33, 970–979. [Google Scholar] [CrossRef] [PubMed]
- Devalla, H.D.; Gélinas, R.; Aburawi, E.H.; Beqqali, A.; Goyette, P.; Freund, C.; Chaix, M.A.; Tadros, R.; Jiang, H.; Le Béchec, A.; et al. TECRL, a new life-threatening inherited arrhythmia gene associated with overlapping clinical features of both LQTS and CPVT. EMBO Mol. Med. 2016, 8, 1390–1408. [Google Scholar] [CrossRef] [PubMed]
- Veerman, C.C.; Mengarelli, I.; Lodder, E.M.; Kosmidis, G.; Bellin, M.; Zhang, M.; Dittmann, S.; Guan, K.; Wilde, A.A.M.; Schulze-Bahr, E.; et al. Switch from fetal to adult SCN5A isoform in human induced pluripotent stem cell-derived cardiomyocytes unmasks the cellular phenotype of a conduction disease-causing mutation. J. Am. Heart Assoc. 2017, 6, e005135. [Google Scholar] [CrossRef] [PubMed]
- Verkerk, A.O. Patch-Clamp Experiments on Ventricular-Like hiPSC-CMs; Department of Medical Biology and Department of Experimental Cardiology, Academic Medical Center, University of Amsterdam: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Wang, Z.; Fermini, B.; Nattel, S. Sustained depolarization-induced outward current in human atrial myocytes: Evidence for a novel delayed rectifier K+ current similar to Kv1.5 cloned channel currents. Circ. Res. 1993, 73, 1061–1076. [Google Scholar] [CrossRef] [PubMed]
- Amos, G.J.; Wettwer, E.; Metzger, F.; Li, Q.; Himmel, H.M.; Ravens, U. Differences between outward currents of human atrial and subepicardial ventricular myocytes. J. Physiol. 1996, 491, 31–50. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, Y.; Wasserstrom, J.A.; Furukawa, T.; Jia, H.; Arentzen, C.E.; Hartz, R.S.; Singer, D.H. Characterization of the sodium current in single human atrial myocytes. Circ. Res. 1992, 71, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.; Koumi, S.; Sakakibara, Y.; Singer, D.H.; Jia, H.; Arentzen, C.E.; Backer, C.L.; Wasserstrom, J.A. An analysis of lidocaine block of sodium current in isolated human atrial and ventricular myocytes. J. Mol. Cell. Cardiol. 1995, 27, 831–846. [Google Scholar] [CrossRef]
- Burashnikov, A.; di Diego, J.M.; Zygmunt, A.C.; Belardinelli, L.; Antzelevitch, C. Atrium-selective sodium channel block as a strategy for suppression of atrial fibrillation: Differences in sodium channel inactivation between atria and ventricles and the role of ranolazine. Circulation 2007, 116, 1449–1457. [Google Scholar] [CrossRef] [PubMed]
- Calloe, K.; Nof, E.; Jespersen, T.; di Diego, J.M.; Chlus, N.; Olesen, S.P.; Antzelevitch, C.; Cordeiro, J.M. Comparison of the effects of a transient outward potassium channel activator on currents recorded from atrial and ventricular cardiomyocytes. J. Cardiovasc. Electr. 2011, 22, 1057–1066. [Google Scholar] [CrossRef] [PubMed]
- Cohen, N.M.; Lederer, W.J. Calcium current in single human cardiac myocytes. J. Cardiovasc. Electr. 1993, 4, 422–437. [Google Scholar] [CrossRef]
- Hatem, S.N.; Coulombe, A.; Balse, E. Specificities of atrial electrophysiology: Clues to a better understanding of cardiac function and the mechanisms of arrhythmias. J. Mol. Cell. Cardiol. 2010, 48, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Giles, W.R.; Imaizumi, Y. Comparison of potassium currents in rabbit atrial and ventricular cells. J. Physiol. 1988, 405, 123–125. [Google Scholar] [CrossRef] [PubMed]
- Melnyk, P.; Zhang, L.; Shrier, A.; Nattel, S. Differential distribution of Kir2.1 and Kir2.3 subunits in canine atrium and ventricle. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H1123–H1133. [Google Scholar] [CrossRef] [PubMed]
- Gaborit, N.; Le Bouter, S.; Szuts, V.; Varro, A.; Escande, D.; Nattel, S.; Demolombe, S. Regional and tissue specific transcript signatures of ion channel genes in the non-diseased human heart. J. Physiol. 2007, 582, 675–693. [Google Scholar] [CrossRef] [PubMed]
- Milstein, M.L.; Musa, H.; Balbuena, D.P.; Anumonwo, J.M.B.; Auerbach, D.S.; Furspan, P.B.; Hou, L.; Hu, B.; Schumacher, S.M.; Vaidyanathan, R.; et al. Dynamic reciprocity of sodium and potassium channel expression in a macromolecular complex controls cardiac excitability and arrhythmia. Proc. Natl. Acad. Sci. USA 2012, 109, E2134–E2143. [Google Scholar] [CrossRef] [PubMed]
- Willis, B.C.; Ponce-Balbuena, D.; Jalife, J. Protein assemblies of sodium and inward rectifier potassium channels control cardiac excitability and arrhythmogenesis. Am. J. Physiol. Heart Circ. Physiol. 2015, 308, H1463–H1473. [Google Scholar] [CrossRef] [PubMed]
- Varghese, A. Reciprocal modulation of IK1-INa extends excitability in cardiac ventricular cells. Front. Physiol. 2016, 7, 542. [Google Scholar] [CrossRef] [PubMed]
- Goversen, B.; de Boer, T.P.; van der Heyden, M.A.G. Commentary: Reciprocal modulation of IK1-INa extends excitability in cardiac ventricular cells. Front. Physiol. 2016, 7, 647. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.D.; Rasmusson, R.L.; Bett, G.C.L. Ionic basis of repolarization of atrial and ventricular specific cell types derived from human induced pluripotent stem cells. Biophys. J. 2016, 110, 343a. [Google Scholar] [CrossRef]
- Marrus, S.; Springer, S.; Johnson, E.; Martinez, R.; Dranoff, E.; Mellor, R.; Yamada, K. Dynamic current clamp experiments define the functional roles of IK1 and Ito,f in human induced pluripotent stem cell derived cardiomyocytes. bioRχiv 2017, 135400. [Google Scholar] [CrossRef]
- Cytocybernetics. Available online: http://cytocybernetics.com/Cybercyte.html (accessed on 31 July 2017).
- Wilders, R. Computer Simulations to Determine the Minimum IK1 Density Required to Obtain a Stable Resting Membrane Potential in Atrial-Like hiPSC-CMs; Department of Medical Biology, Academic Medical Center, University of Amsterdam: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Yajuan, X.; Xin, L.; Zhiyuan, L. A comparison of the performance and application differences between manual and automated patch-clamp techniques. Curr. Chem. Genomics 2012, 6, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Quach, B.; Christini, D. A novel optical dynamic clamp method to make iPSC-CMs a more viable platform for drug screening. In Proceedings of the Program & Abstract Book of the 36th Annual Vincent du Vigneaud Research Symposium, Weill Cornell Medical College, New York, NY, USA, 21 April 2016. [Google Scholar]
- Han, X.; Chow, B.Y.; Zhou, H.; Klapoetke, N.C.; Chuong, A.; Rajimehr, R.; Yang, A.; Baratta, M.V.; Winkle, J.; Desimone, R.; et al. A high-light sensitivity optical neural silencer: Development and application to optogenetic control of non-human primate cortex. Front. Syst. Neurosci. 2011, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Tertoolen, L.G.J.; Braam, S.R.; van Meer, B.J.; Passier, R.; Mummery, C.L. Interpretation of field potentials measured on a multi electrode array in pharmacological toxicity screening on primary and human pluripotent stem cell-derived cardiomyocytes. Biochem. Biophys. Res. Commun. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Shinnawi, R.; Huber, I.; Maizels, L.; Shaheen, N.; Gepstein, A.; Arbel, G.; Tijsen, A.J.; Gepstein, L. Monitoring human-induced pluripotent stem cell-derived cardiomyocytes with genetically encoded calcium and voltage fluorescent reporters. Stem Cell Rep. 2015, 5, 582–596. [Google Scholar] [CrossRef] [PubMed]
- Streckfuss-Bömeke, K.; Wolf, F.; Azizian, A.; Stauske, M.; Tiburcy, M.; Wagner, S.; Hübscher, D.; Dressel, R.; Chen, S.; Jende, J.; et al. Comparative study of human-induced pluripotent stem cells derived from bone marrow cells, hair keratinocytes, and skin fibroblasts. Eur. Heart J. 2013, 34, 2618–2629. [Google Scholar] [CrossRef] [PubMed]
- Lian, X.; Zhang, J.; Azarin, S.M.; Zhu, K.; Hazeltine, L.B.; Bao, X.; Hsiao, C.; Kamp, T.J.; Palecek, S.P. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions. Nat. Protoc. 2013, 8, 162–175. [Google Scholar] [CrossRef] [PubMed]
- Tohyama, S.; Hattori, F.; Sano, M.; Hishiki, T.; Nagahata, Y.; Matsuura, T.; Hashimoto, H.; Suzuki, T.; Yamashita, H.; Satoh, Y.; et al. Distinct metabolic flow enables large-scale purification of mouse and human pluripotent stem cell-derived cardiomyocytes. Cell Stem Cell 2013, 12, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Barry, P.H.; Lynch, J.W. Liquid junction potentials and small cell effects in patch-clamp analysis. J. Membr. Biol. 1991, 121, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, C.M.; Lawson, J.R.; Hunter, P.J.; Nielsen, P.F. The CellML Model Repository. Bioinformatics 2008, 24, 2122–2123. [Google Scholar] [CrossRef] [PubMed]
- Garny, A.; Noble, D.; Hunter, P.J.; Kohl, P. Cellular Open Resource (COR): Current status and future directions. Philos. Trans. A Math. Phys. Eng. Sci. 2009, 367, 1885–1905. [Google Scholar] [CrossRef] [PubMed]
- O’Hara, T.; Virág, L.; Varró, A.; Rudy, Y. Simulation of the undiseased human cardiac ventricular action potential: Model formulation and experimental validation. PLoS Comput. Biol. 2011, 7, e1002061. [Google Scholar] [CrossRef] [PubMed]

| Action Potential Parameter | Fast Beating Cells | Slowly Beating Cells | ||
|---|---|---|---|---|
| CTRL (n = 9) | RA (n = 12) | CTRL (n = 9) | RA (n = 9) | |
| MDP (mV) | −68.2 ± 1.8 | −68.5 ± 0.6 | −70.4 ± 2.8 | −70.8 ± 1.3 |
| Vmax (V/s) | 85.3 ± 29.3 | 40.8 ± 9.9 | 84.1 ± 30.2 | 29.7 ± 5.9 |
| APA (mV) | 105.1 ± 3.8 | 94.7 ± 4.6 | 108.4 ± 5.8 | 82.2 ± 5.7 * |
| AP plateau (mV) | 99.5 ± 4.2 | 86.1 ± 4.8 * | 103.1 ± 4.0 | 72.1 ± 8.5 * |
| APD20 (ms) | 78.1 ± 15.5 | 45.3 ± 5.7 * | 91.5 ± 11.0 | 42.1 ± 8.7 * |
| APD50 (ms) | 124.7 ± 21.9 | 81.5 ± 9.7 | 144.3 ± 17.9 | 79.2 ± 13.7 * |
| APD90 (ms) | 161.7 ± 23.9 | 122.8 ± 13.9 | 181.7 ± 19.6 | 152.4 ± 34.2 |
| Cycle length (ms) | 565 ± 57 | 488 ± 50 | 1392 ± 163 | 2138 ± 502 |
| Action Potential Parameter | CTRL (n = 13) | RA (n = 18) | ||
|---|---|---|---|---|
| without IK1 | with IK1 | without IK1 | with IK1 | |
| MDP (mV) | −67.7 ± 1.6 | −83.3 ± 0.5 † | −70.0 ± 1.4 | −82.1 ± 0.4 † |
| Vmax (V/s) | 78.8 ± 23.4 | 233.8 ± 24.5 † | 70.7 ± 17.7 | 218.3 ± 24.8 † |
| APA (mV) | 99.0 ± 3.9 | 121.9 ± 1.5 † | 84.6 ± 4.5 * | 109.2 ± 3.6 * † |
| AP plateau (mV) | 94.2 ± 4.2 | 113.5 ± 2.3 † | 63.1 ± 6.3 * | 75.0 ± 7.6 * † |
| APD20 (ms) | 74.4 ± 11.4 | 85.5 ± 12.1 † | 27.4 ± 5.5 * | 28.6 ± 7.9 * |
| APD50 (ms) | 116.8 ± 16.4 | 139.6 ± 16.9 † | 50.9 ± 8.4 * | 55.5 ± 11.7 * |
| APD90 (ms) | 151.0 ± 17.8 | 168.0 ± 16.9 † | 102.6 ± 10.2 * | 83.4 ± 11.9 * |
| Experimental Approach | Resting Membrane Potential (mV) | |
|---|---|---|
| Ventricular-Like Model Cell 1 | Atrial-Like Model Cell 1 | |
| Meijer van Putten et al. [5] | −79.9 | −79.2 |
| Rocchetti et al. [15] | −78.1 | −77.6 |
| Bett et al. [14] | −79.1 | −79.5 |
| Jara-Avaca et al. [16] | −79.8 | −81.0 |
| Action Potential Parameter | Meijer van Putten et al. IK1 | Rocchetti et al. IK1 | Bett et al. IK1 | |||
|---|---|---|---|---|---|---|
| Vent | Atr | Vent | Atr | Vent | Atr | |
| MDP (mV) | −83.9 | −84.0 | −79.5 | −79.4 | −82.2 | −82.8 |
| Vmax (V/s) | 129.1 | 209.6 | 87.0 | 149.8 | 130.4 | 210.0 |
| APA (mV) | 134.8 | 138.2 | 120.3 | 129.2 | 132.1 | 136.2 |
| AP plateau (mV) | 112.4 | 110.8 | 107.0 | 106.0 | 101.7 | 100.4 |
| APD20 (ms) | 30.8 | 20.8 | 42.4 | 31.5 | 14.8 | 8.7 |
| APD50 (ms) | 240.9 | 135.1 | 248.5 | 138.3 | 94.8 | 65.8 |
| APD90 (ms) | 306.6 | 183.3 | 311.5 | 189.6 | 144.4 | 96.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verkerk, A.O.; Veerman, C.C.; Zegers, J.G.; Mengarelli, I.; Bezzina, C.R.; Wilders, R. Patch-Clamp Recording from Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: Improving Action Potential Characteristics through Dynamic Clamp. Int. J. Mol. Sci. 2017, 18, 1873. https://doi.org/10.3390/ijms18091873
Verkerk AO, Veerman CC, Zegers JG, Mengarelli I, Bezzina CR, Wilders R. Patch-Clamp Recording from Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: Improving Action Potential Characteristics through Dynamic Clamp. International Journal of Molecular Sciences. 2017; 18(9):1873. https://doi.org/10.3390/ijms18091873
Chicago/Turabian StyleVerkerk, Arie O., Christiaan C. Veerman, Jan G. Zegers, Isabella Mengarelli, Connie R. Bezzina, and Ronald Wilders. 2017. "Patch-Clamp Recording from Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes: Improving Action Potential Characteristics through Dynamic Clamp" International Journal of Molecular Sciences 18, no. 9: 1873. https://doi.org/10.3390/ijms18091873