The Roles of β-Integrin of Chinese Shrimp (Fenneropenaeus chinensis) in WSSV Infection
Abstract
:1. Introduction
2. Results
2.1. Interaction between rFcβInt-ER and Five WSSV Envelope Proteins
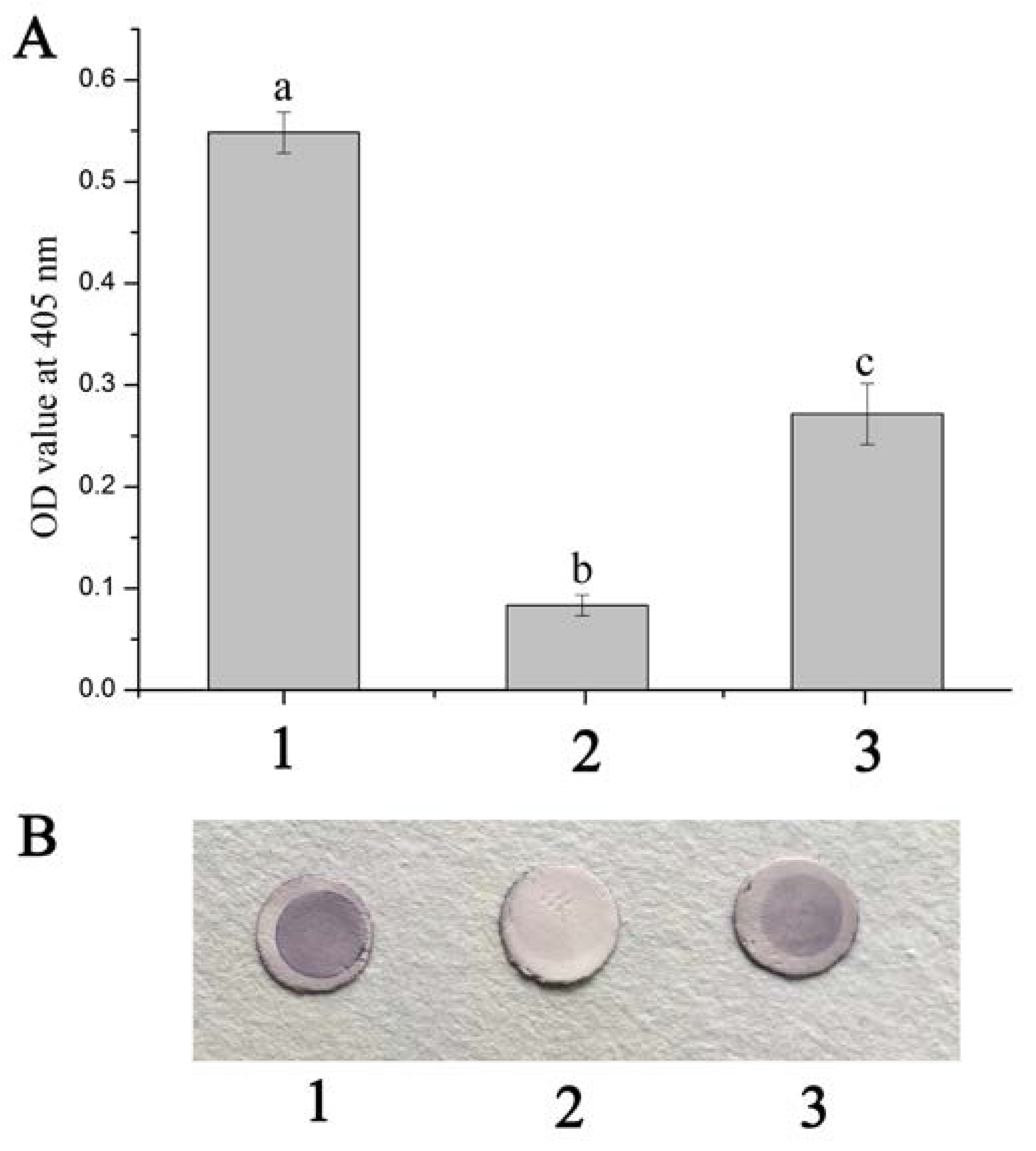
2.2. Blocking Effects of Anti-rFcβInt-ER Antibodies In Vitro
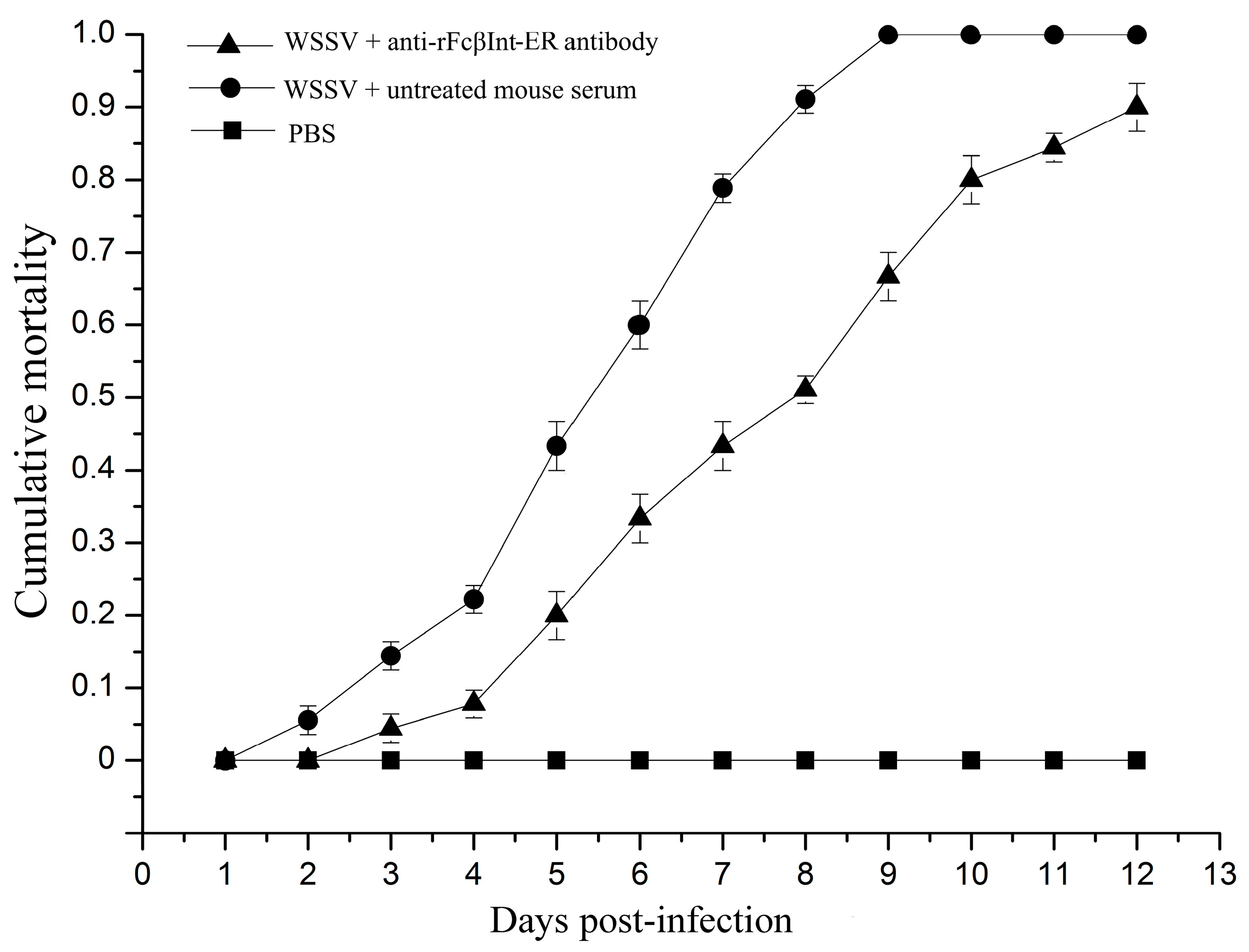
2.3. Infection-Blocking Effect of Anti-rFcβInt-ER Antibodies In Vivo
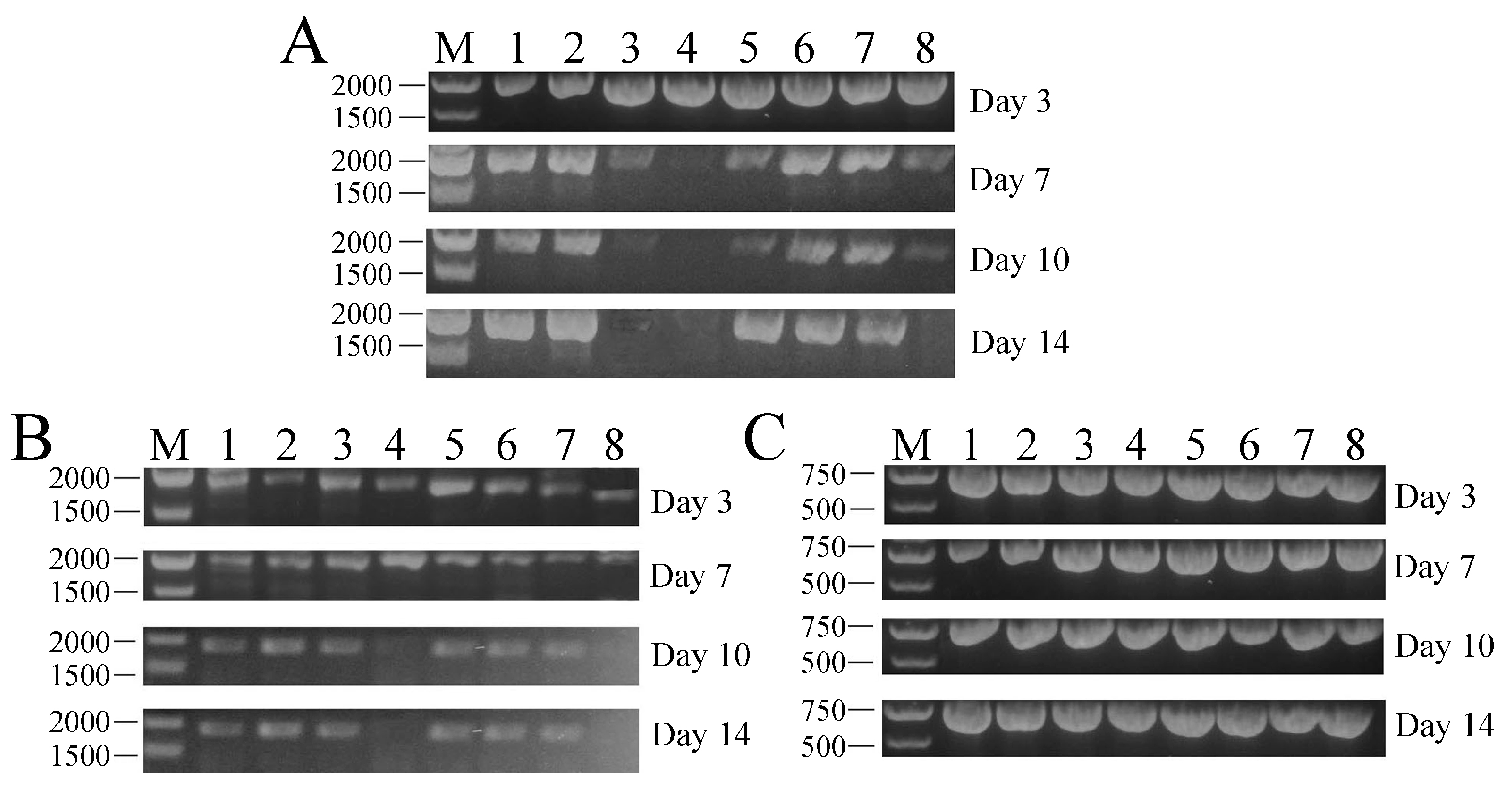
2.4. Tissue Distribution and In Vivo Transcription Analysis of PFcβInt-ER in F. chinensis
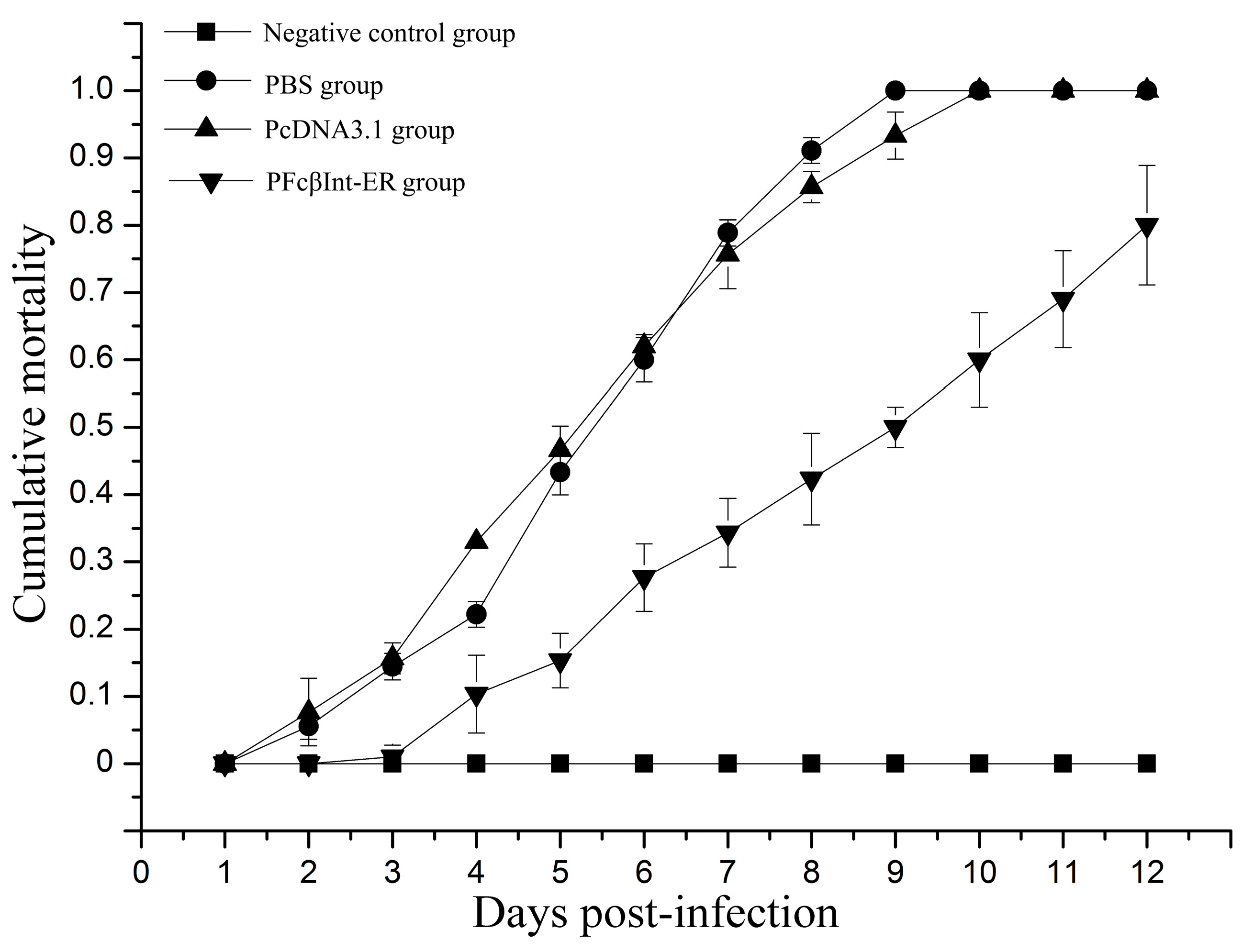
2.5. Neutralizing Effect of PFcβInt-ER against WSSV Infection
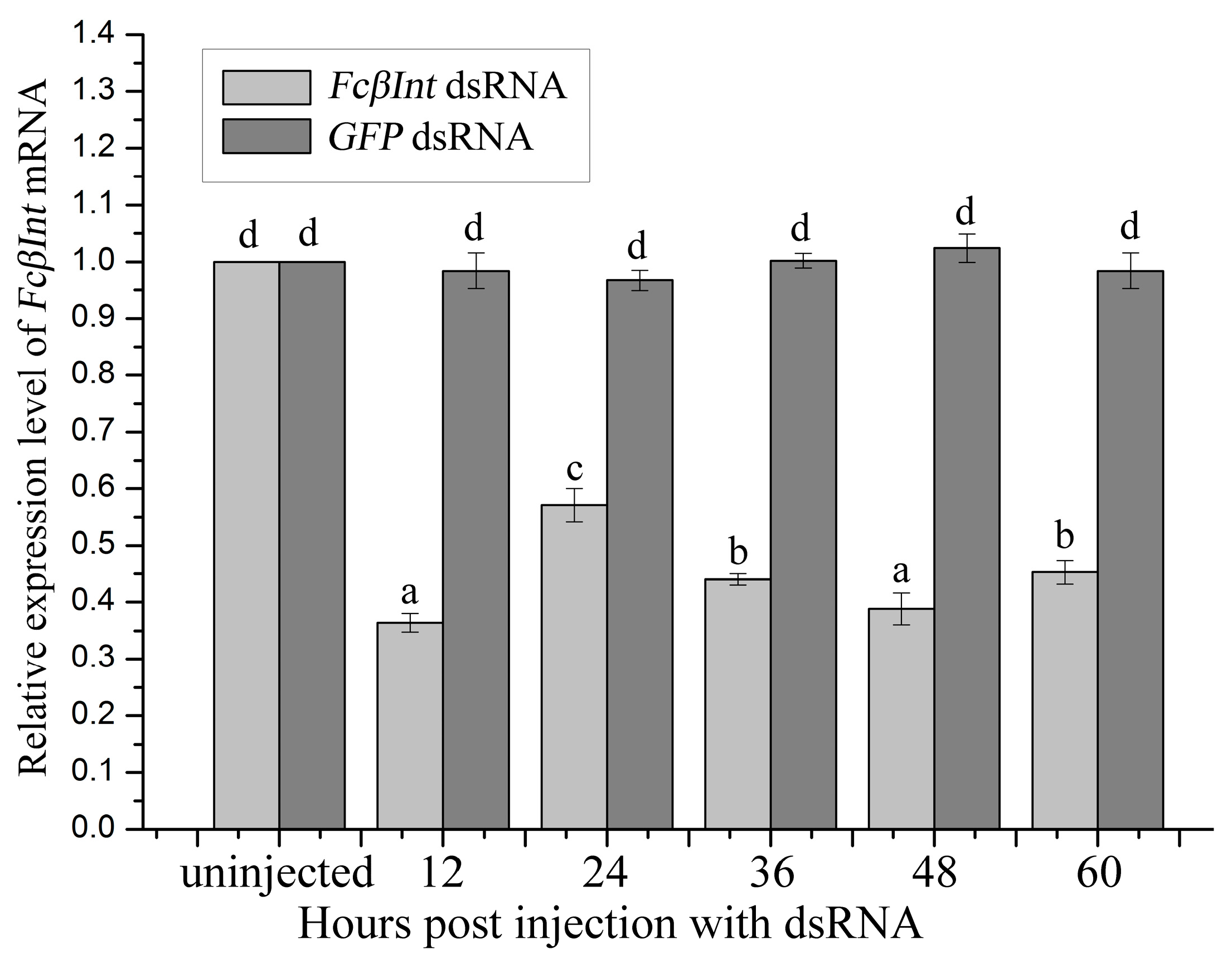
2.6. Gene Silence of FcβInt by dsRNA-Mediated RNAi
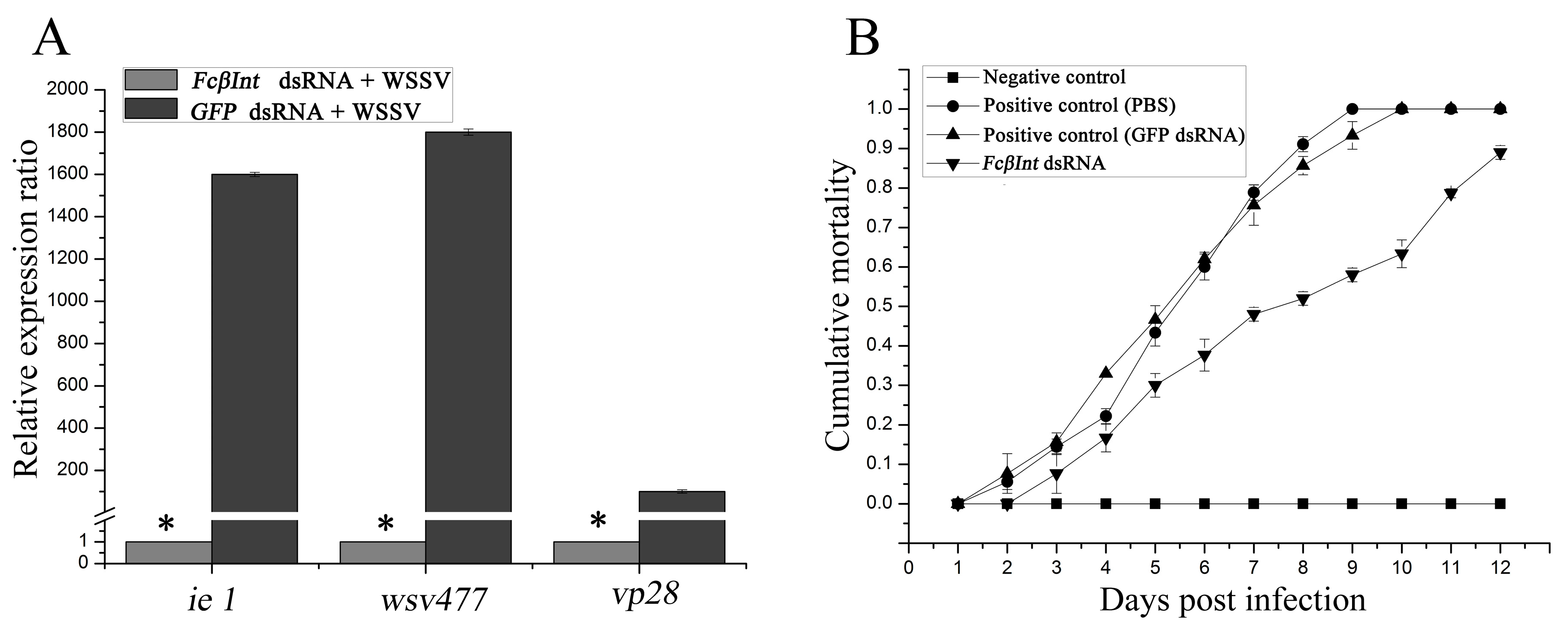
2.7. Inhibition of WSSV Infection after Silencing the FcβInt Gene
3. Discussion
4. Materials and Methods
4.1. Expression of Five WSSV Envelope Proteins
4.2. Detection of Binding Specificity of rFcβInt-ER to WSSV Envelope Proteins by Far-Western
4.3. Preparation of HmFc and Digoxigenin (DIG) Labeled WSSV (WSSV-DIG)
4.4. Blocking Effects of Anti-rFcβInt-ER Antibodies In Vitro and In Vivo
4.5. Preparation of Eukaryotic Expression Plasmid PcDNA3.1-FcβInt-ER (PFcβInt-ER)
4.6. PCR Detection of Plasmid DNA and In Vivo Transcriptional Analysis of PFcβInt-ER in F. chinensis
4.7. Neutralization Assay In Vivo Using PFcβInt-ER
4.8. Gene Silencing of FcβInt
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Arnaout, M.A.; Mahalingam, B.; Xiong, J.P. Integrin structure, allostery, and bidirectional signaling. Annu. Rev. Cell Dev. Biol. 2005, 21, 381–410. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.L. Evolution of the integrin α and β protein families. J. Mol. Evol. 2001, 52, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Takada, Y.; Ye, X.; Simon, S. The integrins. Genome. Biol. 2007, 8, 215. [Google Scholar] [CrossRef] [PubMed]
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Wickham, T.J.; Mathias, P.; Cheresh, D.A.; Nemerow, G.R. Integrins αvβ3 and αvβ5 promote adenovirus internalization but not virus attachment. Cell 1993, 73, 309–319. [Google Scholar] [CrossRef]
- Mou, D.L.; Wang, Y.P.; Huang, C.X.; Li, G.Y.; Pan, L.; Yang, W.S.; Bai, X.F. Cellular entry of Hantaan virus A9 strain: Specific interactions with β3 integrins and a novel 70kDa protein. Biochem. Biophys. Res. Commun. 2006, 339, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Feire, A.L.; Koss, H.; Compton, T. Cellular integrins function as entry receptors for human cytomegalovirus via a highly conserved disintegrin-like domain. Proc. Natl. Acad. Sci. USA 2004, 101, 15470–15475. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.J.; Almond, J.W. Cell receptors for picornaviruses as determinants of cell tropism and pathogenesis. Trends Microbiol. 1998, 6, 198–202. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Liu, Q.H.; Huang, J. Multiple proteins of White spot syndrome virus involved in recognition of β-integrin. J. Biosci. 2014, 39, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Triantafilou, K.; Triantafilou, M. Mechanisms of integrin-mediated virus attachment and internalization process. Crit. Rev. Immunol. 2001, 21, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Paz, A. White spot syndrome virus: An overview on an emergent concern. Vet. Res. 2010, 41, 43. [Google Scholar] [CrossRef] [PubMed]
- Li, L.J.; Yuan, J.F.; Cai, C.A.; Gu, W.G.; Shi, Z.L. Multiple envelope proteins are involved in white spot syndrome virus (WSSV) infection in crayfish. Arch. Virol. 2006, 151, 1309–1317. [Google Scholar] [CrossRef] [PubMed]
- Li, D.F.; Zhang, M.C.; Yang, H.J.; Zhu, Y.B.; Xu, X. β-integrin mediates WSSV infection. Virology 2007, 368, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Wang, X.; Zhan, W. An integrin β subunit of Chinese shrimp Fenneropenaeus chinensis involved in WSSV infection. Aquaculture 2012, 368, 1–9. [Google Scholar] [CrossRef]
- Ruoslahti, E. RGD and other recognition sequences for integrins. Annu. Rev. Cell Dev. Biol. 1996, 12, 697–715. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Zhang, X.; Lin, Q.; Xu, X. Characterization of a novel envelope protein (VP281) of shrimp white spot syndrome virus by mass spectrometry. J. Gen. Virol. 2002, 83, 2385–2392. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P.L.; Nemerow, G.R. Cell integrins: Commonly used receptors for diverse viral pathogens. Trends Microbiol. 2007, 15, 500–507. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; He, J.; Lin, X.; Li, Q.; Pan, D.; Zhang, X.; Xu, X. Complete genome sequence of the shrimp white spot bacilliform virus. J. Virol. 2001, 75, 11811–11820. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.S.; Liu, W.J.; Lee, C.C.; Chou, T.L.; Lee, Y.T.; Wu, T.S.; Wang, A.H.J. A 3D model of the membrane protein complex formed by the white spot syndrome virus structural proteins. PLoS ONE 2010, 5, e10718. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.H.; Zhang, X.L.; Ma, C.Y.; Liang, Y.; Huang, J. VP37 of white spot syndrome virus interact with shrimp cells. Lett. Appl. Microbiol. 2009, 48, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wang, L.; Zhang, X. Identification of white spot syndrome virus (WSSV) envelope proteins involved in shrimp infection. Virology 2005, 332, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xie, X.; Yang, F. Identification and characterization of a prawn white spot syndrome virus gene that encodes an envelope protein VP31. Virology 2005, 340, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lin, Z.; Xu, L.; Yang, F. The RGD motif in VP31 of white spot syndrome virus is involved in cell adhesion. Arch. Virol. 2011, 156, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Gao, Q.; Liang, Y.; Li, C.; Liu, C.; Huang, J. Shrimp arginine kinase being a binding protein of WSSV envelope protein VP31. Chin. J. Oceanol. Limnol. 2016, 34, 1287–1296. [Google Scholar] [CrossRef]
- Li, L.; Lin, S.; Yang, F. Characterization of an envelope protein (VP110) of white spot syndrome virus. J. Gen. Virol. 2006, 87, 1909–1915. [Google Scholar] [CrossRef] [PubMed]
- Van Hulten, M.C.; Witteveldt, J.; Snippe, M.; Vlak, J.M. White spot syndrome virus envelope protein VP28 is involved in the systemic infection of shrimp. Virology 2001, 285, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Nelsen-Salz, B.; Eggers, H.J.; Zimmermann, H. Integrin αvβ3 (vitronectin receptor) is a candidate receptor for the virulent echovirus 9 strain Barty. J. Gen. Virol. 1999, 80, 2311–2313. [Google Scholar] [CrossRef] [PubMed]
- Guglielmi, K.M.; Johnson, E.M.; Stehle, T.; ADermody, T.S. Atachment and cell entry of mammalian orthoreovirus. In Reoviruses: Entry, Assembly and Morphogenesis; Springer Verlag: Berlin, Germany, 2006; pp. 1–38. [Google Scholar]
- Shen, B.; Delaney, M.K.; Du, X. Inside-out, outside-in, and inside–outside-in: G protein signaling in integrin-mediated cell adhesion, spreading, and retraction. Curr. Opin. Cell Biol. 2012, 24, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shao, Y.; Li, C.; Lv, Z.; Wang, H.; Zhang, W.; Zhao, X. A β-integrin from sea cucumber Apostichopus japonicus exhibits LPS binding activity and negatively regulates coelomocyte apoptosis. Fish Shellfish Immunol. 2016, 52, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.M.; Breuer, L.N.; Zhuang, S.; Anderson, S.A.; Nardi, J.B.; Kanost, M.R. A hemocyte-specific integrin required for hemocytic encapsulation in the tobacco hornworm, Manduca sexta. Insect Biochem. Mol. 2005, 35, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, H.; Li, D.; Xu, X. A novel focal adhesion kinase from Marsupenaeus japonicus and its response to WSSV infection. Dev. Comp. Immunol. 2009, 33, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhan, W.; Sheng, X. Neutralizing assay of monoclonal antibodies against white spot syndrome virus (WSSV). Aquaculture 2007, 272, 216–222. [Google Scholar] [CrossRef]
- Lo, C.F.; Leu, J.H.; Ho, C.H.; Chen, C.H.; Peng, S.E.; Chen, Y.T.; Chou, C.M.; Yeh, P.Y.; Huang, H.Y.; Wang, C.H.; et al. Detection of baculovirus associated with white spot syndrome (WSBV) in penaeid shrimps using polymerase chain reaction. Dis. Aquat. Org. 1996, 25, 133–141. [Google Scholar] [CrossRef]
- Zhong, R.; Tang, X.; Zhan, W.; Xing, J.; Sheng, X. Expression kinetics of β-integrin in Chinese shrimp (Fenneropenaeus chinensis) hemocytes following infection with white spot syndrome virus. Fish Shellfish Immunol. 2013, 35, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Tang, X.; Zhan, W. Interaction between white spot syndrome virus VP26 and hemocyte membrane of shrimp, Fenneropenaeus chinensis. Aquaculture 2011, 314, 13–17. [Google Scholar] [CrossRef]
- Lin, Y.C.; Chen, J.C.; Chen, Y.Y.; Liu, C.H.; Cheng, W.; Hsu, C.H.; Tsui, W.C. Characterization of white shrimp Litopenaeus vannamei integrin β and its role in immunomodulation by dsRNA-mediated gene silencing. Dev. Comp. Immunol. 2013, 40, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Tang, X.; Zhan, W. Cloning and characterization of cytoplasmic dynein intermediate chain in Fenneropenaeus chinensis and its essential role in white spot syndrome virus infection. Fish Shellfish Immunol. 2014, 39, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Yingvilasprasert, W.; Supungul, P.; Tassanakajon, A. PmTBC1D20, a Rab GTPase-activating protein from the black tiger shrimp, Penaeus monodon, is involved in white spot syndrome virus infection. Dev. Comp. Immunol. 2014, 42, 302–310. [Google Scholar] [CrossRef] [PubMed]

| Sequence Information | Primers Name | Sequence of Primers (5′-3′) |
|---|---|---|
| CDs of WSSV envelope Protein vp28 | VP28-F | CGGGATCCATGGATCTTTCTTTCACTC |
| VP28-R | GGCCTCGAGCTCGGTCTCAGTGCCAG | |
| CDs of WSSV envelope Protein vp31 | VP31-F | CGGGATCCATGTCTAATGGCGCAAC |
| VP31-R | GGCCTCGAGCTCCTCCTTAAAAGCAGT | |
| CDs of WSSV envelope Protein vp37 | VP37-F | CGGGATCCATGGCGGTAAACTTGG |
| VP37-R | GGCCTCGAGTGTCCAACAATTTAAAAAG | |
| Partial sequence of WSSV envelope Protein vp110 including RGD motif | VP110-F | CGGGATCCACCCACAAAGGACCACC |
| VP110-R | GGCCTCGAGGTCCCTTATTTCTTCCAG | |
| Partial sequence of WSSV envelope Protein vp187 including RGD motif | VP187-F | CGGGATCCGACGACGTTACAAATTTAC |
| VP187-R | GGCCTCGAGCTGAGAGAGGGCACCCGAGC | |
| Universal primer set of PcDNA3.1 plasmid | T7-F | TAATACGACTCACTATAGGG |
| BGH-R | TAGAAGGCACAGTCGAGG | |
| Gene silencing of FcβInt (FcβInt dsRNA) | FcβIntRNAiT7-F | taatacgactcactatagggGTGTTCTGGTCACGGGACTT |
| FcβIntRNAi-R | TGAATGTGTTGGTTGCAGGT | |
| FcβIntRNAi-F | GTGTTCTGGTCACGGGACTT | |
| FcβIntRNAiT7-R | taatacgactcactatagggTGAATGTGTTGGTTGCAGGT | |
| Gene silencing control (GFP dsRNA) | eGFPRNAiT7-F | taatacgactcactatagggGACGTAAACGGCCACAAGTT |
| eGFPRNAi-R | TGTTCTGCTGGTAGTGGTCG | |
| eGFPRNAi-F | GACGTAAACGGCCACAAGTT | |
| eGFPRNAiT7-R | taatacgactcactatagggTGTTCTGCTGGTAGTGGTCG | |
| QPCR for FcβInt | FcβInt-QF | GACCCGCTGAGTGATGTTTC |
| FcβInt-QR | CTTGAACTGCGTCGTGAGG | |
| QPCR for β-actin | β-actin-QF | GAAGTAGCCGCCCTGGTTG |
| β-actin-QR | GGATACCTCGCTTGCTCTGG | |
| QPCR for ie1 | ie1-QF | AGCAAGTGGAGGTGCTATGT |
| ie1-QR | CCATGTCGATCAGTCTCTTC | |
| QPCR for wsv477 | wsv477-QF | GGCCAAGTCATGGAGATCTA |
| wsv477-QR | CCATCCACTTGGTTGCAGTA | |
| QPCR for vp28 | vp28-QF | GGGAACATTCAAGGTGTGGA |
| vp28-QR | GGTGAAGGAGGAGGTGTTGG |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, X.; Zhai, F.; Sheng, X.; Xing, J.; Zhan, W. The Roles of β-Integrin of Chinese Shrimp (Fenneropenaeus chinensis) in WSSV Infection. Int. J. Mol. Sci. 2017, 18, 1465. https://doi.org/10.3390/ijms18071465
Tang X, Zhai F, Sheng X, Xing J, Zhan W. The Roles of β-Integrin of Chinese Shrimp (Fenneropenaeus chinensis) in WSSV Infection. International Journal of Molecular Sciences. 2017; 18(7):1465. https://doi.org/10.3390/ijms18071465
Chicago/Turabian StyleTang, Xiaoqian, Fude Zhai, Xiuzhen Sheng, Jing Xing, and Wenbin Zhan. 2017. "The Roles of β-Integrin of Chinese Shrimp (Fenneropenaeus chinensis) in WSSV Infection" International Journal of Molecular Sciences 18, no. 7: 1465. https://doi.org/10.3390/ijms18071465




