Autophagy Regulates Proteasome Inhibitor-Induced Pigmentation in Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells
Abstract
:1. Introduction
2. Results
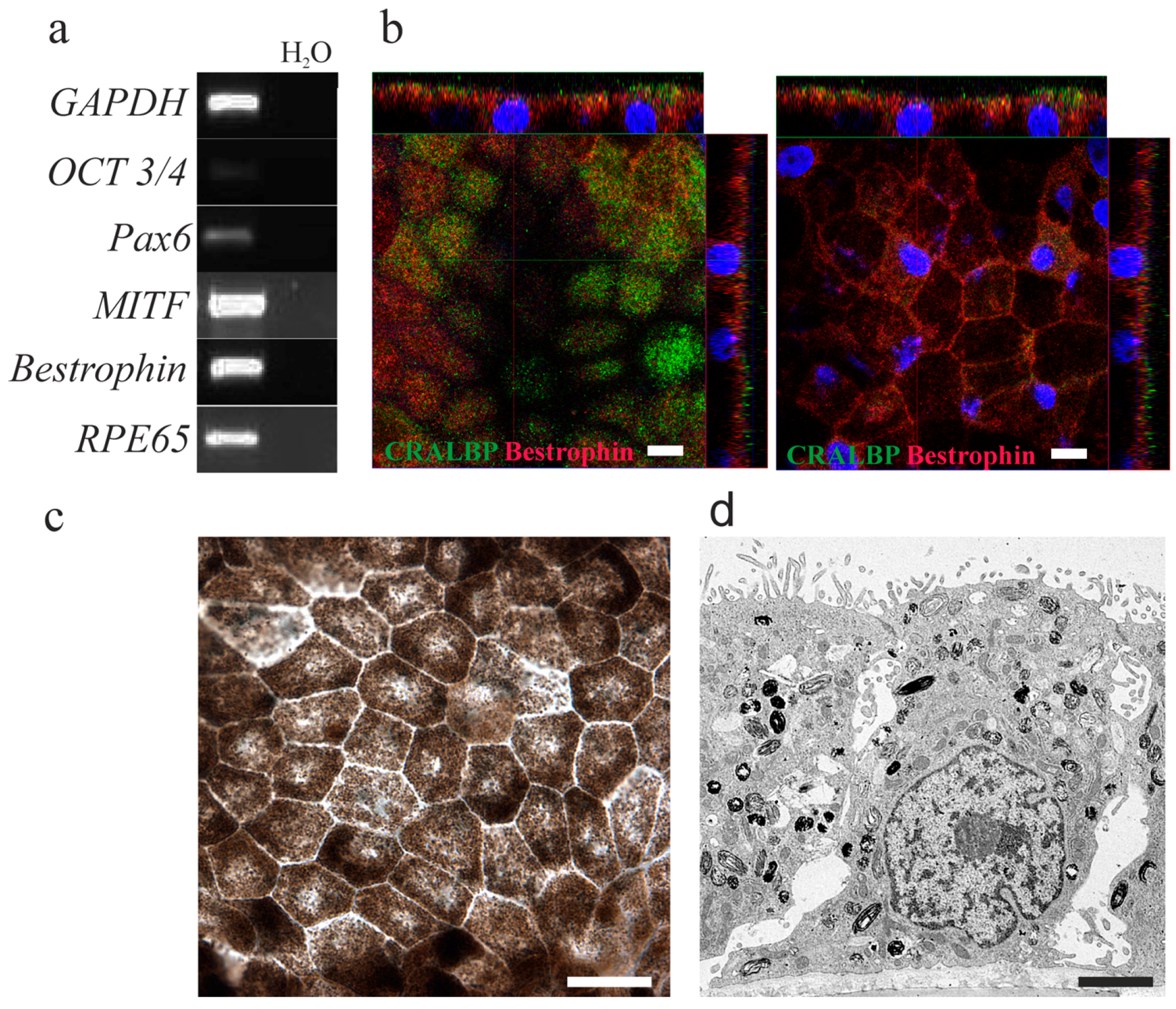
2.1. Human Embryonic Stem Cells-Derived Retinal Pigment Epithelial Cells Show the Typical Retinal Pigment Epithelial Phenotype and Express Retinal Pigment Epithelial Specific Genes
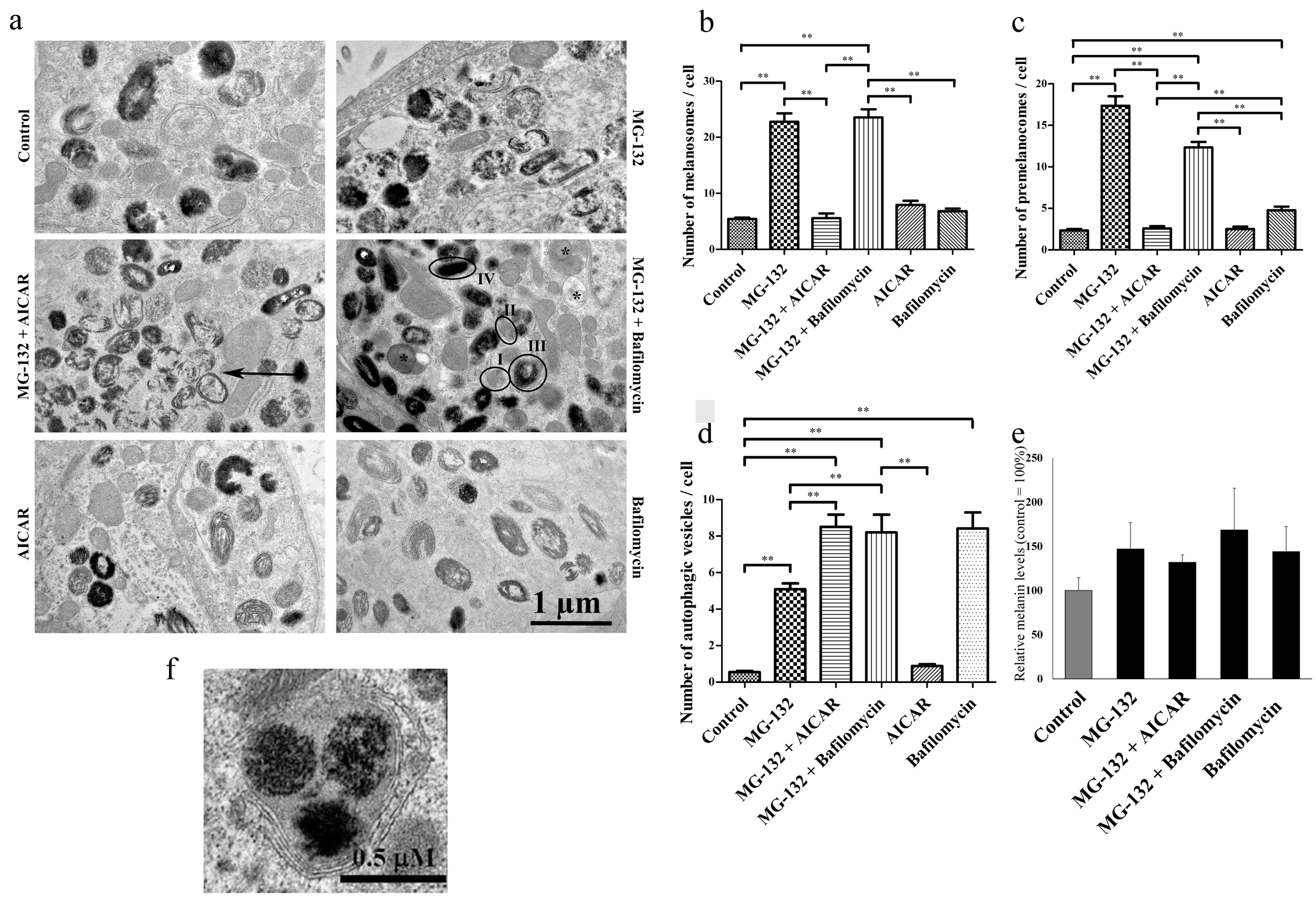
2.2. Proteasomes and Autophagy Regulate Amount of Melanosomes
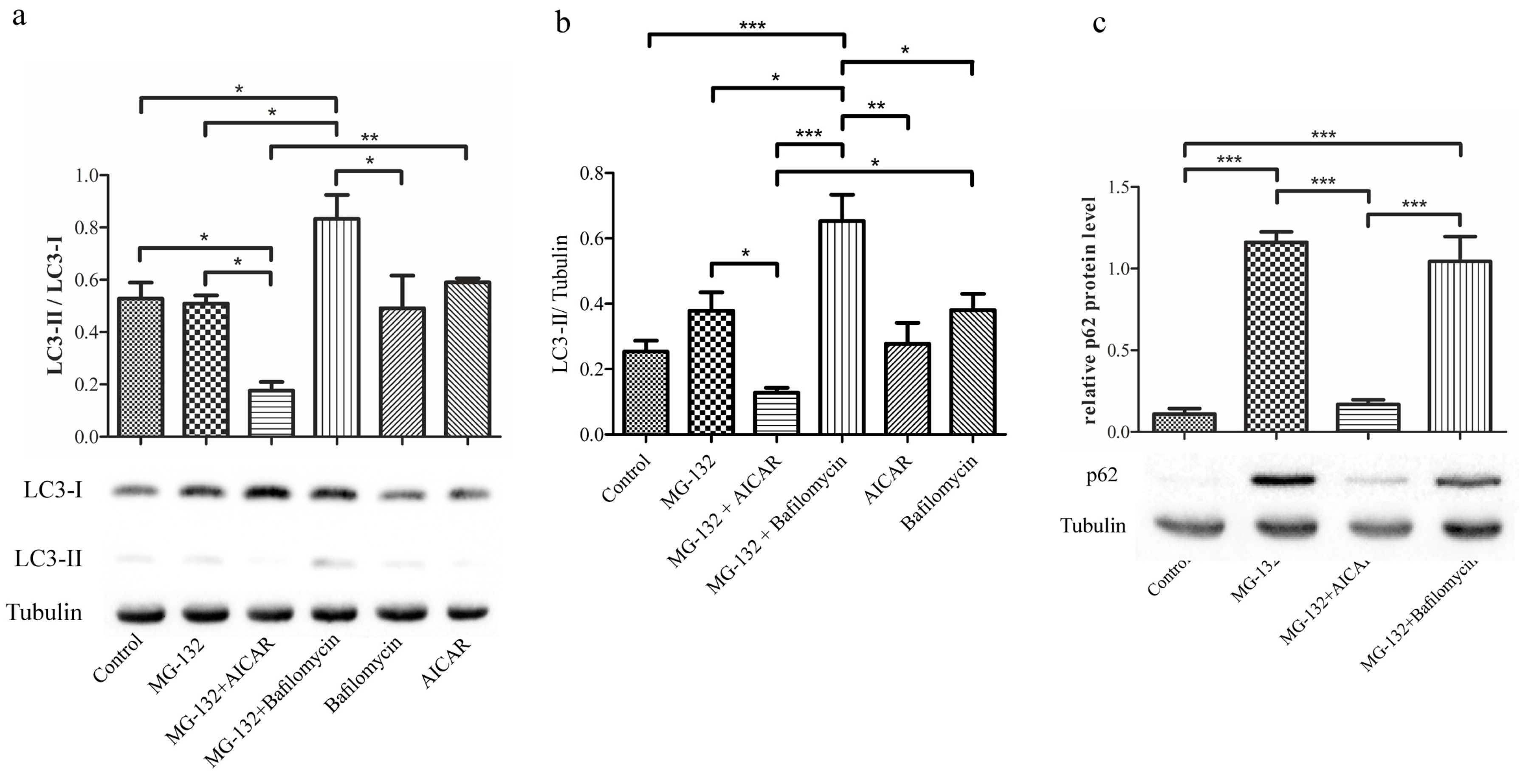
2.3. 5-Aminoimidazole-4-carboxamide Ribonucleotide Decreases Amount of Microtubule-Associated Protein 1A/1B-Light Chain 3 and Sequestosome-1 during Proteasome Inhibition
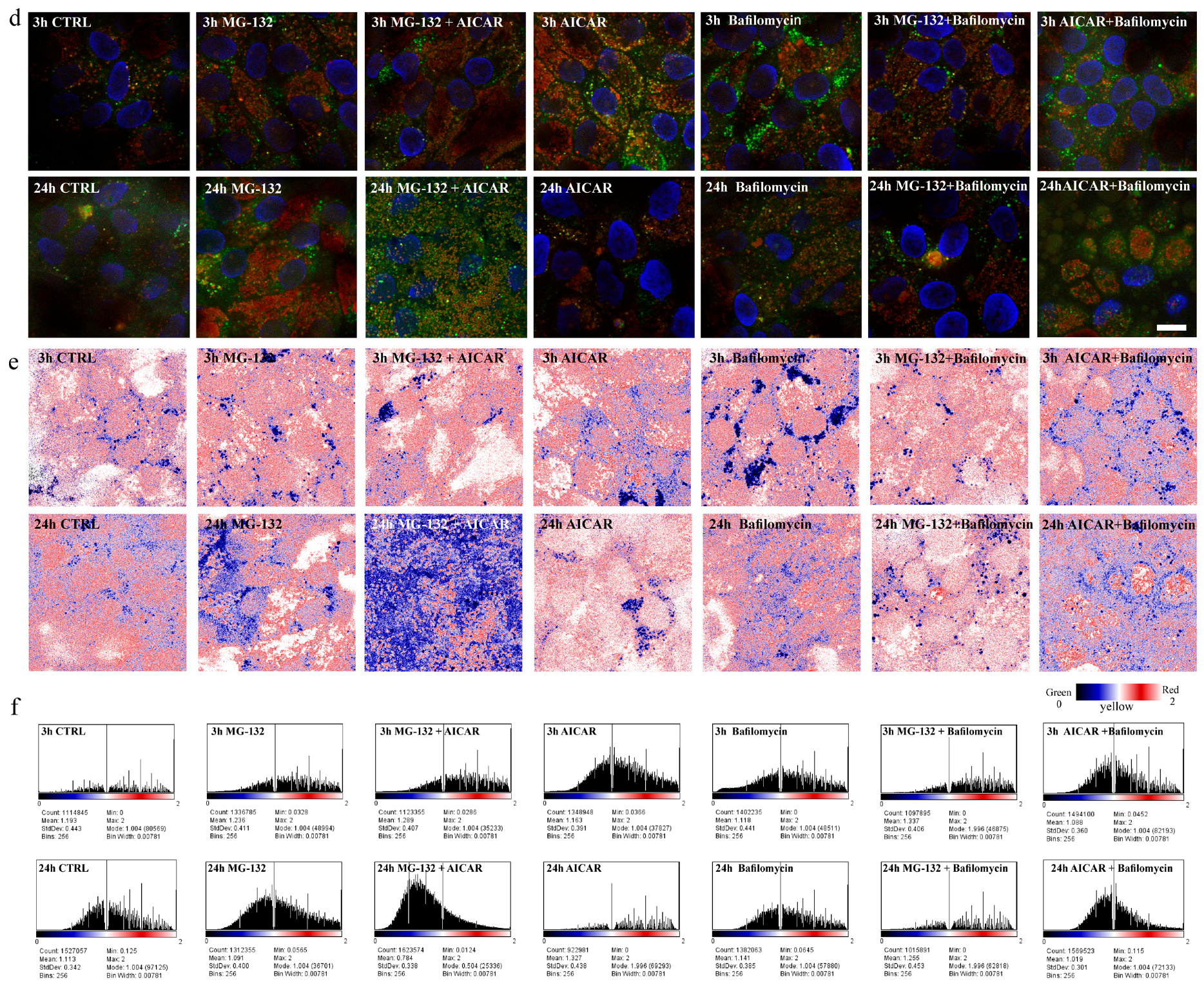
2.4. 5-Aminoimidazole-4-carboxamide Ribonucleotide Treatment Induces Autophagy Flux with Proteasome Inhibition
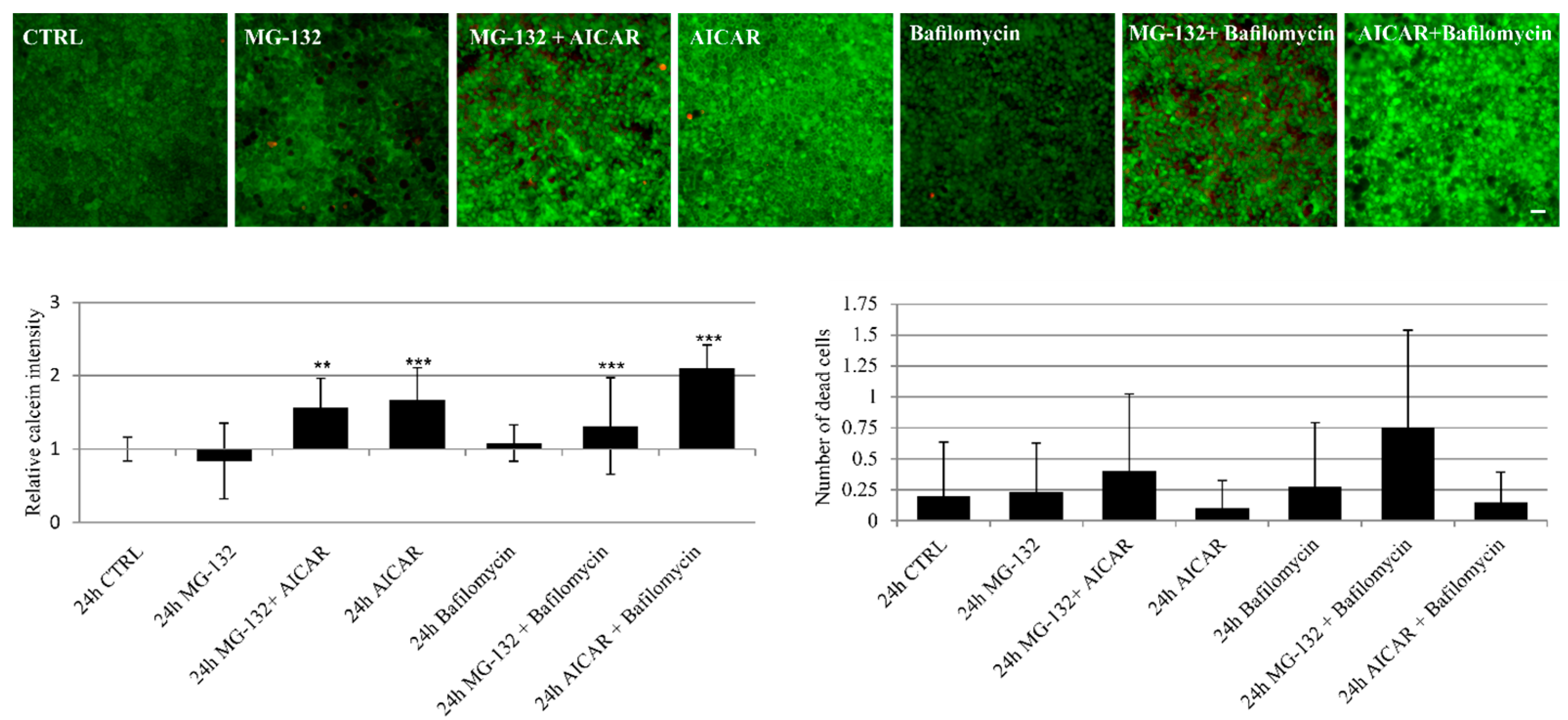
2.5. Proteasome Inhibition and Bafilomycin A1 Induces Melanogenesis
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatments
4.2. Immunofluorescence Labelling
4.3. RNA Extraction, Complementary DNA Synthesis and Reverse Transcription-Polymerase Chain Reaction
4.4. Transmission Electron Microscopy
4.5. Melanin Quantitation by Absorption Spectroscopy
4.6. Western Blot
4.7. Confocal Microscopy Analysis
4.8. Cell Viability
4.9. Ethical Issues
4.10. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Strauss, O. The retinal pigment epithelium in visual function. Physiol. Rev. 2005, 85, 845–881. [Google Scholar] [CrossRef] [PubMed]
- Ryhänen, T.; Hyttinen, J.M.; Kopitz, J.; Rilla, K.; Kuusisto, E.; Mannermaa, E.; Viiri, J.; Holmberg, C.I.; Immonen, I.; Meri, S.; et al. Crosstalk between Hsp70 molecular chaperone, lysosomes and proteasomes in autophagy-mediated proteolysis in human retinal pigment epithelial cells. J. Cell. Mol. Med. 2009, 13, 3616–3631. [Google Scholar] [CrossRef] [PubMed]
- Viiri, J.; Hyttinen, J.M.; Ryhänen, T.; Rilla, K.; Paimela, T.; Kuusisto, E.; Siitonen, A.; Urtti, A.; Salminen, A.; Kaarniranta, K. P62/sequestosome 1 as a regulator of proteasome inhibitor-induced autophagy in human retinal pigment epithelial cells. Mol. Vis. 2010, 16, 1399–1414. [Google Scholar] [PubMed]
- Höhn, A.; Jung, T.; Grimm, S.; Catalgol, B.; Weber, D.; Grune, T. Lipofuscin inhibits the proteasome by binding to surface motifs. Free Radic. Biol. Med. 2011, 50, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Viiri, J.; Amadio, M.; Marchesi, N.; Hyttinen, J.M.; Kivinen, N.; Sironen, R.; Rilla, K.; Akhtar, S.; Provenzani, A.; D’Agostino, V.G.; et al. Autophagy activation clears ELAVL1/HuR-mediated accumulation of SQSTM1/p62 during proteasomal inhibition in human retinal pigment epithelial cells. PLoS ONE 2013, 8, e69563. [Google Scholar] [CrossRef] [PubMed]
- Parzych, K.R.; Klionsky, D.J. An overview of autophagy: Morphology, mechanism, and regulation. Antioxid. Redox Signal. 2014, 20, 460–473. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Zhao, H.; Martinez, J.; Doggett, T.A.; Kolesnikov, A.V.; Tang, P.H.; Ablonczy, Z.; Chan, C.C.; Zhou, Z.; Green, D.R.; et al. Noncanonical autophagy promotes the visual cycle. Cell 2013, 154, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Valapala, M.; Wilson, C.; Hose, S.; Bhutto, I.A.; Grebe, R.; Dong, A.; Greenbaum, S.; Gu, L.; Sengupta, S.; Cano, M.; et al. Lysosomal-mediated waste clearance in retinal pigment epithelial cells is regulated by CRYBA1/betaA3/A1-crystallin via V-ATPase-MTORC1 signaling. Autophagy 2014, 10, 480–496. [Google Scholar] [CrossRef] [PubMed]
- Holz, F.G.; Schütt, F.; Kopitz, J.; Eldred, G.E.; Kruse, F.E.; Völcker, H.E.; Cantz, M. Inhibition of lysosomal degradative functions in RPE cells by a retinoid component of lipofuscin. Investig. Ophthalmol. Vis. Sci. 1999, 40, 737–743. [Google Scholar]
- Bergmann, M.; Schutt, F.; Holz, F.G.; Kopitz, J. Inhibition of the ATP-driven proton pump in RPE lysosomes by the major lipofuscin fluorophore A2-E may contribute to the pathogenesis of age-related macular degeneration. FASEB J. 2004, 18, 562–564. [Google Scholar] [CrossRef] [PubMed]
- Lakkaraju, A.; Finnemann, S.C.; Rodriquez-Boulan, E. The lipofuscin fluorophore A2E perturbs cholesterol metabolism in retinal pigment epithelial cells. Proc. Natl. Acad. Sci. USA 2007, 104, 11026–11031. [Google Scholar] [CrossRef] [PubMed]
- Kaemmerer, E.; Schutt, F.; Krohne, T.U.; Holz, F.G.; Kopitz, J. Effects of lipid peroxidation-related protein modifications on RPE lysosomal functions and POS phagocytosis. Investig. Ophthalmol. Vis. Sci. 2007, 48, 1342–1347. [Google Scholar] [CrossRef] [PubMed]
- Krohne, T.U.; Kaemmerer, E.; Holz, F.G.; Kopitz, J. Lipid peroxidation products reduce lysosomal protease activities in human retinal pigment epithelial cells via two different mechanisms of action. Exp. Eye Res. 2010, 90, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Finnemann, S.C.; Leung, L.W.; Rodriguez-Boulan, E. The lipofuscin component A2E selectively inhibits phagolysosomal degradation of photoreceptor phospholipid by the retinal pigment epithelium. Proc. Natl. Acad. Sci. USA 2002, 99, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cano, M.; Handa, J.T. P62 provides dual cytoprotection against oxidative stress in the retinal pigment epithelium. Biochim. Biophys. Acta 2014, 1843, 1248–1258. [Google Scholar]
- Simon, J.D.; Hong, L.; Peles, D.N. Insights into melanosomes and melanin from some interesting spatial and temporal properties. J. Phys. Chem. B 2008, 112, 13201–13217. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.; Lamah, T.; Kokkinou, D.; Bartz-Schmidt, K.U.; Schraermeyer, U. Melanin protects choroidal blood vessels against light toxicity. Z. Naturforsch. 2006, 61, 427–433. [Google Scholar] [CrossRef]
- Wang, Z.; Dillon, J.; Gaillard, E.R. Antioxidant properties of melanin in retinal pigment epithelial cells. Photochem. Photobiol. 2006, 82, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Sarna, T.; Burke, J.M.; Korytowski, W.; Rόzanowska, M.; Skumatz, C.M.; Zareba, A.; Zareba, M. Loss of melanin from human RPE with aging: Possible role of melanin photooxidation. Exp. Eye Res. 2003, 76, 89–98. [Google Scholar] [CrossRef]
- Zareba, M.; Raciti, M.W.; Henry, M.M.; Sarna, T.; Burke, J.M. Oxidative stress in ARPE-19 cultures: Do melanosomes confer cytoprotection? Free Radic. Biol. Med. 2006, 40, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Burke, J.M.; Henry, M.M.; Zareba, M.; Sarna, T. Photobleaching of melanosomes from retinal pigment epithelium: I. effects on protein oxidation. Photochem. Photobiol. 2007, 83, 920–924. [Google Scholar] [CrossRef] [PubMed]
- Rozankowski, B.; Burke, J.M.; Boulton, M.E.; Sarna, T.; Rozanowska, M. Human RPE melanosomes protect from photosensitized and iron-mediated oxidation but become pro-oxidant in the presence of iron upon photodegradation. Investig. Ophthalmol. Vis. Sci. 2008, 49, 2838–2847. [Google Scholar] [CrossRef] [PubMed]
- Biesemeier, A.; Yoeruek, E.; Eibl, O.; Schraermeyer, U. Iron accumulation in Bruch’s membrane and melanosomes of donor eyes with age-related macular degeneration. Exp. Eye Res. 2015, 137, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Jung, T.; Bader, N.; Grune, T. Lipofuscin: Formation, distribution, and metabolic consequences. Ann. N. Y. Acad. Sci. 2007, 1119, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Kaarniranta, K.; Hyttinen, J.; Ryhänen, T.; Viiri, J.; Paimela, T.; Toropainen, E.; Sorri, I.; Salminen, A. Mechanisms of protein aggregation in the retinal pigment epithelial cells. Front. Biosci. 2010, 2, 1374–1384. [Google Scholar] [CrossRef]
- Julien, S.; Schraermeyer, U. Lipofuscin can be eliminated from the retinal pigment epithelium of monkeys. Neurobiol. Aging 2012, 33, 2390–2397. [Google Scholar] [CrossRef] [PubMed]
- Nociari, M.M.; Lehmann, G.L.; Perez Bay, A.E.; Rady, R.A.; Jiang, Z.; Goicochea, S.; Schreiner, R.; Warren, J.D.; Shan, J.; Adam de Beaumais, S.; et al. Beta cyclodextrins bind, stabilize, and remove lipofuscin bisretinoids from retinal pigment epithelium. Proc. Natl. Acad. Sci. USA 2014, 111, E1402–E1408. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, A.; Oka, M.; Okada, T.; Nakamura, S.; Ueyama, T.; Saito, N.; Hearing, V.J.; Ichihashi, M.; Nishigori, C. Down-regulation of melanogenesis by phospholipase D2 through ubiquitin proteasome-mediated degradation of tyrisinase. J. Biol. Chem. 2004, 279, 27774–27780. [Google Scholar] [CrossRef] [PubMed]
- Ho, H.; Ganesan, A.K. The pleiotropic roles of autophagy regulators in melanogenesis. Pigment Cell Melanoma Res. 2011, 24, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Kalie, E.; Razi, M.; Tooze, S.A. ULK1 regulates melanin levels in MNT-1 cells independently of mTORC1. PLoS ONE 2013, 8, e75313. [Google Scholar] [CrossRef] [PubMed]
- Murase, D.; Hachiya, A.; Takano, K.; Hicks, R.; Visscher, M.O.; Kitahara, T.; Hase, T.; Takema, Y.; Yoshimori, T. Autophagy has a significant role in determining skin color by regulating melanosome degradation in keratinocytes. J. Investig. Dermatol. 2013, 133, 2416–2424. [Google Scholar] [CrossRef] [PubMed]
- Sorkio, A.; Hongisto, H.; Kaarniranta, K.; Uusitalo, H.; Juuti-Uusitalo, K.; Skottman, H. Structure and barrier properties of human embryonic stem cell-derived retinal pigment epithelial cells are affected by extracellular matrix protein coating. Tissue Eng. Part A 2014, 20, 622–634. [Google Scholar] [CrossRef] [PubMed]
- Vaajasaari, H.; Ilmarinen, T.; Juuti-Uusitalo, K.; Rajala, K.; Onnela, N.; Narkilahti, S.; Suuronen, R.; Hyttinen, J.; Uusitalo, H.; Skottman, H. Toward the defined and xeno-free differentiation of functional human pluripotent stem cell-derived retinal pigment epithelial cells. Mol. Vis. 2011, 17, 558–575. [Google Scholar] [PubMed]
- Carr, A.J.; Vugler, A.; Lawrence, J.; Chen, L.L.; Ahmado, A.; Chen, F.K.; Semo, M.; Gias, C.; da Cruz, L.; Moore, H.D.; et al. Molecular characterization and functional analysis of phagocytosis by human embryonic stem cell-derived RPE cells using a novel human retinal assay. Mol. Vis. 2009, 15, 283–295. [Google Scholar] [PubMed]
- Klimanskaya, I.; Hipp, J.; Rezai, K.A.; West, M.; Atala, A.; Lanza, R. Derivation and comparative assessment of retinal pigment epithelium from human embryonic stem cells using transcriptomics. Cloning Stem Cells 2004, 6, 217–245. [Google Scholar] [CrossRef] [PubMed]
- Hirami, Y.; Osakada, F.; Takahashi, K.; Okita, K.; Yamanaka, S.; Ikeda, H.; Yoshimura, N.; Takahashi, M. Generation of retinal cells from mouse and human induced pluripotent stem cells. Neurosci. Lett. 2009, 458, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Deng, X.; Spee, C.; Sonoda, S.; Hsieh, C.L.; Barron, E.; Pera, M.; Hinton, D.R. Polarized secretion of PEDF from human embryonic stem cell-derived RPE promotes retinal progenitor cell survival. Investig. Ophthalmol. Vis. Sci. 2011, 52, 1573–1585. [Google Scholar] [CrossRef] [PubMed]
- Burke, J.M. Epithelial phenotype and the RPE: Is the answer blowing in the Wnt? Prog. Retin. Eye Res. 2008, 27, 579–595. [Google Scholar] [CrossRef] [PubMed]
- Skottman, H. Derivation and characterization of three new human embryonic stem cell lines in Finland. In Vitro Cell. Dev. Biol. Anim. 2010, 46, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Rimpelä, A.K.; Schmitt, M.; Latonen, S.; Hagström, M.; Antopolsky, M.; Manzanares, J.A.; Kidron, H.; Urtti, A. Drug Distribution to Retinal Pigment Epithelium: Studies on Melanin Binding. Cellular Kinetics, and Single Photon Emission Computed Tomography/Computed Tomography Imaging. Mol. Pharm. 2016, 13, 2977–2986. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Cai, J.; Sun, L.; Li, Y.; Qu, J.; Snider, B.J.; Wu, S. Proteasome inhibitors activate autophagy involving inhibition of PI3K-Akt-mTOR pathway as an anti-oxidation defense in human RPE cells. PLoS ONE 2014, 9, e103364. [Google Scholar] [CrossRef] [PubMed]
- Ohkuma, S.; Shimizu, S.; Noto, M.; Sai, Y.; Kinoshita, K.; Tamura, H. Inhibition of cell growth by bafilomycin A1, a selective inhibitor of vacuolar H+-ATPase. In Vitro Cell. Dev. Biol. Anim. 1993, 29A, 862–866. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Lin, M.Z.; Cheng, D.; Braet, F.; Pollock, C.A.; Chen, X.M. Thioredoxin-interacting protein mediates dysfunction of tubular autophagy in diabetic kidneys through inhibiting autophagic flux. Lab. Investig. 2014, 94, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Piippo, N.; Korkmaz, A.; Hytti, M.; Kinnunen, K.; Salminen, A.; Atalay, M.; Kaarniranta, K.; Kauppinen, A. Decline in cellular clearance systems induces inflammasome signaling in human ARPE-19 cells. Biochim. Biophys. Acta 2014, 1843, 3038–3046. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; De Vries, G.W. Alpha2 but not alpha1 AMP-activated protein kinase mediates oxidative stress-induced inhibition of retinal pigment epithelium cell phagocytosis of photoreceptor outer segments. J. Biol. Chem. 2008, 283, 6744–6751. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.J.; Rhim, J.H.; Jang, I.S.; Kim, G.E.; Park, S.C.; Yeo, E.J. Activation of AMP-activated protein kinase stimulates the nuclear localization of glyceraldehyde 3-phosphate dehydrogenase in human diploid fibroblasts. Exp. Mol. Med. 2010, 42, 254–269. [Google Scholar] [CrossRef] [PubMed]
- Kamao, H.; Mandai, M.; Wakamiva, S.; Ishida, J.; Goto, K.; Ono, T.; Suda, T.; Takahashi, M.; Kirvu, J. Objective evaluation of the degree of pigmentation in human induced pluripotent stem cell-derived RPE. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8309–8318. [Google Scholar] [CrossRef] [PubMed]
- Kayatz, P.; Thumann, G.; Luther, T.T.; Jordan, J.F.; Bartz–Schmidt, K.U.; Esser, P.J.; Schraermeyer, U. Oxidation causes melanin fluorescence. Investig. Ophthalmol. Vis. Sci. 2001, 42, 241–246. [Google Scholar]
- Kaarniranta, K.; Sinha, D.; Blasiak, J.; Kauppinen, A.; Veréb, Z.; Salminen, A.; Boulton, M.E.; Petrovski, G. Autophagy and heterophagy dysregulation leads to retinal pigment epithelium dysfunction and development of age-related macular degeneration. Autophagy 2013, 9, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Watabe, H.; Valencia, J.C.; Yasumoto, K.; Kushimoto, T.; Ando, H.; Muller, J.; Vieira, V.D.; Mizoguchi, M.; Appella, E.; Hearing, V.J. Regulation of tyrosinase processing and trafficking by organellar pH and by proteasome activity. J. Biol. Chem. 2004, 279, 7971–7981. [Google Scholar] [CrossRef] [PubMed]
- Reinisalo, M.; Putula, J.; Mannermaa, E.; Urtti, A.; Honkakoski, P. Regulation of the human tyrosinase gene in retinal pigment epithelium cells: The significance of transcription factor orthodenticle homeobox 2 and its polymorphic binding site. Mol. Vis. 2012, 18, 38–54. [Google Scholar] [PubMed]
- Jeong, H.S.; Choi, H.R.; Yun, H.Y.; Baek, K.J.; Kwon, N.S.; Park, K.C.; Park, D.S. Ceramide PC102 inhibits melanin synthesis via proteasomal degradation of microphthalmia-associated transcription factor and tyrosinase. Mol. Cell. Biochem. 2013, 375, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Yun, H.Y.; Baek, K.J.; Kwon, N.S.; Park, K.C.; Kim, D.S. Okadaic acid suppresses melanogenesis via proteasomal degradation of tyrosinase. Biol. Pharm. Bull. 2013, 36, 1503–1508. [Google Scholar] [CrossRef] [PubMed]
- Yu, A.; Malek, T.R. The proteasome regulates receptor-mediated endocytosis of interleukin-2. J. Biol. Chem. 2001, 276, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Van Kerkhof, P.; Alves dos Santos, C.M.; Sachse, M.; Klumperman, J.; Bu, G.; Strous, G.J. Proteasome inhibitors block a late step in lysosomal transport of selected membrane but not soluble proteins. Mol. Biol. Cell 2001, 12, 2556–2566. [Google Scholar] [CrossRef] [PubMed]
- Onnela, N.; Savolainen, V.; Juuti-Uusitalo, K.; Vaajasaari, H.; Skottman, H.; Hyttinen, J. Electric impedance of human embryonic stem cell-derived retinal pigment epithelium. Med. Biol. Eng. Comput. 2012, 50, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Juuti-Uusitalo, K.; Delporte, C.; Grégoire, F.; Perret, J.; Huhtala, H.; Savolainen, V.; Nymark, S.; Hyttinen, J.; Uusitalo, H.; Willermain, F.; et al. Aquaporin expression and function in human pluripotent stem cell-derived retinal pigmented epithelial cells. Investig. Ophthalmol. Vis. Sci. 2013, 54, 3510–3519. [Google Scholar] [CrossRef] [PubMed]
- Kurz, T.; Karlsson, M.; Brunk, U.T.; Nilsson, S.E.; Frennesson, C. ARPE-19 retinal pigment epithelial cells are highly resistant to oxidative stress and exercise strict control over their lysosomal redox-active iron. Autophagy 2009, 5, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Mitter, S.K.; Song, C.; Qi, X.; Mao, H.; Rao, H.; Akin, D.; Lewin, A.; Grant, M.; Dunn, W., Jr.; Ding, J.; et al. Dysregulated autophagy in the RPE is associated with increased susceptibility to oxidative stress and AMD. Autophagy 2014, 10, 1989–2005. [Google Scholar] [CrossRef] [PubMed]
- Decanini, A.; Nordgaard, C.L.; Feng, X.; Ferrington, D.A.; Olsen, T.W. Changes in select redox proteins of the retinal pigment epithelium in age-related macular degeneration. Am. J. Ophthalmol. 2007, 143, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Kay, P.; Yang, Y.C.; Hiscott, P.; Gray, D.; Maminishkis, A.; Paraoan, L. Age-related changes of cystatin C expression and polarized secretion by retinal pigment epithelium: Potential age-related macular degeneration links. Investig. Ophthalmol. Vis. Sci. 2014, 55, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Czarny, P.; Pawlowska, E.; Bialkowska-Warzecha, J.; Kaarniranta, K.; Blasiak, J. Autophagy in DNA damage response. Int. J. Mol. Sci. 2015, 16, 2641–2662. [Google Scholar] [CrossRef] [PubMed]
- Terluk, M.R.; Kapphahn, R.J.; Soukup, L.M.; Gong, H.; Gallardo, C.; Montezuma, S.R.; Ferrington, D.A. Investigating mitochondria as a target for treating age-related macular degeneration. J. Neurosci. 2015, 35, 7304–7311. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Ren, D. Lysosomal physiology. Annu. Rev. Physiol. 2015, 77, 57–80. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.G.; Leapman, R.D.; Zhang, G.; Lai, B.; Valencia, J.C.; Cardarelli, C.O.; Vieira, V.D.; Hearing, V.J.; Gottesman, M.M. Influence of melanosome dynamics on melanoma drug sensitivity. J. Natl. Cancer. Inst. 2009, 101, 1259–1271. [Google Scholar] [CrossRef] [PubMed]
- Koskela, A.; Reinisalo, M.; Petrovski, G.; Sinha, D.; Olmiere, C.; Karjalainen, R.; Kaarniranta, K. Nutraceutical with resveratrol and omega-3 fatty acids induces autophagy in ARPE-19 cells. Nutrients 2016, 8, 284. [Google Scholar] [CrossRef] [PubMed]
| Analysis | Adherent Culture Period (SD) | Entire Culture Period (SD) |
|---|---|---|
| RT-PCR (n = 4) | d104 (83–112) | d230 (235–244) |
| TEM (n = 4) | d127 (36–194) | d279 (158–404) |
| WB (n = 8) | d100 (51–194) | d299 (244–383) |
| IF (n = 79) | d83 (36–112) | d277 (197–356) |
| Live-dead (n = 2) | d83 (36–112) | d277 (197–356) |
| Antibody Name | Abbreviation | Primary/Secondary | Host | Dilution | Cat | Manufacturer |
|---|---|---|---|---|---|---|
| Anti-cellular retinaldehyde-binding protein | CRALBP | primary | mouse monoclonal | 1:1000 | ab15051 | Abcam, Cambridge, UK |
| Rabbit anti-bestrophin | Bestrophin | primary | rabbit monoclonal | 1:500 | ab14928 | Abcam |
| Anti-mouse IgG Alexa Fluor 488 | - | secondary polyclonal | donkey | 1:800 | A21202 | Molecular Probes, Life Technologies, Paisley, UK |
| Anti-rabbit IgG Alexa Fluor 568 | - | secondary polyclonal | goat | 1:800 | A11011 | Molecular Probes, Life Technologies |
| Gene | Primer Sequences (5′ > 3′) | Tm | |
|---|---|---|---|
| Forward | Reverse | ||
| GAPDH | GTT CGA CAG TCA GCC GCA TC | GGA ATT TGC CAT GGG TGG A | 55 |
| OCT 3/4 | CGTGAAGCTGGAGAAGGAGAAGCTG | AAGGGCCGCAGCTTACACATGTTC | 55 |
| PAX6 | AAC AGA CAC AGC CCT CAC AAA CA | CGG GAA CTT GAA CTG GAA CTG AC | 60 |
| MITF | AAG TCC TGA GCT TGC CAT GT | GGC AGA CCT TGG TTT CCA TA | 52 |
| Bestrophin | GAATTTGCAGGTGTCCCTGT | ATCAGGAGGACGAGGAGGAT | 60 |
| RPE65 | TCC CCA ATA CAA CTG CCA CT | CAC CACC ACA CTC AGA ACT A | 52 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juuti-Uusitalo, K.; Koskela, A.; Kivinen, N.; Viiri, J.; Hyttinen, J.M.T.; Reinisalo, M.; Koistinen, A.; Uusitalo, H.; Sinha, D.; Skottman, H.; et al. Autophagy Regulates Proteasome Inhibitor-Induced Pigmentation in Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells. Int. J. Mol. Sci. 2017, 18, 1089. https://doi.org/10.3390/ijms18051089
Juuti-Uusitalo K, Koskela A, Kivinen N, Viiri J, Hyttinen JMT, Reinisalo M, Koistinen A, Uusitalo H, Sinha D, Skottman H, et al. Autophagy Regulates Proteasome Inhibitor-Induced Pigmentation in Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells. International Journal of Molecular Sciences. 2017; 18(5):1089. https://doi.org/10.3390/ijms18051089
Chicago/Turabian StyleJuuti-Uusitalo, Kati, Ali Koskela, Niko Kivinen, Johanna Viiri, Juha M. T. Hyttinen, Mika Reinisalo, Arto Koistinen, Hannu Uusitalo, Debasish Sinha, Heli Skottman, and et al. 2017. "Autophagy Regulates Proteasome Inhibitor-Induced Pigmentation in Human Embryonic Stem Cell-Derived Retinal Pigment Epithelial Cells" International Journal of Molecular Sciences 18, no. 5: 1089. https://doi.org/10.3390/ijms18051089





