Lignin from Micro- to Nanosize: Applications
Abstract
:1. Introduction
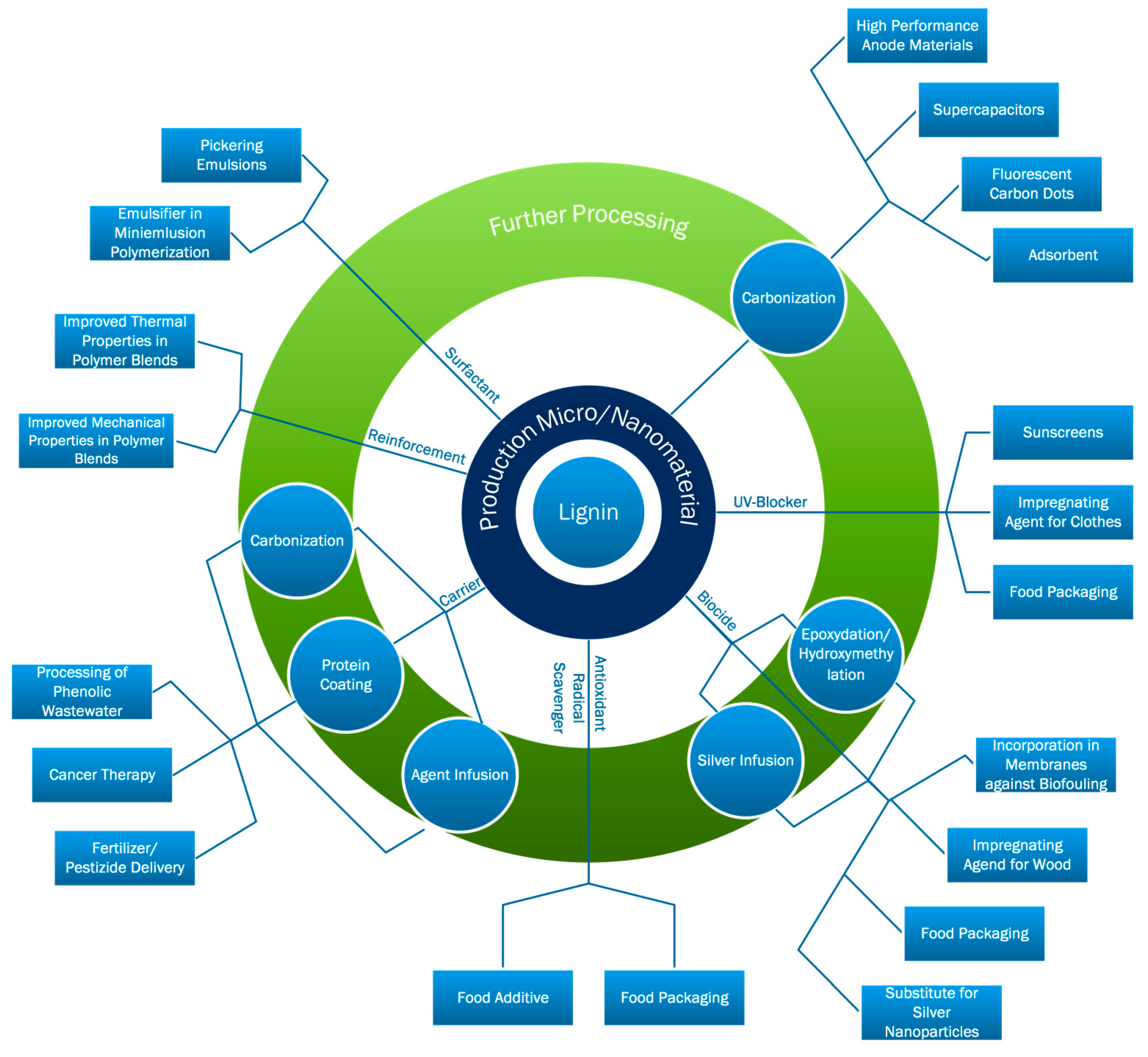
2. Application Overview for Lignin from Micro- to Nanosize
3. Application of Lignin Nanoparticles (LNPs) as Reinforcement
4. Applications of Lignin Nanoparticles as Ultraviolet (UV) Blocker
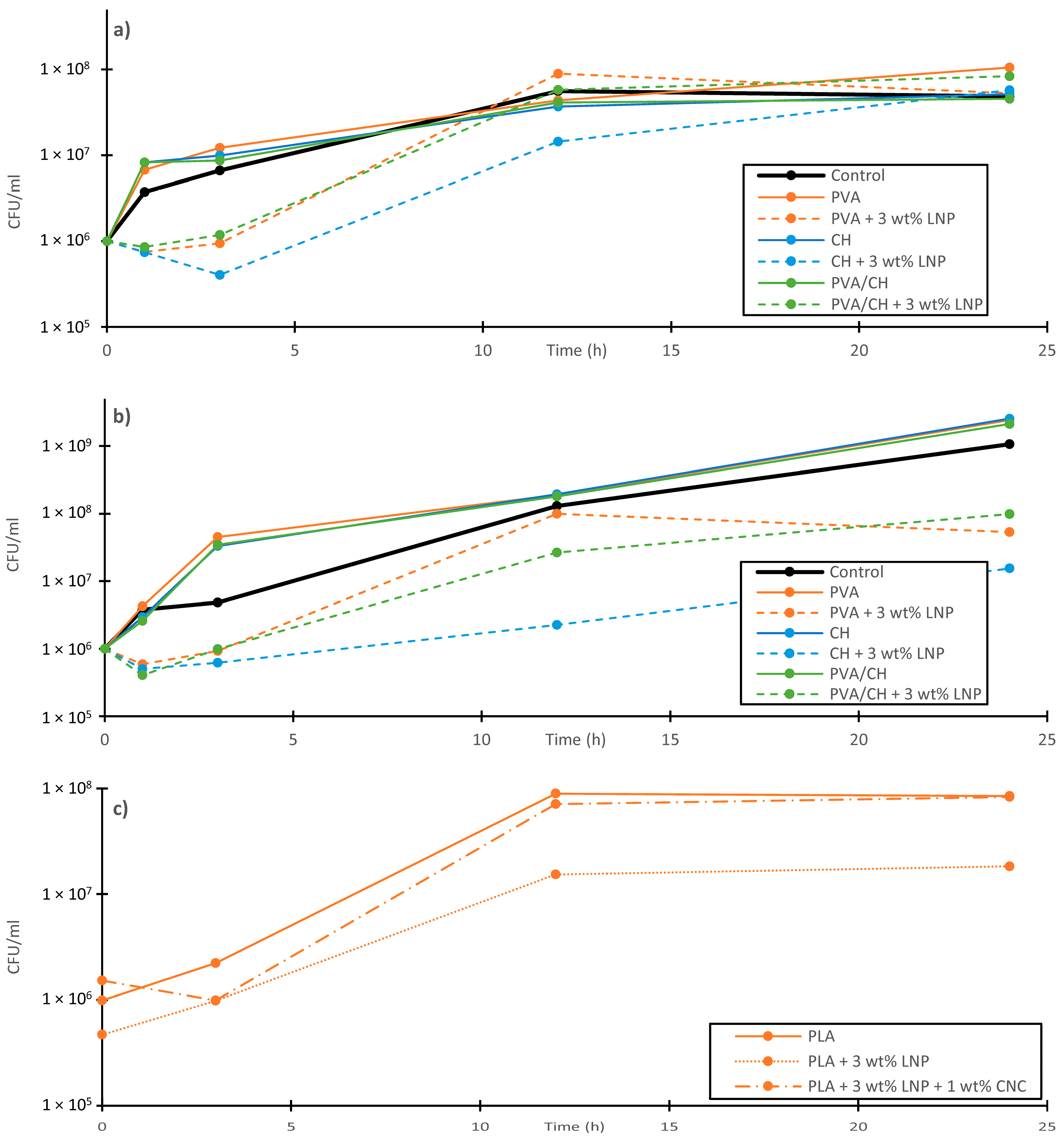
5. Application of Lignin Nanoparticles as Biocide
6. Applications of Lignin Nanoparticles as Antioxidants/Radical Scavengers
7. Application of Lignin Nanoparticles as Surfactants in Pickering Emulsions
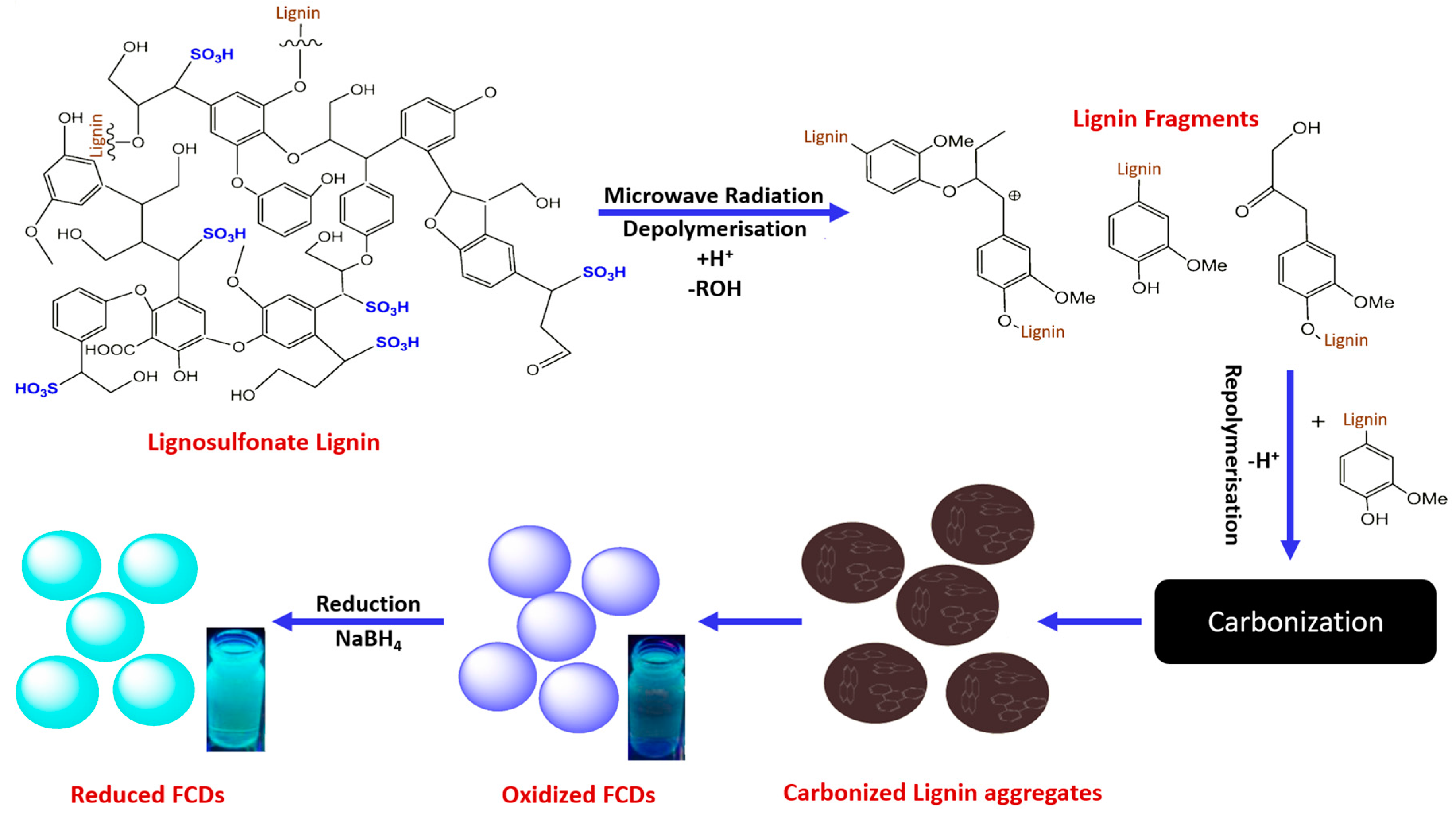
8. Applications of Carbonized Lignin Nano Fibers and Particles
9. Application of Lignin Nanoparticles as Nano- and Microcarrier
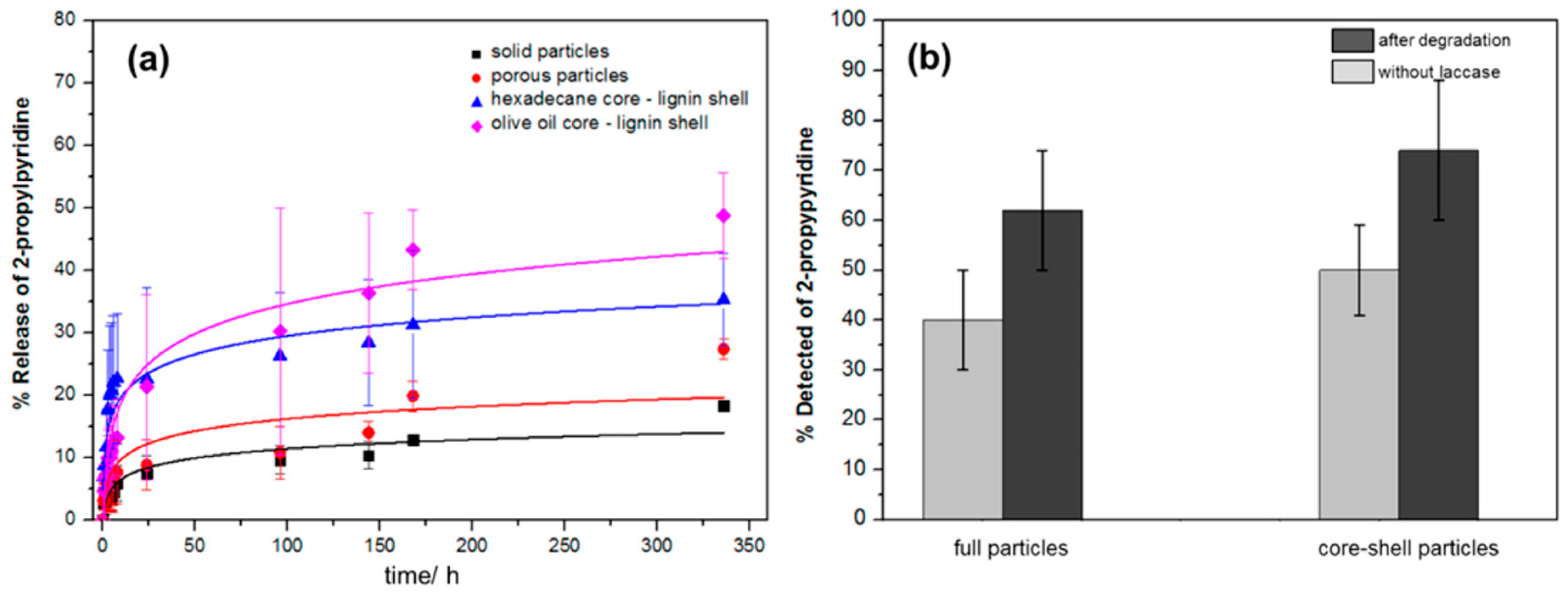
9.1. Capsules, Hollow and Porous Structures
9.2. Solid and Porous Particles/Structures
10. Conclusions and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AL | Alkali lignin |
| ATRP | Atom transfer radical polymerization |
| BHA | Butylated hydroxyanisole |
| Bio-PTT | Bio-poly(trimethylene terephthalate) |
| BZL | Benzazulene |
| CAP | Capecitabine |
| CAT | Cellulose triacetate |
| CFU | Colony-forming unit |
| CH | Chitosan |
| CNC | Cellulose nanocrystals |
| CNT | Carbon nanotube |
| DEAEMA | 2-(diethyl-amino)ethyl methacrylate |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| g-PLA | Glycidyl methacrylate grafted polylactic acid |
| HeLa | Human cervical adenocarcinoma |
| KL | Kraft lignin |
| LNP | Lignin nanoparticles |
| LNT | Lignin nanotubes |
| NMP | Nitroxide-mediated radical polymerization |
| OS | Organosolv lignin |
| PBS | Phosphate-buffered saline |
| PDAC | Polydiallyldimethylammonium chloride |
| PEG | Poly(ethylene glycol) diglycidyl ether |
| PLA | Polylactic acid |
| PLR | Picloram |
| PS | Polystyrene |
| PVA | Polyvinyl alcohol |
| RAFT | Reversible addition–fragmentation chain transfer |
| rFCDs | Reduced fluorescence carbon dots |
| RSA | Radical scavenging activity |
| RSV | Resveratrol |
| SFN | Sorafenib |
| SPF | Sun protection factor |
| SR101 | Hydrophilic fluorescent dye sulforhodamine |
| SRSA | Superoxide radical scavenging activity |
| TDI | 2,4-Toluene diisocyanate |
| WG | Wheat gluten |
References
- Zhang, M.L.; Fan, Y.T.; Xing, Y.; Pan, C.M.; Zhang, G.S.; Lay, J.J. Enhanced biohydrogen production from cornstalk wastes with acidification pretreatment by mixed anaerobic cultures. Biomass Bioenergy 2007, 31, 250–254. [Google Scholar] [CrossRef]
- Tuck, C.O.; Perez, E.; Horvath, I.T.; Sheldon, R.A.; Poliakoff, M. Valorization of Biomass: Deriving More Value from Waste. Science 2012, 337, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin Biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Sannigrahi, P.; Ragauskas, A.J. Characterization of Fermentation Residues from the Production of Bio-Ethanol from Lignocellulosic Feedstocks. J. Biobased Mater. Bioenergy 2011, 5, 514–519. [Google Scholar] [CrossRef]
- Holladay, J.E.; White, J.F.; Bozell, J.J.; Johnson, D. Top Value-Added Chemicals from Biomass-Volume II, Results of Screening for Potential Candidates from Biorefinery Lignin; United States Department of Energy: Richland, WA, USA, 2007; Volume II.
- Graichen, F.H.M.; Grigsby, W.J.; Hill, S.J.; Raymond, L.G.; Sanglard, M.; Smith, D.A.; Thorlby, G.J.; Torr, K.M.; Warnes, J.M. Yes, we can make money out of lignin and other bio-based resources. Ind. Crops Prod. 2017, 106, 74–85. [Google Scholar] [CrossRef]
- Calvo-Flores, F.G.; Dobado, J.A. Lignin as Renewable Raw Material. ChemSusChem 2010, 3, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.P.; Bura, R.; Mabee, W.E.; Berlin, A.; Pan, X.; Saddler, J.N. Substrate Pretreatment: The Key to Effective Enzymatic Hydrolysis of Lignocellulosics? In Biofuels; Springer: Berlin/Heidelberg, Germany, 2007; Volume 108, pp. 67–93. [Google Scholar]
- Li, C.; Zhao, X.; Wang, A.; Huber, G.W.; Zhang, T. Catalytic Transformation of Lignin for the Production of Chemicals and Fuels. Chem. Rev. 2015, 115, 11559–11624. [Google Scholar] [CrossRef] [PubMed]
- Gellerstedt, G.; Henriksson, G. Lignins: Major Sources, Structure and Properties. In Monomers, Polymers and Composites from Renewable Resources; Elsevier: Amsterdam, The Netherlands, 2008; pp. 201–224. [Google Scholar]
- Ragauskas, A.J.; Beckham, G.T.; Biddy, M.J.; Chandra, R.; Chen, F.; Davis, M.F.; Davison, B.H.; Dixon, R.A.; Gilna, P.; Keller, M.; et al. Lignin valorization: Improving lignin processing in the biorefinery. Science 2014, 344, 1246843. [Google Scholar] [CrossRef] [PubMed]
- Hussain, F. Review article: Polymer-matrix Nanocomposites, Processing, Manufacturing, and Application: An Overview. J. Compos. Mater. 2006, 40, 1511–1575. [Google Scholar] [CrossRef]
- Beisl, S.; Miltner, A.; Friedl, A. Lignin from Micro- to Nanosize: Production Methods. Int. J. Mol. Sci. 2017, 18, 1244. [Google Scholar] [CrossRef] [PubMed]
- Doherty, W.O.S.; Mousavioun, P.; Fellows, C.M. Value-adding to cellulosic ethanol: Lignin polymers. Ind. Crops Prod. 2011, 33, 259–276. [Google Scholar] [CrossRef] [Green Version]
- Laurichesse, S.; Avérous, L. Chemical modification of lignins: Towards biobased polymers. Prog. Polym. Sci. 2014, 39, 1266–1290. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Dominici, F.; Kenny, J.M.M.; Puglia, D. Effect of processing conditions and lignin content on thermal, mechanical and degradative behavior of lignin nanoparticles/polylactic (acid) bionanocomposites prepared by melt extrusion and solvent casting. Eur. Polym. J. 2015, 71, 126–139. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Dominici, F.; Giovanale, G.; Mazzaglia, A.; Balestra, G.M.; Kenny, J.M.; Puglia, D. Synergic effect of cellulose and lignin nanostructures in PLA based systems for food antibacterial packaging. Eur. Polym. J. 2016, 79, 1–12. [Google Scholar] [CrossRef]
- Auras, R.; Harte, B.; Selke, S. An Overview of Polylactides as Packaging Materials. Macromol. Biosci. 2004, 4, 835–864. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Kenny, J.M.; Puglia, D. Structure and properties of biodegradable wheat gluten bionanocomposites containing lignin nanoparticles. Ind. Crops Prod. 2015, 74, 348–356. [Google Scholar] [CrossRef]
- Kayserilioǧlu, B.Ş.; Bakir, U.; Yilmaz, L.; Akkaş, N. Drying Temperature and Relative Humidity Effects on Wheat Gluten Film Properties. J. Agric. Food Chem. 2003, 51, 964–968. [Google Scholar] [CrossRef] [PubMed]
- Tunc, S.; Angellier, H.; Cahyana, Y.; Chalier, P.; Gontard, N.; Gastaldi, E. Functional properties of wheat gluten/montmorillonite nanocomposite films processed by casting. J. Memb. Sci. 2007, 289, 159–168. [Google Scholar] [CrossRef]
- Duval, A.; Molina-Boisseau, S.; Chirat, C. Comparison of Kraft lignin and lignosulfonates addition to wheat gluten-based materials: Mechanical and thermal properties. Ind. Crops Prod. 2013, 49, 66–74. [Google Scholar] [CrossRef]
- Gontard, N.; Duchez, C.; Cuq, J.-L.; Gulibert, S. Edible composite films of wheat gluten and lipids: Water vapour permeability and other physical properties. Int. J. Food Sci. Technol. 2007, 29, 39–50. [Google Scholar] [CrossRef]
- Mojumdar, S.C.; Moresoli, C.; Simon, L.C.; Legge, R.L. Edible wheat gluten (WG) protein films. J. Therm. Anal. Calorim. 2011, 104, 929–936. [Google Scholar] [CrossRef]
- Del Saz-Orozco, B.; Oliet, M.; Alonso, M.V.; Rojo, E.; Rodríguez, F. Formulation optimization of unreinforced and lignin nanoparticle-reinforced phenolic foams using an analysis of variance approach. Compos. Sci. Technol. 2012, 72, 667–674. [Google Scholar] [CrossRef]
- Landrock, A.H. Handbook of Plastic Foams: Types, Properties, Manufacture and Applications; Elsevier: Amsterdam, The Netherlands, 1995. [Google Scholar]
- Shen, H.; Nutt, S. Mechanical characterization of short fiber reinforced phenolic foam. Compos. Part A Appl. Sci. Manuf. 2003, 34, 899–906. [Google Scholar] [CrossRef]
- Shen, H.; Lavoie, A.J.; Nutt, S.R. Enhanced peel resistance of fiber reinforced phenolic foams. Compos. Part A Appl. Sci. Manuf. 2003, 34, 941–948. [Google Scholar] [CrossRef]
- Nevárez, L.A.M.; Casarrubias, L.B.; Celzard, A.; Fierro, V.; Muñoz, V.T.; Davila, A.C.; Lubian, J.R.T.; Sánchez, G.G. Biopolymer-based nanocomposites: Effect of lignin acetylation in cellulose triacetate films. Sci. Technol. Adv. Mater. 2011, 12, 45006. [Google Scholar] [CrossRef] [PubMed]
- Arthanareeswaran, G.; Thanikaivelan, P.; Srinivasn, K.; Mohan, D.; Rajendran, M. Synthesis, characterization and thermal studies on cellulose acetate membranes with additive. Eur. Polym. J. 2004, 40, 2153–2159. [Google Scholar] [CrossRef]
- Yang, W.; Dominici, F.; Fortunati, E.; Kenny, J.M.; Puglia, D. Effect of lignin nanoparticles and masterbatch procedures on the final properties of glycidyl methacrylate-g-poly (lactic acid) films before and after accelerated UV weathering. Ind. Crops Prod. 2015, 77, 833–844. [Google Scholar] [CrossRef]
- Yang, W.; Owczarek, J.S.S.; Fortunati, E.; Kozanecki, M.; Mazzaglia, A.; Balestra, G.M.M.; Kenny, J.M.M.; Torre, L.; Puglia, D. Antioxidant and antibacterial lignin nanoparticles in polyvinyl alcohol/chitosan films for active packaging. Ind. Crops Prod. 2016, 94, 800–811. [Google Scholar] [CrossRef]
- Nair, S.S.; Sharma, S.; Pu, Y.; Sun, Q.; Pan, S.; Zhu, J.Y.; Deng, Y.; Ragauskas, A.J. High Shear Homogenization of Lignin to Nanolignin and Thermal Stability of Nanolignin-Polyvinyl Alcohol Blends. ChemSusChem 2014, 7, 3513–3520. [Google Scholar] [CrossRef] [PubMed]
- Kubo, S.; Kadla, J.F. The formation of strong intermolecular interactions in immiscible blends of poly(vinyl alcohol) (PVA) and lignin. Biomacromolecules 2003, 4, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Chen, W.; Liu, Y.; Bai, S.; Wang, Q. Thermal melt processing to prepare halogen-free flame retardant poly(vinyl alcohol). Polym. Degrad. Stab. 2014, 109, 261–269. [Google Scholar] [CrossRef]
- Gupta, A.K.; Mohanty, S.; Nayak, S.K. Influence of addition of vapor grown carbon fibers on mechanical, thermal and biodegradation properties of lignin nanoparticle filled bio-poly(trimethylene terephthalate) hybrid nanocomposites. RSC Adv. 2015, 5, 56028–56036. [Google Scholar] [CrossRef]
- Zhang, J. Study of poly(trimethylene terephthalate) as an engineering thermoplastics material. J. Appl. Polym. Sci. 2004, 91, 1657–1666. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, properties and uses of a new class of materials. Mater. Sci. Eng. R Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Cai, X.; Riedl, B.; Zhang, S.Y.; Wan, H. The impact of the nature of nanofillers on the performance of wood polymer nanocomposites. Compos. Part A Appl. Sci. Manuf. 2008, 39, 727–737. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Mohanty, A.K.; Singh, S.P.; Ng, P.K.W. Effect of the processing methods on the performance of polylactide films: Thermocompression versus solvent casting. J. Appl. Polym. Sci. 2006, 101, 3736–3742. [Google Scholar] [CrossRef]
- Jiang, C.; He, H.; Jiang, H.; Ma, L.; Jia, D.M. Nano-lignin filled natural rubber composites: Preparation and characterization. Express Polym. Lett. 2013, 7, 480–493. [Google Scholar] [CrossRef] [Green Version]
- Lanzalunga, O.; Bietti, M. Photo- and radiation chemical induced degradation of lignin model compounds. J. Photochem. Photobiol. B Biol. 2000, 56, 85–108. [Google Scholar] [CrossRef]
- Barsberg, S.; Elder, T.; Felby, C. Lignin−Quinone Interactions: Implications for Optical Properties of Lignin. Chem. Mater. 2003, 15, 649–655. [Google Scholar] [CrossRef]
- Yearla, S.R.; Padmasree, K. Preparation and characterisation of lignin nanoparticles: Evaluation of their potential as antioxidants and UV protectants. J. Exp. Nanosci. 2016, 11, 289–302. [Google Scholar] [CrossRef]
- Li, H.; Deng, Y.; Wu, H.; Ren, Y.; Qiu, X.; Zheng, D.; Li, C. Self-assembly of kraft lignin into nanospheres in dioxane-water mixtures. Holzforschung 2016, 70, 725–731. [Google Scholar] [CrossRef]
- Zimniewska, M.; Kozłowski, R.; Batog, J. Nanolignin Modified Linen Fabric as a Multifunctional Product. Mol. Cryst. Liq. Cryst. 2008, 484, 43/(409)–50/(416). [Google Scholar] [CrossRef]
- Qian, Y.; Qiu, X.; Zhu, S. Sunscreen Performance of Lignin from Different Technical Resources and Their General Synergistic Effect with Synthetic Sunscreens. ACS Sustain. Chem. Eng. 2016, 4, 4029–4035. [Google Scholar] [CrossRef]
- Qian, Y.; Qiu, X.; Zhu, S. Lignin: A nature-inspired sun blocker for broad-spectrum sunscreens. Green Chem. 2015, 17, 320–324. [Google Scholar] [CrossRef]
- Qian, Y.; Zhong, X.; Li, Y.; Qiu, X. Fabrication of uniform lignin colloidal spheres for developing natural broad-spectrum sunscreens with high sun protection factor. Ind. Crops Prod. 2017, 101, 54–60. [Google Scholar] [CrossRef]
- Cazacu, G.; Capraru, M.; Popa, V.I. Advances in Natural Polymers; Thomas, S., Visakh, P.M., Mathew, A.P., Eds.; Advanced Structured Materials; Springer: Berlin/Heidelberg, Germany, 2013; Volume 18. [Google Scholar]
- Nada, A.M.A.; El-Diwany, A.I.; Elshafei, A.M. Infrared and antimicrobial studies on different lignins. Acta Biotechnol. 1989, 9, 295–298. [Google Scholar] [CrossRef]
- Zemek, J.; Košíková, B.; Augustín, J.; Joniak, D. Antibiotic properties of lignin components. Folia Microbiol. (Praha) 1979, 24, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Sláviková, E.; Košíková, B. Inhibitory effect of lignin by-products of pulping on yeast growth. Folia Microbiol. Off. J. Inst. Microbiol. Acad. Sci. Czech Repub. 1994, 39, 241–243. [Google Scholar] [CrossRef]
- Telysheva, G.; Dizhbite, T.; Lebedeva, G.; Nikoleava, V. Lignin products for decontamination of environment objects from pathogenic microorganisms and pollutants. In Proceedings of the 7th ILI Forum, Barcelona, Spain, 27–28 April 2005; pp. 71–74. [Google Scholar]
- Panáček, A.; Kolář, M.; Večeřová, R.; Prucek, R.; Soukupová, J.; Kryštof, V.; Hamal, P.; Zbořil, R.; Kvítek, L. Antifungal activity of silver nanoparticles against Candida spp. Biomaterials 2009, 30, 6333–6340. [Google Scholar] [CrossRef] [PubMed]
- Lara, H.H.; Garza-Treviño, E.N.; Ixtepan-Turrent, L.; Singh, D.K. Silver nanoparticles are broad-spectrum bactericidal and virucidal compounds. J. Nanobiotechnol. 2011, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; AlSalhi, M.S.; Siddiqui, M.K.J. Silver nanoparticle applications and human health. Clin. Chim. Acta 2010, 411, 1841–1848. [Google Scholar] [CrossRef] [PubMed]
- Stern, S.T.; McNeil, S.E. Nanotechnology Safety Concerns Revisited. Toxicol. Sci. 2008, 101, 4–21. [Google Scholar] [CrossRef] [PubMed]
- Jeong, E.; Im, W.-T.; Kim, D.-H.; Kim, M.-S.; Kang, S.; Shin, H.-S.; Chae, S.-R. Different susceptibilities of bacterial community to silver nanoparticles in wastewater treatment systems. J. Environ. Sci. Heal. Part A 2014, 49, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Walser, T.; Limbach, L.K.; Brogioli, R.; Erismann, E.; Flamigni, L.; Hattendorf, B.; Juchli, M.; Krumeich, F.; Ludwig, C.; Prikopsky, K.; et al. Persistence of engineered nanoparticles in a municipal solid-waste incineration plant. Nat. Nanotechnol. 2012, 7, 520–524. [Google Scholar] [CrossRef] [PubMed]
- Popa, V.I.; Cǎpraru, A.M.; Grama, S.; Mǎluţan, T. Nanoparticles based on modified lignins with biocide properties. Cellul. Chem. Technol. 2011, 45, 221–226. [Google Scholar]
- Gîlcă, I.-A.; Popa, V.I. Study on Biocidal Properties of Some Nanoparticles Based on Epoxy Lignin. Cellul. Chem. Technol. 2013, 47, 3–4. [Google Scholar]
- Schaad, N.W.; Committee, A.P.S.B. Laboratory Guide for Identification of Plant Pathogenic Bacteria; APS Press: St. Paul, MN, USA, 1988. [Google Scholar]
- Quattrucci, A.; Ovidi, E.; Tiezzi, A.; Vinciguerra, V.; Balestra, G.M. Biological control of tomato bacterial speck using Punica granatum fruit peel extract. Crop Prot. 2013, 46, 18–22. [Google Scholar] [CrossRef]
- Waleron, M.; Waleron, K.; Lojkowska, E. Characterization of Pectobacterium carotovorum subsp. odoriferum causing soft rot of stored vegetables. Eur. J. Plant Pathol. 2014, 139, 457–469. [Google Scholar] [CrossRef]
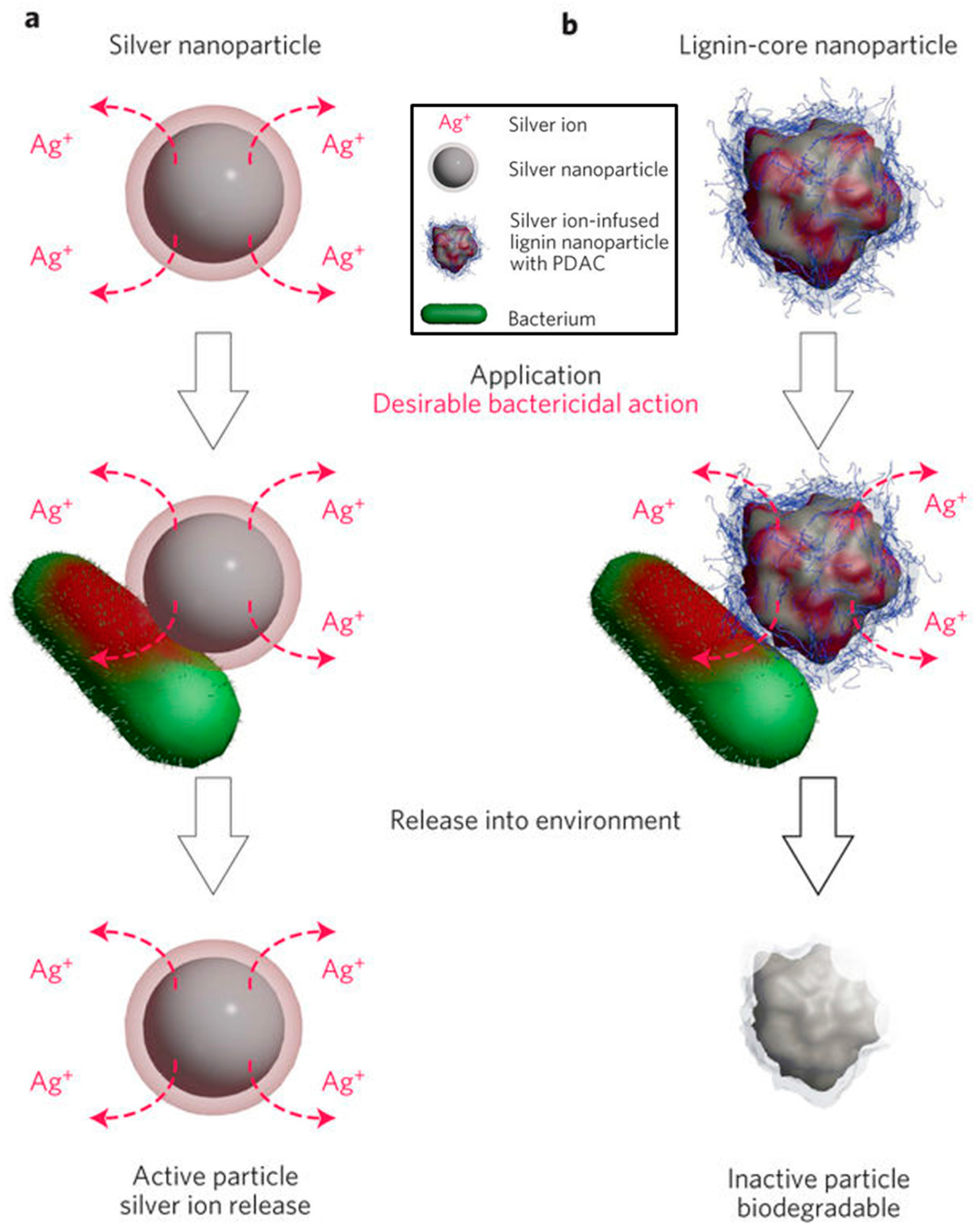
- Richter, A.P.; Brown, J.S.; Bharti, B.; Wang, A.; Gangwal, S.; Houck, K.; Cohen Hubal, E.A.; Paunov, V.N.; Stoyanov, S.D.; Velev, O.D. An environmentally benign antimicrobial nanoparticle based on a silver-infused lignin core. Nat. Nanotechnol. 2015, 10, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Richter, A.P.; Bharti, B.; Armstrong, H.B.; Brown, J.S.; Plemmons, D.; Paunov, V.N.; Stoyanov, S.D.; Velev, O.D. Synthesis and characterization of biodegradable lignin nanoparticles with tunable surface properties. Langmuir 2016, 32, 6468–6477. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.-J.; Chu, L.-H.; Gau, R.-J. Free radical-scavenging properties of lignin. Nutr. Cancer 1998, 30, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Dizhbite, T.; Telysheva, G.; Jurkjane, V.; Viesturs, U. Characterization of the radical scavenging activity of lignins - natural antioxidants. Bioresour. Technol. 2004, 95, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Hsu, B.; Coupar, I.M.; Ng, K. Antioxidant activity of hot water extract from the fruit of the Doum palm, Hyphaene thebaica. Food Chem. 2006, 98, 317–328. [Google Scholar] [CrossRef]
- Lu, Q.; Zhu, M.; Zu, Y.; Liu, W.; Yang, L.; Zhang, Y.; Zhao, X.; Zhang, X.; Zhang, X.; Li, W. Comparative antioxidant activity of nanoscale lignin prepared by a supercritical antisolvent (SAS) process with non-nanoscale lignin. Food Chem. 2012, 135, 63–67. [Google Scholar] [CrossRef]
- Pan, X.; Kadla, J.F.; Ehara, K.; Gilkes, N.; Saddler, J.N. Organosolv Ethanol Lignin from Hybrid Poplar as a Radical Scavenger: Relationship between Lignin Structure, Extraction Conditions, and Antioxidant Activity. J. Agric. Food Chem. 2006, 54, 5806–5813. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Wei, Q.; Li, Z. Preparation and evaluation of the free radical scavenging activities of nanoscale lignin biomaterials. BioResources 2014, 9, 6699–6706. [Google Scholar] [CrossRef]
- Li, Z.; Ge, Y. Extraction of lignin from sugar cane bagasse and its modification into a high performance dispersant for pesticide formulations. J. Braz. Chem. Soc. 2011, 22, 1866–1871. [Google Scholar] [CrossRef]
- Byun, Y.; Kim, Y.T.; Whiteside, S. Characterization of an antioxidant polylactic acid (PLA) film prepared with α-tocopherol, BHT and polyethylene glycol using film cast extruder. J. Food Eng. 2010, 100, 239–244. [Google Scholar] [CrossRef]
- Domenek, S.; Louaifi, A.; Guinault, A.; Baumberger, S. Potential of Lignins as Antioxidant Additive in Active Biodegradable Packaging Materials. J. Polym. Environ. 2013, 21, 692–701. [Google Scholar] [CrossRef] [Green Version]
- Maag, H. Fatty acid derivatives: Important surfactants for household, cosmetic and industrial purposes. J. Am. Oil Chem. Soc. 1984, 61, 259–267. [Google Scholar] [CrossRef]
- Morán, C.; Clapés, P.; Comelles, F.; García, T.; Pérez, L.; Vinardell, P.; Mitjans, M.; Infante, M.R. Chemical Structure/Property Relationship in Single-Chain Arginine Surfactants. Langmuir 2001, 17, 5071–5075. [Google Scholar] [CrossRef]
- Von Rybinski, W.; Hill, K. Alkyl Polyglycosides—Properties and Applications of a new Class of Surfactants. Angew. Chem. Int. Ed. 1998, 37, 1328–1345. [Google Scholar] [CrossRef]
- Silmore, K.S.; Gupta, C.; Washburn, N.R. Tunable Pickering emulsions with polymer-grafted lignin nanoparticles (PGLNs). J. Colloid Interface Sci. 2016, 466, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Binks, B.P. Particles as surfactants—Similarities and differences. Curr. Opin. Colloid Interface Sci. 2002, 7, 21–41. [Google Scholar] [CrossRef]
- Nypelö, T.E.; Carrillo, C.A.; Rojas, O.J. Lignin supracolloids synthesized from (W/O) microemulsions: Use in the interfacial stabilization of Pickering systems and organic carriers for silver metal. Soft Matter 2015, 11, 2046–2054. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Yang, Y.; Yang, R.; Wang, C. Alkaline lignin extracted from furfural residues for pH-responsive Pickering emulsions and their recyclable polymerization. Green Chem. 2012, 14, 3230. [Google Scholar] [CrossRef]
- Saigal, T.; Dong, H.; Matyjaszewski, K.; Tilton, R.D. Pickering Emulsions Stabilized by Nanoparticles with Thermally Responsive Grafted Polymer Brushes. Langmuir 2010, 26, 15200–15209. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, N.J.; Anna, S.L.; Saigal, T.; Tilton, R.D.; Walker, L.M. Interfacial Dynamics and Rheology of Polymer-Grafted Nanoparticles at Air–Water and Xylene–Water Interfaces. Langmuir 2012, 28, 8052–8063. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.; Sarbu, T.; Sirk, K.; Lowry, G.V.; Matyjaszewski, K.; Tilton, R.D. Oil-in-Water Emulsions Stabilized by Highly Charged Polyelectrolyte-Grafted Silica Nanoparticles. Langmuir 2005, 21, 9873–9878. [Google Scholar] [CrossRef] [PubMed]
- Matyjaszewski, K. Atom Transfer Radical Polymerization (ATRP): Current Status and Future Perspectives. Macromolecules 2012, 45, 4015–4039. [Google Scholar] [CrossRef]
- Nicolas, J.; Guillaneuf, Y.; Bertin, D.; Gigmes, D.; Charleux, B. Nitroxide-Mediated Polymerization. Polym. Sci. A Compr. Ref. 2012, 3, 277–350. [Google Scholar]
- Chiefari, J.; Chong, Y.K.B.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.L.; Moad, G.; et al. Living Free-Radical Polymerization by Reversible Addition−Fragmentation Chain Transfer: The RAFT Process. Macromolecules 1998, 31, 5559–5562. [Google Scholar] [CrossRef]
- Moad, G.; Rizzardo, E.; Thang, S.H. Radical addition–fragmentation chemistry in polymer synthesis. Polymer 2008, 49, 1079–1131. [Google Scholar] [CrossRef]
- Gupta, C.; Washburn, N.R. Polymer-Grafted Lignin Surfactants Prepared via Reversible Addition–Fragmentation Chain-Transfer Polymerization. Langmuir 2014, 30, 9303–9312. [Google Scholar] [CrossRef] [PubMed]
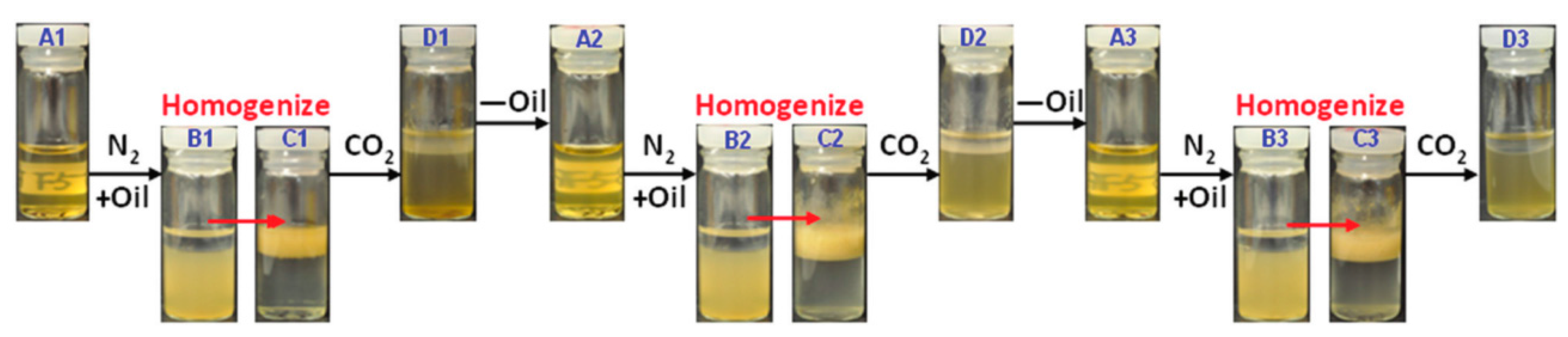
- Qian, Y.; Zhang, Q.; Qiu, X.; Zhu, S. CO2-responsive diethylaminoethyl-modified lignin nanoparticles and their application as surfactants for CO2/N2-switchable Pickering emulsions. Green Chem. 2014, 16, 4963–4968. [Google Scholar] [CrossRef]
- Inagaki, M.; Yang, Y.; Kang, F. Carbon Nanofibers Prepared via Electrospinning. Adv. Mater. 2012, 24, 2547–2566. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.C.; Saha, A.; Jana, N.R.; Sarkar, R. Fluorescent Carbon Nanoparticles: Synthesis, Characterization, and Bioimaging Application. J. Phys. Chem. C 2009, 113, 18546–18551. [Google Scholar] [CrossRef]
- Schuster, J.; He, G.; Mandlmeier, B.; Yim, T.; Lee, K.T.; Bein, T.; Nazar, L.F. Spherical Ordered Mesoporous Carbon Nanoparticles with High Porosity for Lithium-Sulfur Batteries. Angew. Chem. 2012, 124, 3651–3655. [Google Scholar] [CrossRef]
- Zhang, L.; Aboagye, A.; Kelkar, A.; Lai, C.; Fong, H. A review: Carbon nanofibers from electrospun polyacrylonitrile and their applications. J. Mater. Sci. 2014, 49, 463–480. [Google Scholar] [CrossRef]
- Kadla, J.; Kubo, S.; Venditti, R.; Gilbert, R.; Compere, A.; Griffith, W. Lignin-based carbon fibers for composite fiber applications. Carbon N. Y. 2002, 40, 2913–2920. [Google Scholar] [CrossRef]
- Hu, S.; Hsieh, Y.-L. Ultrafine microporous and mesoporous activated carbon fibers from alkali lignin. J. Mater. Chem. A 2013, 1, 11279. [Google Scholar] [CrossRef]
- Hu, S.; Zhang, S.; Pan, N.; Hsieh, Y.-L. High energy density supercapacitors from lignin derived submicron activated carbon fibers in aqueous electrolytes. J. Power Sources 2014, 270, 106–112. [Google Scholar] [CrossRef]
- Lai, C.; Zhou, Z.; Zhang, L.; Wang, X.; Zhou, Q.; Zhao, Y.; Wang, Y.; Wu, X.-F.; Zhu, Z.; Fong, H. Free-standing and mechanically flexible mats consisting of electrospun carbon nanofibers made from a natural product of alkali lignin as binder-free electrodes for high-performance supercapacitors. J. Power Sources 2014, 247, 134–141. [Google Scholar] [CrossRef]
- Wang, S.X.; Yang, L.; Stubbs, L.P.; Li, X.; He, C. Lignin-derived fused electrospun carbon fibrous mats as high performance anode materials for lithium ion batteries. ACS Appl. Mater. Interfaces 2013, 5, 12275–12282. [Google Scholar] [CrossRef] [PubMed]
- Gonugunta, P.; Vivekanandhan, S.; Mohanty, A.K.; Misra, M. A Study on Synthesis and Characterization of Biobased Carbon Nanoparticles from Lignin. World J. Nano Sci. Eng. 2012, 2, 148–153. [Google Scholar] [CrossRef]
- Yiamsawas, D.; Beckers, S.J.; Lu, H.; Landfester, K.; Wurm, F.R. Morphology-Controlled Synthesis of Lignin Nanocarriers for Drug Delivery and Carbon Materials. ACS Biomater. Sci. Eng. 2017, 3, 2375–2383. [Google Scholar] [CrossRef]
- Sun, Y.-P.; Zhou, B.; Lin, Y.; Wang, W.; Fernando, K.A.S.; Pathak, P.; Meziani, M.J.; Harruff, B.A.; Wang, X.; Wang, H.; et al. Quantum-Sized Carbon Dots for Bright and Colorful Photoluminescence. J. Am. Chem. Soc. 2006, 128, 7756–7757. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-P.; Wang, X.; Lu, F.; Cao, L.; Meziani, M.J.; Luo, P.G.; Gu, L.; Veca, L.M. Doped Carbon Nanoparticles as a New Platform for Highly Photoluminescent Dots. J. Phys. Chem. C. Nanomater. Interfaces 2008, 112, 18295–18298. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Wang, X.; Meziani, M.J.; Lu, F.; Wang, H.; Luo, P.G.; Lin, Y.; Harruff, B.A.; Veca, L.M.; Murray, D.; et al. Carbon Dots for Multiphoton Bioimaging. J. Am. Chem. Soc. 2007, 129, 11318–11319. [Google Scholar] [CrossRef] [PubMed]
- Rai, S.; Singh, B.K.; Bhartiya, P.; Singh, A.; Kumar, H.; Dutta, P.K.; Mehrotra, G.K. Lignin derived reduced fluorescence carbon dots with theranostic approaches: Nano-drug-carrier and bioimaging. J. Lumin. 2017, 190, 492–503. [Google Scholar] [CrossRef]
- Chen, W.; Hu, C.; Yang, Y.; Cui, J.; Liu, Y. Rapid Synthesis of Carbon Dots by Hydrothermal Treatment of Lignin. Materials (Basel) 2016, 9, 184. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Han, B.; Wang, Z.; Gao, C.; Peng, C.; Shen, J. Hollow chitosan-alginate multilayer microcapsules as drug delivery vehicle: Doxorubicin loading and in vitro and in vivo studies. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.T.; Tzortzis, G.; Charalampopoulos, D.; Khutoryanskiy, V.V. Production and Evaluation of Dry Alginate-Chitosan Microcapsules as an Enteric Delivery Vehicle for Probiotic Bacteria. Biomacromolecules 2011, 12, 2834–2840. [Google Scholar] [CrossRef] [PubMed]
- Broaders, K.E.; Pastine, S.J.; Grandhe, S.; Fréchet, J.M.J. Acid-degradable solid-walled microcapsules for pH-responsive burst-release drug delivery. Chem. Commun. 2011, 47, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Subirade, M. Elaboration and Characterization of Soy/Zein Protein Microspheres for Controlled Nutraceutical Delivery. Biomacromolecules 2009, 10, 3327–3334. [Google Scholar] [CrossRef] [PubMed]
- Chun, H.; Kim, C.-H.; Cho, Y.-H. Microencapsulation of Lactobacillus plantarum DKL 109 using External Ionic Gelation Method. Korean J. Food Sci. Anim. Resour. 2014, 34, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Tomaro-Duchesneau, C.; Saha, S.; Malhotra, M.; Coussa-Charley, M.; Kahouli, I.; Jones, M.L.; Labbé, A.; Prakash, S. Probiotic Ferulic Acid Esterase Active Lactobacillus fermentum NCIMB 5221 APA Microcapsules for Oral Delivery: Preparation and in Vitro Characterization. Pharmaceuticals 2012, 5, 236–248. [Google Scholar] [CrossRef] [PubMed]
- Caruso, F. (Ed.) Colloids and Colloid Assemblies; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2003. [Google Scholar]
- Venkatesan, P.; Manavalan, R.; Valliappan, K. Microencapsulateation: A Vital Technique in Novel Drug Delivery System. J. Pharm. Sci. Res. 2009, 1, 26–35. [Google Scholar]
- Singh, M.N.; Hemant, K.S.Y.; Ram, M.; Shivakumar, H.G. Microencapsulation: A promising technique for controlled drug delivery. Res. Pharm. Sci. 2010, 5, 65–77. [Google Scholar] [PubMed]
- Tsuji, K. Microencapsulation of pesticides and their improved handling safety. J. Microencapsul. 2001, 18, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Green, D.W.; Mann, S.; Oreffo, R.O.C. Mineralized polysaccharide capsules as biomimetic microenvironments for cell, gene and growth factor delivery in tissue engineering. Soft Matter 2006, 2, 732–737. [Google Scholar] [CrossRef]
- Kurosaki, T.; Kitahara, T.; Fumoto, S.; Nishida, K.; Yamamoto, K.; Nakagawa, H. Chondroitin Sulfate Capsule System for Efficient and Secure Gene Delivery. J. Pharm. Pharm. Sci. 2010, 13, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Desai, K.G.H.; Jin Park, H. Recent Developments in Microencapsulation of Food Ingredients. Dry. Technol. 2005, 23, 1361–1394. [Google Scholar] [CrossRef]
- Herzfeldt, C.D.; Kümmel, R. Dissociation constants, solubilities and dissolution rates of some selected nonsteroidal antiinflammatories. Drug Dev. Ind. Pharm. 1983, 9, 767–793. [Google Scholar] [CrossRef]
- Vayssières, L.; Chanéac, C.; Tronc, E.; Jolivet, J.P. Size Tailoring of Magnetite Particles Formed by Aqueous Precipitation: An Example of Thermodynamic Stability of Nanometric Oxide Particles. J. Colloid Interface Sci. 1998, 205, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Yiamsawas, D.; Baier, G.; Thines, E.; Landfester, K.; Wurm, F.R. Biodegradable lignin nanocontainers. RSC Adv. 2014, 4, 11661–11663. [Google Scholar] [CrossRef]
- Tortora, M.; Cavalieri, F.; Mosesso, P.; Ciaffardini, F.; Melone, F.; Crestini, C. Ultrasound Driven Assembly of Lignin into Microcapsules for Storage and Delivery of Hydrophobic Molecules. Biomacromolecules 2014, 15, 1634–1643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, N.; Dempere, L.A.; Tong, Z. Synthesis of pH-Responsive Lignin-Based Nanocapsules for Controlled Release of Hydrophobic Molecules. ACS Sustain. Chem. Eng. 2016, 4, 5204–5211. [Google Scholar] [CrossRef]
- Chen, C.-K.; Wang, Q.; Jones, C.H.; Yu, Y.; Zhang, H.; Law, W.-C.; Lai, C.K.; Zeng, Q.; Prasad, P.N.; Pfeifer, B.A.; et al. Synthesis of pH-Responsive Chitosan Nanocapsules for the Controlled Delivery of Doxorubicin. Langmuir 2014, 30, 4111–4119. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Zhang, F.; Zhang, S.; Pollack, S.F.; Elsabahy, M.; Fan, J.; Wooley, K.L. Poly(ethylene oxide)-block-Polyphosphoester-graft-Paclitaxel Conjugates with Acid-Labile Linkages as a pH-Sensitive and Functional Nanoscopic Platform for Paclitaxel Delivery. Adv. Healthc. Mater. 2014, 3, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Mina, M.V.; Puzyk, I.P.; Puzyk, M.V. The effect of acids on fluorescence of coumarin-6 in organic solvents. Opt. Spectrosc. 2013, 114, 244–246. [Google Scholar] [CrossRef]
- Nghi, D.H.; Bittner, B.; Kellner, H.; Jehmlich, N.; Ullrich, R.; Pecyna, M.J.; Nousiainen, P.; Sipila, J.; Huong, L.M.; Hofrichter, M.; et al. The Wood Rot Ascomycete Xylaria polymorpha Produces a Novel GH78 Glycoside Hydrolase That Exhibits α-l-Rhamnosidase and Feruloyl Esterase Activities and Releases Hydroxycinnamic Acids from Lignocelluloses. Appl. Environ. Microbiol. 2012, 78, 4893–4901. [Google Scholar] [CrossRef] [PubMed]
- Dowding, P.J.; Atkin, R.; Vincent, B.; Bouillot, P. Oil Core–Polymer Shell Microcapsules Prepared by Internal Phase Separation from Emulsion Droplets. I. Characterization and Release Rates for Microcapsules with Polystyrene Shells. Langmuir 2004, 20, 11374–11379. [Google Scholar] [CrossRef] [PubMed]
- Ten, E.; Ling, C.; Wang, Y.; Srivastava, A.; Dempere, L.A.; Vermerris, W. Lignin Nanotubes As Vehicles for Gene Delivery into Human Cells. Biomacromolecules 2014, 15, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Magrez, A.; Kasas, S.; Salicio, V.; Pasquier, N.; Seo, J.W.; Celio, M.; Catsicas, S.; Schwaller, B.; Forró, L. Cellular Toxicity of Carbon-Based Nanomaterials. Nano Lett. 2006, 6, 1121–1125. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhao, Y.; Sun, B.; Chen, C. Understanding the Toxicity of Carbon Nanotubes. Acc. Chem. Res. 2013, 46, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Qian, Y.; Huang, J.; Yang, D.; Deng, Y.; Qiu, X. Fabrication of Lignosulfonate Vesicular Reverse Micelles to Immobilize Horseradish Peroxidase. Ind. Eng. Chem. Res. 2016, 55, 2731–2737. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, J. Encapsulation of the Herbicide Picloram by Using Polyelectrolyte Biopolymers as Layer-by-Layer Materials. J. Agric. Food Chem. 2013, 61, 3789–3796. [Google Scholar] [CrossRef] [PubMed]
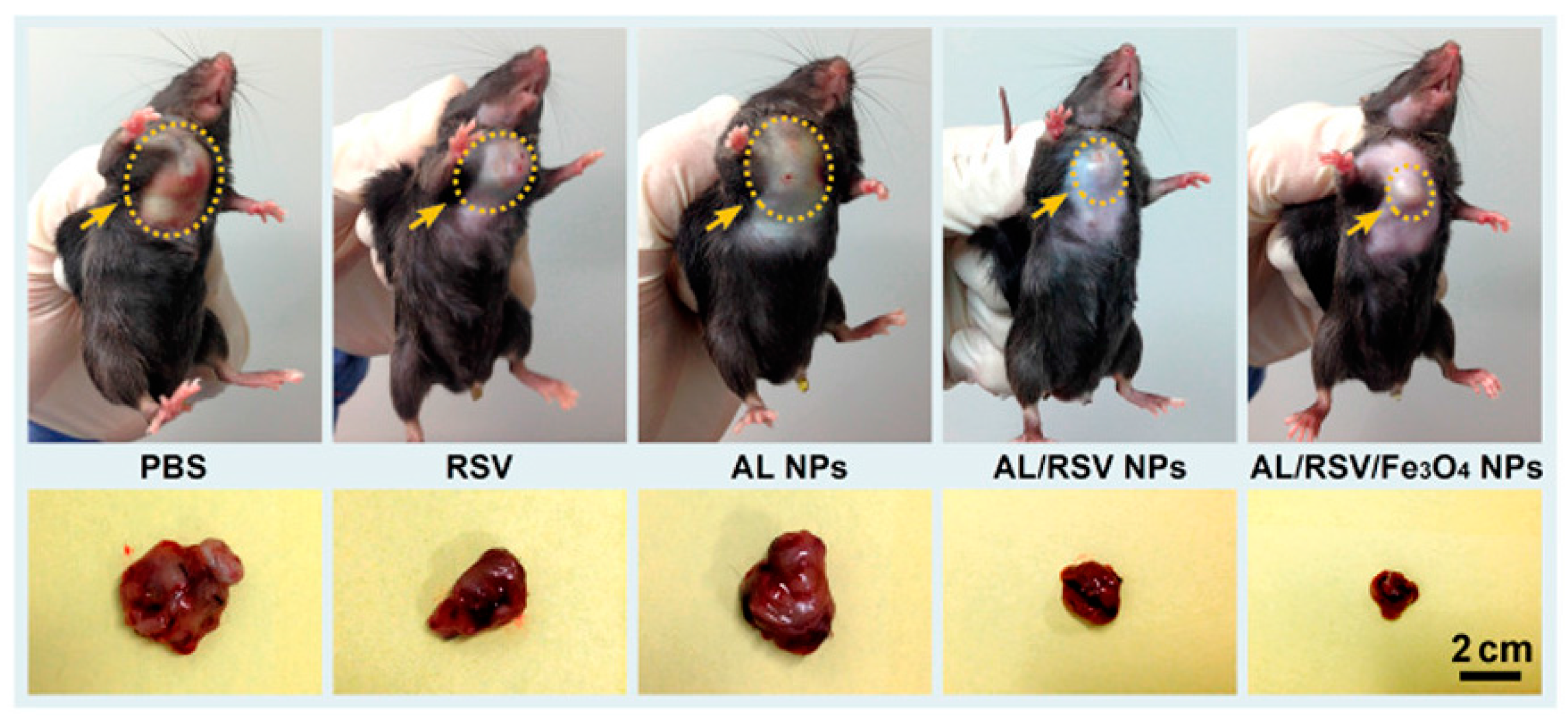
- Figueiredo, P.; Lintinen, K.; Kiriazis, A.; Hynninen, V.; Liu, Z.; Bauleth-Ramos, T.; Rahikkala, A.; Correia, A.; Kohout, T.; Sarmento, B.; et al. In vitro evaluation of biodegradable lignin-based nanoparticles for drug delivery and enhanced antiproliferation effect in cancer cells. Biomaterials 2017, 121, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Liu, R.; Hu, L.Q.; Zou, Z.F.; Si, C.L. Lignin Nanoparticle as a Novel Green Carrier for the Efficient Delivery of Resveratrol. ACS Sustain. Chem. Eng. 2017, 5, 8241–8249. [Google Scholar] [CrossRef]




| Investigated in | Neat Polymer | Production Method | Application Field and Neat Properties | Investigated Properties by Addition of Lignin Nanoparticles (LNPs) |
|---|---|---|---|---|
| [16,17] | Polylactic acid (PLA) |
|
|
|
| [19] | Wheat gluten (WG) |
|
|
|
| [25] | Phenolic foams |
|
|
|
| [29] | Cellulose triacetate (CTA) |
|
|
|
| [31] | Glycidyl methacrylate grafted PLA |
|
|
|
| [32,33] | Poly vinyl alcohol (PVA)/chitosan (CH) |
|
|
|
| [36] | Bio-poly (trimethylene terephthalate) |
|
|
|
| Source | Polymer | Filler Composition and Concentration | Tensile Strength σ (MPa) | Young’s Modulus E (MPa) | Elongation at Break εb (%) |
|---|---|---|---|---|---|
| [19] | Wheat gluten | neat | 5.5 ± 0.8 | 180.5 ± 57.9 | 297 ± 11 |
| 3 wt % LNP | 13.3 ± 1.6 | 553.2 ± 56.3 | 28 ± 10 | ||
| [32] | PVA | neat | 45.7 ± 1.3 | 1140 ± 220 | 164 ± 15 |
| 3 wt % LNP | 51.4 ± 3.3 | 2100 ± 130 | 30.6 ± 8.5 | ||
| [17] | PLA | neat | 44.0 ± 4.6 | 2010 ± 210 | 15.0 |
| 3 wt % LNP | 41.1 ± 1.9 | 1390 ± 60 | 66.3 | ||
| 1 wt % LNP + 3 wt % CNC | 53.6 ± 6.9 | 2500 ± 170 | 7.3 | ||
| [31] | PLA/g-PLA | neat PLA | 44.4 ± 4.3 | 1950 ± 250 | 16.9 ± 4.0 |
| PLA + 1 wt % LNP | 48.6 ± 3.5 | 2150 ± 130 | 26.8 ± 4.8 | ||
| g-PLA+ 1 wt % LNP | 47.2 ± 3.1 | 1630 ± 110 | 108 ± 20 | ||
| PLA/g-PLA + 1 wt % LNP | 56.4 ± 3.3 | 2120 ± 140 | 20.4 ± 8.9 | ||
| [16] | PLA | neat | 44.3 ± 4.6 | 1960 ± 230 | 17.0 ± 3.8 |
| PLA + 1 wt % LNP | 48.6 ± 3.4 | 2150 ± 130 | 26.7 ± 4.8 | ||
| PLA + 3 wt % LNP | 40.9 ± 2.1 | 1380 ± 60 | 66.7 ± 4.0 | ||
| [41] | Natural rubber | neat | 25.24 ± 0.38 | 2.00 ± 0.03 2 | 654 ± 13 |
| 7 wt % LNP 1 | 29.24 ± 0.59 | 2.95 ± 0.10 2 | 658 ± 20 | ||
| [36] | bio-PTT | neat | 51.49 ± 0.5 | 2058 ± 37 | - |
| 1.5 wt % LNP | 59.16 ± 0.7 | 2227 ± 47 | - | ||
| 1.5 wt % LNP + 7 wt % carbon fibers | 61.74 ± 0.6 | 2309 ± 9 | - |
| Source | Lignin Type | Grafting Method | Grafted Polymers | Stabilized Emulsion |
|---|---|---|---|---|
| [91] | KL | RAFT | polyacrylamide | water/hexanes 1 |
| poly(acrylic acid) | ||||
| [80] | KL | RAFT | polyacrylamide | water/cyclohexane |
| [92] | AL | ATRP | 2-(diethyl-amino)ethyl methacrylate | water/decane |
| Sample | Carbonization Temperature (°C) | O (%) | C (%) | BET Surface Area (m2/g) | Average Pore Volume (cm3/g) | Average Pore Width (Å) |
|---|---|---|---|---|---|---|
| Pristine lignin | - | 27 | 73 | 5 | 0.017 | 16.5 |
| Porous LNP | - | 21.6 | 78.4 | 15 | 0.020 | 34.7 |
| Pristine lignin | 800 | 14.1 | 85.9 | 29 | 0.023 | 18.5 |
| Porous LNP | 600 | 17.2 | 82.8 | 202 | 0.123 | 14.7 |
| Porous LNP | 800 | 8 | 92 | 552 | 0.274 | 14.8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beisl, S.; Friedl, A.; Miltner, A. Lignin from Micro- to Nanosize: Applications. Int. J. Mol. Sci. 2017, 18, 2367. https://doi.org/10.3390/ijms18112367
Beisl S, Friedl A, Miltner A. Lignin from Micro- to Nanosize: Applications. International Journal of Molecular Sciences. 2017; 18(11):2367. https://doi.org/10.3390/ijms18112367
Chicago/Turabian StyleBeisl, Stefan, Anton Friedl, and Angela Miltner. 2017. "Lignin from Micro- to Nanosize: Applications" International Journal of Molecular Sciences 18, no. 11: 2367. https://doi.org/10.3390/ijms18112367





