Immunohistochemical Analysis of the Natural Killer Cell Cytotoxicity Pathway in Human Abdominal Aortic Aneurysms
Abstract
:1. Introduction
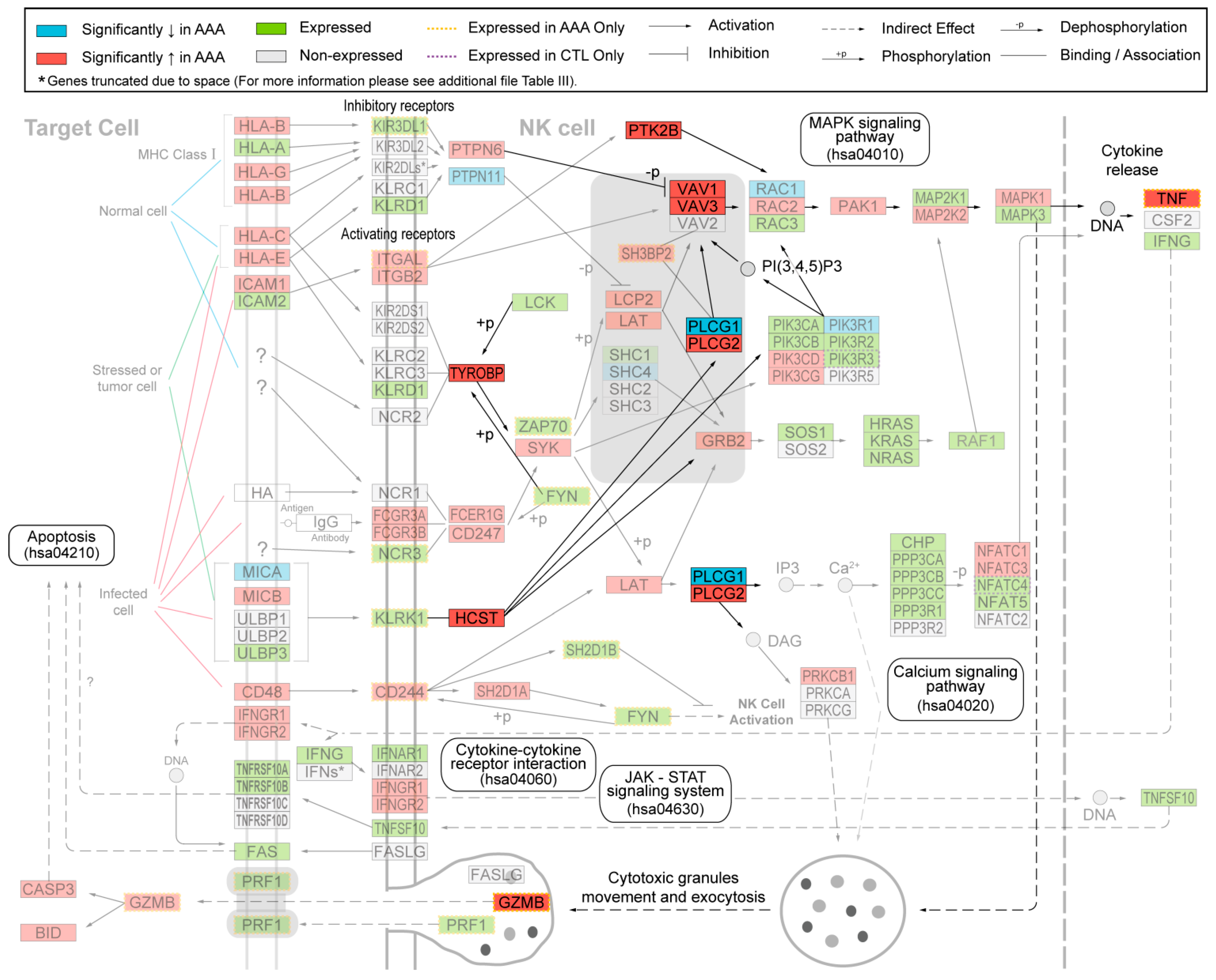
2. Results
2.1. Immunohistochemical Analysis Demonstrates Staining of Members of NK Cell Mediated Cytotoxicity Pathway in Human AAA Tissue
| Case ID | Age (Years) | Sex | Cause of Death | Classification |
|---|---|---|---|---|
| ME0101 | 53 | Male | Gunshot Wound | Control |
| ME0105 | 53 | Male | Unknown | Control |
| ME0205 | 78 | Male | Cardiac Arrest | Control |
| ME0501 | 69 | Female | Fall (Head Trauma) | Control |
| ME0503 | 54 | Male | Cardiac Arrest | Control |
| ME0505 | 59 | Female | Cardiovascular | Control |
| ME1001 | 88 | Female | Trauma | Control |
| ME1003 | 44 | Male | Overdose/Cardiovascular | Control |
| WSU052 | 70 | Male | NA | AAA |
| WSU060 | 70 | Male | NA | AAA |
| WSU068 | 72 | Male | NA | AAA |
| WSU075 | 67 | Male | NA | AAA |
| WSU080 | 64 | Female | NA | AAA |
| WSU081 | 69 | Male | NA | AAA |

2.2. Immunohistochemical Analyses with Double Staining Show Co-Localization of Proteins Participating in the Early and Late Steps of the NK Pathway Activation
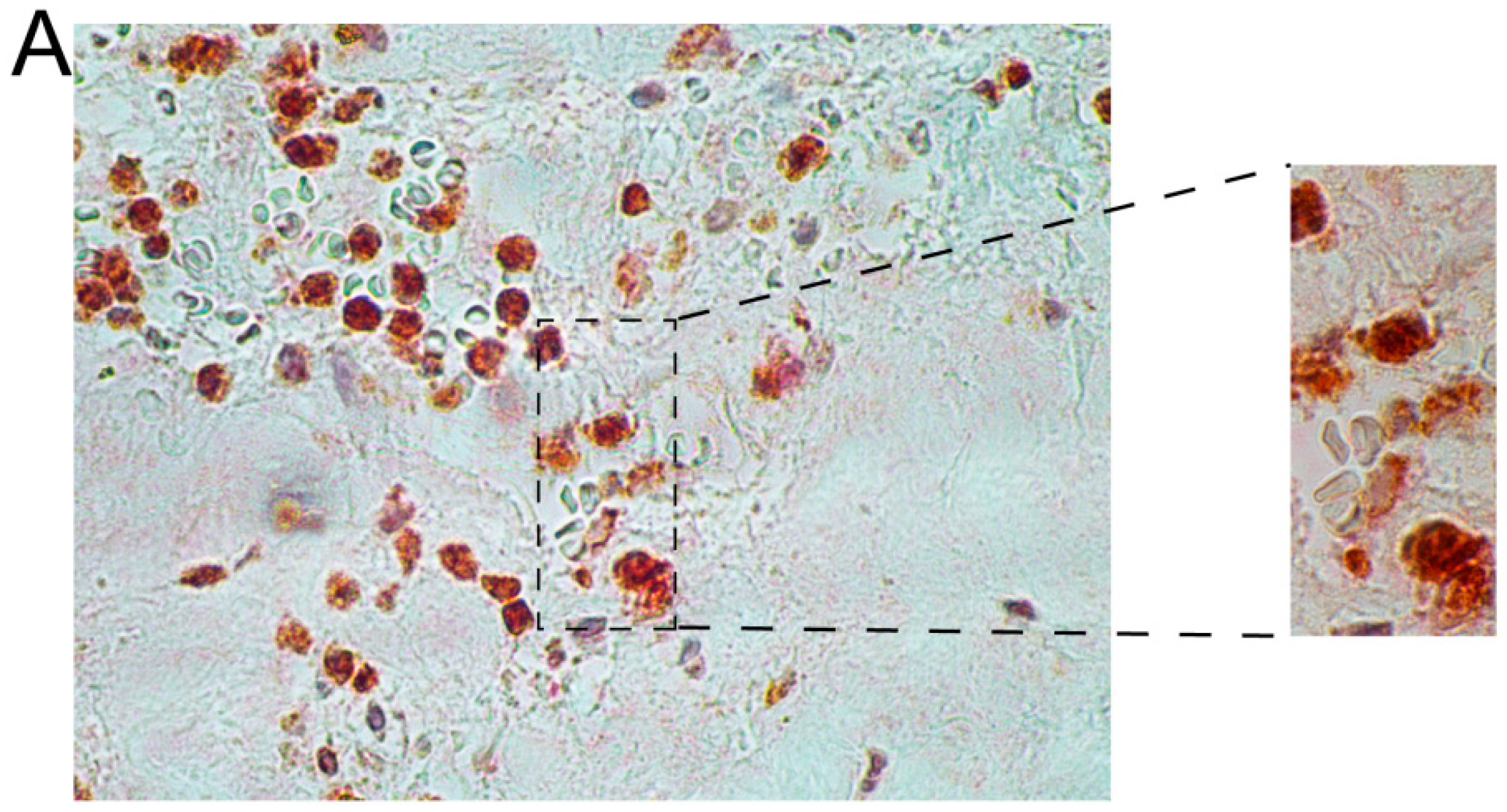
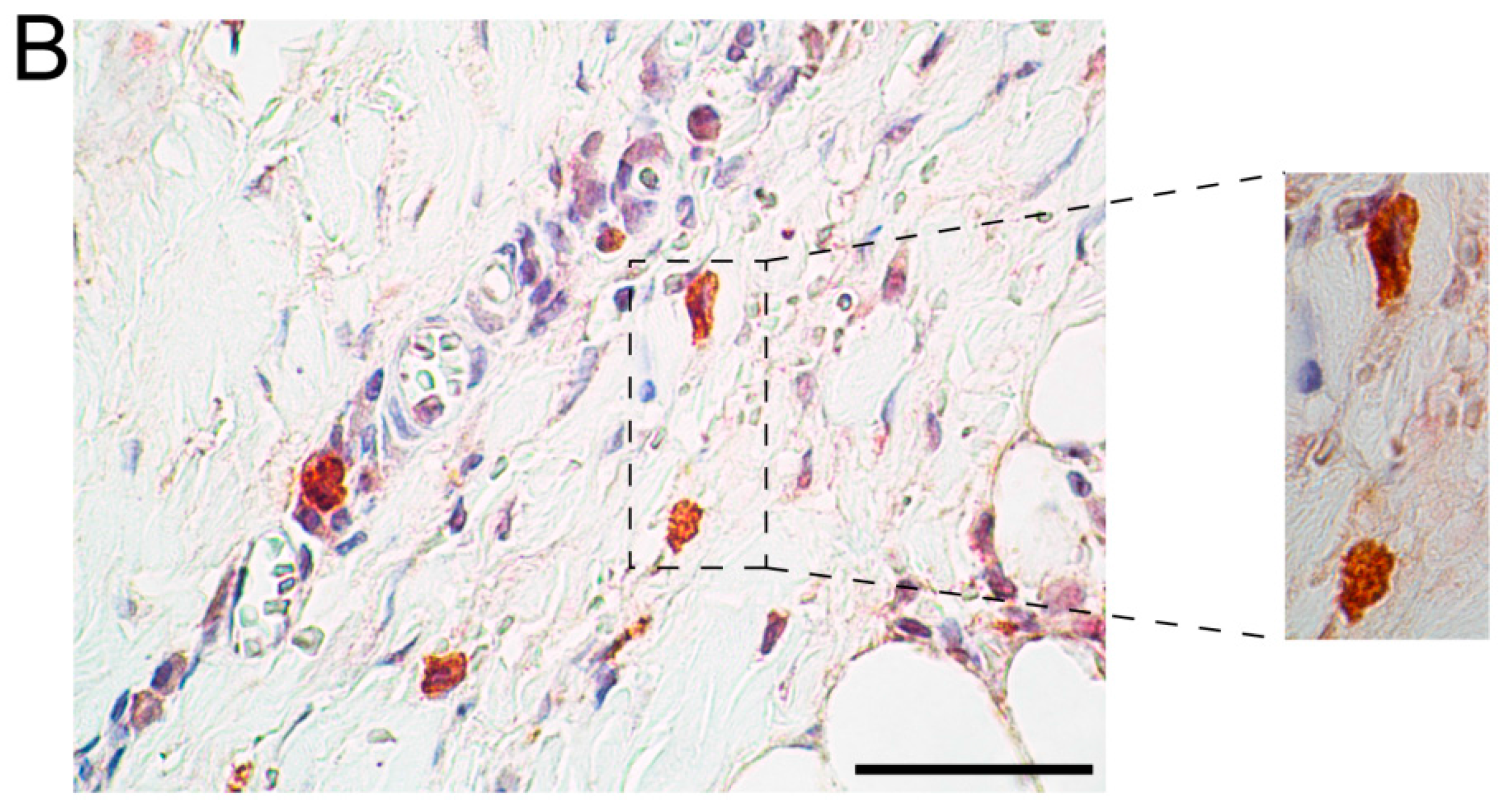
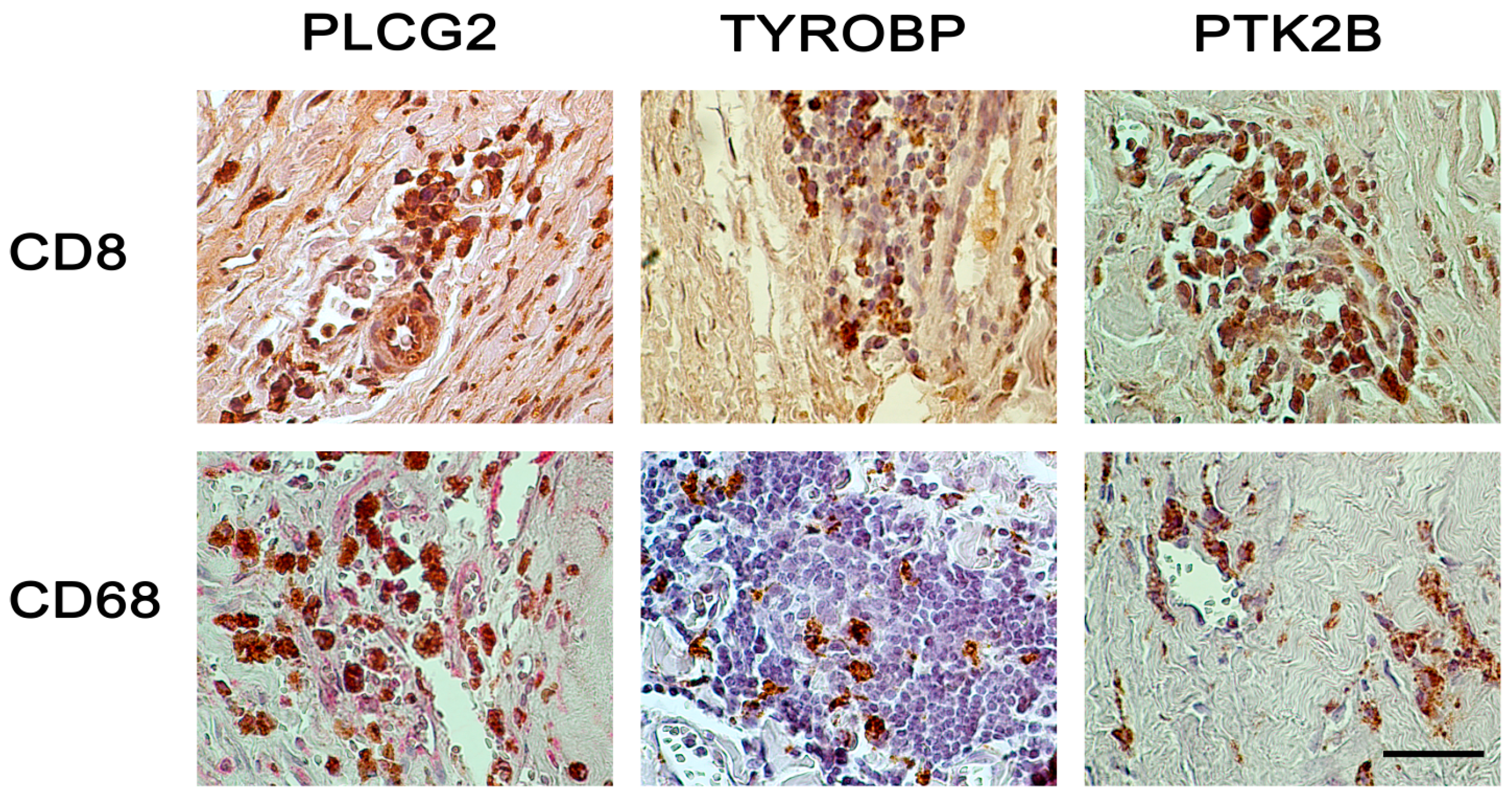
3. Discussion
4. Experimental Section
4.1. Human Aortic Tissue Samples
4.2. Microarray-Based Gene Expression Studies
4.3. Immunohistochemical Analysis
| Gene Symbol * | Gene ID * | Protein Symbols | Full Name | Catalog Number | Supplier | Species |
|---|---|---|---|---|---|---|
| PTK2B | 2185 | PTK2B, PYK2, PKB, PTK | Protein tyrosine kinase 2 β | ab55358 ¶ | Abcam, Cambridge, MA, USA | Rabbit polyclonal |
| LAT | 27040 | LAT, pp36 | Linker for activation of T cells | ab32070 § | Abcam, Cambridge, MA, USA | Rabbit monoclonal |
| VAV3 | 10451 | VAV3 | Vav 3 guanine nucleotide exchange factor | ab40889 | Abcam, Cambridge, MA, USA | Rabbit polyclonal |
| RAC2 | 5880 | RAC2, HSPC022, p21-Rac2, EN-7 | Ras-related C3 botulinum toxin substrate 2 (rho family, small GTP binding protein Rac2) | ab2244 † | Abcam, Cambridge, MA, USA | Goat polyclonal |
| TNF | 7124 | TNF, TNFA, TNFSF2, DIF | Tumor necrosis factor | ab6671 | Abcam, Cambridge, MA, USA | Rabbit polyclonal |
| CD244 | 51744 | CD244, 2B4, NAIL, NKR2B4, SLAMF4 | CD244 molecule, natural killer cell receptor 2B4 | HPA010628 † | Sigma-Aldrich, St. Louis, MO, USA | Rabbit polyclonal |
| TYROBP | 7305 | DAP12, KARAP, PLOSL | TYRO protein tyrosine kinase binding protein | sc-20783 ¶ | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Rabbit polyclonal |
| HCST | 10870 | DAP10, KAP10, PIK3AP | Hematopoietic cell signal transducer | sc-10531 ¶ | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Goat polyclonal |
| SH2D1A | 4068 | SAP, LYP, DSHP, MTCP1, EBVS | SH2 domain containing 1A | sc-8333 † | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Rabbit polyclonal |
| VAV1 | 7409 | VAV1, VAV | Vav 1 guanine nucleotide exchange factor | sc-132 | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Rabbit polyclonal |
| PLCG1 | 5335 | PLCG1, PLC1, NCKAP3, PLC148 | Phospholipase C, gamma 1 | sc-7290 | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Mouse monoclonal |
| PLCG2 | 5336 | PLCG2, FCAS3, APLAID | Phospholipase C, gamma 2 (phosphatidylinositol-specific) | sc-5283 ¶ | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Mouse monoclonal |
| RAC1 | 5879 | RAC1, MIG5, TC-25, p21-Rac1 | Ras-related C3 botulinum toxin substrate 1 (rho family, small GTP binding protein Rac1) | 05-389 † | Millipore Corporation, Billerica, MA, USA | Mouse monoclonal |
| RAF1 | 5894 | RAF1, NS5, CRAF | v-raf-1 murine leukemia viral oncogene homolog 1 | sc-7267 † | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Mouse monoclonal |
| GZMB | 3002 | GZMB, HLP, CCPI, CGL1, SECT, CTLA1, CTSGL1 | Granzyme B (granzyme 2, cytotoxic T-lymphocyte-associated serine esterase 1) | sc-1969 ¶ | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | Goat polyclonal |
| CD8A | 925 | CD8, MAL, p32, Leu2 | CD8a molecule | M7103 ¶ | Dako, Glostrup, Denmark | Mouse monoclonal |
| CD68 | 968 | CD68, GP110, LAMP4, SCARD1 | CD68 molecule | M0876 ¶ | Dako, Glostrup, Denmark | Mouse monoclonal |
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Emeto, T.I.; Seto, S.W.; Golledge, J. Targets for medical therapy to limit abdominal aortic aneurysm progression. Curr. Drug Targets 2014, 15, 860–873. [Google Scholar] [CrossRef] [PubMed]
- Lederle, F.A. Abdominal aortic aneurysm. Ann. Intern. Med. 2009, 150, ITC5-1. [Google Scholar] [CrossRef] [PubMed]
- Nischan, J.; Lenk, G.M.; Boddy, A.M.; Lillvis, J.H.; Tromp, G.; Kuivaniemi, H. Abdominal aortic aneurysms—A complex genetic disease. In Aneurysms Types, Risks, Formation and Treatment; Laurent, A., Morel, E., Eds.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2009; pp. 1–59. [Google Scholar]
- Boddy, A.M.; Lenk, G.M.; Lillvis, J.H.; Nischan, J.; Kyo, Y.; Kuivaniemi, H. Basic research studies to understand aneurysm disease. Drug News Perspect. 2008, 21, 142–148. [Google Scholar] [PubMed]
- Abdul-Hussien, H.; Hanemaaijer, R.; Kleemann, R.; Verhaaren, B.F.; van Bockel, J.H.; Lindeman, J.H. The pathophysiology of abdominal aortic aneurysm growth: Corresponding and discordant inflammatory and proteolytic processes in abdominal aortic and popliteal artery aneurysms. J. Vasc. Surg. 2010, 51, 1479–1487. [Google Scholar] [CrossRef] [PubMed]
- Bobryshev, Y.V.; Lord, R.S. Vascular-associated lymphoid tissue (VALT) involvement in aortic aneurysm. Atherosclerosis 2001, 154, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.E.; Haines, G.K.; Rizzo, R.J.; Radosevich, J.A.; Pope, R.M.; Robinson, P.G.; Pearce, W.H. Human abdominal aortic aneurysms: Immunophenotypic analysis suggesting an immune-mediated response. Am. J. Pathol. 1990, 137, 1199–1213. [Google Scholar] [PubMed]
- McCormick, M.L.; Gavrila, D.; Weintraub, N.L. Role of oxidative stress in the pathogenesis of abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Estrelinha, M.; Hinterseher, I.; Kuivaniemi, H. Gene expression studies in human abdominal aortic aneurysm. Rev. Vasc. Med. 2014, 2, 77–82. [Google Scholar] [CrossRef]
- Lenk, G.M.; Tromp, G.; Weinsheimer, S.; Gatalica, Z.; Berguer, R.; Kuivaniemi, H. Whole genome expression profiling reveals a significant role for immune function in human abdominal aortic aneurysms. BMC Genomics 2007, 8, 237. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Smyth, M.J.; Cretney, E.; Kelly, J.M.; Westwood, J.A.; Street, S.E.; Yagita, H.; Takeda, K.; van Dommelen, S.L.; Degli-Esposti, M.A.; Hayakawa, Y. Activation of NK cell cytotoxicity. Mol. Immunol. 2005, 42, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Vivier, E.; Nunes, J.A.; Vely, F. Natural killer cell signaling pathways. Science 2004, 306, 1517–1519. [Google Scholar] [CrossRef] [PubMed]
- Forester, N.D.; Cruickshank, S.M.; Scott, D.J.; Carding, S.R. Functional characterization of T cells in abdominal aortic aneurysms. Immunology 2005, 115, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Henderson, E.L.; Geng, Y.J.; Sukhova, G.K.; Whittemore, A.D.; Knox, J.; Libby, P. Death of smooth muscle cells and expression of mediators of apoptosis by T lymphocytes in human abdominal aortic aneurysms. Circulation 1999, 99, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Hinterseher, I.; Erdman, R.; Donoso, L.A.; Vrabec, T.R.; Schworer, C.M.; Lillvis, J.H.; Boddy, A.M.; Derr, K.; Golden, A.; Bowen, W.D.; et al. Role of complement cascade in abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Hinterseher, I.; Erdman, R.; Elmore, J.R.; Stahl, E.; Pahl, M.C.; Derr, K.; Golden, A.; Lillvis, J.H.; Cindric, M.C.; Jackson, K.; et al. Novel pathways in the pathobiology of human abdominal aortic aneurysms. Pathobiology 2013, 80, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lillvis, J.H.; Erdman, R.; Schworer, C.M.; Golden, A.; Derr, K.; Gatalica, Z.; Cox, L.A.; Shen, J.; Vander Heide, R.S.; Lenk, G.M.; et al. Regional expression of HOXA4 along the aorta and its potential role in human abdominal aortic aneurysms. BMC Physiol. 2011, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Lillvis, J.H.; Kyo, Y.; Tromp, G.; Lenk, G.M.; Li, M.; Lu, Q.; Igo, R.P.J.; Sakalihasan, N.; Ferrell, R.E.; Schworer, C.M.; et al. Analysis of positional candidate genes in the AAA1 susceptibility locus for abdominal aortic aneurysms on chromosome 19. BMC Med. Genet. 2011, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Pahl, M.C.; Derr, K.; Gabel, G.; Hinterseher, I.; Elmore, J.R.; Schworer, C.M.; Peeler, T.C.; Franklin, D.P.; Gray, J.L.; Carey, D.J.; et al. MicroRNA expression signature in human abdominal aortic aneurysms. BMC Med. Genomics 2012, 5, 25. [Google Scholar] [CrossRef] [PubMed]
- Okigaki, M.; Davis, C.; Falasca, M.; Harroch, S.; Felsenfeld, D.P.; Sheetz, M.P.; Schlessinger, J. Pyk2 regulates multiple signaling events crucial for macrophage morphology and migration. Proc. Natl. Acad. Sci. USA 2003, 100, 10740–10745. [Google Scholar] [CrossRef] [PubMed]
- Owen, K.A.; Pixley, F.J.; Thomas, K.S.; Vicente-Manzanares, M.; Ray, B.J.; Horwitz, A.F.; Parsons, J.T.; Beggs, H.E.; Stanley, E.R.; Bouton, A.H. Regulation of lamellipodial persistence, adhesion turnover, and motility in macrophages by focal adhesion kinase. J. Cell Biol. 2007, 179, 1275–1287. [Google Scholar] [CrossRef] [PubMed]
- Forester, N.D.; Cruickshank, S.M.; Scott, D.J.; Carding, S.R. Increased natural killer cell activity in patients with an abdominal aortic aneurysm. Br. J. Surg. 2006, 93, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Hansson, G.K. Inflammation and immunity in diseases of the arterial tree: Players and layers. Circ. Res. 2015, 116, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Chan, W.L.; Pejnovic, N.; Liew, T.V.; Hamilton, H. Predominance of Th2 response in human abdominal aortic aneurysm: Mistaken identity for IL-4-producing NK and NKT cells? Cell. Immunol. 2005, 233, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Biros, E.; Moran, C.S.; Rush, C.M.; Gabel, G.; Schreurs, C.; Lindeman, J.H.; Walker, P.J.; Nataatmadja, M.; West, M.; Holdt, L.M.; et al. Differential gene expression in the proximal neck of human abdominal aortic aneurysm. Atherosclerosis 2014, 233, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Juvonen, J.; Surcel, H.M.; Satta, J.; Teppo, A.M.; Bloigu, A.; Syrjala, H.; Airaksinen, J.; Leinonen, M.; Saikku, P.; Juvonen, T. Elevated circulating levels of inflammatory cytokines in patients with abdominal aortic aneurysm. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2843–2847. [Google Scholar] [CrossRef] [PubMed]
- Rush, C.; Nyara, M.; Moxon, J.V.; Trollope, A.; Cullen, B.; Golledge, J. Whole genome expression analysis within the angiotensin II-apolipoprotein E deficient mouse model of abdominal aortic aneurysm. BMC Genomics 2009, 10, 298. [Google Scholar] [CrossRef] [PubMed]
- Golledge, A.L.; Walker, P.; Norman, P.E.; Golledge, J. A systematic review of studies examining inflammation associated cytokines in human abdominal aortic aneurysm samples. Dis. Markers 2009, 26, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, C.M.; Ang, L.S.; Boivin, W.A.; Cooper, D.M.; Williams, S.J.; Zhao, H.; Hendel, A.; Folkesson, M.; Swedenborg, J.; Allard, M.F.; et al. Perforin-independent extracellular granzyme B activity contributes to abdominal aortic aneurysm. Am. J. Pathol. 2010, 176, 1038–1049. [Google Scholar] [CrossRef] [PubMed]
- Seko, Y.; Sato, O.; Takagi, A.; Tada, Y.; Matsuo, H.; Yagita, H.; Okumura, K.; Yazaki, Y. Perforin-secreting killer cell infiltration in the aortic tissue of patients with atherosclerotic aortic aneurysm. Jpn. Circ. J. 1997, 61, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Ocana, E.; Bohorquez, J.C.; Perez-Requena, J.; Brieva, J.A.; Rodriguez, C. Characterisation of T and B lymphocytes infiltrating abdominal aortic aneurysms. Atherosclerosis 2003, 170, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Newman, K.M.; Jean-Claude, J.; Li, H.; Ramey, W.G.; Tilson, M.D. Cytokines that activate proteolysis are increased in abdominal aortic aneurysms. Circulation 1994, 90, II224–II227. [Google Scholar] [PubMed]
- Xiong, W.; MacTaggart, J.; Knispel, R.; Worth, J.; Persidsky, Y.; Baxter, B.T. Blocking TNF-α attenuates aneurysm formation in a murine model. J. Immunol. 2009, 183, 2741–2746. [Google Scholar] [CrossRef] [PubMed]
- Bumdelger, B.; Kokubo, H.; Kamata, R.; Fujii, M.; Ishida, M.; Ishida, T.; Yoshizumi, M. Induction of Timp1 in smooth muscle cells during development of abdominal aortic aneurysms. Hiroshima J. Med. Sci. 2013, 62, 63–67. [Google Scholar] [PubMed]
- Chen, K.; Li, W.; Major, J.; Rahaman, S.O.; Febbraio, M.; Silverstein, R.L. Vav guanine nucleotide exchange factors link hyperlipidemia and a prothrombotic state. Blood 2011, 117, 5744–5750. [Google Scholar] [CrossRef] [PubMed]
- Harb, D.; Bujold, K.; Febbraio, M.; Sirois, M.G.; Ong, H.; Marleau, S. The role of the scavenger receptor CD36 in regulating mononuclear phagocyte trafficking to atherosclerotic lesions and vascular inflammation. Cardiovasc. Res. 2009, 83, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Pelham, C.J.; Agrawal, D.K. Emerging roles for triggering receptor expressed on myeloid cells receptor family signaling in inflammatory diseases. Expert Rev. Clin. Immunol. 2014, 10, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, S.O.; Zhou, G.; Silverstein, R.L. Vav protein guanine nucleotide exchange factor regulates CD36 protein-mediated macrophage foam cell formation via calcium and dynamin-dependent processes. J. Biol. Chem. 2011, 286, 36011–36019. [Google Scholar] [CrossRef] [PubMed]
- Roda-Navarro, P.; Reyburn, H.T. The traffic of the NKG2D/Dap10 receptor complex during natural killer (NK) cell activation. J. Biol. Chem. 2009, 284, 16463–16472. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Lee, G.S.; Brady, J.; Datta, S.; Katan, M.; Sheikh, A.; Martins, M.S.; Bunney, T.D.; Santich, B.H.; Moir, S.; et al. A hypermorphic missense mutation in PLCG2, encoding phospholipase Cγ2, causes a dominantly inherited autoinflammatory disease with immunodeficiency. Am. J. Hum. Genet. 2012, 91, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.C.; Lanier, L.L. NK cell development, homeostasis and function: Parallels with CD8+ T cells. Nat. Rev. Immunol. 2011, 11, 645–657. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information: Gene Expression Omnibus. Available online: http://www.ncbi.nlm.nih.gov/geo/ (accessed on 8 December 2014).
- Barrett, T.; Troup, D.B.; Wilhite, S.E.; Ledoux, P.; Rudnev, D.; Evangelista, C.; Kim, I.F.; Soboleva, A.; Tomashevsky, M.; Edgar, R. NCBI GEO: Mining tens of millions of expression profiles—Database and tools update. Nucleic Acids Res. 2007, 35, D760–D765. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information Home Page. Available online: http://www.ncbi.nlm.nih.gov/ (accessed on 8 December 2014).
- Tromp, G.; Gatalica, Z.; Skunca, M.; Berguer, R.; Siegel, T.; Kline, R.A.; Kuivaniemi, H. Elevated expression of matrix metalloproteinase-13 in abdominal aortic aneurysms. Ann. Vasc. Surg. 2004, 18, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 Years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- HUGO Gene Nomenclature Committee. Genenames.org, A Curated Online Repository of HGNC Approved Gene Nomenclature. Available online: http://www.genenames.org/ (accessed on 25 January 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hinterseher, I.; Schworer, C.M.; Lillvis, J.H.; Stahl, E.; Erdman, R.; Gatalica, Z.; Tromp, G.; Kuivaniemi, H. Immunohistochemical Analysis of the Natural Killer Cell Cytotoxicity Pathway in Human Abdominal Aortic Aneurysms. Int. J. Mol. Sci. 2015, 16, 11196-11212. https://doi.org/10.3390/ijms160511196
Hinterseher I, Schworer CM, Lillvis JH, Stahl E, Erdman R, Gatalica Z, Tromp G, Kuivaniemi H. Immunohistochemical Analysis of the Natural Killer Cell Cytotoxicity Pathway in Human Abdominal Aortic Aneurysms. International Journal of Molecular Sciences. 2015; 16(5):11196-11212. https://doi.org/10.3390/ijms160511196
Chicago/Turabian StyleHinterseher, Irene, Charles M. Schworer, John H. Lillvis, Elizabeth Stahl, Robert Erdman, Zoran Gatalica, Gerard Tromp, and Helena Kuivaniemi. 2015. "Immunohistochemical Analysis of the Natural Killer Cell Cytotoxicity Pathway in Human Abdominal Aortic Aneurysms" International Journal of Molecular Sciences 16, no. 5: 11196-11212. https://doi.org/10.3390/ijms160511196





