Physical Exercise Promotes Recovery of Neurological Function after Ischemic Stroke in Rats
Abstract
:1. Introduction
2. Results
2.1. Physical Exercise Improves Recovery of Neurological Function (NF)
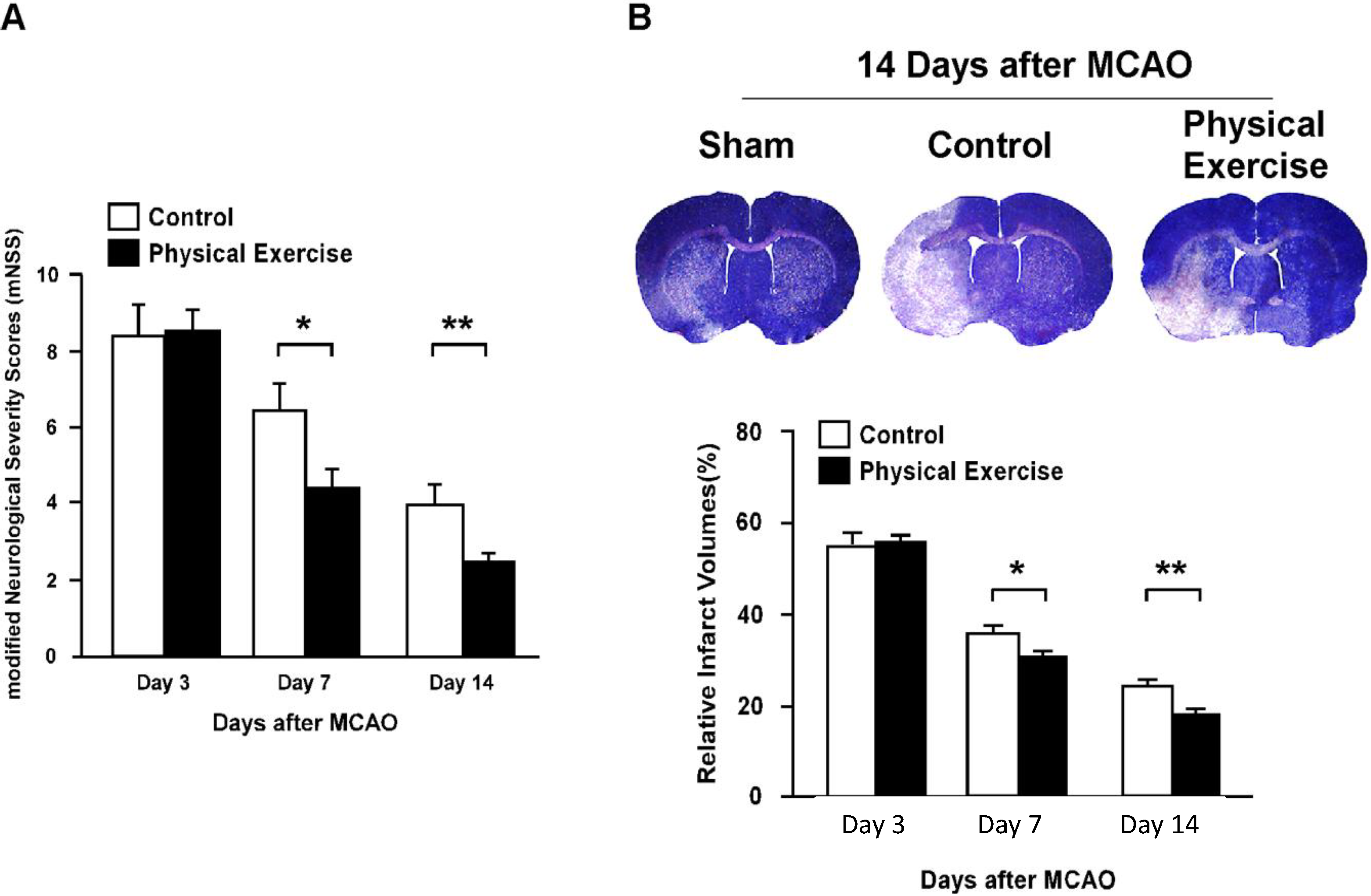
2.2. Physical Exercise Reduces the Volume of the Infarct Area after an Ischemic Stroke
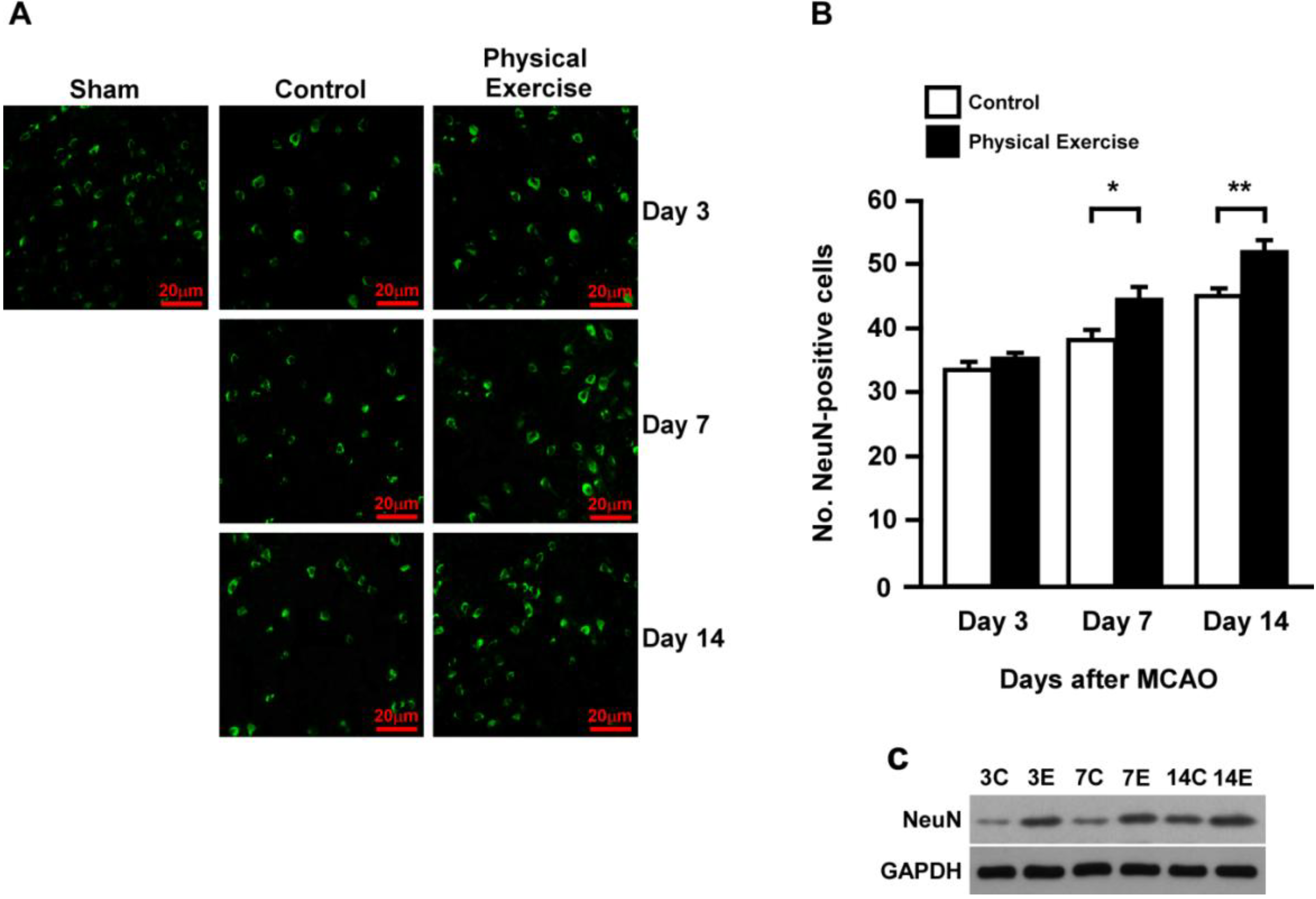
2.3. Physical Exercise Increases Newborn Neurons and Neural Progenitor Cells
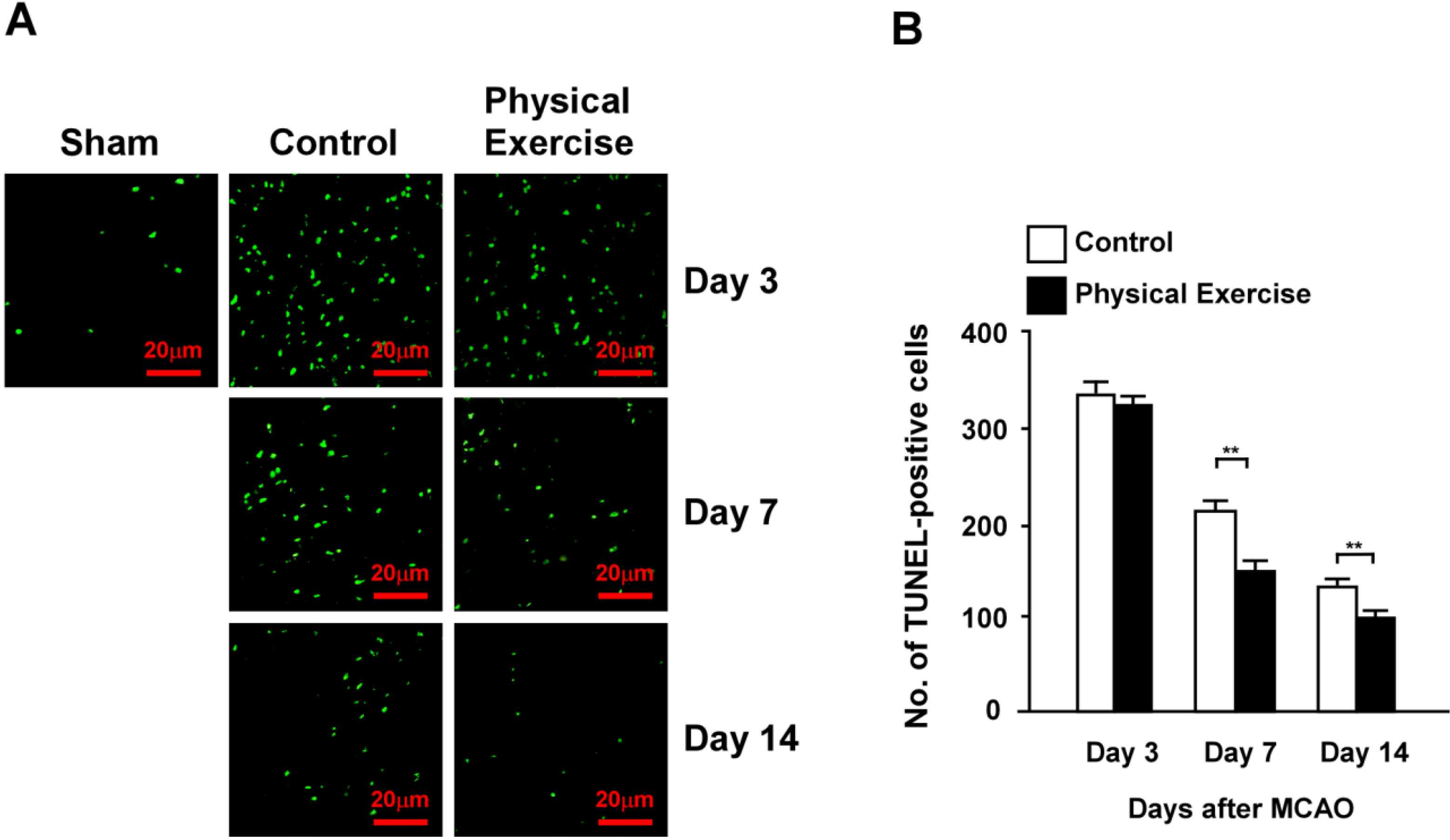
2.4. Physical Exercise Decreases Apoptosis in the Peri-Infarct Region

2.5. Physical Exercise Activates Insulin-Like Growth Factor-1 (IGF-1)/Akt Signaling in the Peri-Infarct Region

2.6. Discussion
2.6.1. Physical Exercise Promotes Nestin- and NeuN-Positive Cells, and Attenuates Apoptosis in the Peri-Infarct Region
2.6.2. Physical Exercise Can Promote the Expression of IGF-1 in the Peri-Infarct Region
3. Experimental Section
3.1. Animals and Treatments
3.2. Exercise Training and Function Testing
3.3. Tissue Preparation for Histochemistry
3.4. Nissl Staining
3.5. Immunofluorescence Staining
3.6. Deoxynucleotidyl Transferase-Mediated dUTP in Situ Nick-End Labeling (TUNEL) Assay
3.7. Western Blotting Analysis
3.8. Image Analysis and Quantification
3.9. Statistical Analysis
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhang, P.; Zhang, Y.; Zhang, J.; Wu, Y.; Jia, J.; Wu, J.; Hu, Y. Early exercise protects against cerebral ischemic injury through inhibiting neuron apoptosis in cortex in rats. Int. J. Mol. Sci. 2013, 14, 6074–6089. [Google Scholar] [CrossRef]
- Knecht, S.; Hesse, S.; Oster, P. Rehabilitation after stroke. Dtsch. Arztebl. Int. 2011, 108, 600–606. [Google Scholar]
- Zhang, L.; Hu, X.; Luo, J.; Li, L.; Chen, X.; Huang, R.; Pei, Z. Physical exercise improves functional recovery through mitigation of autophagy, attenuation of apoptosis and enhancement of neurogenesis after MCAO in rats. BMC Neurosci. 2013, 14, 46–53. [Google Scholar] [CrossRef]
- De la Perez, O.N.; Davalos, A. Neuroprotection in cerebral infarction: The opportunity of new studies. Cerebrovasc. Dis. 2007, 24, 153–156. [Google Scholar] [CrossRef]
- Shimodozono, M.; Noma, T.; Nomoto, Y.; Hisamatsu, N.; Kamada, K.; Miyata, R.; Matsumoto, S.; Ogata, A.; Etoh, S.; Basford, J.R.; et al. Benefits of a repetitive facilitative exercise program for the upper paretic extremity after subacute stroke: A randomized controlled trial. Neurorehabil. Neural Repair. 2012, 27, 296–305. [Google Scholar]
- Eadie, B.D.; Redila, V.A.; Christie, B.R. Voluntary exercise alters the cytoarchitecture of the adult dentate gyrus by increasing cellular proliferation, dendritic complexity, and spine density. J. Comp. Neurol. 2005, 486, 39–47. [Google Scholar] [CrossRef]
- Dragert, K.; Zehr, E.P. High-intensity unilateral dorsiflexor resistance training results in bilateral neuromuscular plasticity after stroke. Exp. Brain Res. 2013, 225, 93–104. [Google Scholar] [CrossRef]
- Lee, S.U.; Kim, D.Y.; Park, S.H.; Choi, D.H.; Park, H.W.; Han, T.R. Mild to moderate early exercise promotes recovery from cerebral ischemia in rats. Can. J. Neurol. Sci. 2009, 36, 443–449. [Google Scholar]
- Carro, E.; Trejo, J.L.; Busiguina, S.; Torres-Aleman, I. Circulating insulin-like growth factor 1 mediates the protective effects of physical exercise against brain insults of different etiology and anatomy. J. Neurosci. 2001, 21, 5678–5684. [Google Scholar]
- Trejo, J.L.; Carro, E.; Torres-Aleman, I. Circulating insulin-like growth factor 1 mediates exercise-induced increases in the number of new neurons in the adult hippocampus. J. Neurosci. 2001, 21, 1628–1634. [Google Scholar]
- De Smedt, A.; Brouns, R.; Uyttenbogaart, M.; de Raedt, S.; Moens, M.; Wilczak, N.; Luijckx, G.J.; de Keyser, J. Insulin-like growth factor I serum levels influence ischemic stroke outcome. Stroke 2011, 8, 2180–2185. [Google Scholar]
- Russo, V.C.; Gluckman, P.; Feldman, E.L.; Werther, G.A. The insulin-like growth factor system and its pleiotropic functions in the brain. Endocr. Rev. 2005, 26, 916–943. [Google Scholar] [CrossRef]
- Yamashita, T.; Ninomiya, M.; Hernández, A.P.; García-Verdugo, J.M.; Sunabori, T.; Sakaguchi, M.; Adachi, K.; Kojima, T.; Hirota, Y.; Kawase, T.; et al. Subventricular zone-derived neuroblasts migrate and differentiate into mature neurons in the post-stroke adultstriatum. J. Neurosci. 2006, 26, 6627–6636. [Google Scholar] [CrossRef]
- Franke, T.F.; Hornik, C.P.; Segev, L.; Shostak, G.A.; Sugimoto, C. PI3K/Akt and apoptosis: Size matters. Oncogene 2003, 22, 8983–8998. [Google Scholar] [CrossRef]
- Hanada, M.; Feng, J.; Hemmings, B.A. Structure, regulation and function of PKB/AKT—A major therapeutic target. Biochim. Biophys. Acta 2004, 1697, 3–16. [Google Scholar] [CrossRef]
- Saha, B.; Peron, S.; Murray, K.; Jaber, M.; Gaillard, A. Cortical lesion stimulates adult subventricular zone neural progenitor cell proliferation and migration to the site of injury. Stem Cell Res. 2013, 11, 965–977. [Google Scholar] [CrossRef]
- Thored, P.; Arvidsson, A.; Cacci, E.; Ahlenius, H.; Kallur, T.; Darsalia, V.; Ekdahl, C.T.; Kokaia, Z.; Lindvall, O. Persistent production of neurons from adult brain stem cells during recovery after stroke. Stem Cells 2006, 24, 739–747. [Google Scholar] [CrossRef]
- Brandt, M.D.; Maass, A.; Kempermann, G.; Storch, A. Physical exercise increases Notch activity, proliferation and cell cycle exit of type-3 progenitor cells in adult hippocampal neurogenesis. Eur. J. Neurosci. 2010, 32, 1256–1264. [Google Scholar] [CrossRef]
- Chung, H.; Li, E.; Kim, Y.; Kim, S.; Park, S. Multiple signaling pathways mediate ghrelin-induced proliferation of hippocampal neural stem cells. J. Endocrinol. 2013, 218, 49–59. [Google Scholar] [CrossRef]
- Rahmani, A.; Kheradmand, D.; Keyhanvar, P.; Shoae-Hassani, A.; Darbandi-Azar, A. Neurogenesis and increase in differentiated neural cell survival via phosphorylation of Akt1 after fluoxetine treatment of stem cells. Biomed. Res. Int. 2013, 582526, 1–9. [Google Scholar]
- Chan, P.H. Future targets and cascades for neuroprotective strategies. Stroke 2004, 35, 2748–2750. [Google Scholar] [CrossRef]
- Fresno Vara, J.A.; Casado, E.; de Castro, J.; Cejas, P.; Belda-Iniesta, C.; Gonzalez-Baron, M. PI3K/Akt signalling pathway and cancer. Cancer Treat Rev. 2004, 30, 193–204. [Google Scholar] [CrossRef]
- O’Kusky, J.; Ye, P. Neurodevelopmental effects of insulin-like growth factor signaling. Front. Neuroendocrinol. 2012, 33, 230–251. [Google Scholar] [CrossRef]
- Arsenijevic, Y.; Weiss, S.; Schneider, B.; Aebisher, P. Insulin-like growth factor-I is necessary for stem cell proliferation and demonstrates distinct actions of epidermal growth factor and fibroblast growth factor-2. J. Neurosci. 2001, 15, 7194–7202. [Google Scholar]
- Vicario-Abejón, C.; Yusta-Boyo, M.J.; Fernández-Moreno, C.; de Pablo, F. Locally born olfactory bulb stem cells proliferate in response to insulin-related factors and require endogenous insulin-like growth factor-I for differentiation into neurons and glia. J. Neurosci. 2003, 23, 895–906. [Google Scholar]
- Suh, H.S.; Zhao, M.L.; Derico, L.; Choi, N.; Lee, S.C. Insulin-like growth factor 1 and 2 (IGF1, IGF2) expression in human microglia: Differential regulation by inflammatory mediators. J. Neuroinflamm. 2013, 10, 37. [Google Scholar] [CrossRef]
- Hoehn, B.D.; Palmer, T.D.; Steinberg, G.K. Neurogenesis in rats after focal cerebral ischemia is enhanced by indomethacin. Stroke 2005, 36, 2718–2724. [Google Scholar] [CrossRef]
- Hu, X.Q.; Zheng, H.Q.; Yan, T.B.; Pan, S.Q.; Fang, J.; Jiang, R.S.; Ma, S.F. Physical exercise induces expression of CD31 and facilitates neural function recovery in rats with focal cerebral infarction. Neurol. Res. 2010, 32, 397–402. [Google Scholar] [CrossRef]
- Zea Longa, E.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar] [CrossRef]
- Leasure, J.L.; Grider, M. The effect of mild post-stroke exercise on reactive neurogenesis and recovery of somatosensation in aged rats. Exp. Neurol. 2010, 226, 58–67. [Google Scholar] [CrossRef]
- Borlongan, C.V.; Randall, T.S.; Cahill, D.W.; Sanberg, P.R. Asymmetrical motor behavior in rats with unilateral striatal excitotoxic lesions as revealed by the elevated body swing test. Brain Res. 1995, 676, 231–234. [Google Scholar]
- Schallert, T.; Kozlowski, D.A.; Humm, J.L.; Cocke, R.R. Use-dependent structural events in recovery of function. Adv. Neurol. 1997, 73, 229–238. [Google Scholar]
- Germano, A.F.; Dixon, C.E.; D’ Avella, D.; Hayes, R.L.; Tomasello, F. Behavior deficits following experimental subarachnoid hemorrhage in the rat. J. Neurotrauma 1994, 11, 345–353. [Google Scholar] [CrossRef]
- Imai, H.; Harland, J.; McCulloch, J.; Graham, D.I.; Brown, S.M.; Macrae, I.M. Specific expression of the cell cycle regulation proteins, GADD34 and PCNA, in the peri-infarct zone after focal cerebral ischemia in the rat. Eur. J. Neurosci. 2002, 15, 1929–1936. [Google Scholar] [CrossRef]
- Swanson, R.A.; Morton, M.T.; Tsao-Wu, G.; Savalos, R.A.; Davidson, C.; Sharp, F.R. A semiautomated method for measuring brain infarct volume. J. Cereb. Blood Flow Metab. 1990, 10, 290–293. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.S.; Li, J.J.; Xing, S.H.; Li, C.; Li, Y.L.; Dang, C.; Fan, Y.H.; Yu, J.; Pei, Z.; et al. Autophagosomes accumulation is associated with β-amyloid deposits and secondary damage in the thalamus after focal cortical infarction in hypertension rats. J. Neurochem. 2012, 120, 564–573. [Google Scholar] [CrossRef]
- Kronenberg, G.; Reuter, K.; Steiner, B.; Brandt, M.D.; Jessberger, S.; Yamaguchi, M.; Kempermann, G. Subpopulations of proliferating cells of the adult hippocampus respond differently to physiologic neurogenic stimuli. J. Comp. Neurol. 2003, 467, 455–463. [Google Scholar] [CrossRef]
- Dang, J.; Mitkari, B.; Kipp, M.; Beyer, C. Gonadal steroids prevent cell damage and stimulate behavioral recovery after transient middle cerebral artery occlusion in male and female rats. Brain Behav. Immun. 2011, 25, 715–726. [Google Scholar] [CrossRef]
- Agulla, J.; Brea, D.; Campos, F.; Sobrino, T.; Argibay, B.; Al-Soufi, W.; Blanco, M.; Castillo, J.; Ramos-Cabrer, P. In vivo theranostics at the peri-infarct region in cerebral ischemia. Theranostics 2013, 4, 90–105. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zheng, H.-Q.; Zhang, L.-Y.; Luo, J.; Li, L.-L.; Li, M.; Zhang, Q.; Hu, X.-Q. Physical Exercise Promotes Recovery of Neurological Function after Ischemic Stroke in Rats. Int. J. Mol. Sci. 2014, 15, 10974-10988. https://doi.org/10.3390/ijms150610974
Zheng H-Q, Zhang L-Y, Luo J, Li L-L, Li M, Zhang Q, Hu X-Q. Physical Exercise Promotes Recovery of Neurological Function after Ischemic Stroke in Rats. International Journal of Molecular Sciences. 2014; 15(6):10974-10988. https://doi.org/10.3390/ijms150610974
Chicago/Turabian StyleZheng, Hai-Qing, Li-Ying Zhang, Jing Luo, Li-Li Li, Menglin Li, Qingjie Zhang, and Xi-Quan Hu. 2014. "Physical Exercise Promotes Recovery of Neurological Function after Ischemic Stroke in Rats" International Journal of Molecular Sciences 15, no. 6: 10974-10988. https://doi.org/10.3390/ijms150610974



