An Environmentally Benign Protocol for Aqueous Synthesis of Tetrahydrobenzo[b]Pyrans Catalyzed by Cost-Effective Ionic Liquid
Abstract
:1. Introduction
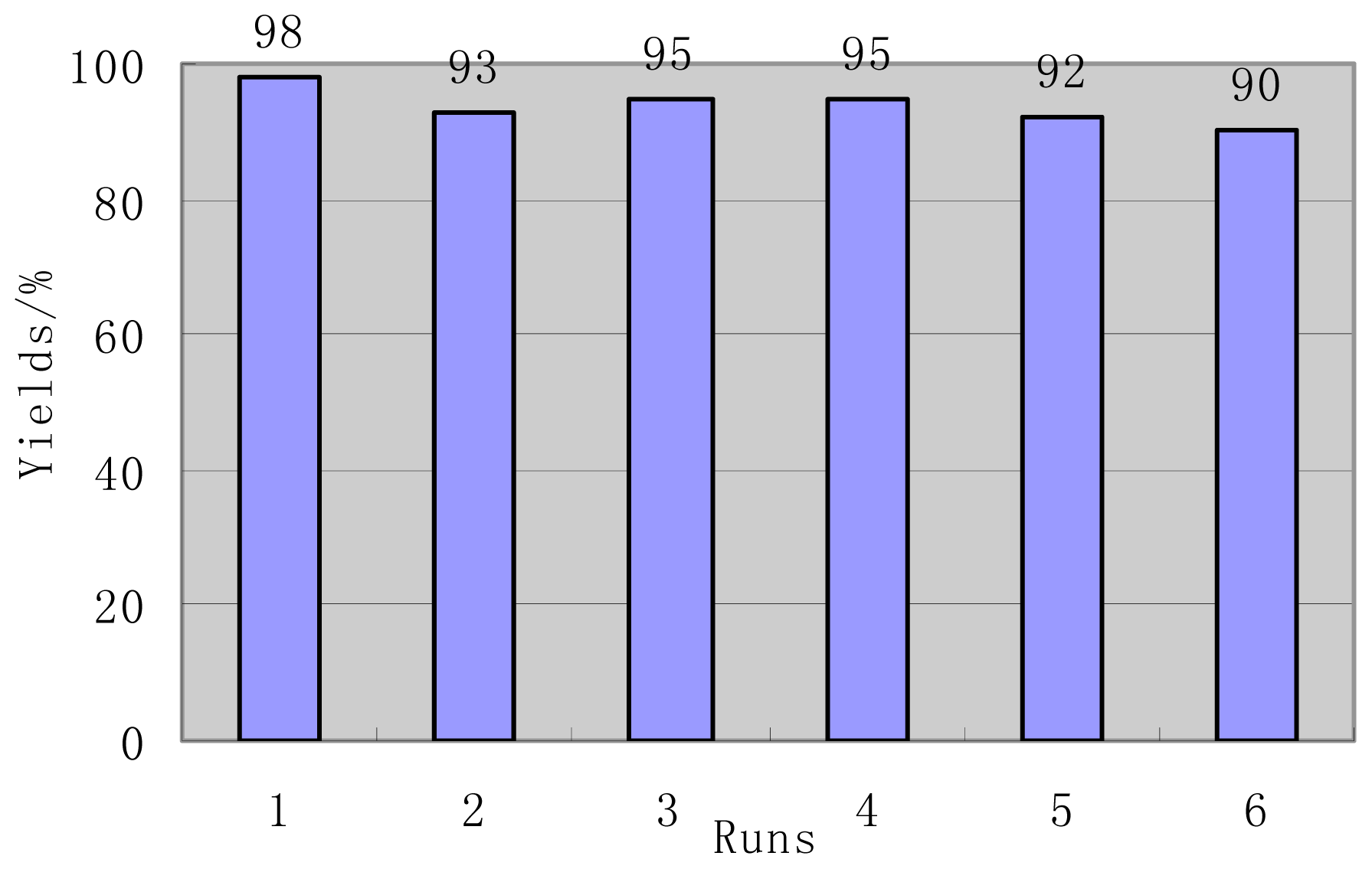
2. Results and Discussion
3. Experimental Section
3.1. Materials and General Methods
3.2. General Procedure for the Synthesis of the ChCl-Based Ionic Liquids
3.3. Typical Procedure for One-Pot Synthesis of Tetrahydrobenzo[b]Pyrans
3.4. Spectroscopic Data of Typical Products
4. Conclusions
Supplementary Information
ijms-15-06897-s001.pdfAcknowledgments
Conflicts of Interest
References
- Hafez, E.A.A.; Elnagdi, M.H.; Elagemey, A.G.A.; EI-Taweel, F.M.A.A. Nitriles in heterocyclic synthesis-novel synthesis of benzo[c]coumarin and of benzo[c]pyrano[3,2-c]quinoline derivatives. Heterocycles 1987, 26, 903–907. [Google Scholar]
- Armesto, D.; Horspool, W.M.; Martin, N.; Ramos, A.; Seaone, C. Synthesis of cyclobutenes by the novel photochemical ring contraction 4-sbustituted 2-amino-3,5-dicyano-6-phenyl-4H-pyrans. J. Org. Chem 1989, 54, 3069–3072. [Google Scholar]
- Ellis, G.P. The Chemistry of Heterocyclic Compounds Chromenes, Chromanes and Chromones; Weissberger, A., Taylor, E.C., Eds.; John Wiley: New York, NY, USA, 1977; pp. 11–139. [Google Scholar]
- Martin, N.; Martin, G.; Seoane, C.; Maco, J.L.; Albert, A.; Cano, F.H. Michael addition of malononitrile to α-acetylcinnamamides. Ann. Chem 1993, 7, 801. [Google Scholar]
- Marco, J.L.; Martin, N.; Grau, A.M.; Seoane, C.; Albert, A.; Cano, F.H. Development of methods for the synthesis of chiral, highly functionalized 2-aminno-4-aryl-4H-pyrans. Tetrahedron 1994, 50, 3509. [Google Scholar]
- Green, G.R.; Evans, J.M.; Vong, A.K. Comprehensive Heterocyclic Chemistry II; Ktritsky, A.R., Rees, C.W., Scriven, E.F.V., Eds.; Permagon Press: Oxford UK, 1995; Volume 5, p. 469. [Google Scholar]
- Jin, T.S.; Wang, A.Q.; Wang, X.; Zhang, J.S.; Li, T.S. A clean one-pot synthesis of tetrahydrobenzo[b]pyran derivatives catalyzed by hexadecyltrimethyl ammonium bromide in aqueous media. Synlett 2004, 5, 871–873. [Google Scholar]
- Bonsignore, L.; Loy, G.; Secci, D.; Calignano, A. Synthesis and pharmacological activity of 2-oxo-(2H) 1-benzopyran-3-carboxamide derivatives. Eur. J. Med. Chem 1993, 28, 517–520. [Google Scholar]
- Devi, I.; Bhuyan, P.J. Sodium bromide catalysed one-pot synthesis of tetrahydrobenzo[b]pyrans via a three-component cyclocondensation under microwave irradiation and solvent free conditions. Tetrahedron Lett 2004, 45, 8625–8627. [Google Scholar]
- Konkoy, C.S.; Fick, D.B.; Cai, S.X.; Lan, N.C.; Keana, J.F.W. Substituted 5-oxo-5,6,7,8-tetrahydro-4h-1-benzopyrans and benzothiopyrans and the use thereof as potentiators of ampa. PCT Int. Appl WO0075123, 2000. [Google Scholar]Chem. Abstr 2001, 134, 29313a.
- Dömling, A. Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev 2006, 106, 17–89. [Google Scholar]
- Ganem, B. Strategies for innovation in multicomponent reaction design. Acc. Chem. Res 2009, 42, 463–472. [Google Scholar]
- Toure, B.B.; Hall, D.G. Natural product synthesis using multicomponent reaction strategies. Chem. Rev 2009, 109, 4439–4486. [Google Scholar]
- Tahmassebi, D.; Bryson, J.A.; Binz, S.I. 1,4-Diazabicyclo[2.2.2]octane as an efficient catalyst for a clean, one-pot synthesis of tetrahydrobenzo[b]pyran derivatives via multicomponent reaction in aqueous media. Synth. Commun 2011, 41, 2701–2711. [Google Scholar]
- Lian, X.Z.; Huang, Y.; Li, Y.Q.; Zheng, W.J. A green synthesis of tetrahydrobenzo[b]pyran derivatives through three-component condensation using N-methylimidazole as organocatalyst. Monatsh. Chem 2008, 139, 129–131. [Google Scholar]
- Balalaie, S.; Sheikh-Ahmadi, M.; Bararjanian, M. Tetra-methyl ammonium hydroxide: An efficient and versatile catalyst for the one-pot synthesis of tetrahydrobenzo[b]pyran derivatives in aqueous media. Catal. Commun 2007, 8, 1724–1728. [Google Scholar]
- Balalaie, S.; Bararjanian, M.; Sheikh-Ahmadi, M. Diammonium hydrogen phosphate: An efficient and versatile catalyst for the one-pot synthesis of tetrahydrobenzo[b]pyran derivatives in aqueous media. Synth. Commun 2007, 37, 1097–1108. [Google Scholar]
- Pore, D.M.; Undale, K.A.; Dongare, B.B.; Desai, U.V. Potassium phosphate catalyzed a rapid three-component synthesis of tetrahydrobenzo[b]pyran at ambient temperature. Catal. Lett 2009, 132, 104–108. [Google Scholar]
- Katkar, S.S.; Lande, M.K.; Arbad, B.R.; Gaikwad, S.T. A recyclable and highly effective ZnO-beta zeolite as a catalyst for one-pot three-component synthesis of tetrahydrobenzo[b]pyrans. Chin. J. Chem 2011, 29, 199–202. [Google Scholar]
- Rathod, S.; Arbad, B.; Land, M. Preparation, characterization, and catalytic application of a nanosized Ce1Mgx Zr1−x O2 solid heterogeneous catalyst for the synthesis of Tetrahydrobenzo[b]pyran derivatives. Chin. J. Catal 2010, 31, 631–636. [Google Scholar]
- Tabatabaeian, K.; Heidari, H.; Mamaghani, M.; Mahmoodi, N.O. Ru(II) complexes bearing tertiary phosphine ligands: A novel and efficient homogeneous catalyst for one-pot synthesis of dihydropyrano[3,2-c]chromene and tetrahydrobenzo[b]pyran derivatives. Appl. Organometal. Chem 2012, 26, 56–61. [Google Scholar]
- Hekmatshoar, R.; Majedi, S.; Bakhtiari, K. Sodium selenate catalyzed simple and efficient synthesis of tetrahydro benzo[b]pyran derivatives. Catal. Commun 2008, 9, 307–310. [Google Scholar]
- Balalaie, S.; Bararjanian, M.; Amani, A.M.; Movassagh, B. (S)-Proline as a neutral and efficient catalyst for the one-pot synthesis of tetrahydrobenzo[b]pyran derivatives in aqueous media. Synlett 2006, 263–266. [Google Scholar]
- Li, Y.; Chen, H.; Shi, C.; Ji, S. Efficient one-pot synthesis of spirooxindole derivatives catalyzed by l-proline in aqueous medium. J. Comb. Chem 2010, 12, 231–237. [Google Scholar]
- Khurana, J.M.; Nand, B.; Saluja, P. DBU: A highly efficient catalyst for one-pot synthesis of substituted 3,4-dihydropyrano[3,2-c]chromenes, dihydropyrano[4,3-b]pyranes, 2-amino-4H-benzo[h]chromenes and 2-amino-4H benzo[g]chromenes in aqueous medium. Tetrahedron 2010, 66, 5637–5641. [Google Scholar]
- Ziarani, G.M.; Abbasi, A.; Badiei, A.; Aslani, Z. An efficient synthesis of tetrahydrobenzo[b]pyran derivatives using sulfonic acid functionalized silica as an efficient catalyst. E-J. Chem 2011, 8, 293–295. [Google Scholar]
- Nemouchi, S.; Boulcina, R.; Carboni, B.; Debache, A. Phenylboronic acid as an efficient and convenient catalyst for a three-component synthesis of tetrahydrobenzo[b]pyrans. C. R. Chim 2012, 15, 394–397. [Google Scholar]
- Oskooie, H.A.; Heravi, M.M.; Karimi, N.; Zadeh, M.E. Versatile catalyst for the one-pot synthesis of tetrahydrobenzo[b]pyran derivatives. Synth. Commun 2011, 41, 436–440. [Google Scholar]
- Sabitha, G.; Arundhathi, K.; Sudhakar, K.; Sastry, B.S.; Yadav, J.S. Cerium(III) chloride-catalyzed one-pot synthesis of tetrahydrobenzo[b]pyrans. Synth. Commun 2009, 39, 433–442. [Google Scholar]
- Khaksar, S.; Rouhollahpour, A.; Talesh, S.M. A facile and efficient synthesis of 2-amino-3-cyano-4H-chromenes and tetrahydrobenzo[b]pyrans using 2,2,2-trifluoroethanol as a metal-free and reusable medium. J. Fluor. Chem 2012, 141, 11–15. [Google Scholar]
- Feng, C.; Wang, Q.; Lu, C.; Yang, G.; Chen, Z. Green Synthesis of Tetrahydrobenzo[b]Pyrans by Microwave Assisted Multi-Component One-Pot Reactions in PEG-400. Comb. Chem. High Throughput Screen 2012, 15, 100–103. [Google Scholar]
- Li, J.T.; Xu, W.Z.; Yang, L.C.; Li, T.S. One-pot synthesis of 2-amino-4-aryl-3- carbalkoxy-7,7-dimethyl-5,6,7,8-tetrahydrobenzo[b]pyran derivatives catalyzed by KF/basic Al2O3 under ultrasound irradiation. Synth. Commun 2004, 34, 4565–4571. [Google Scholar]
- Zhang, Q.; Zhang, S.; Deng, Y. Recent advances in ionic liquid catalysis. Green Chem 2011, 13, 2619–2637. [Google Scholar]
- Niedermeyer, H.; Hallett, J.P.; Villar-Garcia, I.J.; Hunt, P.A.; Welton, T. Mixtures of ionic liquids. Chem. Soc. Rev 2012, 41, 7780–7802. [Google Scholar]
- Pereira, M.M.A. Immobilized Ionic Liquids in Organic Chemistry. Curr. Org. Chem 2012, 16, 1680–1710. [Google Scholar]
- Tang, S.; Baker, G.A.; Zhao, H. Ether- and alcohol-functionalized task-specific ionic liquids: Attractive properties and applications. Chem. Soc. Rev 2012, 41, 4030–4066. [Google Scholar]
- Payagala, T.; Armstrong, D.W. Chiral ionic liquids: A compendium of syntheses and applications 2005–2012. Chirality 2012, 24, 17–53. [Google Scholar]
- Isambert, N.; Duque, M.M.S.; Plaquevent, J.-C.; Génisson, Y.; Rodriguez, J.; Constantieux, T. Multicomponent reactions and ionic liquids: A perfect synergy for eco-compatible heterocyclic synthesis. Chem. Soc. Rev 2011, 40, 1347–1357. [Google Scholar]
- Ying, A.-G.; Ye, W.-D.; Liu, L.; Wu, G.-F.; Chen, X.-Z.; Qian, S.; Zhang, Q.-P. Progress in the application of ionic liquids to organic synthesis. Chin. J. Org. Chem 2008, 28, 2081–2094. [Google Scholar]
- Ying, A.-G.; Chen, X.-Z.; Ye, W.-D.; Zhang, D.-F.; Liu, L.; Chen, J.-H. Application of ionic liquids in organic synthesis promoted by microwave irradiation. Prog. Chem 2008, 20, 1642–1650. [Google Scholar]
- Salvi, P.P.; Mandhare, A.M.; Sartape, A.S.; Pawar, D.K.; Han, H.S.; Kolekar, S.S. An efficient protocol for synthesis of tetrahydrobenzo[b]pyrans using amino functionalized ionic liquid. C. R. Chim 2011, 14, 878–882. [Google Scholar]
- Wen, L.-R.; Xie, H.-Y.; Li, M. A basic ionic liquid catalyzed reaction of benzothiazole, aldehydes, and 5,5-dimethyl-1,3-cyclohexanedione: Efficient synthesis of tetrahydrobenzo[b]pyrans. J. Heterocycl. Chem 2009, 46, 954–959. [Google Scholar]
- Zheng, J.; Li, Y. Basic ionic liquid-catalyzed multicomponent synthesis of tetrahydrobenzo[b]pyrans and pyrano[c]chromenes. Mendeleev Commun 2011, 21, 280–281. [Google Scholar]
- Shaterian, H.R.; Arman, M.; Rigi, F. Domino Knoevenagel condensation, Michael addition, and cyclization using ionic liquid, 2-hydroxyethylammonium formate, as a recoverable catalyst. J. Mol. Liq 2011, 158, 145–150. [Google Scholar]
- Ruβ, C.; König, B. Low melting mixtures in organic synthesis—An alternative to ionic liquids? Green Chem 2012, 14, 2969–2982. [Google Scholar]
- Zhang, Q.; Vigier, K.D.O.; Royer, S.; Jérôme, F. Deep eutectic solvents: Syntheses, properties and applications. Chem. Soc. Rev 2012, 41, 7108–7146. [Google Scholar]
- Abelló, S.; Medina, F.; Rodríguez, X.; Cesteros, Y.; Salagre, P.; Sueiras, J.E.S.; Tichit, D.; Coq, B. Supported choline hydroxide (ionic liquid) as heterogeneous catalyst for aldol condensation reactions. Chem. Commun 2004, 1096–1097. [Google Scholar]
- Ying, A.-G.; Liu, L.; Wu, G.-F.; Chen, G.; Chen, X.-Z.; Ye, W.-D. Aza-Michael addition of aliphatic or aromatic amines to alpha,beta-unsaturated compounds catalyzed by a DBU-derived ionic liquid under solvent-free conditions. Tetrahedron Lett 2009, 50, 1653–1657. [Google Scholar]
- Ying, A.-G.; Wang, L.-M.; Deng, H.-X.; Chen, J.-H.; Chen, X.-Z.; Ye, W.-D. Green and efficient aza-Michael additions of aromatic amines to alpha,beta-unsaturated ketones catalyzed by DBU based task-specific ionic liquids without solvent. Arkivoc 2009, 288–298. [Google Scholar]
- Ying, A.-G.; Liu, L.; Wu, G.-F.; Chen, X.-Z.; Ye, W.-D.; Chen, J.-H.; Zhang, K.-Y. Knoevenagel condensation catalyzed by DBU brønsted ionic liquid without solvent. Chem. Res. Chin. Univ 2009, 25, 876–881. [Google Scholar]
- Ying, A.-G.; Wang, L.-M.; Wang, L.-L.; Chen, X.-Z.; Ye, W.-D. Green and efficient Knoevenagel condensation catalysed by a DBU based ionic liquid in water. J. Chem. Res 2010, 30–33. [Google Scholar]
- Ying, A.-G.; Chen, X.-Z.; Wu, C.-L.; Zheng, R.-H.; Liang, H.-D.; Ge, C.-H. Novel task-specific ionic liquids as solvent for Michael addition of methylene active compounds to chalcones without any catalyst. Synth. Commun 2012, 42, 3455–3462. [Google Scholar]
- Ying, A.-G.; Zhang, Q.-H.; Li, H.-M.; Shen, G.-F.; Gong, W.-Z.; He, M.-J. An environmentally benign protocol: Catalyst-free Michael addition of aromatic amines to alpha,beta-unsaturated ketones in glycerol. Res. Chem. Intermed 2013, 39, 517–525. [Google Scholar]
- Ying, A.; Xu, S.; Liu, S.; Ni, Y.; Yang, J.; Wu, C. Novel multiple-acidic ionic liquids: Catalysts for environmentally friendly benign synthesis of trans-beta-nitrostyrenes under solvent-free conditions. Ind. Eng. Chem. Res 2014, 53, 547–552. [Google Scholar]
- Ying, A.; Ni, Y.; Xu, S.; Liu, S.; Yang, J.; Li, R. Novel DABCO based ionic liquids: Green and efficient catalysts with dual catalytic roles for aqueous Knoevenagel condensation. Ind. Eng. Chem. Res 2014, 53, 5678–5682. [Google Scholar]
- Ying, A.; Liu, S.; Ni, Y.; Qiu, F.; Xu, S.; Tang, W. Ionic tagged DABCO grafted on magnetic nanoparticles: A water-compatible catalyst for aqueous aza-Michael addition of amines to α,β-unsatuated amides. Catal. Sci. Technol 2014. [Google Scholar] [CrossRef]
- Gao, S.; Tsai, C.H.; Tseng, C.; Yao, C.F. Fluoride ion catalyzed multicomponent reactions for efficient synthesis of 4H-chromene and N-arylquinoline derivatives in aqueous media. Tetrahedron 2008, 64, 9143–9149. [Google Scholar]
- Banerjee, S.; Horn, A.; Khatri, H.; Sereda, G. A green one-pot multicomponent synthesis of 4H-pyrans and polysubstituted aniline derivatives of biological, pharmacological, and optical applications using silica nanoparticles as reusable catalyst. Tetrahedron Lett 2011, 52, 1878–1881. [Google Scholar]



 | |||||
|---|---|---|---|---|---|
| Entry | Ionic Liquids | Mol % | Solvents (10 mL) | Time (h) | Yield (%) b |
| 1 | [bmim]BF4 | 20 | Water | 5 | 17 |
| 2 | [bmim]PF6 | 20 | Water | 5 | 8 |
| 3 | [DBU][Ac] | 20 | Water | 2 | 31 |
| 4 | [DBU][Lac] | 20 | Water | 2 | 28 |
| 5 | [Ch][Ac] | 20 | Water | 3 | 52 |
| 6 | [Ch][Pr] | 20 | Water | 3 | 47 |
| 7 | [Ch][Bu] | 20 | Water | 3 | 55 |
| 8 | [Ch][Lac] | 20 | Water | 3 | 55 |
| 9 | [Ch][OTf] | 20 | Water | 3 | 38 |
| 10 | [Ch][OH] | 20 | Water | 2 | 95 |
| 11 | [bmim]OH | 20 | Water | 3 | 86 |
| 12 | [Ch][OH] | 10 | Water | 2 | 96 |
| 13 | [Ch][OH] | 5 | Water | 5 | 83 |
| 14 | [Ch][OH] | 30 | Water | 2 | 95 |
| 15 | [Ch][OH] | 50 | Water | 2 | 96 |
| 16 c | [Ch][OH] | 10 | CH2Cl2 | 5 | Trace |
| 17 c | [Ch][OH] | 10 | THF | 3 | Trace |
| 18 c | [Ch][OH] | 10 | Ethanol | 2 | 82 |
| 19 c | [Ch][OH] | 10 | Methanol | 2 | 68 |
 | ||||||
|---|---|---|---|---|---|---|
| Entry | R1 | Time (h) | Products | Yields (%) a | Mp (°C) Measured | Mp (°C) References |
| 1 | C6H5 | 1 | 2a | 92 | 232–234 | 234–235 [57] |
| 4-NO2-C6H4 | 30 min | 2b | 88 | 176–177 | 177–178 [27] | |
| 2 | 3-NO2-C6H4 | 20 min | 2c | 95 | 213–214 | 213–215 [27] |
| 3 | 4-Cl-C6H4 | 1 | 2d | 86 | 212–214 | 209–210 [19] |
| 4 | 4-F-C6H4 | 10 min | 2e | 90 | 198–200 | 200 [28] |
| 5 | 4-Br-C6H4 | 1 | 2f | 91 | 165–168 | 169–170[30] |
| 6 | 4-CN-C6H4 | 30 min | 2g | 90 | 227–229 | 224–226 [18] |
| 7 | 4-OH-C6H4 | 2 | 2h | 86 | 206–298 | 207–209 [28] |
| 8 | 4-MeO-C6H4 | 3 | 2i | 95 | 198–200 | 199–200 [28] |
| 9 | 4-N(Me)2-C6H4 | 5 | 2j | 98 | 216–218 | 217–218 [57] |
| 10 | 3,4-2(MeO)-C6H3 | 3 | 2k | 92 | 184–185 | 182–184 [14] |
| 11 c | 3-Pyridine | 10 | 2l | 0 | - | - |
| 12 | 2-Furyl | 3 | 2m | 82 | 223–226 | 222–224 [57] |
| 13 c | 2-Naphthyl | 10 | 2n | 0 | - | - |
| 14 | 2-Thienyl | 1 | 2o | 85 | 210–212 | 210–212 [18] |
| 15 b | C6H5-CH=CH- | 5 | 2p | 68 | 205–207 | 208–210 [27] |
| 16 b | CH3CH2 | 3 | 2q | 85 | 190–192 | 190–194 [22] |
| 17 b | CH3CH2CH2 | 3 | 2r | 79 | 192–194 | 192–193 [58] |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hu, H.; Qiu, F.; Ying, A.; Yang, J.; Meng, H. An Environmentally Benign Protocol for Aqueous Synthesis of Tetrahydrobenzo[b]Pyrans Catalyzed by Cost-Effective Ionic Liquid. Int. J. Mol. Sci. 2014, 15, 6897-6909. https://doi.org/10.3390/ijms15046897
Hu H, Qiu F, Ying A, Yang J, Meng H. An Environmentally Benign Protocol for Aqueous Synthesis of Tetrahydrobenzo[b]Pyrans Catalyzed by Cost-Effective Ionic Liquid. International Journal of Molecular Sciences. 2014; 15(4):6897-6909. https://doi.org/10.3390/ijms15046897
Chicago/Turabian StyleHu, Huanan, Fangli Qiu, Anguo Ying, Jianguo Yang, and Haiping Meng. 2014. "An Environmentally Benign Protocol for Aqueous Synthesis of Tetrahydrobenzo[b]Pyrans Catalyzed by Cost-Effective Ionic Liquid" International Journal of Molecular Sciences 15, no. 4: 6897-6909. https://doi.org/10.3390/ijms15046897





