Prevention of Intra-Abdominal Adhesion by Bi-Layer Electrospun Membrane
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Bi-Layer Electrospun Fibrous Membrane
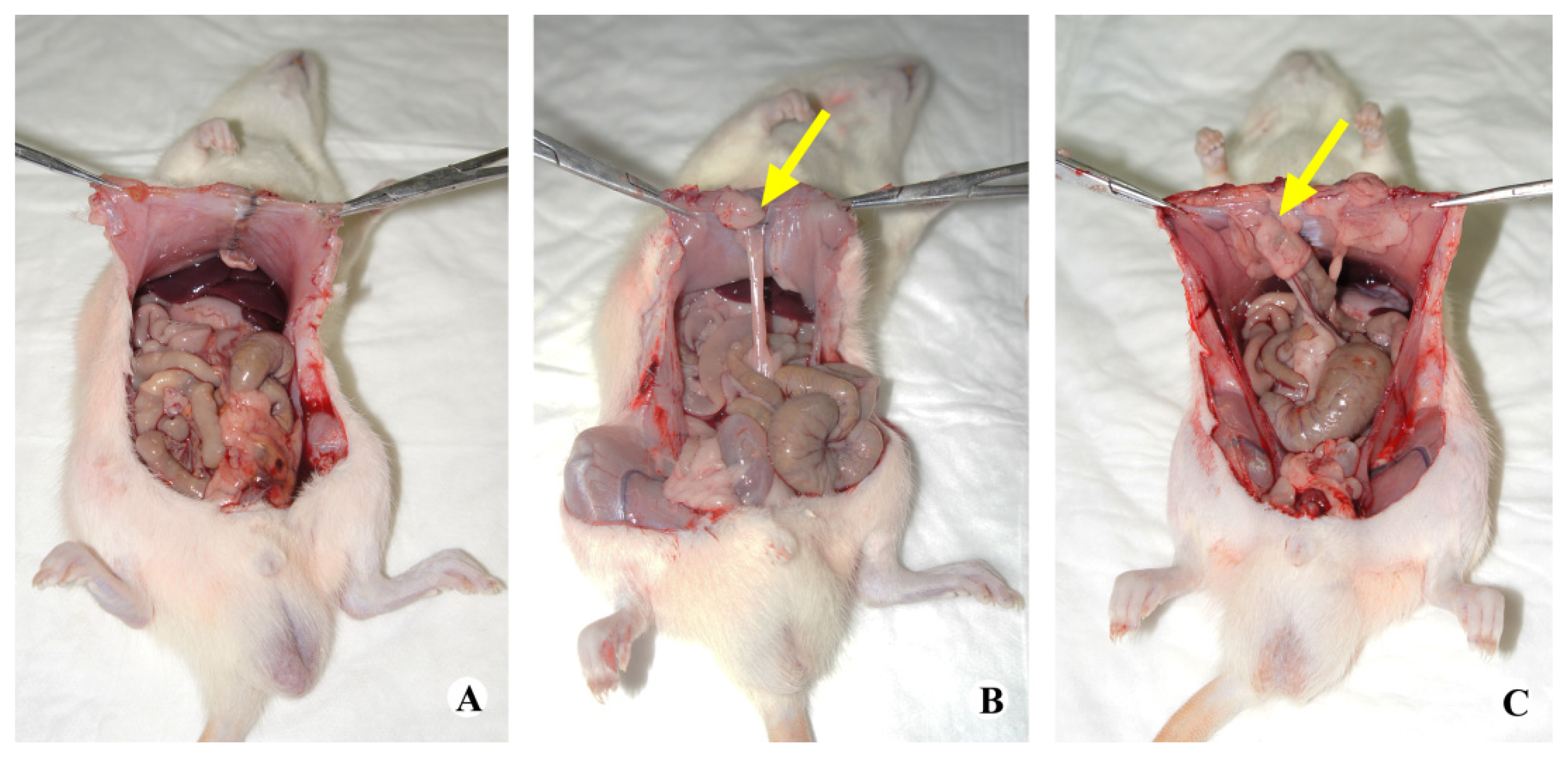
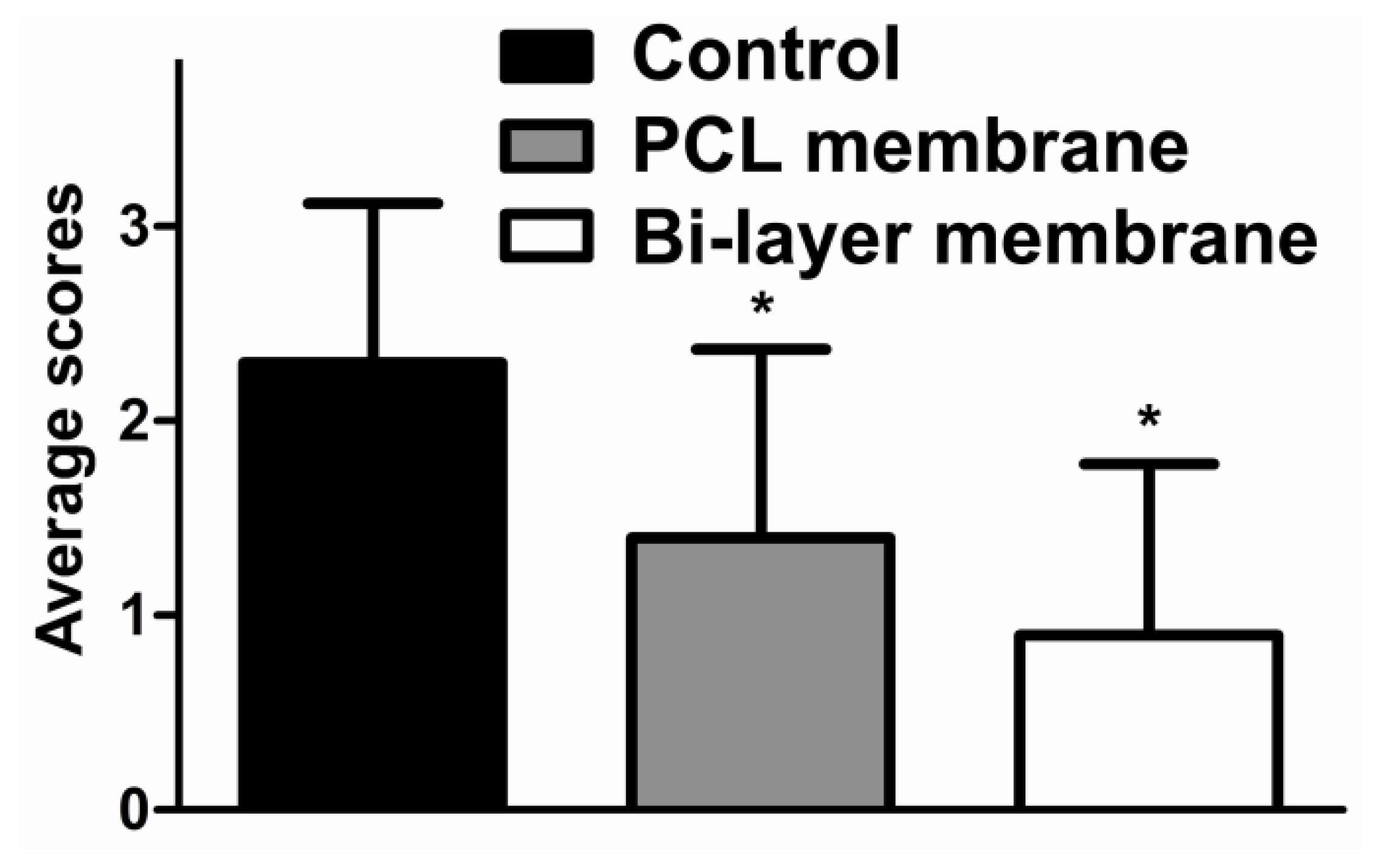
2.2. Gross Observation
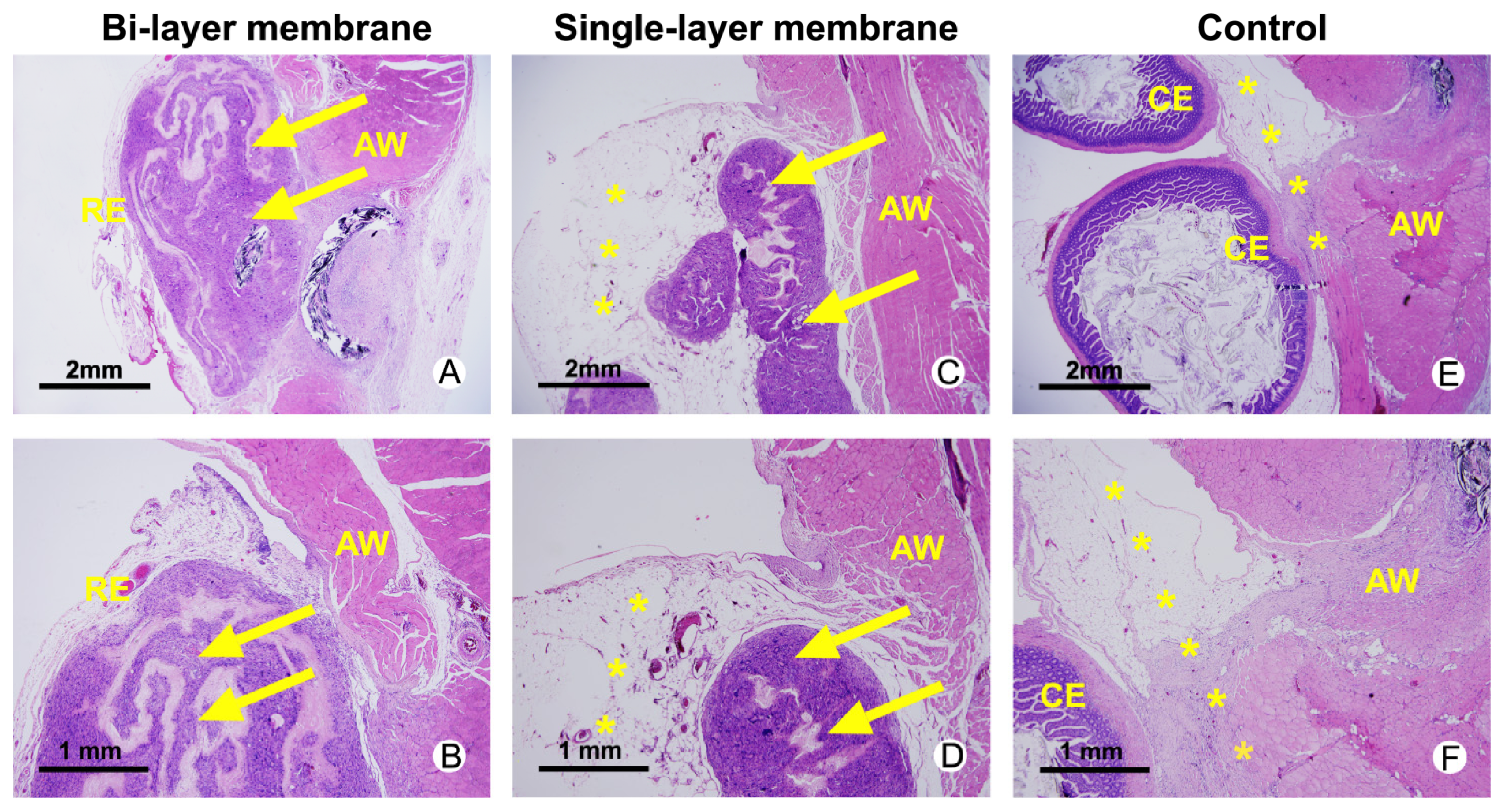
2.3. Histological Assessments
2.4. Discussion
3. Experimental Section
3.1. Preparation of Bi-Layer Electrospun Fibrous Membrane
3.2. Rat Cecum Abrasion Surgical Model
3.3. Macroscopic Evaluation
3.4. Histological Evaluation
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Sahin, M.; Cakir, M.; Avsar, F.M.; Tekin, A.; Kucukkartallar, T.; Akoz, M. The effects of anti-adhesion materials in preventing postoperative adhesion in abdominal cavity (anti-adhesion materials for postoperative adhesions). Inflammation 2007, 30, 244–249. [Google Scholar]
- Markogiannakis, H.; Messaris, E.; Dardamanis, D.; Pararas, N.; Tzertzemelis, D.; Giannopoulos, P.; Larentzakis, A.; Lagoudianakis, E.; Manouras, A.; Bramis, I. Acute mechanical bowel obstruction: Clinical presentation, etiology, management and outcome. World J. Gastroenterol 2007, 13, 432–437. [Google Scholar]
- Lee, J.H.; Go, A.K.; Oh, S.H.; Lee, K.E.; Yuk, S.H. Tissue anti-adhesion potential of ibuprofen-loaded plla–peg diblock copolymer films. Biomaterials 2005, 26, 671–678. [Google Scholar]
- Lee, M.W.; Hung, C.L.; Cheng, J.C.; Wang, Y.J. A new anti-adhesion film synthesized from polygalacturonic acid with 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide crosslinker. Biomaterials 2005, 26, 3793–3799. [Google Scholar]
- Yeo, Y.; Bellas, E.; Highley, C.B.; Langer, R.; Kohane, D.S. Peritoneal adhesion prevention with an in situ cross-linkable hyaluronan gel containing tissue-type plasminogen activator in a rabbit repeated-injury model. Biomaterials 2007, 28, 3704–3713. [Google Scholar]
- Binda, M.; Koninckx, P. Prevention of adhesion formation in a laparoscopic mouse model should combine local treatment with peritoneal cavity conditioning. Hum. Reprod 2009, 24, 1473–1479. [Google Scholar]
- Diamond, M.P.; Decherney, A.H. Pathogenesis of adhesion formation/reformation: Application to reproductive pelvic surgery. Microsurgery 2005, 8, 103–107. [Google Scholar]
- Cashman, J.; Burt, H.; Springate, C.; Gleave, J.; Jackson, J. Camptothecin-loaded films for the prevention of postsurgical adhesions. Inflamm. Res 2004, 53, 355–362. [Google Scholar]
- Hopkins, M.P.; von Gruenigen, V.E.; Holda, S.; Weber, B. The effect of intermittent-release intraperitoneal chemotherapy on wound healing. Am. J. Obstet. Gynecol 1997, 176, 819–825. [Google Scholar]
- Yeo, Y.; Adil, M.; Bellas, E.; Astashkina, A.; Chaudhary, N.; Kohane, D.S. Prevention of peritoneal adhesions with an in situ cross-linkable hyaluronan hydrogel delivering budesonide. J. Controll. Release 2007, 120, 178–185. [Google Scholar]
- Tsai, S.; Fang, J.; Yang, C.; Chen, J.; Su, L.; Jan, S. Preparation and evaluation of a hyaluronate-collagen film for preventing post-surgical adhesion. J. Int. Med. Res 2005, 33, 68–76. [Google Scholar]
- Reijnen, M.M.; Bleichrodt, R.P.; van Goor, H. Pathophysiology of intra-abdominal adhesion and abscess formation, and the effect of hyaluronan. Bri. J. Surg 2003, 90, 533–541. [Google Scholar]
- Collins, M.N.; Birkinshaw, C. Hyaluronic acid based scaffolds for tissue engineering—A review. Carbohydr. Polym 2013, 92, 1262–1279. [Google Scholar]
- Yeo, Y.; Ito, T.; Bellas, E.; Highley, C.B.; Marini, R.; Kohane, D.S. In situ cross-linkable hyaluronan hydrogels containing polymeric nanoparticles for preventing postsurgical adhesions. Ann. Surg 2007, 245, 819–824. [Google Scholar]
- Liu, Y.; Shu, X.Z.; Prestwich, G.D. Reduced postoperative intra-abdominal adhesions using carbylan-sx, a semisynthetic glycosaminoglycan hydrogel. Fertil. Steril 2007, 87, 940–948. [Google Scholar]
- Collins, M.N.; Birkinshaw, C. Comparison of the effectiveness of four different crosslinking agents with hyaluronic acid hydrogel films for tissue-culture applications. J. Appl. Polym. Sci 2007, 104, 3183–3191. [Google Scholar]
- Liu, S.; Zhao, J.; Ruan, H.; Tang, T.; Liu, G.; Yu, D.; Cui, W.; Fan, C. Biomimetic sheath membrane via electrospinning for anti-adhesion of repaired tendon. Biomacromolecules 2012, 13, 3611–3619. [Google Scholar]
- Holmdahl, L. The role of fibrinolysis in adhesion formation. Eur. J. Surg. Suppl. Acta Chir. Suppl 1997, 577, 24–31. [Google Scholar]
- Yeo, Y.; Highley, C.B.; Bellas, E.; Ito, T.; Marini, R.; Langer, R.; Kohane, D.S. In situ cross-linkable hyaluronic acid hydrogels prevent post-operative abdominal adhesions in a rabbit model. Biomaterials 2006, 27, 4698–4705. [Google Scholar]
- Tan, H.; Li, H.; Rubin, J.P.; Marra, K.G. Controlled gelation and degradation rates of injectable hyaluronic acid-based hydrogels through a double crosslinking strategy. J. Tissue Eng. Regen. Med 2011, 5, 790–797. [Google Scholar]
- Patterson, J.; Siew, R.; Herring, S.W.; Lin, A.S.; Guldberg, R.; Stayton, P.S. Hyaluronic acid hydrogels with controlled degradation properties for oriented bone regeneration. Biomaterials 2010, 31, 6772–6781. [Google Scholar]
- Liakakos, T.; Thomakos, N.; Fine, P.M.; Dervenis, C.; Young, R.L. Peritoneal adhesions: Etiology, pathophysiology, and clinical significance. Dig. Surg 2001, 18, 260–273. [Google Scholar]
- Gago, L.; Saed, G.M.; Chauhan, S.; Elhammady, E.F.; Diamond, M.P. Seprafilm (modified hyaluronic acid and carboxymethylcellulose) acts as a physical barrier. Fertil. Steril 2003, 80, 612–616. [Google Scholar]
- Falabella, C.A.; Melendez, M.M.; Weng, L.; Chen, W. Novel macromolecular crosslinking hydrogel to reduce intra-abdominal adhesions. J. Surg. Res 2010, 159, 772–778. [Google Scholar]
- Lo, H.Y.; Kuo, H.T.; Huang, Y.Y. Application of polycaprolactone as an anti-adhesion biomaterial film. Artif. Organs 2010, 34, 648–653. [Google Scholar]
- Buckenmaier, C., 3rd; Pusateri, A.E.; Harris, R.A.; Hetz, S.P. Comparison of antiadhesive treatments using an objective rat model. Am. Surg 1999, 65, 274–282. [Google Scholar]
- Yaacobi, Y.; Israel, A.A.; Goldberg, E.P. Prevention of postoperative abdominal adhesions by tissue precoating with polymer solutions. J. Surg. Res 1993, 55, 422–426. [Google Scholar]
- Burns, J.W.; Skinner, K.; Colt, J.; Sheidlin, A. Prevention of tissue injury and postsurgical adhesions by precoating tissues with yaluronic acid solutions. J. Surg. Res 1995, 59, 644–652. [Google Scholar]
- Zong, X.; Li, S.; Chen, E.; Garlick, B.; Kim, K.; Fang, D.; Chiu, J.; Zimmerman, T.; Brathwaite, C.; Hsiao, B.S. Prevention of postsurgery-induced abdominal adhesions by electrospun bioabsorbable nanofibrous poly (lactide-co-glycolide)-based membranes. Ann. Surg 2004, 240, 910–915. [Google Scholar]

© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jiang, S.; Wang, W.; Yan, H.; Fan, C. Prevention of Intra-Abdominal Adhesion by Bi-Layer Electrospun Membrane. Int. J. Mol. Sci. 2013, 14, 11861-11870. https://doi.org/10.3390/ijms140611861
Jiang S, Wang W, Yan H, Fan C. Prevention of Intra-Abdominal Adhesion by Bi-Layer Electrospun Membrane. International Journal of Molecular Sciences. 2013; 14(6):11861-11870. https://doi.org/10.3390/ijms140611861
Chicago/Turabian StyleJiang, Shichao, Wei Wang, Hede Yan, and Cunyi Fan. 2013. "Prevention of Intra-Abdominal Adhesion by Bi-Layer Electrospun Membrane" International Journal of Molecular Sciences 14, no. 6: 11861-11870. https://doi.org/10.3390/ijms140611861



