Apparent Polyploidization after Gamma Irradiation: Pitfalls in the Use of Quantitative Polymerase Chain Reaction (qPCR) for the Estimation of Mitochondrial and Nuclear DNA Gene Copy Numbers
Abstract
:1. Introduction
2. Results and Discussion
2.1. qPCR Yield from Cells after Radiation Exposure
2.2. qPCR Yield from Isolated DNA after Radiation Exposure
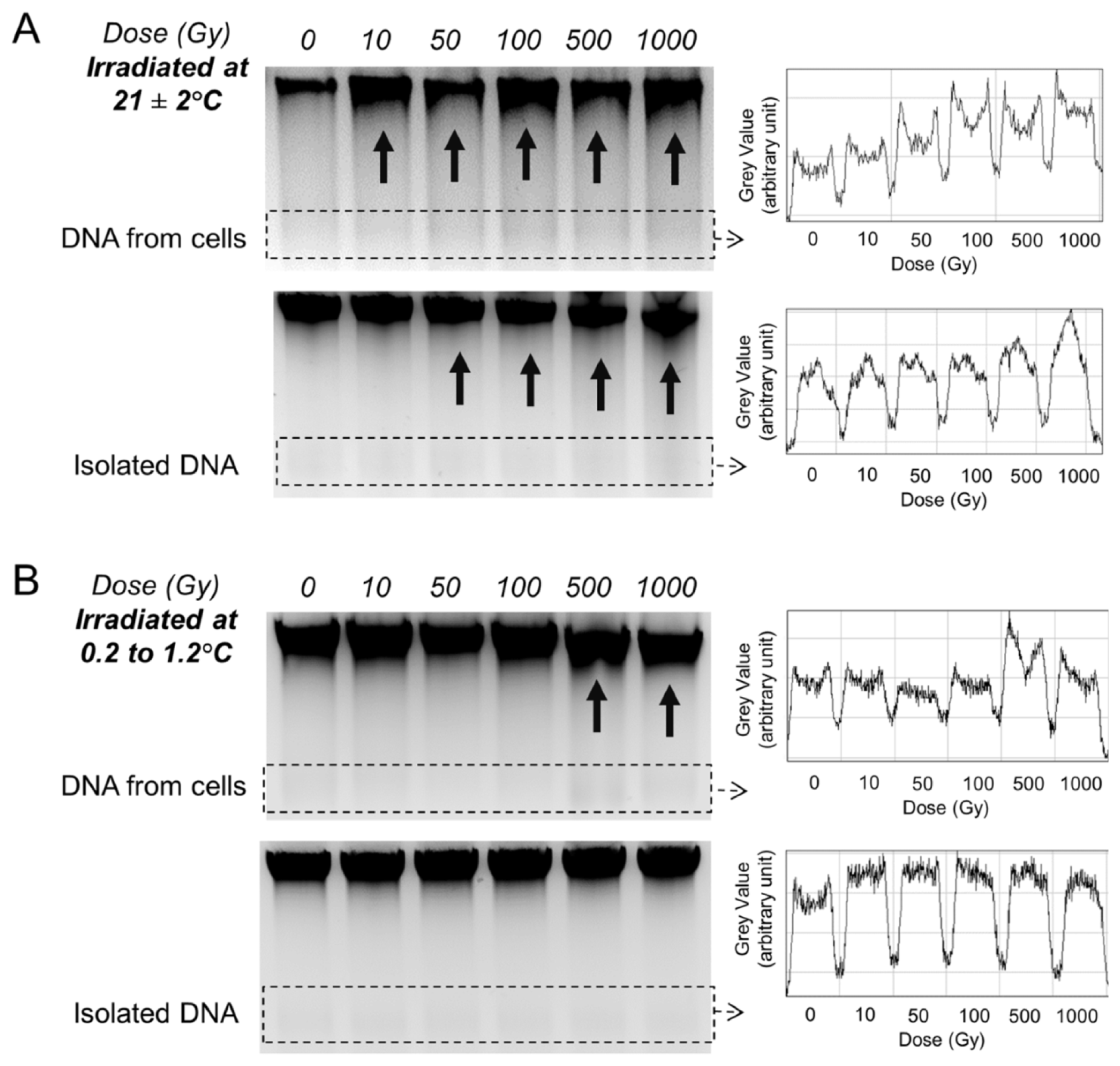
2.3. Gel Analysis of DNA Template Fragmentation after Radiation Exposure
2.4. qPCR Yield from Cells after Radiation Exposure at Low Temperature
2.5. Gel Analysis of DNA Template Fragmentation after Radiation Exposure at Low Temperature
3. Experimental Section
3.1. Sample Preparation
3.2. Gamma Irradiation
3.3. DNA Isolation and Gel Electrophoresis
3.4. qPCR
4. Conclusions
Supplementary Information
ijms-14-11544-s001.pdfAcknowledgments
Conflict of Interest
References
- Iborra, F.; Kimura, H.; Cook, P. The functional organization of mitochondrial genomes in human cells. BMC Biol 2004, 2, 9. [Google Scholar]
- Evdokimovskiĭ, E.V.; Patrushev, M.V.; Ushakova, T.E.; Gaziev, A.I. Sharp changes in the copy number of mtDNA and its transcription in the blood cells of X-ray irradiated mice are observed, and mtDNA fragments appear in the blood serum. Radiats Biol. Radioecol 2007, 47, 402–407. [Google Scholar]
- Gubina, N.; Merekina, O.; Ushakova, T. Mitochondrial DNA transcription in mouse liver, skeletal muscle, and brain following lethal X-ray irradiation. Biochem. Moscow 2010, 75, 777–783. [Google Scholar]
- Malakhova, L.; Bezlepkin, V.G.; Antipova, V.; Ushakova, T.; Fomenko, L.; Sirota, N.; Gaziev, A.I. The increase in mitochondrial DNA copy number in the tissues of gamma-irradiated mice. Cell Mol. Biol. Lett 2005, 10, 721–732. [Google Scholar]
- Wen, Q.; Hu, Y.; Ji, F.; Qian, G. Mitochondrial DNA alterations of peripheral lymphocytes in acute lymphoblastic leukemia patients undergoing total body irradiation therapy. Radiat. Oncol 2011, 6, 133. [Google Scholar]
- Zhang, H.; Maguire, D.; Swarts, S.; Sun, W.; Yang, S.; Wang, W.; Liu, C.; Zhang, M.; Zhang, D.; Zhang, L.; et al. Replication of murine mitochondrial DNA following irradiation. Adv. Exp. Med. Biol 2009, 645, 43–48. [Google Scholar]
- Zhou, X.; Li, N.; Wang, Y.; Wang, Y.; Zhang, X.; Zhang, H. Effects of X-irradiation on mitochondrial DNA damage and its supercoiling formation change. Mitochondrion 2011, 11, 886–892. [Google Scholar]
- Kulkarni, R.; Marples, B.; Balasubramaniam, M.; Thomas, R.A.; Tucker, J.D. Mitochondrial gene expression changes in normal and mitochondrial mutant cells after exposure to ionizing radiation. Radiat. Res 2010, 173, 635–644. [Google Scholar]
- Wang, L.; Kuwahara, Y.; Li, L.; Baba, T.; Shin, R.W.; Ohkubo, Y.; Ono, K.; Fukumoto, M. Analysis of Common Deletion (CD) and a novel deletion of mitochondrial DNA induced by ionizing radiation. Int. J. Radiat. Biol 2007, 83, 433–442. [Google Scholar]
- Bai, R.-K.; Wong, L.-J.C. Simultaneous detection and quantification of mitochondrial DNA deletion(s), depletion, and over-replication in patients with mitochondrial disease. J. Mol. Diagn 2005, 7, 613–622. [Google Scholar]
- Schon, E.A. Complements of the house. J. Clin. Invest 2004, 114, 760–762. [Google Scholar]
- Malik, A.N.; Shahni, R.; Rodriguez-de-Ledesma, A.; Laftah, A.; Cunningham, P. Mitochondrial DNA as a non-invasive biomarker: Accurate quantification using real time quantitative PCR without co-amplification of pseudogenes and dilution bias. Biochem. Biophys. Res. Commun 2011, 412, 1–7. [Google Scholar]
- Malik, A.N.; Czajka, A. Is mitochondrial DNA content a potential biomarker of mitochondrial dysfunction? Mitochondrion 2013, in press. [Google Scholar]
- Kam, W.W.Y.; McNamara, A.L.; Lake, V.; Banos, C.; Davies, J.B.; Kuncic, Z.; Banati, R.B. Predicted ionization in mitochondria and observed acute changes in the mitochondrial transcriptome after gamma irradiation: A Monte Carlo simulation and quantitative PCR study. Mitochondrion 2013, in press. [Google Scholar]
- Kam, W.W.Y.; Middleton, R.; Lake, V.; Banati, R.B. Green fluorescent protein alters the transcriptional regulation of human mitochondrial genes after gamma irradiation. J. Fluoresc. 2013, in press. [Google Scholar]
- Sikorsky, J.A.; Primerano, D.A.; Fenger, T.W.; Denvir, J. Effect of DNA damage on PCR amplification efficiency with the relative threshold cycle method. Biochem. Biophys. Res. Commun 2004, 323, 823–830. [Google Scholar]
- Brisco, M.J.; Latham, S.; Bartley, P.A.; Morley, A.A. Incorporation of measurement of DNA integrity into qPCR assays. BioTechniques 2010, 49, 893–897. [Google Scholar]
- Meyer, J. QPCR: A tool for analysis of mitochondrial and nuclear DNA damage in ecotoxicology. Ecotoxicology 2010, 19, 804–811. [Google Scholar]
- Morales, A.; Miranda, M.; Sanchez-Reyes, A.; Biete, A.; Fernandez-Checa, J.C. Oxidative damage of mitochondrial and nuclear DNA induced by ionizing radiation in human hepatoblastoma cells. Int. J. Radiat. Oncol. Biol. Phys 1998, 42, 191–203. [Google Scholar]
- Elmroth, K.; Erkell, L.J.; Hultborn, R. Influence of temperature on radiation-induced inhibition of DNA supercoiling. Radiat. Res 1999, 152, 137–143. [Google Scholar]
- Dang, L.; Lisowska, H.; Manesh, S.S.; Sollazzo, A.; Deperas-Kaminska, M.; Staaf, E.; Haghdoost, S.; Brehwens, K.; Wojcik, A. Radioprotective effect of hypothermia on cells—A multiparametric approach to delineate the mechanisms. Int. J. Radiat. Biol 2012, 88, 507–514. [Google Scholar]
- Brzozowska, K.; Johannes, C.; Obe, G.; Hentschel, R.; Morand, J.; Moss, R.; Wittig, A.; Sauerwein, W.; Liniecki, J.; Szumiel, I.; et al. Effect of temperature during irradiation on the level of micronuclei in human peripheral blood lymphocytes exposed to X-rays and neutrons. Int. J. Radiat. Biol 2009, 85, 891–899. [Google Scholar]
- Lundgren-Eriksson, L.; Hultborn, R.; Henriksson, R. Hypothermic modulation of doxorubicin, cisplatin and radiation cytotoxicity in vitro. Anticancer Res 2001, 21, 3275–3280. [Google Scholar]
- Elmroth, K.; Nygren, J.; Erkell, L.J.; Hultborn, R. Effect of hypothermic irradiation of the growth characteristics of two human cell lines. Anticancer Res 2000, 20, 3429–3433. [Google Scholar]
- Baird, B.J.; Dickey, J.S.; Nakamura, A.J.; Redon, C.E.; Parekh, P.; Griko, Y.V.; Aziz, K.; Georgakilas, A.G.; Bonner, W.M.; Martin, O.A. Hypothermia postpones DNA damage repair in irradiated cells and protects against cell killing. Mutat Res 2011, 711, 142–149. [Google Scholar]
- Lundgren-Eriksson, L.; Palm, A.; Ryd, W.; Warnhammar, E.; Hultborn, R. Radio-and chemotoxicity in mice during hypothermia. Anticancer Res 2001, 21, 3269–3274. [Google Scholar]
- Henner, W.D.; Grunberg, S.M.; Haseltine, W.A. Sites and structure of gamma radiation-induced DNA strand breaks. J. Biol. Chem 1982, 257, 11750–11754. [Google Scholar]
- Clayton, D.A. Transcription and replication of mitochondrial DNA. Hum. Reprod 2000, 15, 11–17. [Google Scholar]
- Elmroth, K.; Nygren, J.; Erkell, L.J.; Hultborn, R. Radiation-induced double-strand breaks in mammalian DNA: Influence of temperature and DMSO. Int. J. Radiat. Biol 2000, 76, 1501–1508. [Google Scholar]
- Elmroth, K.; Nygren, J.; Stenerlow, B.; Hultborn, R. Chromatin- and temperature-dependent modulation of radiation-induced double-strand breaks. Int. J. Radiat. Biol 2003, 79, 809–816. [Google Scholar]
- Liu, X.; Zou, H.; Slaughter, C.; Wang, X. DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 1997, 89, 175–184. [Google Scholar]
- Larsen, B.D.; Rampalli, S.; Burns, L.E.; Brunette, S.; Dilworth, F.J.; Megeney, L.A. Caspase 3/caspase-activated DNase promote cell differentiation by inducing DNA strand breaks. Proc. Natl. Acad. Sci. USA 2010, 107, 4230–4235. [Google Scholar]
- Hanawalt, P.C.; Spivak, G. Transcription-coupled DNA repair: Two decades of progress and surprises. Nat. Rev. Mol. Cell Biol 2008, 9, 958–970. [Google Scholar]
- Cha, R.S.; Thilly, W.G. Specificity, efficiency, and fidelity of PCR. Genome Res 1993, 3, S18–S29. [Google Scholar]
- Pinto, M.; Prise, K.M.; Michael, B.D. Quantification of radiation induced DNA double-strand breaks in human fibroblasts by PFGE: Testing the applicability of random breakage models. Int. J. Radiat. Biol 2002, 78, 375–388. [Google Scholar]
- Chan, Y.L.; Roebuck, D.J.; Yuen, M.P.; Yeung, K.W.; Lau, K.Y.; Li, C.K.; Chik, K.W. Long-term cerebral metabolite changes on proton magnetic resonance spectroscopy in patients cured of acute lymphoblastic leukemia with previous intrathecal methotrexate and cranial irradiation prophylaxis. Int. J. Radiat. Oncol. Biol. Phys 2001, 50, 759–763. [Google Scholar]
- Steen, R.G.; Spence, D.; Wu, S.J.; Xiong, X.P.; Kun, L.E.; Merchant, T.E. Effect of therapeutic ionizing radiation on the human brain. Ann. Neurol 2001, 50, 787–795. [Google Scholar]
- Tanriover, N.; Ulu, M.O.; Sar, M.; Uzan, M. Anaplastic oligoastrocytoma: Previous treatment as a possible cause in a child with acute lymphoblastic leukemia. Childs Nervous Syst 2007, 23, 469–473. [Google Scholar]
- Indo, H.P.; Inanami, O.; Koumura, T.; Suenaga, S.; Yen, H.C.; Kakinuma, S.; Matsumoto, K.; Nakanishi, I.; St Clair, W.; St Clair, D.K.; et al. Roles of mitochondria-generated reactive oxygen species on X-ray-induced apoptosis in a human hepatocellular carcinoma cell line, HLE. Free Radic. Res 2012, 46, 1029–1043. [Google Scholar]
- Epperly, M.W.; Melendez, J.A.; Zhang, X.; Nie, S.; Pearce, L.; Peterson, J.; Franicola, D.; Dixon, T.; Greenberger, B.A.; Komanduri, P.; et al. Mitochondrial targeting of a catalase transgene product by plasmid liposomes increases radioresistance in vitro and in vivo. Radiat. Res 2009, 171, 588–595. [Google Scholar]
- Epperly, M.W.; Defilippi, S.; Sikora, C.; Gretton, J.; Kalend, A.; Greenberger, J.S. Intratracheal injection of manganese superoxide dismutase (MnSOD) plasmid/liposomes protects normal lung but not orthotopic tumors from irradiation. Gene Ther 2000, 7, 1011–1018. [Google Scholar]
- Yamamori, T.; Yasui, H.; Yamazumi, M.; Wada, Y.; Nakamura, Y.; Nakamura, H.; Inanami, O. Ionizing radiation induces mitochondrial reactive oxygen species production accompanied by upregulation of mitochondrial electron transport chain function and mitochondrial content under control of the cell cycle checkpoint. Free Radic. Biol. Med 2012, 53, 260–270. [Google Scholar]
- Epperly, M.W.; Gretton, J.E.; Sikora, C.A.; Jefferson, M.; Bernarding, M.; Nie, S.; Greenberger, J.S. Mitochondrial localization of superoxide dismutase is required for decreasing radiation-induced cellular damage. Radiat. Res 2003, 160, 568–578. [Google Scholar]
- Pearce, L.L.; Epperly, M.W.; Greenberger, J.S.; Pitt, B.R.; Peterson, J. Identification of respiratory complexes I and III as mitochondrial sites of damage following exposure to ionizing radiation and nitric oxide. Nitric Oxide 2001, 5, 128–136. [Google Scholar]
- Prithivirajsingh, S.; Story, M.D.; Bergh, S.A.; Geara, F.B.; Kian Ang, K.; Ismail, S.M.; Stevens, C.W.; Buchholz, T.A.; Brock, W.A. Accumulation of the common mitochondrial DNA deletion induced by ionizing radiation. FEBS Lett 2004, 571, 227–232. [Google Scholar]
- Richter, C.; Park, J.W.; Ames, B.N. Normal oxidative damage to mitochondrial and nuclear DNA is extensive. Proc. Natl. Acad. Sci. USA 1988, 85, 6465–6467. [Google Scholar]
- May, A.; Bohr, V.A. Gene-specific repair of γ-ray-induced DNA strand breaks in colon cancer cells: No coupling to transcription and no removal from the mitochondrial genome. Biochem. Biophys. Res. Commun 2000, 269, 433–437. [Google Scholar]
- Cheng, K.T.; Hou, W.C.; Huang, Y.C.; Wang, L.F. Baicalin induces differential expression of cytochrome C oxidase in human lung H441 cell. J. Agric. Food Chem 2003, 51, 7276–7279. [Google Scholar]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem 2009, 55, 611–622. [Google Scholar]
- Kitano, T.; Umetsu, K.; Tian, W.; Osawa, M. Two universal primer sets for species identification among vertebrates. Int. J. Legal Med 2007, 121, 423–427. [Google Scholar]
- Rorbach, J.; Yusoff, A.A.; Tuppen, H.; Abg-Kamaludin, D.P.; Chrzanowska-Lightowlers, Z.M.A.; Taylor, R.W.; Turnbull, D.M.; McFarland, R.; Lightowlers, R.N. Overexpression of human mitochondrial valyl tRNA synthetase can partially restore levels of cognate mt-tRNAVal carrying the pathogenic C25U mutation. Nucleic Acids Res 2008, 36, 3065–3074. [Google Scholar]
- Garbian, Y.; Ovadia, O.; Dadon, S.; Mishmar, D. Gene expression patterns of oxidative phosphorylation complex I subunits are organized in clusters. PLoS One 2010, 5, e9985. [Google Scholar]
- Owens, K.M.; Kulawiec, M.; Desouki, M.M.; Vanniarajan, A.; Singh, K.K. Impaired OXPHOS complex III in breast cancer. PLoS One 2011, 6, e23846. [Google Scholar]
- Uchiumi, T.; Ohgaki, K.; Yagi, M.; Aoki, Y.; Sakai, A.; Matsumoto, S.; Kang, D. ERAL1 is associated with mitochondrial ribosome and elimination of ERAL1 leads to mitochondrial dysfunction and growth retardation. Nucleic Acids Res 2010, 38, 5554–5568. [Google Scholar]
- Wilkening, S.; Stahl, F.; Bader, A. Comparison of primary human hepatocytes and hepatoma cell line HEPG2 with regard to their biotransformation properties. Drug Metab. Dispos 2003, 31, 1035–1042. [Google Scholar]
- Ma, X.; Gao, F.; Rusie, A.; Hemingway, J.; Ostmann, A.B.; Sroga, J.M.; Jegga, A.G.; Das, S.K. Decidual cell polyploidization necessitates mitochondrial activity. PLoS One 2011, 6, e26774. [Google Scholar]



| Coding origin | Gene name | Forward primer | Reverse primer | Reference |
|---|---|---|---|---|
| Mitochondria | 12S | CCCAAACTGGGATTAGATACCC | GTTTGCTGAAGATGGCGGTA | [50] |
| 16S | GCCTGTTTACCAAAAACATCAC | CTCCATAGGGTCTTCTCGTCTT | [50] | |
| MTTV | CTGGAAAGTGCACTTGGACG | GGGTAAATGGTTTGGCTAAGG | [51] | |
| MTTL1 | TATACCCACACCCACCCAAG | GCGATTAGAATGGGTACAAT | [51] | |
| Complex I subunit ND6 | GGATCCTCCCGAATCAAC | GTAGGATTGGTGCTGTGG | [52] | |
| Complex III subunit CYB | TGAAACTTCGGCTCACTCCT | AATGTATGGGATGGCGGATA | [53] | |
| Complex IV subunit 2 | CAGGAAATAGAAACCGTCTGAACTATCCTG | CTGTGGTTTGCTCCACAGATTTCAGTGCAT | [48] | |
| Complex V subunit 8 | ATGGCCCACCATAATTACCC | GCAATGAATGAAGCGAACAG | [54] | |
| Nucleus | Actin | GTGGGGCGCCCCAGGCACCA | CTCCTTAATGTCACGCACGATTTC | [48] |
| Complex I subunit 5 | GAGAAGCTGGCTATGGTTAAAGCG | CCACTAATGGCTCCCATAGTTTCC | [52] | |
| Complex III subunit CYC1 | CCAAAACCATACCCCAACAG | TATGCCAGCTTCCGACTCTT | [53] | |
| Complex V subunit O | ACCCAAGGAGTCGTTTCTGC | TTAGACAATCTCCCGCATAGC | [55] | |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kam, W.W.Y.; Lake, V.; Banos, C.; Davies, J.; Banati, R. Apparent Polyploidization after Gamma Irradiation: Pitfalls in the Use of Quantitative Polymerase Chain Reaction (qPCR) for the Estimation of Mitochondrial and Nuclear DNA Gene Copy Numbers. Int. J. Mol. Sci. 2013, 14, 11544-11559. https://doi.org/10.3390/ijms140611544
Kam WWY, Lake V, Banos C, Davies J, Banati R. Apparent Polyploidization after Gamma Irradiation: Pitfalls in the Use of Quantitative Polymerase Chain Reaction (qPCR) for the Estimation of Mitochondrial and Nuclear DNA Gene Copy Numbers. International Journal of Molecular Sciences. 2013; 14(6):11544-11559. https://doi.org/10.3390/ijms140611544
Chicago/Turabian StyleKam, Winnie W. Y., Vanessa Lake, Connie Banos, Justin Davies, and Richard Banati. 2013. "Apparent Polyploidization after Gamma Irradiation: Pitfalls in the Use of Quantitative Polymerase Chain Reaction (qPCR) for the Estimation of Mitochondrial and Nuclear DNA Gene Copy Numbers" International Journal of Molecular Sciences 14, no. 6: 11544-11559. https://doi.org/10.3390/ijms140611544




