Astrocytic Vesicle Mobility in Health and Disease
Abstract
:1. Introduction
2. Gliotransmitter-Loaded Secretory Vesicles
2.1. Amino Acid-Loaded Vesicles
2.2. ATP-Loaded Vesicles
2.3. Peptide-Loaded Vesicles
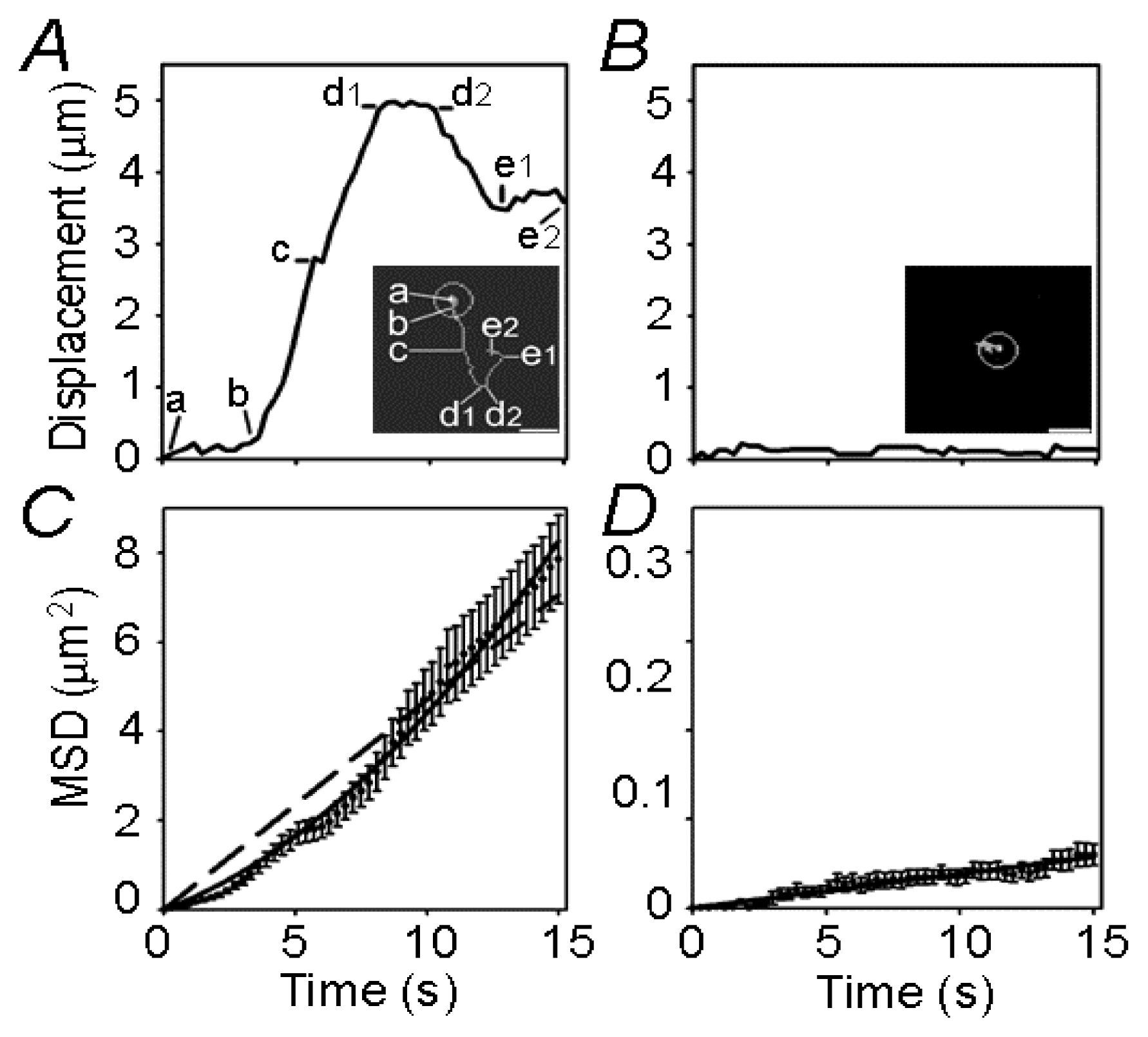
3. Endocytotic Vesicles
4. Vesicles Transporting Aquaporins
5. Vesicles Delivering Plasma Membrane Transporters and Receptors
6. Conclusions
Acknowledgements
Conflict of Interest
References
- Nedergaard, M.; Ransom, B.; Goldman, S. New roles for astrocytes: Redefining the functional architecture of the brain. Trends Neurosci 2003, 26, 523–530. [Google Scholar]
- Stevens, B. Neuron-astrocyte signaling in the development and plasticity of neural circuits. Neurosignals 2008, 16, 278–288. [Google Scholar]
- Haydon, P. GLIA: Listening and talking to the synapse. Nat. Rev. Neurosci 2001, 2, 185–193. [Google Scholar]
- Anderson, C.; Nedergaard, M. Astrocyte-mediated control of cerebral microcirculation. Trends Neurosci 2003, 26, 340–344, ; author reply 344–345.. [Google Scholar]
- Zonta, M.; Angulo, M.; Gobbo, S.; Rosengarten, B.; Hossmann, K.; Pozzan, T.; Carmignoto, G. Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nat. Neurosci 2003, 6, 43–50. [Google Scholar]
- Gordon, G.; Mulligan, S.; MacVicar, B. Astrocyte control of the cerebrovasculature. Glia 2007, 55, 1214–1221. [Google Scholar]
- Dong, Y.; Benveniste, E. Immune function of astrocytes. Glia 2001, 36, 180–190. [Google Scholar]
- De Keyser, J.; Zeinstra, E.; Frohman, E. Are astrocytes central players in the pathophysiology of multiple sclerosis? Arch. Neurol 2003, 60, 132–136. [Google Scholar]
- Abbott, N.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci 2006, 7, 41–53. [Google Scholar]
- Ke, C.; Poon, W.S.; Ng, H.K.; Pang, J.C.; Chan, Y. Heterogeneous responses of aquaporin-4 in oedema formation in a replicated severe traumatic brain injury model in rats. Neurosci. Lett 2001, 301, 21–24. [Google Scholar]
- Nase, G.; Helm, P.J.; Enger, R.; Ottersen, O.P. Water entry into astrocytes during brain edema formation. Glia 2008, 56, 895–902. [Google Scholar]
- Risher, W.C.; Andrew, R.D.; Kirov, S.A. Real-time passive volume responses of astrocytes to acute osmotic and ischemic stress in cortical slices and in vivo revealed by two-photon microscopy. Glia 2009, 57, 207–221. [Google Scholar]
- Parpura, V.; Zorec, R. Gliotransmission: Exocytotic release from astrocytes. Brain Res. Rev 2010, 63, 83–92. [Google Scholar]
- Osborne, K.D.; Lee, W.; Malarkey, E.B.; Irving, A.J.; Parpura, V. Dynamic imaging of cannabinoid receptor 1 vesicular trafficking in cultured astrocytes. ASN Neuro 2009, 1, e00022. [Google Scholar]
- Kreft, M.; Stenovec, M.; Rupnik, M.; Grilc, S.; Krzan, M.; Potokar, M.; Pangrsic, T.; Haydon, P.; Zorec, R. Properties of Ca(2+)-dependent exocytosis in cultured astrocytes. Glia 2004, 46, 437–445. [Google Scholar]
- Guček, A.; Vardjan, N.; Zorec, R. Exocytosis in astrocytes: Transmitter release and membrane signal regulation. Neurochem. Res 2012, 37, 2351–2363. [Google Scholar]
- Zorec, R.; Araque, A.; Carmignoto, G.; Haydon, P.G.; Verkhratsky, A.; Parpura, V. Astroglial excitability and gliotransmission: An appraisal of Ca2+ as a signalling route. ASN Neuro 2012, 4, e00080. [Google Scholar]
- Parpura, V.; Heneka, M.T.; Montana, V.; Oliet, S.H.; Schousboe, A.; Haydon, P.G.; Stout, R.F.; Spray, D.C.; Reichenbach, A.; Pannicke, T.; et al. Glial cells in (patho)physiology. J. Neurochem 2012, 121, 4–27. [Google Scholar]
- Potokar, M.; Kreft, M.; Pangrsic, T.; Zorec, R. Vesicle mobility studied in cultured astrocytes. Biochem. Biophys. Res. Commun 2005, 329, 678–683. [Google Scholar]
- Potokar, M.; Kreft, M.; Li, L.; Daniel Andersson, J.; Pangrsic, T.; Chowdhury, H.; Pekny, M.; Zorec, R. Cytoskeleton and vesicle mobility in astrocytes. Traffic 2007, 8, 12–20. [Google Scholar]
- Vardjan, N.; Gabrijel, M.; Potokar, M.; Svajger, U.; Kreft, M.; Jeras, M.; de Pablo, Y.; Faiz, M.; Pekny, M.; Zorec, R. IFN-γ-induced increase in the mobility of MHC class II compartments in astrocytes depends on intermediate filaments. J. Neuroinflamm 2012, 9, e144. [Google Scholar]
- Pangrsic, T.; Potokar, M.; Stenovec, M.; Kreft, M.; Fabbretti, E.; Nistri, A.; Pryazhnikov, E.; Khiroug, L.; Giniatullin, R.; Zorec, R. Exocytotic release of ATP from cultured astrocytes. J. Biol. Chem 2007, 282, 28749–28758. [Google Scholar]
- Bezzi, P.; Gundersen, V.; Galbete, J.; Seifert, G.; Steinhäuser, C.; Pilati, E.; Volterra, A. Astrocytes contain a vesicular compartment that is competent for regulated exocytosis of glutamate. Nat. Neurosci 2004, 7, 613–620. [Google Scholar]
- Parpura, V.; Baker, B.; Jeras, M.; Zorec, R. Regulated exocytosis in astrocytic signal integration. Neurochem. Int 2010, 57, 451–459. [Google Scholar]
- Martineau, M.; Galli, T.; Baux, G.; Mothet, J. Confocal imaging and tracking of the exocytotic routes for d-serine-mediated gliotransmission. Glia 2008, 56, 1271–1284. [Google Scholar]
- Coco, S.; Calegari, F.; Pravettoni, E.; Pozzi, D.; Taverna, E.; Rosa, P.; Matteoli, M.; Verderio, C. Storage and release of ATP from astrocytes in culture. J. Biol. Chem 2003, 278, 1354–1362. [Google Scholar]
- Krzan, M.; Stenovec, M.; Kreft, M.; Pangrsic, T.; Grilc, S.; Haydon, P.; Zorec, R. Calcium-dependent exocytosis of atrial natriuretic peptide from astrocytes. J. Neurosci 2003, 23, 1580–1583. [Google Scholar]
- Jean, Y.; Lercher, L.; Dreyfus, C. Glutamate elicits release of BDNF from basal forebrain astrocytes in a process dependent on metabotropic receptors and the PLC pathway. Neuron Glia Biol 2008, 4, 35–42. [Google Scholar]
- Potokar, M.; Stenovec, M.; Jorgačevski, J.; Holen, T.; Kreft, M.; Ottersen, O.P.; Zorec, R. Regulation of AQP4 surface expression via vesicle mobility in astrocytes. Glia 2013, 61, 917–928. [Google Scholar]
- Martineau, M.; Shi, T.; Puyal, J.; Knolhoff, A.M.; Dulong, J.; Gasnier, B.; Klingauf, J.; Sweedler, J.V.; Jahn, R.; Mothet, J.P. Storage and uptake of d-serine into astrocytic synaptic-like vesicles specify gliotransmission. J. Neurosci 2013, 33, 3413–3423. [Google Scholar]
- Potokar, M.; Stenovec, M.; Kreft, M.; Kreft, M.; Zorec, R. Stimulation inhibits the mobility of recycling peptidergic vesicles in astrocytes. Glia 2008, 56, 135–144. [Google Scholar]
- Potokar, M.; Stenovec, M.; Gabrijel, M.; Li, L.; Kreft, M.; Grilc, S.; Pekny, M.; Zorec, R. Intermediate filaments attenuate stimulation-dependent mobility of endosomes/lysosomes in astrocytes. Glia 2010, 58, 1208–1219. [Google Scholar]
- Potokar, M.; Lacovich, V.; Chowdhury, H.H.; Kreft, M.; Zorec, R. Rab4 and Rab5 GTPase are required for directional mobility of endocytic vesicles in astrocytes. Glia 2012, 60, 594–604. [Google Scholar]
- Stenovec, M.; Milošević, M.; Petrušić, V.; Potokar, M.; Stević, Z.; Prebil, M.; Kreft, M.; Trkov, S.; Andjus, P.R.; Zorec, R. Amyotrophic lateral sclerosis immunoglobulins G enhance the mobility of Lysotracker-labelled vesicles in cultured rat astrocytes. Acta Physiol. (Oxf) 2011, 203, 457–471. [Google Scholar]
- Trkov, S.; Stenovec, M.; Kreft, M.; Potokar, M.; Parpura, V.; Davletov, B.; Zorec, R. Fingolimod—A sphingosine-like molecule inhibits vesicle mobility and secretion in astrocytes. Glia 2012, 60, 1406–1416. [Google Scholar]
- Stenovec, M.; Kreft, M.; Grilc, S.; Potokar, M.; Kreft, M.; Pangrsic, T.; Zorec, R. Ca2+-dependent mobility of vesicles capturing anti-VGLUT1 antibodies. Exp. Cell Res 2007, 313, 3809–3818. [Google Scholar]
- Parpura, V.; Basarsky, T.; Liu, F.; Jeftinija, K.; Jeftinija, S.; Haydon, P. Glutamate-mediated astrocyte-neuron signalling. Nature 1994, 369, 744–747. [Google Scholar]
- Montana, V.; Ni, Y.; Sunjara, V.; Hua, X.; Parpura, V. Vesicular glutamate transporter-dependent glutamate release from astrocytes. J. Neurosci 2004, 24, 2633–2642. [Google Scholar]
- Montana, V.; Malarkey, E.; Verderio, C.; Matteoli, M.; Parpura, V. Vesicular transmitter release from astrocytes. Glia 2006, 54, 700–715. [Google Scholar]
- Bergami, M.; Santi, S.; Formaggio, E.; Cagnoli, C.; Verderio, C.; Blum, R.; Berninger, B.; Matteoli, M.; Canossa, M. Uptake and recycling of pro-BDNF for transmitter-induced secretion by cortical astrocytes. J. Cell Biol 2008, 183, 213–221. [Google Scholar]
- Cassé, F.; Bardou, I.; Danglot, L.; Briens, A.; Montagne, A.; Parcq, J.; Alahari, A.; Galli, T.; Vivien, D.; Docagne, F. Glutamate controls tPA recycling by astrocytes, which in turn influences glutamatergic signals. J. Neurosci 2012, 32, 5186–5199. [Google Scholar]
- Crippa, D.; Schenk, U.; Francolini, M.; Rosa, P.; Verderio, C.; Zonta, M.; Pozzan, T.; Matteoli, M.; Carmignoto, G. Synaptobrevin2-expressing vesicles in rat astrocytes: Insights into molecular characterization, dynamics and exocytosis. J. Physiol 2006, 570, 567–582. [Google Scholar]
- Wacker, I.; Kaether, C.; Krömer, A.; Migala, A.; Almers, W.; Gerdes, H. Microtubule-dependent transport of secretory vesicles visualized in real time with a GFP-tagged secretory protein. J. Cell Sci 1997, 110, 1453–1463. [Google Scholar]
- Burke, N.; Han, W.; Li, D.; Takimoto, K.; Watkins, S.; Levitan, E. Neuronal peptide release is limited by secretory granule mobility. Neuron 1997, 19, 1095–10102. [Google Scholar]
- Tvaruskó, W.; Bentele, M.; Misteli, T.; Rudolf, R.; Kaether, C.; Spector, D.; Gerdes, H.; Eils, R. Time-resolved analysis and visualization of dynamic processes in living cells. Proc. Natl. Acad. Sci. USA 1999, 96, 7950–7955. [Google Scholar]
- Duncan, R.; Greaves, J.; Wiegand, U.; Matskevich, I.; Bodammer, G.; Apps, D.; Shipston, M.; Chow, R. Functional and spatial segregation of secretory vesicle pools according to vesicle age. Nature 2003, 422, 176–180. [Google Scholar]
- Danbolt, N.C. Glutamate uptake. Prog. Neurobiol 2001, 65, 1–105. [Google Scholar]
- Li, D.; Hérault, K.; Silm, K.; Evrard, A.; Wojcik, S.; Oheim, M.; Herzog, E.; Ropert, N. Lack of evidence for vesicular glutamate transporter expression in mouse astrocytes. J. Neurosci 2013, 33, 4434–4455. [Google Scholar]
- Zhang, Q.; Pangrsic, T.; Kreft, M.; Krzan, M.; Li, N.; Sul, J.; Halassa, M.; van Bockstaele, E.; Zorec, R.; Haydon, P. Fusion-related release of glutamate from astrocytes. J. Biol. Chem 2004, 279, 12724–12733. [Google Scholar]
- Zhang, Q.; Fukuda, M.; van Bockstaele, E.; Pascual, O.; Haydon, P. Synaptotagmin IV regulates glial glutamate release. Proc. Natl. Acad. Sci. USA 2004, 101, 9441–9446. [Google Scholar]
- Jeftinija, S.D.; Jeftinija, K.V.; Stefanovic, G.; Liu, F. Neuroligand-evoked calcium-dependent release of excitatory amino acids from cultured astrocytes. J. Neurochem 1996, 66, 676–684. [Google Scholar]
- Parpura, V.; Liu, F.; Brethorst, S.; Jeftinija, K.; Jeftinija, S.; Haydon, P.G. Alpha-latrotoxin stimulates glutamate release from cortical astrocytes in cell culture. FEBS Lett 1995, 360, 266–270. [Google Scholar]
- Almqvist, J.; Huang, Y.; Laaksonen, A.; Wang, D.; Hovmöller, S. Docking and homology modeling explain inhibition of the human vesicular glutamate transporters. Protein Sci 2007, 16, 1819–1829. [Google Scholar]
- Aravanis, A.M.; Pyle, J.L.; Tsien, R.W. Single synaptic vesicles fusing transiently and successively without loss of identity. Nature 2003, 423, 643–647. [Google Scholar]
- Gasparini, S.; Saviane, C.; Voronin, L.L.; Cherubini, E. Silent synapses in the developing hippocampus: Lack of functional AMPA receptors or low probability of glutamate release? Proc. Natl. Acad. Sci. USA 2000, 97, 9741–9746. [Google Scholar]
- Volterra, A.; Meldolesi, J. Astrocytes, from brain glue to communication elements: The revolution continues. Nat. Rev. Neurosci 2005, 6, 626–640. [Google Scholar]
- Pelkmans, L.; Zerial, M. Kinase-regulated quantal assemblies and kiss-and-run recycling of caveolae. Nature 2005, 436, 128–133. [Google Scholar]
- Zimmermann, H. Signalling via ATP in the nervous system. Trends Neurosci 1994, 17, 420–426. [Google Scholar]
- Guthrie, P.B.; Knappenberger, J.; Segal, M.; Bennett, M.V.; Charles, A.C.; Kater, S.B. ATP released from astrocytes mediates glial calcium waves. J. Neurosci 1999, 19, 520–528. [Google Scholar]
- Wang, Z.; Haydon, P.G.; Yeung, E.S. Direct observation of calcium-independent intercellular ATP signaling in astrocytes. Anal. Chem 2000, 72, 2001–2007. [Google Scholar]
- Stout, C.E.; Costantin, J.L.; Naus, C.C.; Charles, A.C. Intercellular calcium signaling in astrocytes via ATP release through connexin hemichannels. J. Biol. Chem 2002, 277, 10482–10488. [Google Scholar]
- Stehberg, J.; Moraga-Amaro, R.; Salazar, C.; Becerra, A.; Echeverría, C.; Orellana, J.A.; Bultynck, G.; Ponsaerts, R.; Leybaert, L.; Simon, F.; et al. Release of gliotransmitters through astroglial connexin 43 hemichannels is necessary for fear memory consolidation in the basolateral amygdala. FASEB J 2012, 26, 3649–3657. [Google Scholar]
- Blum, A.E.; Walsh, B.C.; Dubyak, G.R. Extracellular osmolarity modulates G protein-coupled receptor-dependent ATP release from 1321N1 astrocytoma cells. Am. J. Physiol. Cell Physiol 2010, 298, C386–C396. [Google Scholar]
- Calegari, F.; Coco, S.; Taverna, E.; Bassetti, M.; Verderio, C.; Corradi, N.; Matteoli, M.; Rosa, P. A regulated secretory pathway in cultured hippocampal astrocytes. J. Biol. Chem 1999, 274, 22539–22547. [Google Scholar]
- Zhang, Z.; Chen, G.; Zhou, W.; Song, A.; Xu, T.; Luo, Q.; Wang, W.; Gu, X.S.; Duan, S. Regulated ATP release from astrocytes through lysosome exocytosis. Nat. Cell. Biol 2007, 9, 945–953. [Google Scholar]
- Jaiswal, J.K.; Fix, M.; Takano, T.; Nedergaard, M.; Simon, S.M. Resolving vesicle fusion from lysis to monitor calcium-triggered lysosomal exocytosis in astrocytes. Proc. Natl. Acad. Sci. USA 2007, 104, 14151–14156. [Google Scholar]
- Li, D.; Ropert, N.; Koulakoff, A.; Giaume, C.; Oheim, M. Lysosomes are the major vesicular compartment undergoing Ca2+-regulated exocytosis from cortical astrocytes. J. Neurosci 2008, 28, 7648–7658. [Google Scholar]
- Verderio, C.; Cagnoli, C.; Bergami, M.; Francolini, M.; Schenk, U.; Colombo, A.; Riganti, L.; Frassoni, C.; Zuccaro, E.; Danglot, L.; et al. TI-VAMP/VAMP7 is the SNARE of secretory lysosomes contributing to ATP secretion from astrocytes. Biol. Cell 2012, 104, 213–228. [Google Scholar]
- Belai, A.; Burnstock, G. Pattern of distribution and co-localization of NOS and ATP in the myenteric plexus of human fetal stomach and intestine. Neuroreport 2000, 11, 5–8. [Google Scholar]
- Bodin, P.; Burnstock, G. Evidence that release of adenosine triphosphate from endothelial cells during increased shear stress is vesicular. J. Cardiovasc. Pharmacol 2001, 38, 900–908. [Google Scholar]
- Pascual, O.; Casper, K.B.; Kubera, C.; Zhang, J.; Revilla-Sanchez, R.; Sul, J.Y.; Takano, H.; Moss, S.J.; McCarthy, K.; Haydon, P.G. Astrocytic purinergic signaling coordinates synaptic networks. Science 2005, 310, 113–116. [Google Scholar]
- Haydon, P.G.; Carmignoto, G. Astrocyte control of synaptic transmission and neurovascular coupling. Physiol. Rev 2006, 86, 1009–1031. [Google Scholar]
- Zhang, J.M.; Wang, H.K.; Ye, C.Q.; Ge, W.; Chen, Y.; Jiang, Z.L.; Wu, C.P.; Poo, M.M.; Duan, S. ATP released by astrocytes mediates glutamatergic activity-dependent heterosynaptic suppression. Neuron 2003, 40, 971–982. [Google Scholar]
- Han, W.; Ng, Y.; Axelrod, D.; Levitan, E. Neuropeptide release by efficient recruitment of diffusing cytoplasmic secretory vesicles. Proc. Natl. Acad. Sci. USA 1999, 96, 14577–14582. [Google Scholar]
- Taraska, J.; Perrais, D.; Ohara-Imaizumi, M.; Nagamatsu, S.; Almers, W. Secretory granules are recaptured largely intact after stimulated exocytosis in cultured endocrine cells. Proc. Natl. Acad. Sci. USA 2003, 100, 2070–2075. [Google Scholar]
- Jahn, R.; Südhof, T. Membrane fusion and exocytosis. Annu. Rev. Biochem 1999, 68, 863–911. [Google Scholar]
- Valtorta, F.; Meldolesi, J.; Fesce, R. Synaptic vesicles: Is kissing a matter of competence? Trends Cell Biol 2001, 11, 324–328. [Google Scholar]
- Nogami, M.; Shiga, J.; Takatsu, A.; Endo, N.; Ishiyama, I. Immunohistochemistry of atrial natriuretic peptide in brain infarction. Histochem. J 2001, 33, 87–90. [Google Scholar]
- Autry, A.E.; Adachi, M.; Nosyreva, E.; Na, E.S.; Los, M.F.; Cheng, P.F.; Kavalali, E.T.; Monteggia, L.M. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 2011, 475, 91–95. [Google Scholar]
- Zarate, C.A.; Singh, J.B.; Carlson, P.J.; Brutsche, N.E.; Ameli, R.; Luckenbaugh, D.A.; Charney, D.S.; Manji, H.K. A randomized trial of an N-methyl-d-aspartate antagonist in treatment-resistant major depression. Arch. Gen. Psychiatry 2006, 63, 856–864. [Google Scholar]
- Potokar, M.; Kreft, M.; Lee, S.; Takano, H.; Haydon, P.; Zorec, R. Trafficking of astrocytic vesicles in hippocampal slices. Biochem. Biophys. Res. Commun 2009, 390, 1192–1196. [Google Scholar]
- Pekny, M.; Eliasson, C.; Chien, C.; Kindblom, L.; Liem, R.; Hamberger, A.; Betsholtz, C. GFAP-deficient astrocytes are capable of stellation in vitro when cocultured with neurons and exhibit a reduced amount of intermediate filaments and an increased cell saturation density. Exp. Cell Res 1998, 239, 332–343. [Google Scholar]
- Wilhelmsson, U.; Li, L.; Pekna, M.; Berthold, C.; Blom, S.; Eliasson, C.; Renner, O.; Bushong, E.; Ellisman, M.; Morgan, T.; Pekny, M. Absence of glial fibrillary acidic protein and vimentin prevents hypertrophy of astrocytic processes and improves post-traumatic regeneration. J. Neurosci 2004, 24, 5016–5021. [Google Scholar]
- Eliasson, C.; Sahlgren, C.; Berthold, C.; Stakeberg, J.; Celis, J.; Betsholtz, C.; Eriksson, J.; Pekny, M. Intermediate filament protein partnership in astrocytes. J. Biol. Chem 1999, 274, 23996–24006. [Google Scholar]
- Haidet-Phillips, A.M.; Hester, M.E.; Miranda, C.J.; Meyer, K.; Braun, L.; Frakes, A.; Song, S.; Likhite, S.; Murtha, M.J.; Foust, K.D.; et al. Astrocytes from familial and sporadic ALS patients are toxic to motor neurons. Nat. Biotechnol 2011, 29, 824–828. [Google Scholar]
- Eisen, A. Amyotrophic lateral sclerosis: A 40-year personal perspective. J. Clin. Neurosci 2009, 16, 505–512. [Google Scholar]
- Ekestern, E. Neurotrophic factors and amyotrophic lateral sclerosis. Neurodegener Dis 2004, 1, 88–100. [Google Scholar]
- Pehar, M.; Cassina, P.; Vargas, M.R.; Castellanos, R.; Viera, L.; Beckman, J.S.; Estévez, A.G.; Barbeito, L. Astrocytic production of nerve growth factor in motor neuron apoptosis: Implications for amyotrophic lateral sclerosis. J. Neurochem 2004, 89, 464–473. [Google Scholar]
- Urushitani, M.; Sik, A.; Sakurai, T.; Nukina, N.; Takahashi, R.; Julien, J.P. Chromogranin-mediated secretion of mutant superoxide dismutase proteins linked to amyotrophic lateral sclerosis. Nat. Neurosci 2006, 9, 108–118. [Google Scholar]
- Rothstein, J.D.; van Kammen, M.; Levey, A.I.; Martin, L.J.; Kuncl, R.W. Selective loss of glial glutamate transporter GLT-1 in amyotrophic lateral sclerosis. Ann. Neurol 1995, 38, 73–84. [Google Scholar]
- van Damme, P.; Dewil, M.; Robberecht, W.; van den Bosch, L. Excitotoxicity and amyotrophic lateral sclerosis. Neurodegener. Dis 2005, 2, 147–159. [Google Scholar]
- Zhang, R.; Gascon, R.; Miller, R.G.; Gelinas, D.F.; Mass, J.; Hadlock, K.; Jin, X.; Reis, J.; Narvaez, A.; McGrath, M.S. Evidence for systemic immune system alterations in sporadic amyotrophic lateral sclerosis (sALS). J. Neuroimmunol 2005, 159, 215–224. [Google Scholar]
- Garbuzova-Davis, S.; Saporta, S.; Haller, E.; Kolomey, I.; Bennett, S.P.; Potter, H.; Sanberg, P.R. Evidence of compromised blood-spinal cord barrier in early and late symptomatic SOD1 mice modeling ALS. PLoS One 2007, 2, e1205. [Google Scholar]
- Di Giorgio, F.P.; Carrasco, M.A.; Siao, M.C.; Maniatis, T.; Eggan, K. Non-cell autonomous effect of glia on motor neurons in an embryonic stem cell-based ALS model. Nat. Neurosci 2007, 10, 608–614. [Google Scholar]
- Nagai, M.; Re, D.B.; Nagata, T.; Chalazonitis, A.; Jessell, T.M.; Wichterle, H.; Przedborski, S. Astrocytes expressing ALS-linked mutated SOD1 release factors selectively toxic to motor neurons. Nat. Neurosci 2007, 10, 615–622. [Google Scholar]
- Hasegawa, H.; Ma, T.; Skach, W.; Matthay, M.A.; Verkman, A.S. Molecular cloning of a mercurial-insensitive water channel expressed in selected water-transporting tissues. J. Biol. Chem 1994, 269, 5497–5500. [Google Scholar]
- Amiry-Moghaddam, M.; Ottersen, O.P. The molecular basis of water transport in the brain. Nat. Rev. Neurosci 2003, 4, 991–1001. [Google Scholar]
- Jung, J.S.; Bhat, R.V.; Preston, G.M.; Guggino, W.B.; Baraban, J.M.; Agre, P. Molecular characterization of an aquaporin cDNA from brain: Candidate osmoreceptor and regulator of water balance. Proc. Natl. Acad. Sci. USA 1994, 91, 13052–13056. [Google Scholar]
- Nielsen, S.; Nagelhus, E.A.; Amiry-Moghaddam, M.; Bourque, C.; Agre, P.; Ottersen, O.P. Specialized membrane domains for water transport in glial cells: High-resolution immunogold cytochemistry of aquaporin-4 in rat brain. J. Neurosci 1997, 17, 171–180. [Google Scholar]
- Badaut, J.; Lasbennes, F.; Magistretti, P.J.; Regli, L. Aquaporins in brain: Distribution, physiology, and pathophysiology. J. Cereb. Blood Flow Metab 2002, 22, 367–378. [Google Scholar]
- Arciénega, I.I.; Brunet, J.F.; Bloch, J.; Badaut, J. Cell locations for AQP1, AQP4 and 9 in the non-human primate brain. Neuroscience 2010, 167, 1103–1114. [Google Scholar]
- Nagelhus, E.A.; Veruki, M.L.; Torp, R.; Haug, F.M.; Laake, J.H.; Nielsen, S.; Agre, P.; Ottersen, O.P. Aquaporin-4 water channel protein in the rat retina and optic nerve: Polarized expression in Müller cells and fibrous astrocytes. J. Neurosci 1998, 18, 2506–2519. [Google Scholar]
- Neely, J.D.; Christensen, B.M.; Nielsen, S.; Agre, P. Heterotetrameric composition of aquaporin-4 water channels. Biochemistry 1999, 38, 11156–11163. [Google Scholar]
- Badaut, J.; Nehlig, A.; Verbavatz, J.; Stoeckel, M.; Freund-Mercier, M.J.; Lasbennes, F. Hypervascularization in the magnocellular nuclei of the rat hypothalamus: Relationship with the distribution of aquaporin-4 and markers of energy metabolism. J. Neuroendocrinol 2000, 12, 960–969. [Google Scholar]
- Badaut, J.; Verbavatz, J.M.; Freund-Mercier, M.J.; Lasbennes, F. Presence of aquaporin-4 and muscarinic receptors in astrocytes and ependymal cells in rat brain: A clue to a common function? Neurosci. Lett 2000, 292, 75–78. [Google Scholar]
- Arima, H.; Yamamoto, N.; Sobue, K.; Umenishi, F.; Tada, T.; Katsuya, H.; Asai, K. Hyperosmolar mannitol simulates expression of aquaporins 4 and 9 through a p38 mitogen-activated protein kinase-dependent pathway in rat astrocytes. J. Biol. Chem 2003, 278, 44525–44534. [Google Scholar]
- Yamamoto, N.; Yoneda, K.; Asai, K.; Sobue, K.; Tada, T.; Fujita, Y.; Katsuya, H.; Fujita, M.; Aihara, N.; Mase, M.; et al. Alterations in the expression of the AQP family in cultured rat astrocytes during hypoxia and reoxygenation. Brain Res. Mol. Brain Res 2001, 90, 26–38. [Google Scholar]
- Manley, G.T.; Fujimura, M.; Ma, T.; Noshita, N.; Filiz, F.; Bollen, A.W.; Chan, P.; Verkman, A.S. Aquaporin-4 deletion in mice reduces brain edema after acute water intoxication and ischemic stroke. Nat. Med 2000, 6, 159–163. [Google Scholar]
- Papadopoulos, M.C.; Manley, G.T.; Krishna, S.; Verkman, A.S. Aquaporin-4 facilitates reabsorption of excess fluid in vasogenic brain edema. FASEB J 2004, 18, 1291–1293. [Google Scholar]
- Gunnarson, E.; Zelenina, M.; Axehult, G.; Song, Y.; Bondar, A.; Krieger, P.; Brismar, H.; Zelenin, S.; Aperia, A. Identification of a molecular target for glutamate regulation of astrocyte water permeability. Glia 2008, 56, 587–596. [Google Scholar]
- Nicchia, G.P.; Ficarella, R.; Rossi, A.; Giangreco, I.; Nicolotti, O.; Carotti, A.; Pisani, F.; Estivill, X.; Gasparini, P.; Svelto, M.; et al. D184E mutation in aquaporin-4 gene impairs water permeability and links to deafness. Neuroscience 2011, 197, 80–88. [Google Scholar]
- Hirt, B.; Gleiser, C.; Eckhard, A.; Mack, A.F.; Müller, M.; Wolburg, H.; Löwenheim, H. All functional aquaporin-4 isoforms are expressed in the rat cochlea and contribute to the formation of orthogonal arrays of particles. Neuroscience 2011, 189, 79–92. [Google Scholar]
- Moe, S.E.; Sorbo, J.G.; Sogaard, R.; Zeuthen, T.; Petter Ottersen, O.; Holen, T. New isoforms of rat Aquaporin-4. Genomics 2008, 91, 367–377. [Google Scholar]
- Nicchia, G.P.; Rossi, A.; Mola, M.G.; Procino, G.; Frigeri, A.; Svelto, M. Actin cytoskeleton remodeling governs aquaporin-4 localization in astrocytes. Glia 2008, 56, 1755–1766. [Google Scholar]
- Thrane, A.S.; Rappold, P.M.; Fujita, T.; Torres, A.; Bekar, L.K.; Takano, T.; Peng, W.; Wang, F.; Thrane, V.R.; Enger, R.; et al. Critical role of aquaporin-4 (AQP4) in astrocytic Ca2+ signaling events elicited by cerebral edema. Proc. Natl. Acad. Sci. USA 2011, 108, 846–851. [Google Scholar]
- Pangrsic, T.; Potokar, M.; Haydon, P.; Zorec, R.; Kreft, M. Astrocyte swelling leads to membrane unfolding, not membrane insertion. J. Neurochem 2006, 99, 514–523. [Google Scholar]
- Takano, T.; Kang, J.; Jaiswal, J.K.; Simon, S.M.; Lin, J.H.; Yu, Y.; Li, Y.; Yang, J.; Dienel, G.; Zielke, H.R.; et al. Receptor-mediated glutamate release from volume sensitive channels in astrocytes. Proc. Natl. Acad. Sci. USA 2005, 102, 16466–16471. [Google Scholar]
- Pasantes-Morales, H.; Franco, R.; Ochoa, L.; Ordaz, B. Osmosensitive release of neurotransmitter amino acids: Relevance and mechanisms. Neurochem. Res 2002, 27, 59–65. [Google Scholar]
- Soos, J.M.; Morrow, J.; Ashley, T.A.; Szente, B.E.; Bikoff, E.K.; Zamvil, S.S. Astrocytes express elements of the class II endocytic pathway and process central nervous system autoantigen for presentation to encephalitogenic T cells. J. Immunol 1998, 161, 5959–5966. [Google Scholar]
- Stenovec, M.; Kreft, M.; Grilc, S.; Pangrsic, T.; Zorec, R. EAAT2 density at the astrocyte plasma membrane and Ca(2+)-regulated exocytosis. Mol. Membr. Biol 2008, 25, 203–215. [Google Scholar]
- Fontana, A.; Fierz, W.; Wekerle, H. Astrocytes present myelin basic protein to encephalitogenic T-cell lines. Nature 1984, 307, 273–276. [Google Scholar]
- Shrikant, P.; Benveniste, E.N. The central nervous system as an immunocompetent organ: Role of glial cells in antigen presentation. J. Immunol 1996, 157, 1819–1822. [Google Scholar]
- Barois, N.; Forquet, F.; Davoust, J. Actin microfilaments control the MHC class II antigen presentation pathway in B cells. J. Cell Sci 1998, 111, 1791–1800. [Google Scholar]
- Wubbolts, R.; Fernandez-Borja, M.; Jordens, I.; Reits, E.; Dusseljee, S.; Echeverri, C.; Vallee, R.B.; Neefjes, J. Opposing motor activities of dynein and kinesin determine retention and transport of MHC class II-containing compartments. J. Cell Sci 1999, 112, 785–795. [Google Scholar]
- Vyas, J.M.; Kim, Y.M.; Artavanis-Tsakonas, K.; Love, J.C.; van der Veen, A.G.; Ploegh, H.L. Tubulation of class II MHC compartments is microtubule dependent and involves multiple endolysosomal membrane proteins in primary dendritic cells. J. Immunol 2007, 178, 7199–7210. [Google Scholar]
- Vascotto, F.; Lankar, D.; Faure-André, G.; Vargas, P.; Diaz, J.; le Roux, D.; Yuseff, M.I.; Sibarita, J.B.; Boes, M.; Raposo, G.; et al. The actin-based motor protein myosin II regulates MHC class II trafficking and BCR-driven antigen presentation. J. Cell Biol 2007, 176, 1007–1019. [Google Scholar]
- Junyent, F.; de Lemos, L.; Utrera, J.; Paco, S.; Aguado, F.; Camins, A.; Pallàs, M.; Romero, R.; Auladell, C. Content and traffic of taurine in hippocampal reactive astrocytes. Hippocampus 2011, 21, 185–197. [Google Scholar]
- Gabrijel, M.; Kreft, M.; Zorec, R. Monitoring lysosomal fusion in electrofused hybridoma cells. Biochim. Biophys. Acta 2008, 1778, 483–490. [Google Scholar]
- Jaiswal, J.K.; Andrews, N.W.; Simon, S.M. Membrane proximal lysosomes are the major vesicles responsible for calcium-dependent exocytosis in nonsecretory cells. J. Cell Biol 2002, 159, 625–635. [Google Scholar]
- de Keyser, J.; Zeinstra, E.; Mostert, J.; Wilczak, N. Beta 2-adrenoceptor involvement in inflammatory demyelination and axonal degeneration in multiple sclerosis. Trends Pharmacol. Sci 2004, 25, 67–71. [Google Scholar]
- Frohman, E.M.; Vayuvegula, B.; Gupta, S.; van den Noort, S. Norepinephrine inhibits gamma-interferon-induced major histocompatibility class II (Ia) antigen expression on cultured astrocytes via beta-2-adrenergic signal transduction mechanisms. Proc. Natl. Acad. Sci. USA 1988, 85, 1292–1296. [Google Scholar]
- Vesce, S.; Bezzi, P.; Volterra, A. The active role of astrocytes in synaptic transmission. Cell Mol. Life Sci 1999, 56, 991–1000. [Google Scholar]
- Amara, S.G.; Fontana, A.C. Excitatory amino acid transporters: Keeping up with glutamate. Neurochem. Int 2002, 41, 313–318. [Google Scholar]
- O’Kane, R.L.; Martínez-López, I.; DeJoseph, M.R.; Viña, J.R.; Hawkins, R.A. Na(+)-dependent glutamate transporters (EAAT1, EAAT2, and EAAT3) of the blood-brain barrier. A mechanism for glutamate removal. J. Biol. Chem 1999, 274, 31891–31895. [Google Scholar]
- Robinson, M.B. Regulated trafficking of neurotransmitter transporters: Common notes but different melodies. J. Neurochem 2002, 80, 1–11. [Google Scholar]
- Huang, Y.H.; Bergles, D.E. Glutamate transporters bring competition to the synapse. Curr. Opin. Neurobiol 2004, 14, 346–352. [Google Scholar]
- Chun, J.; Brinkmann, V. A mechanistically novel, first oral therapy for multiple sclerosis: The development of fingolimod (FTY720, Gilenya). Discov. Med 2011, 12, 213–228. [Google Scholar]
- Foster, C.A.; Howard, L.M.; Schweitzer, A.; Persohn, E.; Hiestand, P.C.; Balatoni, B.; Reuschel, R.; Beerli, C.; Schwartz, M.; Billich, A. Brain penetration of the oral immunomodulatory drug FTY720 and its phosphorylation in the central nervous system during experimental autoimmune encephalomyelitis: Consequences for mode of action in multiple sclerosis. J. Pharmacol. Exp. Ther 2007, 323, 469–475. [Google Scholar]
- Murphy, S.; Pearce, B.; Jeremy, J.; Dandona, P. Astrocytes as eicosanoid-producing cells. Glia 1988, 1, 241–245. [Google Scholar]



| Vesicle type (cargo) | Velocity (μm/s) (spontaneous) | Velocity (μm/s) (stimulation) | References | Comment * |
|---|---|---|---|---|
| Recycling (VGLUT1) | 0.05 ± 0.02 | 0.13 ± 0.01 (ATP) 0.08 ± 0.00 (Iono) | Stenovec et al., 2007 [36] | Cell culture, rat |
| Recycling (VGLUT1) WT | 0.06 ± 0.00 | ND | Potokar et al., 2010 [32] | Cell culture, mouse |
| Recycling (VGLUT1) GFAP−/−Vim−/− | 0.08 ± 0.00 | ND | Potokar et al., 2010 [32] | Cell culture, mouse |
| Recycling (VGLUT1) | 0.028 ± 0.001 | ND | Potokar et al., 2009 [81] | Hippocampal slices, mouse |
| Recycling (ANP) WT | 0.07 ± 0.00 | ND | Potokar et al., 2010 [32] | Cell culture, mouse |
| Recycling (ANP) GFAP−/−Vim−/− | 0.07 ± 0.00 | ND | Potokar et al., 2010 [32] | Cell culture, mouse |
| Recycling (ANP) | 0.037± 0.001 | ND | Potokar et al., 2009 [81] | Hippocampal slices, mouse |
| Recycling (ANP) | 0.06 ± 0.001 | 0.02 ± 0.002 (Iono), 0.03 ± 0.001 (ATP) | Potokar et al., 2008 [31] | Cell culture, rat |
| Secretory vesicle (ANP) | 0.40 ± 0.007 0.29 ± 0.01 | ND 0.27 ± 0.01 (Iono) | Potokar et al., 2005 [19]; Potokar et al., 2007 [20] | Cell culture, rat and mouse |
| Secretory vesicle (VGLUT1) Secretory vesicles (Sb2) | 0.19 ± 0.02 0.65 ± 0.04 | ND ND | Trkov et al., 2012 [35] Crippa et al.,2006 [42] | Cell culture, rat Cell culture, rat |
| Endo./lyso. WT | 0.04 ± 0.00 | ND | Potokar et al., 2010 [32] | Cell culture, mouse |
| Endo./lyso. GFAP−/−Vim−/− | 0.04 ± 0.00 | ND | Potokar et al., 2010 [32] | Cell culture, mouse |
| Late endo./lyso. WT | 0.058 ± 0.001 | 0.052 ± 0.001 (ATP) | Vardjan et al., 2012 [21] | Cell culture, mouse |
| Late endo./lyso. GFAP−/−Vim−/− | 0.050 ± 0.001 | 0.049 ± 0.001 (ATP) | Vardjan et al., 2012 [21] | Cell culture, mouse |
| Late endo./lyso. (MHC-II) WT | 0.079 ± 0.001 | 0.057 ± 0.001 (ATP) | Vardjan et al., 2012 [21] | Cell culture, mouse |
| Late endo./lyso. (MHC-II) GFAP−/−Vim−/− | 0.053 ± 0.001 | 0.047 ± 0.001 (ATP) | Vardjan et al., 2012 [21] | Cell culture, mouse |
| Late endo./lyso. (AQP4) | 0.04 ± 0.00, 0.06 ± 0.00 | ND | Potokar et al., 2013 [29] | Cell culture, rat |
| Endo./lyso. (CB1 receptor) | 0.11 | ND | Osborne et al., 2009 [14] | Cell culture, rat |
| Endo./lyso. (JAGGED/Notch) | 0.06 ± 0.00 | ND | Stenovec et al., unpublished | Cell culture, mouse |
| Endo./lyso. | 0.11 ± 0.02 | 0.067±0.001 (Fingo) | Trkov et al., 2012 [35] | Cell culture, rat |
| Endo./lyso | 0.21 ± 0.00 | 0.23 ± 0.00 (ALS IgG) | Stenovec et al., 2011 [34] | Cell culture, rat |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Potokar, M.; Vardjan, N.; Stenovec, M.; Gabrijel, M.; Trkov, S.; Jorgačevski, J.; Kreft, M.; Zorec, R. Astrocytic Vesicle Mobility in Health and Disease. Int. J. Mol. Sci. 2013, 14, 11238-11258. https://doi.org/10.3390/ijms140611238
Potokar M, Vardjan N, Stenovec M, Gabrijel M, Trkov S, Jorgačevski J, Kreft M, Zorec R. Astrocytic Vesicle Mobility in Health and Disease. International Journal of Molecular Sciences. 2013; 14(6):11238-11258. https://doi.org/10.3390/ijms140611238
Chicago/Turabian StylePotokar, Maja, Nina Vardjan, Matjaž Stenovec, Mateja Gabrijel, Saša Trkov, Jernej Jorgačevski, Marko Kreft, and Robert Zorec. 2013. "Astrocytic Vesicle Mobility in Health and Disease" International Journal of Molecular Sciences 14, no. 6: 11238-11258. https://doi.org/10.3390/ijms140611238





