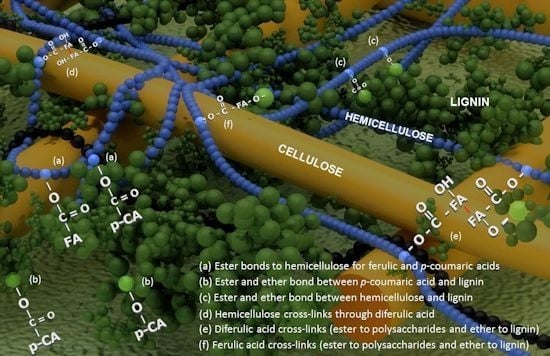
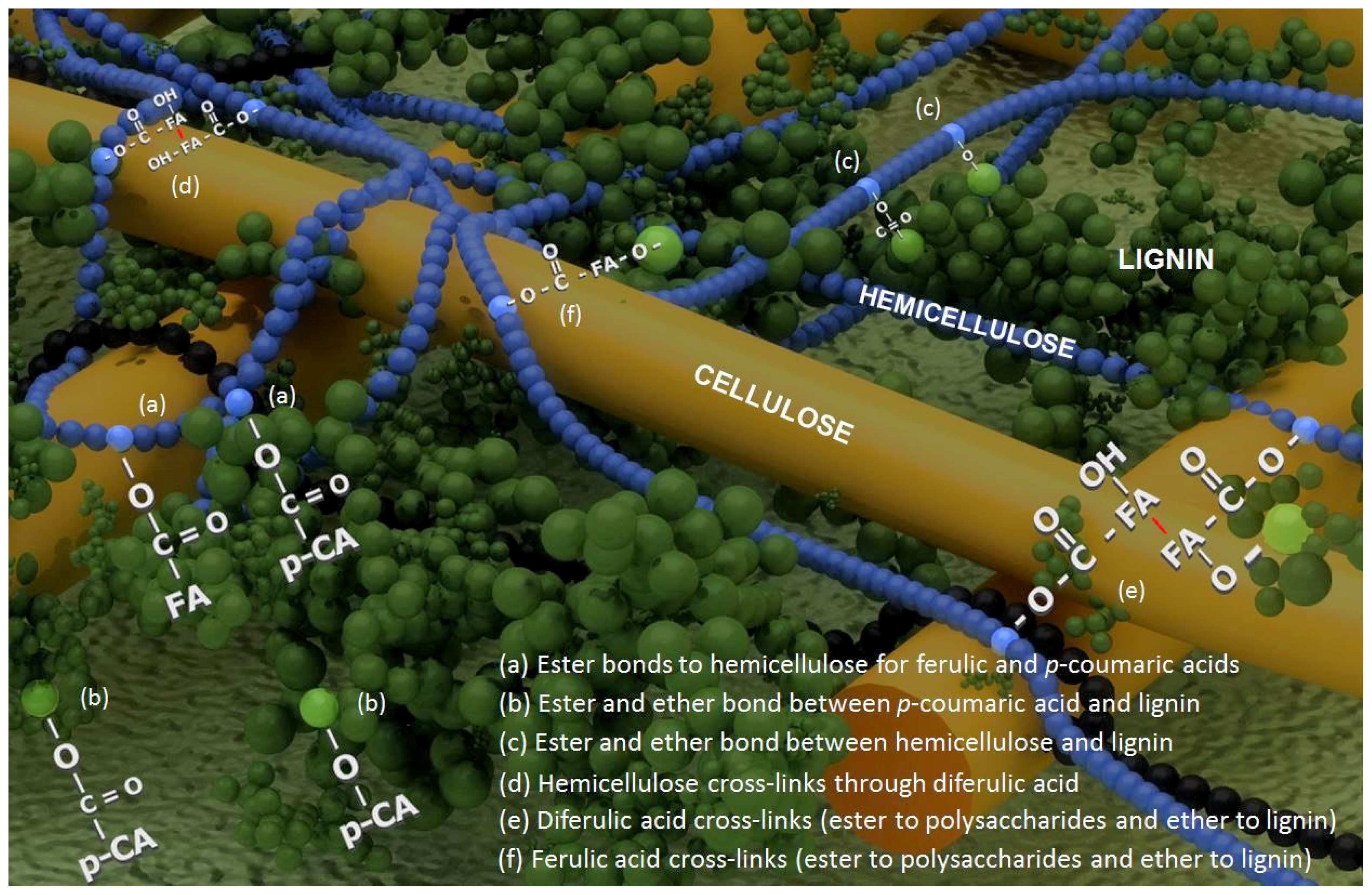
Impact of Cell Wall Composition on Maize Resistance to Pests and Diseases
Abstract
:1. Introduction
2. Cell Wall Polysaccharides
3. Cell Wall Bound Hydroxycinnamates
4. Lignin
5. Conclusions
Acknowledgments
Abbreviations
| FA | Ferulate |
| pCA | p-Coumarate |
| DFA | Diferulates |
| NDF | neutral detergent fiber |
| ADF | acid detergent fiber |
| ADL | acid detergent lignin |
| HRGPs | hydroxyproline-rich glycoprotein |
| ECB | European corn borer |
| SWCB | Southwestern corn borer |
| SCB | Sugarcane borer |
| MCB | Mediterranean corn borer |
| MW | Maize weevil |
| SRC | Southern rust of corn |
| MLB | Maydis leaf blight |
References
- Brett, C.T.; Waldron, K. Cell Wall Formation. In Physiology and Biochemistry of Plant Cell Walls; Brett, C.T., Waldron, K., Eds.; Chapman & Hall: London, UK, 1996; pp. 75–111. [Google Scholar]
- Hazen, S.P.; Hawley, R.M.; Davis, G.L.; Henrissat, B.; Walton, J.D. Quantitative trait loci and comparative genomics of cereal cell wall composition. Plant Physiol 2003, 132, 263–271. [Google Scholar]
- Carpita, N.; McCann, M. The Cell Wall. In Biochemistry and Molecular Biology of Plants; Buchanan, B.B., Gruissem, W., Jones, R.L., Eds.; American Society of Plant Physiologists: Rockville, MD, USA, 2000; pp. 52–108. [Google Scholar]
- Carpita, N.C.; Gibeaut, D.M. Structural models of primary cell walls in flowering consistency of molecular structure with the physical properties of the walls during growth. Plant J 1993, 3, 1–30. [Google Scholar]
- Cosgrove, D.J. Assembly and enlargement of the primary cell wall in plants. Annu. Rev. Cell Dev. Biol 1997, 13, 171–201. [Google Scholar]
- Whilstler, R.L.; Richards, E.L. Hemicelluloses. In The Carbohydrates; Pigman, W., Horton, D., Eds.; Academic Press: New York, NY, USA, 1970; Volume 2a, pp. 447–469. [Google Scholar]
- Hartley, R.D. p-Coumaric and ferulic components of cell-walls of ryegrass and their relationships with lignin and digestibility. J. Sci. Food Agric 1972, 23, 1347–1354. [Google Scholar]
- Scalbert, A.; Monties, B.; Lallemand, J.Y.; Guittet, E.; Rolando, C. Ether linkage between phenolic-acids and lignin fractions from wheta straw. Phytochemistry 1985, 24, 1359–1362. [Google Scholar]
- Ralph, J.; Grabber, J.H.; Hatfield, R.D. Lignin-ferulate cross-links in grasses. Active incorporation of ferulate polysaccharide esters into ryegrass lignins. Carbohydr. Res 1995, 275, 167–178. [Google Scholar]
- Barros-Ríos, J.; Malvar, R.A.; Santiago, R. Función de la pared celular del maíz (Zea mays L.) como mecanismos de defensa frente a la plaga del taladro (Ostrinia nubilalis Hüb y Sesamia nonagrioides Lef.). Rev. Educ. Bioquím 2011, 30, 132–142. [Google Scholar]
- Rojanaridpiched, C.; Gracen, V.E.; Everett, H.L.; Coors, J.G.; Pugh, B.F.; Bouthyette, P. Multiple factor resistance in maize to European corn borer. Maydica 1984, 29, 305–315. [Google Scholar]
- Ostrander, B.M.; Coors, J.G. Relationship between plant composition and European corn borer resistance in three maize populations. Crop Sci 1997, 37, 1741–1745. [Google Scholar]
- Buendgen, M.R.; Coors, J.G.; Grombacher, A.W.; Russell, W.A. European corn borer resistance and cell wall composition of three maize populations. Crop Sci 1990, 30, 505–510. [Google Scholar]
- Beeghly, H.H.; Coors, J.G.; Lee, M. Plant fiber composition and resistance to European corn borer in four maize populations. Maydica 1997, 42, 297–303. [Google Scholar]
- Martin, S.A.; Darrah, L.L.; Hibbard, B.E. Divergent selection for rind penetrometer resistance and its effects on European corn borer damage and stalk traits in corn. Crop Sci 2004, 44, 711–717. [Google Scholar]
- Cardinal, A.J.; Lee, M. Genetic relationships between resistance to stalk-tunneling by the European corn borer and cell-wall components in maize population B73 × B52. Theor. Appl. Genet 2005, 111, 1–7. [Google Scholar]
- Krakowsky, M.D.; Lee, M.; Holland, J.B. Genotypic correlation and multivariate QTL analyses for cell wall components and resistance to stalk tunneling by the European corn borer in maize. Crop Sci 2007, 47, 485–490. [Google Scholar]
- Krakowsky, M.D.; Lee, M.; Woodman-Clikeman, W.L.; Long, M.J.; Sharopova, N. QTL mapping of resistance to stalk tunneling by the European corn borer in RILs of maize population B73 × De811. Crop Sci 2004, 44, 274–282. [Google Scholar]
- Bergvinson, D.J.; Arnason, J.T.; Hamilton, R.I. Phytochemical changes during recurrent selection for resistance to the European corn borer. Crop Sci 1997, 37, 1567–1572. [Google Scholar]
- Bergvinson, D.J.; Arnason, J.T.; Pietrzak, L.N. Localization and quantification of cell wall phenolics in European corn borer resistant and susceptible maize inbreds. Can. J. Bot. Rev. Can. Bot 1994, 72, 1243–1249. [Google Scholar]
- Bergvinson, D.J.; Arnason, J.T.; Hamilton, R.I.; Mihm, J.A.; Jewell, D.C. Determining leaf toughness and its role in maize resistance to the European corn borer (Lepidoptera, Pyralidae). J. Econ. Entomol 1994, 87, 1743–1748. [Google Scholar]
- Hedin, P.A.; Davis, F.M.; Williams, W.P.; Salin, M.L. Possible factors of leaf feeding resistance in corn to the Southwestern corn borer. J. Agric. Food Chem 1984, 32, 262–267. [Google Scholar]
- Hedin, P.A.; Williams, W.P.; Davis, F.M.; Buckley, P.M. Roles of amino acids, protein, and fiber in leaf feeding resistance of corn to the fall armyworm. J. Chem. Ecol 1990, 16, 1977–1995. [Google Scholar]
- Hedin, P.A.; Davis, F.M.; Williams, W.P.; Hicks, R.P.; Fisher, T.H. Hemicellulose is an important leaf-feeding resistance factor in corn to the fall armyworm. J. Chem. Ecol 1996, 22, 1655–1668. [Google Scholar]
- Williams, W.P.; Davis, F.M.; Buckley, P.M.; Hedin, P.A.; Baker, G.T.; Luthe, D.S. Factors associated with resistance to fall armyworm (Lepidoptera: Noctuidae) and southwestern corn borer (Lepidoptera: Crambidae) in corn at different vegetative stages. J. Econ. Entomol 1998, 91, 1471–1480. [Google Scholar]
- Ramputh, A.I. Soluble and Cell Wall Bound Phenolic Mediated Insect Resistance in Corn and Sorghum. Ph.D. Dissertation, Ottawa-Carleton Institute of Biology, Ottawa, ON, Canada, 2002. [Google Scholar]
- Santiago, R.; Butron, A.; Arnason, J.T.; Reid, L.M.; Souto, X.C.; Malvar, R.A. Putative role of pith cell wall phenylpropanoids in Sesamia nonagrioides (Lepidoptera: Noctuidae) resistance. J. Agric. Food Chem 2006, 54, 2274–2279. [Google Scholar]
- Santiago, R.; Butron, A.; Revilla, P.; Ana Malvar, R. Is the basal area of maize internodes involved in borer resistance? BMC Plant Biol 2011, 11, 137. [Google Scholar] [Green Version]
- Santiago, R.; Butron, A.; Reid, L.M.; Arnason, J.T.; Sandoya, G.; Souto, X.C.; Malvar, R.A. Diferulate content of maize sheaths is associated with resistance to the Mediterranean corn borer Sesamia nonagrioides (Lepidoptera: Noctuidae). J. Agric. Food Chem 2006, 54, 9140–9144. [Google Scholar]
- Santiago, R.; Sandoya, G.; Butron, A.; Barros, J.; Malvar, R.A. Changes in phenolic concentrations during recurrent selection for resistance to the Mediterranean corn borer (Sesamia nonagrioides Lef.). J. Agric. Food Chem 2008, 56, 8017–8022. [Google Scholar]
- Rodriguez, V.M.; Santiago, R.; Ana Malvar, R.; Butron, A. Inducible maize defense mechanisms against the corn borer Sesamia nonagrioides: A transcriptome and biochemical approach. Mol. Plant Microbe Interact 2012, 25, 61–68. [Google Scholar]
- Barros-Rios, J.; Malvar, R.A.; Jung, H.J.; Santiago, R. Cell wall composition as a maize defense mechanism against corn borers. Phytochemistry 2011, 72, 365–371. [Google Scholar]
- Classen, D.; Arnason, J.T.; Serratos, J.A.; Lambert, J.D.H.; Nozzolillo, C.; Philogene, B.J.R. Correlation of phenolic acid content of maize to resistance to Sitophilus zeamais, the maize weevil, in CIMMYTs collections. J. Chem. Ecol 1990, 16, 301–315. [Google Scholar]
- Arnason, J.T.; Baum, B.; Gale, J.; Lambert, J.D.H.; Bergvinson, D.; Philogene, B.J.R.; Serratos, J.A.; Mihm, J.; Jewell, D.C. Variation in resistance of mexican landraces to maize weevil Sitophilus zeamais, in relation to taxonomic and biochemical parameters. Euphytica 1994, 74, 227–236. [Google Scholar]
- Garcia-Lara, S.; Bergvinson, D.J.; Burt, A.J.; Ramputh, A.I.; Diaz-Pontones, D.M.; Arnason, J.T. The role of pericarp cell wall components in maize weevil resistance. Crop Sci 2004, 44, 1546–1552. [Google Scholar]
- Bergvinson, D.; Garcia-Lara, S. Genetic approaches to reducing losses of stored grain to insects and diseases. Curr. Opin. Plant Biol 2004, 7, 480–485. [Google Scholar]
- García-Lara, S.; Burt, A.J.; Arnason, J.T.; Bergvinson, D.J. QTL mapping of tropical maize grain components associated with maize weevil resistance. Crop Sci 2010, 50, 815. [Google Scholar]
- Santiago, R.; Reid, L.M.; Arnason, J.T.; Zhu, X.; Martinez, N.; Malvar, R.A. Phenolics in maize genotypes differing in susceptibility to Gibberella stalk rot (Fusarium graminearum schwabe). J. Agric. Food Chem 2007, 55, 5186–5193. [Google Scholar]
- Liu, X.Y.; Jin, J.Y.; He, P.; Gao, W.; Li, W.J. Effect of potassium chloride on lignin metabolism and its relation to resistance of corn to stalk rot. Sci. Agric. Sin 2007, 40, 2780–2787. [Google Scholar]
- Bily, A.C.; Reid, L.M.; Taylor, J.H.; Johnston, D.; Malouin, C.; Burt, A.J.; Bakan, B.; Regnault-Roger, C.; Pauls, K.P.; Arnason, J.T.; Philogene, B.J.R. Dehydrodimers of ferulic acid in maize grain pericarp and aleurone: Resistance factors to Fusarium graminearum. Phytopathology 2003, 93, 712–719. [Google Scholar]
- Assabgui, R.A.; Reid, L.M.; Hamilton, R.I.; Arnason, J.T. Correlation of kernel E-ferulic acid content of maize with resitance to Fusarium graminearum. Phytopathology 1993, 83, 949–953. [Google Scholar]
- Cao, A.; Reid, L.M.; Butron, A.; Malvar, R.A.; Souto, X.C.; Santiago, R. Role of hydroxycinnamic acids in the infection of maize silks by Fusarium graminearum Schwabe. Mol. Plant Microbe Interact 2011, 24, 1020–1026. [Google Scholar]
- Sampietro, D.A.; Fauguel, C.M.; Vattuone, M.A.; Presello, D.A.; Catalán, C.A.N. Phenylpropanoids from maize pericarp: Resistance factors to kernel infection and fumonisin accumulation by Fusarium verticillioides. Eur. J. Plant Pathol 2012, 135, 105–113. [Google Scholar]
- Mideros, S.X.; Windham, G.L.; Williams, W.P.; Nelson, R.J. Tissue-specific components of resistance to aspergillus ear rot of maize. Phytopathology 2012, 102, 787–793. [Google Scholar]
- Spangler, L.M. Impact of Lignin and Caffeoyl Coenzyme a O-methyltransferase 1 on Aspergillus flavusGrowth in Maize Cobs. Master’s Thesis, The Pennsylvania State University, 2008. [Google Scholar]
- Ji, H.C.; Yamakawa, T. Cell wall contents and estimation of genetic parameters for polysora rust resistance in tropical maize (Zea mays L.). J. Fac. Agric. Kyushu Univ 2011, 56, 231–236. [Google Scholar]
- Lyons, P.C.; Hipskind, J.; Vincent, J.R.; Nicholson, R.L. Phenylpropanoid dissemination in maize resistant or susceptible to Helminthosporium maydis. Maydica 1993, 38, 175–181. [Google Scholar]
- Brodeur-Campbell, S.E.; Vucetich, J.A.; Richter, D.L.; Waite, T.A.; Rosemier, J.N.; Tsai, C.-J. Insect herbivory on low-lignin transgenic aspen. Environ. Entomol 2006, 35, 1696–1701. [Google Scholar]
- Schroeder, F.C.; del Campo, M.L.; Grant, J.B.; Weibel, D.B.; Smedley, S.R.; Bolton, K.L.; Meinwald, J.; Eisner, T. Pinoresinol: A lignol of plant origin serving for defense in a caterpillar. Proc. Natl. Acad. Sci. USA 2006, 103, 15497–15501. [Google Scholar]
- Read, J.; Stokes, A. Plant biomechanics in an ecological context. Am. J. Bot 2006, 93, 1546–1565. [Google Scholar]
- Clissold, F.J. The Biomechanics of Chewing and Plant Fracture: Mechanisms and Implications. In Advances in Insect Physiology: Insect Mechanics and Control; Casas, J., Simpson, S.J., Eds.; Elsevier Science Serials: Amsterdam, The Netherlands, 2007; Volume 34, pp. 317–372. [Google Scholar]
- Bell, A.A. Biochemical mechanisms of disease resistance. Annu. Rev. Plant Physiol. Plant Mol. Biol 1981, 32, 21–81. [Google Scholar]
- Friend, J. Plant phenolics, lignification and plant disease. Prog. Phytochem 1981, 7, 197–261. [Google Scholar]
- Nicholson, R.L.; Hammerschmidt, R. Phenolic compounds and their role in disease resistance. Annu. Rev. Phytopathol 1992, 30, 369–389. [Google Scholar]
- Matern, U.; Grimmig, B.; Kneusel, R.E. Plant cell wall reinforcement in the disease resistance response. Molecular composition and regulation. Can. J. Bot. Rev. Can. Bot 1995, 73, S511–S517. [Google Scholar]
- Grabber, J.H.; Hatfield, R.D.; Ralph, J. Diferulate cross-links impede the enzymatic degradation of non-lignified maize walls. J. Sci. Food Agric 1998, 77, 193–200. [Google Scholar]
- Taylor, N.G. Cellulose biosynthesis and deposition in higher plants. New Phytol 2008, 178, 239–252. [Google Scholar]
- Somerville, C.; Bauer, S.; Brininstool, G.; Facette, M.; Hamann, T.; Milne, J.; Osborne, E.; Parezdez, A.; Persson, S.; Raab, T.; et al. Towards a systems approach to understanding plant cell walls. Science 2004, 306, 2206–2211. [Google Scholar]
- Carpita, N.C. Structure and biogenesis of the cell walls of grasses. Annu. Rev. Plant Physiol. Plant Mol. Biol 1996, 47, 445–476. [Google Scholar]
- Van Soest, P. Nutritional Ecology of the Ruminant; Cornell University Press: Ithaca, NY, USA; p. 1994.
- Grombacher, A.W.; Russell, W.A.; Guthrie, W.D. Resistance to first generation European corn borer (Lepidoptera, Pyralidae) and DIMBOA concentration in midwhorl leaves of the BS9 maize synthetic. J. Kansas Entomol. Soc 1989, 62, 103–107. [Google Scholar]
- Santiago, R.; Souto, X.C.; Sotelo, J.; Butron, A.; Malvar, R.A. Relationship between maize stem structural characteristics and resistance to pink stem borer (Lepidoptera: Noctuidae) attack. J. Econ. Entomol 2003, 96, 1563–1570. [Google Scholar]
- Butron, A.; Malvar, R.A.; Cartea, M.E.; Ordas, A.; Velasco, P. Resistance of maize inbreds to pink stem borer. Crop Sci 1999, 39, 102–107. [Google Scholar]
- Ordas, B.; Butron, A.; Soengas, P.; Ordas, A.; Malvar, R.A. Antibiosis of the pith maize to Sesamia nonagrioides (Lepidoptera: Noctuidae). J. Econ. Entomol 2002, 95, 1044–1048. [Google Scholar]
- Rawlings, J.O. Applied Regression Analysis: A Research Tool, 2nd ed; Springer: New York, NY, USA, 1988. [Google Scholar]
- Cardinal, A.J.; Lee, M.; Moore, K.J. Genetic mapping and analysis of quantitative trait loci affecting fiber and lignin content in maize. Theor. Appl. Genet 2003, 106, 866–874. [Google Scholar]
- Cordero, A.; Malvar, R.A.; Butron, A.; Revilla, P.; Velasco, P.; Ordas, A. Population dynamics and life-cycle of corn borers in south Atlantic European coast. Maydica 1998, 43, 5–12. [Google Scholar]
- Velasco, P.; Revilla, P.; Monetti, L.; Butron, A.; Ordas, A.; Malvar, R.A. Corn borers (Lepidoptera: Noctuidae; Crambidae) in Northwestern Spain: Population dynamics and distribution. Maydica 2007, 52, 195–203. [Google Scholar]
- Hilal, A. Etude du développement de Sesamia nonagrioides et établissement de modèles pour la prévision de ses populations dans la nature. Eur. Mediterranean Plant Prot. Organ. Bull 1981, 11, 107–112. [Google Scholar]
- Larue, P. La Sésamie du maïs (Sesamia nonagrioides Lef.). Dégáts et actualisation de la lutte. Def. Veg 1984, 227, 163–179. [Google Scholar]
- Ordas, B.; Malvar, R.A.; Santiago, R.; Butron, A. QTL mapping for Mediterranean corn borer resistance in European flint germplasm using recombinant inbred lines. BMC Genomics 2010, 11, 174. [Google Scholar]
- Ordas, B.; Malvar, R.A.; Santiago, R.; Sandoya, G.; Romay, M.C.; Butron, A. Mapping of QTL for resistance to the Mediterranean corn borer attack using the intermated B73 × Mo17 (IBM) population of maize. Theor. Appl. Genet 2009, 119, 1451–1459. [Google Scholar]
- Krakowsky, M.D.; Brinkman, M.J.; Woodman-Clikeman, W.L.; Lee, M. Genetic components of resistance to stalk tunneling by the European corn borer in maize. Crop Sci 2002, 42, 1309–1315. [Google Scholar]
- Wild, C.P.; Turner, P.C. The toxicology of aflatoxins as a basis for public health decisions. Mutagenesis 2002, 17, 471–481. [Google Scholar]
- Melching, J.S. Corn Rusts: Types, Races, and Destructive Potential. Proceedings of the 30th Annual Corn and Sorghum Research Conference, Washington DC, USA, 1975; pp. 90–115.
- Logrieco, A.; Bottalico, A.; Mule, G.; Moretti, A.; Perrone, G. Epidemiology of toxigenic fungi and their associated mycotoxins for some Mediterranean crops. Eur. J. Plant Pathol 2003, 109, 645–667. [Google Scholar]
- Munkvold, G.P. Epidemiology of Fusarium diseases and their mycotoxins in maize ears. Eur. J. Plant Pathol 2003, 109, 705–713. [Google Scholar]
- Sutton, J.C.; Baliko, W.; Funnell, H.S. Evidence for translocation of zearalenone in corn plants colonized by Fusarium graminearum. Can. J. Plant Sci 1976, 56, 7–12. [Google Scholar]
- Grabber, J.H.; Ralph, J.; Lapierre, C.; Barrière, Y. Genetic and molecular basis of grass cell-wall degradability. I. Lignin-cell wall matrix interactions. C. R. Biol 2004, 327, 455–465. [Google Scholar]
- Ralph, J.; Hatfield, R.D.; Quideau, S.; Helm, R.F.; Grabber, J.H.; Jung, H.J.G. Pathway of p-coumaric acid incorporation into maize lignin as revealed by NMR. J. Am. Chem. Soc 1994, 116, 9448–9456. [Google Scholar]
- Grabber, J.H.; Quideau, S.; Ralph, J. p-coumaroylated syringyl units in maize lignin: Implications for beta-ether cleavage by thioacidolysis. Phytochemistry 1996, 43, 1189–1194. [Google Scholar]
- Bunzel, M.; Funk, C.; Steinhart, H. Semipreparative isolation of dehydrodiferulic and dehydrotriferulic acids as standard substances from maize bran. J. Sep. Sci 2004, 27, 1080–1086. [Google Scholar]
- Bunzel, M.; Heuermann, B.; Kim, H.; Ralph, J. Peroxidase-catalyzed oligomerization of ferulic acid esters. J. Agric. Food Chem 2008, 56, 10368–10375. [Google Scholar]
- Jacquet, G.; Pollet, B.; Lapierre, C. New ether-liked ferulic acid coniferyl alcohol dimers identified in grass straws. J. Agric. Food Chem 1995, 43, 2746–2751. [Google Scholar]
- Iiyama, K.; Lam, T.B.T.; Stone, B.A. Covalent cross-links in the cell wall. Plant Physiol 1994, 104, 315–320. [Google Scholar]
- Bartolome, B.; Faulds, C.B.; Tuohy, M.; Hazlewood, G.P.; Gilbert, H.J.; Williamson, G. Influence of different xylanases on the activity of ferulic acid esterase on wheat bran. Biotechnol. Appl. Biochem 1995, 22, 65–73. [Google Scholar]
- Hatfield, R.; Ralph, J.; Grabber, J.H. A potential role for sinapyl p-coumarate as a radical transfer mechanism in grass lignin formation. Planta 2008, 228, 919–928. [Google Scholar]
- Takahama, U.; Oniki, T. Enhancement of peroxidase-dependent oxidation of sinapyl alcohol by an apoplastic component, 4-coumaric acid ester isolated from epicotyls of Vigna angularis L. Plant Cell Physiol 1997, 38, 456–462. [Google Scholar]
- Mueller-Harvey, I.; Hartley, R.D.; Harris, P.J.; Curzon, E.H. Linkage of p-coumaryl and feruloyl groups to cell wall polysaccharides of barley straw. Carbohydr. Res 1986, 148, 71–85. [Google Scholar]
- Morrison, T.A.; Jung, H.G.; Buxton, D.R.; Hatfield, R.D. Cell-wall composition of maize internodes of varying maturity. Crop Sci 1998, 38, 455–460. [Google Scholar]
- Hatfield, R.D.; Chaptman, A.K. Comparing-corn types for differences in cell wall characteristics and p-coumaroylation of lignin. J. Agric. Food Chem 2009, 57, 4243–4249. [Google Scholar]
- Scobbie, L.; Russell, W.; Provan, G.J.; Chesson, A. The newly extended maize internode. A model study of secondary cell wall formation and consequences for digestibility. J. Sci. Food Agric 1993, 61, 217–225. [Google Scholar]
- MacAdam, J.W.; Grabber, J.H. Relationship of growth cessation with the formation of diferulate cross-links and p-coumaroylated lignins in tall fescue leaf blades. Planta 2002, 215, 785–93. [Google Scholar]
- Jung, H.-J.G. Maize stem tissues: Ferulate deposition in developing internode cell walls. Phytochemistry 2003, 63, 543–549. [Google Scholar]
- Fontaine, A.S.; Briand, M.; Barriere, Y. Genetic variation and QTL mapping of para-coumaric and ferulic acid contents in maize stover at silage harvest. Maydica 2003, 48, 75–82. [Google Scholar]
- Mechin, V.; Argillier, O.; Menanteau, V.; Barriere, Y.; Mila, I.; Pollet, B.; Lapierre, C. Relationship of cell wall composition to in vitro cell wall digestibility of maize inbred line stems. J. Sci. Food Agric 2000, 80, 574–580. [Google Scholar]
- Harris, P.J.; Hartley, R.D. Detection of bound ferulic acid in cell walls of the Gramineae by ultraviolet fluorescence microscopy. Nature 1976, 259, 508–510. [Google Scholar]
- Waldron, K.W.; Parr, A.J.; Ng, A.; Ralph, J. Cell wall esterified phenolic dimers: Identification and quantification by reverse phase high performance liquid chromatography and diode array detection. Phytochem. Anal 1996, 7, 305–312. [Google Scholar]
- Saulnier, L.; Thibault, J.F. Ferulic acid and diferulic acids as components of sugar-beet pectins and maize bran heteroxylans. J. Sci. Food Agric 1999, 79, 396–402. [Google Scholar]
- Bunzel, M.; Ralph, J.; Funk, C.; Steinhart, H. Isolation and identification of a ferulic acid dehydrotrimer from saponified maize bran insoluble fiber. Eur. Food Res. Technol 2003, 217, 128–133. [Google Scholar]
- Funk, C.; Ralph, J.; Steinhart, H.; Bunzel, M. Isolation and structural characterisation of 8-O-4/8-O-4- and 8-8/8-O-4-coupled dehydrotriferulic acids from maize bran. Phytochemistry 2005, 66, 363–371. [Google Scholar]
- Bunzel, M.; Ralph, J.; Bruening, P.; Steinhart, H. Structural identification of dehydrotriferulic and dehydrotetraferulic acids isolated from insoluble maize bran fiber. J. Agric. Food Chem 2006, 54, 6409–6418. [Google Scholar]
- Rouau, X.; Cheynier, V.; Surget, A.; Gloux, D.; Barron, C.; Meudec, E.; Louis-Montero, J.; Criton, M. A dehydrotrimer of ferulic acid from maize bran. Phytochemistry 2003, 63, 899–903. [Google Scholar]
- Piber, M.; Koehler, P. Identification of dehydro-ferulic acid-tyrosine in rye and wheat: Evidence for a covalent cross-link between arabinoxylans and proteins. J. Agric. Food Chem 2005, 53, 5276–5284. [Google Scholar]
- Zarra, I.; Sanchez, M.; Queijeiro, E.; Pena, M.J.; Revilla, G. The cell wall stiffening mechanism in Pinus pinaster Aiton: Regulation by apoplastic levels of ascorbate and hydrogen peroxide. J. Sci. Food Agric 1999, 79, 416–420. [Google Scholar]
- Fry, S.C. Phenolic components of the primary cell wall and their possible role in the hormonal regulation of growth. Planta 1979, 146, 343–351. [Google Scholar]
- Kamisaka, S.; Takeda, S.; Takahashi, K.; Shibata, K. Diferulic and ferulic acid in the cell wall of avena coleoptiles. Their relationships to mechanical properties of the cell wall. Physiol. Plant 1990, 78, 1–7. [Google Scholar]
- Azuma, T.; Okita, N.; Nanmori, T.; Yasuda, T. Relationship between the deposition of phenolic acids in the cell walls and the cessation of rapid growth in internodes of floating rice. Plant Prod. Sci 2005, 8, 447–453. [Google Scholar]
- Hossain, M.T.; Soga, K.; Wakabayashi, K.; Kamisaka, S.; Fujii, S.; Yamamoto, R.; Hoson, T. Modification of chemical properties of cell walls by silicon and its role in regulation of the cell wall extensibility in oat leaves. J. Plant Physiol 2007, 164, 385–393. [Google Scholar]
- Hatfield, R.D.; Ralph, J.; Grabber, J.H. Cell wall structural foundations: Molecular basis for improving forage digestibilities. Crop Sci 1999, 39, 27–37. [Google Scholar]
- Jung, H.G.; Lamb, J.F.S. Stem morphological and cell wall traits associated with divergent in vitro neutral detergent fiber digestibility in alfalfa clones. Crop Sci 2006, 46, 2054–2061. [Google Scholar]
- Jung, H.G.; Casler, M.D. Maize stem tissues: Impact of development on cell wall degradability. Crop Sci 2006, 46, 1801–1809. [Google Scholar]
- Eraso, F.; Hartley, R.D. Monomeric and dimeric phenolic constituents of plant cell walls possible factors influencing wall biodegradability. J. Sci. Food Agric 1990, 51, 163–170. [Google Scholar]
- Iiyama, K.; Lam, T.B.T. Structural characteristics of cell walls of forage grasses—Their nutritional evaluation for ruminants—Review. Asian Austr. J. Anim. Sci 2001, 14, 862–879. [Google Scholar]
- Lam, T.B.T.; Iiyama, K.; Stone, B.A. Hot alkali-labile linkages in the walls of the forage grass Phalaris aquatica and Lolium perenne and their relation to in vitro wall digestibility. Phytochemistry 2003, 64, 603–607. [Google Scholar]
- Martin, M.M. The evolution of cellulose digestion in insects. Philos. Trans. R. Soc. Lond. Ser. B 1991, 333, 281–288. [Google Scholar]
- McGinnis, A.J.; Kasting, R. Dietary cellulose effect on food consumption and growth of a grasshopper. Can. J. Zool 1967, 45, 165–167. [Google Scholar]
- Raubenheimer, D.; Simpson, S.J. The geometry of compensatory feeding in the locust. Anim. Behav 1993, 45, 953–964. [Google Scholar]
- Scriber, J.M.; Slansky, F. The nutritional ecology of immature insects. Annu. Rev. Entomol 1981, 26, 183–211. [Google Scholar]
- Fry, S.C. Cross-linking of matrix polymers in the growing cell walls of angiosperms. Annu. Rev. Plant Physiol. Plant Mol. Biol 1986, 37, 165–186. [Google Scholar]
- Santiago, R.; Malvar, R.A. Role of dehydrodiferulates in maize resistance to pests and diseases. Int. J. Mol. Sci 2010, 11, 691–703. [Google Scholar]
- Malvar, R.A.; Butrón, A.; Ordás, B.; Santiago, R. Causes of Natural Resistance to Stem Borers in Maize. In Crop Protection Research Advances; Williams, P.V., Ed.; Nova Science Publishers Inc.: New York, NY, USA, 2008; pp. 57–100. [Google Scholar]
- Groh, S.; Gonzalez-de-Leon, D.; Khairallah, M.M.; Jiang, C.; Bergvinson, D.; Bohn, M.; Hoisington, D.A.; Melchinger, A.E. QTL mapping in tropical maize: III. Genomic regions for resistance to Diatraea spp and associated traits in two RIL populations. Crop Sci 1998, 38, 1062–1072. [Google Scholar]
- Barros-Rios, J. Fortificación de la Pared Celular Como Mecanismo de Defensa Frente a la Plaga del Taladro en el Maíz. Ph.D. Dissertation, University of Vigo, Pontevedra, Spain, 2012. [Google Scholar]
- Markham, R.H.; Bosque-Pérez, N.A.; Borgemeister, C.; Meikle, W.G. Developing pest management strategies for the maize weevil, Sitophilus zeamais, and the large grain borer, Prostephanus truncatus, in the humid and sub-humid tropics. FAO Plant Prot. Bull 1994, 42, 125–136. [Google Scholar]
- Pingali, P.L.; Pandey, S. Meeting World Maize Needs: Technology Opportunities and Priorities for the Public Sector. In World Maize Facts and Trends. Meeting World Maize Needs: Technological Opportunities and Priorities for the Public Sector; Pingali, P.L., Ed.; International Maize and Wheat Improvement Center: Texcoco, Mexico, 2001. [Google Scholar]
- Burr, S.J.; Fry, S.C. Feruloylated arabinoxylans are oxidatively cross-linked by extracellular maize peroxidase but not by horseradish peroxidase. Mol. Plant 2009, 2, 883–892. [Google Scholar]
- Boutigny, A.-L.; Barreau, C.; Atanasova-Penichon, V.; Verdal-Bonnin, M.-N.; Pinson-Gadais, L.; Richard-Forget, F. Ferulic acid, an efficient inhibitor of type B trichothecene biosynthesis and Tri gene expression in Fusarium liquid cultures. Mycol. Res 2009, 113, 746–753. [Google Scholar]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin biosynthesis. Annu. Rev. Plant Biol 2003, 54, 519–46. [Google Scholar]
- Bonawitz, N.D.; Chapple, C. The genetics of lignin biosynthesis: Connecting genotype to phenotype. Annu. Rev. Genet 2010, 44, 337–363. [Google Scholar]
- Baucher, M.; Monties, B.; van Montagu, M.; Boerjan, W. Biosynthesis and genetic engineering of lignin. Crit. Rev. Plant Sci 1998, 17, 125–197. [Google Scholar]
- Barrière, Y.; Riboulet, C.; Méchin, V.; Maltese, S.; Pignon, M.; Cardinal, A.; Lapierre, C.; Lübberstedt, T.; Martinant, J.P. Genetics and Genomics of Lignification in Grass Cell Walls based on Maize as Model Species. In Genes, Genomes and Genomics; Global Science Books: Isleworth, UK, 2007; pp. 133–156. [Google Scholar]
- Coley, P.D. Herbivory and defensive characteristics of tree species in a lowland tropical forest. Ecol. Monogr 1983, 53, 209–233. [Google Scholar]
- Raupp, M.J. Effects of leaf toughness on mandibular wear of the leaf beetle, Plagiodera versicolora. Ecol. Entomol 1985, 10, 73–79. [Google Scholar]
- Felton, G.W.; Donato, K.; Delvecchio, R.J.; Duffey, S.S. Activation of plant foliar oxidases by insect feeding reduces nutritive quality of foliage for noctuid herbibores. J. Chem. Ecol 1989, 15, 2667–2694. [Google Scholar]
- Stevenson, P.C.; Anderson, J.C.; Blaney, W.M.; Simmonds, M.S.J. Developmental inhibition od Spodoptera litura (Fab) larvae by a novel caffeoylquinic acid form the wild groundnut, Arachis paraguariensis (Chod et Hassl). J. Chem. Ecol 1993, 19, 2917–2933. [Google Scholar]
- McMullen, M.D.; Frey, M.; Degenhardt, J. Genetics and biochemistry of insect resistance in maize. Handbook Maize 2009. [Google Scholar] [CrossRef]
- Vance, C.P.; Kirk, T.K.; Sherwood, R.T. Lignification as a mechanism of disease resistance. Annu. Rev. Phytopathol 1980, 18, 259–288. [Google Scholar]
- Stange, R.R.; Ralph, J.; Peng, J.P.; Sims, J.J.; Midland, S.L.; McDonald, R.E. Acidolysis and hot water extraction provide new insights into the composition of the induced “lignin-like” material from squash fruit. Phytochemistry 2001, 57, 1005–1011. [Google Scholar]
- Zhang, S.-H.; Yang, Q.; Ma, R.-C. Erwinia carotovora ssp. Carotovora infection induced “defense lignin” accumulation and lignin biosynthetic gene expression in chinese cabbage (Brassica rapa L. ssp pekinensis). J. Integr. Plant Biol 2007, 49, 993–1002. [Google Scholar]
- Karkonen, A.; Koutaniemi, S. Lignin biosynthesis studies in plant tissue cultures. J. Integr. Plant Biol 2010, 52, 176–185. [Google Scholar]
- Ride, J.P. The effect of induced lignification on the resistance of wheat cell walls to fungal degradation. Physiol. Plant Pathol 1980, 16, 187–192. [Google Scholar]
| Pest or disease | Tissue/organ | Cell wall component | References |
|---|---|---|---|
| Ostrinia nubilalis (ECB) | whorl tissue and leaf sheath, leaf blade and stalk | NDF, ADF and lignin | [11,12] |
| ECB | stalks and leaf sheaths | crude fiber, cellulose, hemicellulose, NDF, ADF, NDF adjusted by ADL, ADL, lignin and silica | [13–18] |
| ECB | immature and mature leaf blade and leaf sheath, rind, node, and pith | crude fibre and hydroxycinnamates | [19–21] |
| Diatraea grandiosella (SWCB) and Spodoptera frugiperda (FAW) | whorl leaves and leaves | hemicellulose, cellulose, lignin | [22–25] |
| SWCB and Diatraea saccharalis (SCB) | leaves | hydroxycinnamates | [26] |
| Sesamia nonagrioides (MCB) | stalk, pith, and leaf-sheaths | hydroxycinnamates | [27–31] |
| MCB and ECB | pith and rind | cell wall polyssacharides, lignin and hydroxycinnamates | [32] |
| Sithophilus zeamais (MW) | grain and pericarp | protein and hydroxycinnamates | [33–37] |
| Fusarium graminearum | pith | Hydroxycinnamates lignin | [38,39] |
| F. graminearum | grain, pericarp and aleurone | hydroxycinnamates | [40,41] |
| F. graminearum | silks | hemicellulose | [42] |
| Fusarium verticillioides | pericarp | hydroxycinnamates | [43] |
| Aspergillus flavus | grain | fiber and carbohydrates | [44] |
| A. flavus | cob | lignin content and composition | [45] |
| Puccinia polysora | leaves | NDF and ADF | [46] |
| Helminthosporium maydis | leaves | hydroxycinnamates | [47] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Santiago, R.; Barros-Rios, J.; Malvar, R.A. Impact of Cell Wall Composition on Maize Resistance to Pests and Diseases. Int. J. Mol. Sci. 2013, 14, 6960-6980. https://doi.org/10.3390/ijms14046960
Santiago R, Barros-Rios J, Malvar RA. Impact of Cell Wall Composition on Maize Resistance to Pests and Diseases. International Journal of Molecular Sciences. 2013; 14(4):6960-6980. https://doi.org/10.3390/ijms14046960
Chicago/Turabian StyleSantiago, Rogelio, Jaime Barros-Rios, and Rosa A. Malvar. 2013. "Impact of Cell Wall Composition on Maize Resistance to Pests and Diseases" International Journal of Molecular Sciences 14, no. 4: 6960-6980. https://doi.org/10.3390/ijms14046960